Assessment of the Therapeutic Efficacy of Silver Nanoparticles against Secondary Cystic Echinococcosis in BALB/c Mice
Abstract
1. Introduction
2. Materials and Methods
2.1. Preparation of Sidr Plant Extract
2.2. Synthesis and Characterization of Green Silver Nanoparticles
2.3. Liver Samples Collection and Viability Test
2.4. In Vivo Toxicity of AgNPs
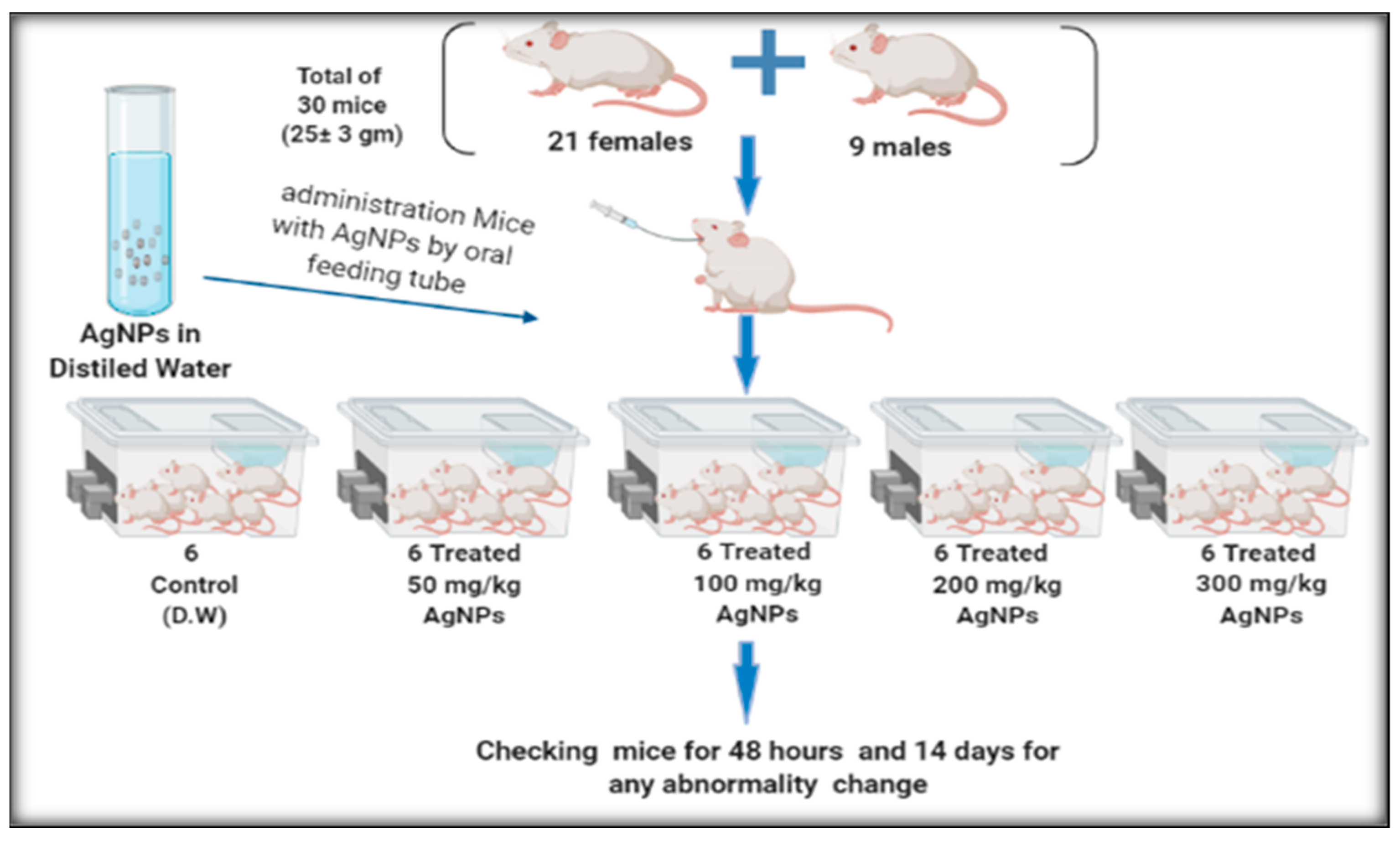
2.4.1. Experimental Animals
2.4.2. Acute (Short Term) Toxicity of AgNps Testing
2.5. In Vivo Effect of AgNps on Protoscoleces
2.5.1. Infection of BALB/c Mice with Secondary Hydatid Cysts
2.5.2. Treatment of the Infected BALB/c Mice with AgNPs
2.6. Histological Preparation
2.7. Statistical Analysis
3. Results
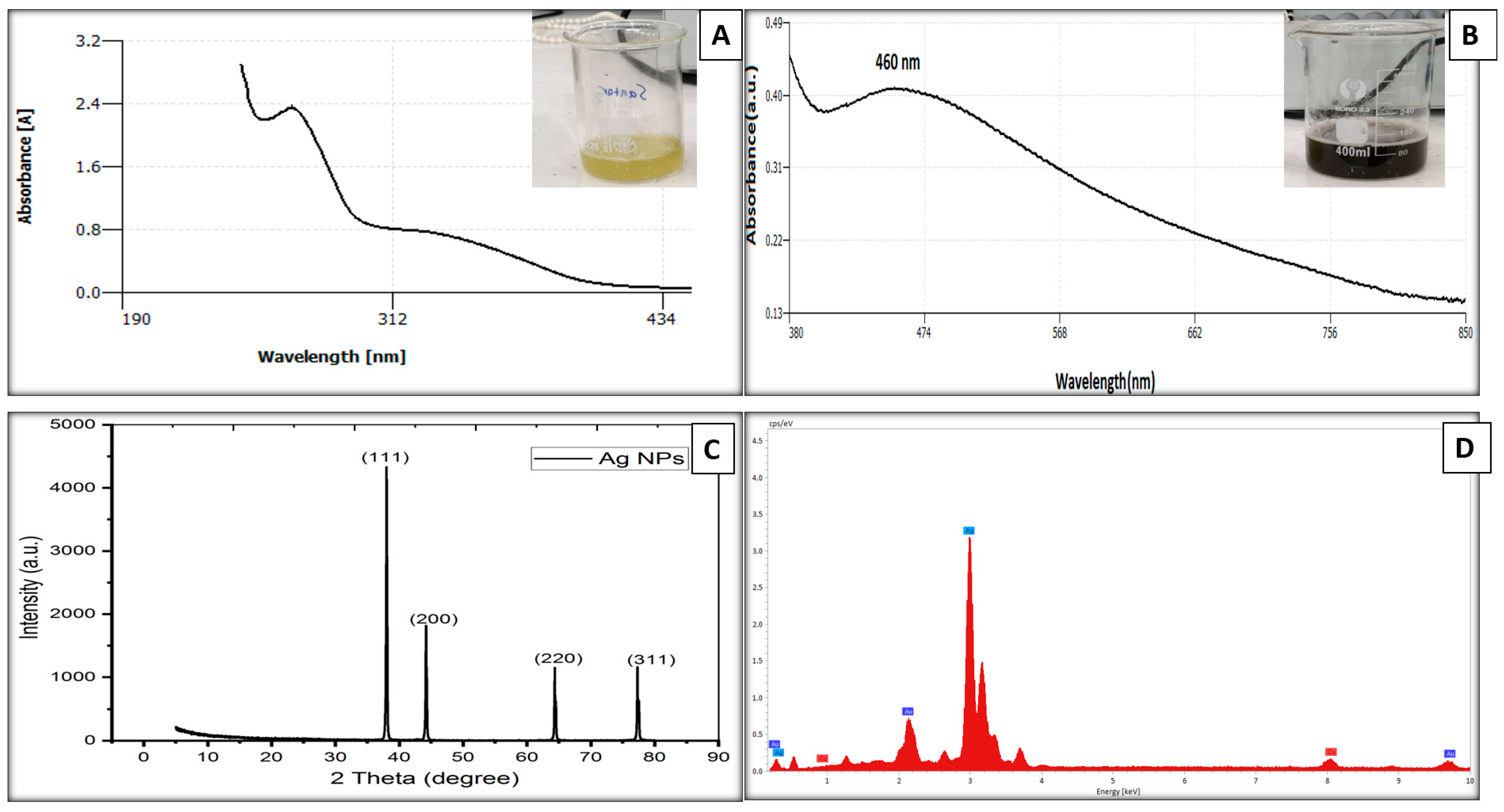
3.1. Biosynthesis of Silver Nanoparticles
3.1.1. Plant Extracts UV-Vis Spectrum
3.1.2. Nanoparticles Characterization
3.2. Viability of the Collected Protoscoleces
3.3. The Toxicity of Ag Nps
Acute (Short Term) Toxicity of AgNPs in BALB/c Mice
3.4. In Vivo Aspect of Echinococcosis
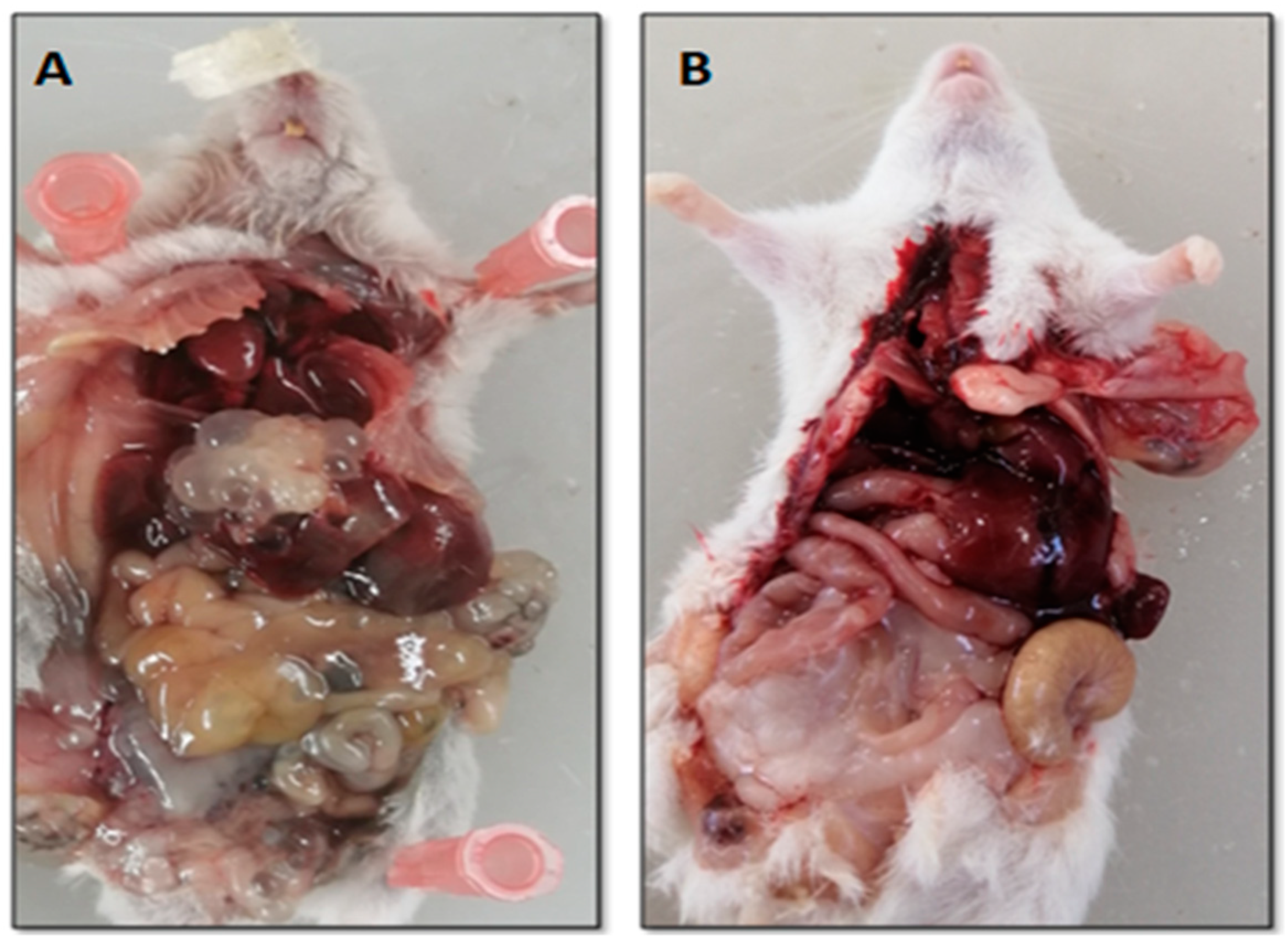
3.4.1. In Vivo Secondary Hydatid Cyst Infection
3.4.2. In Vivo Efficiency of AgNPs
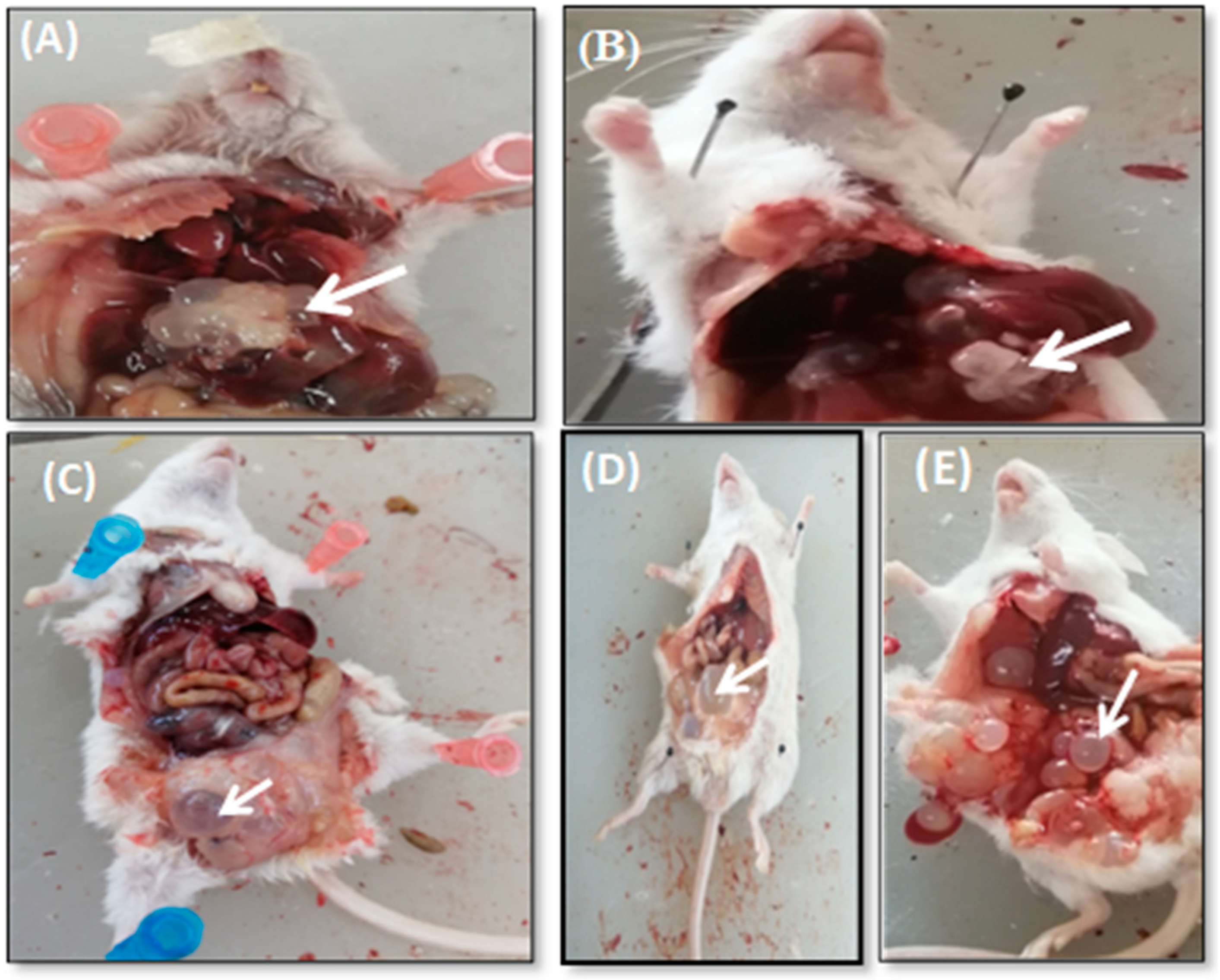
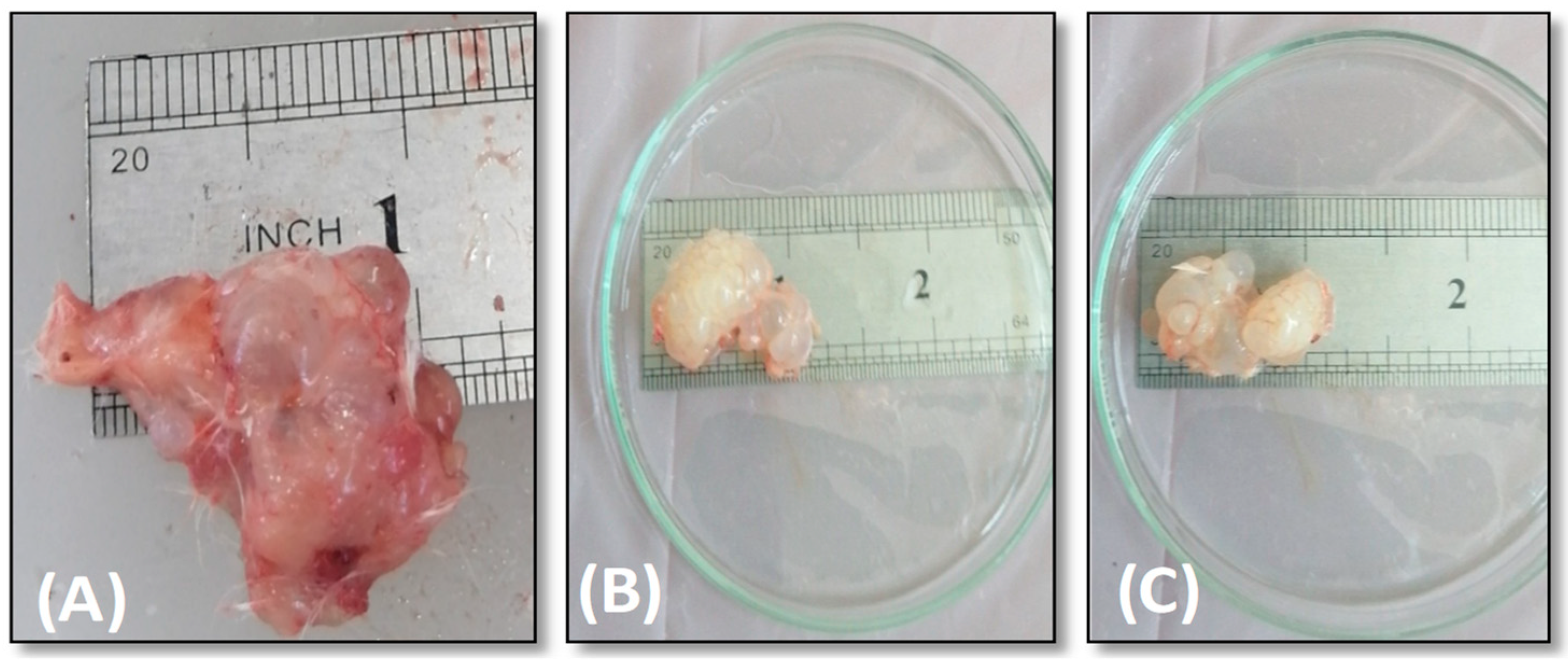
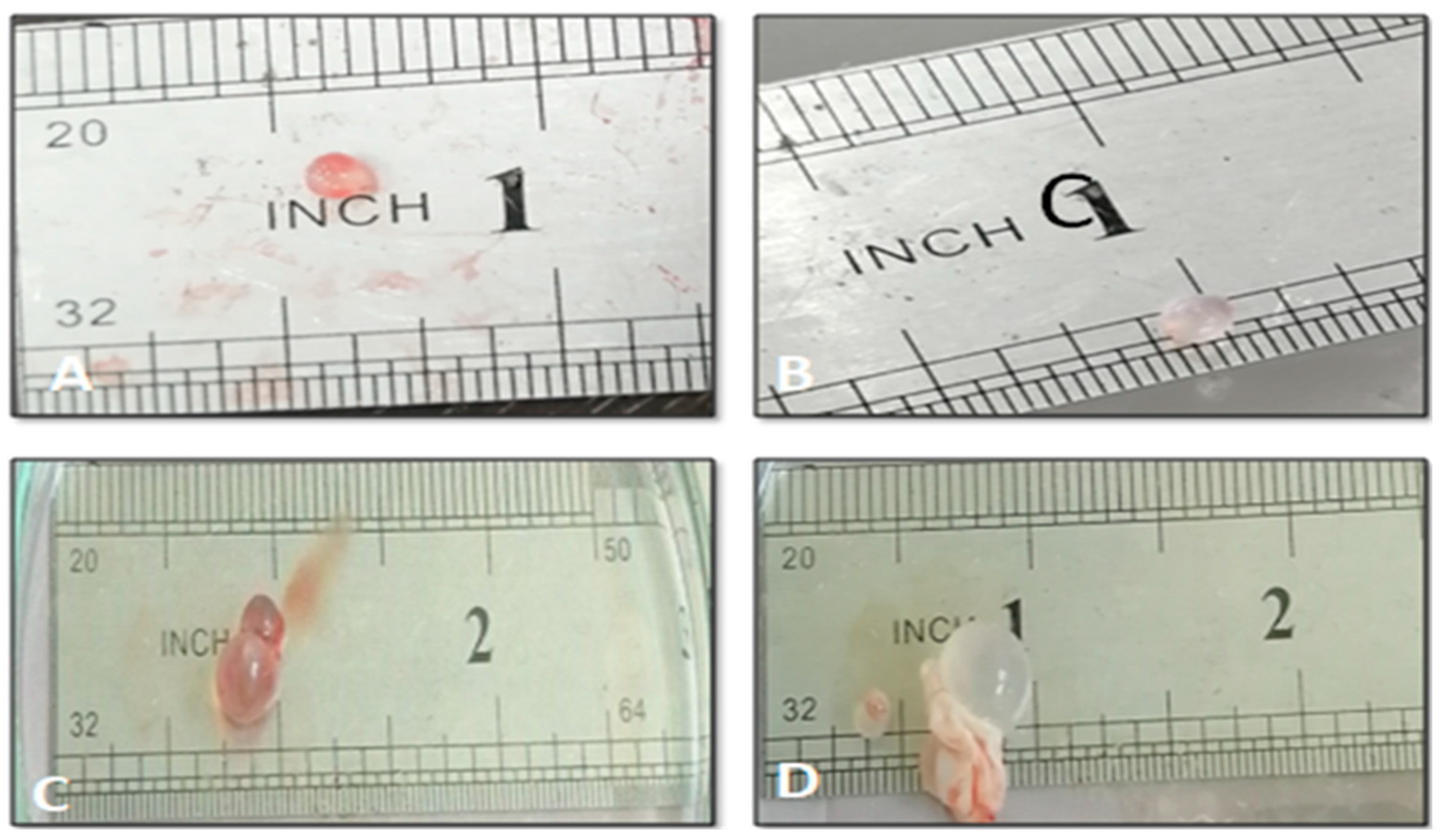
The Effect of AgNPs against Hydatid Cyst
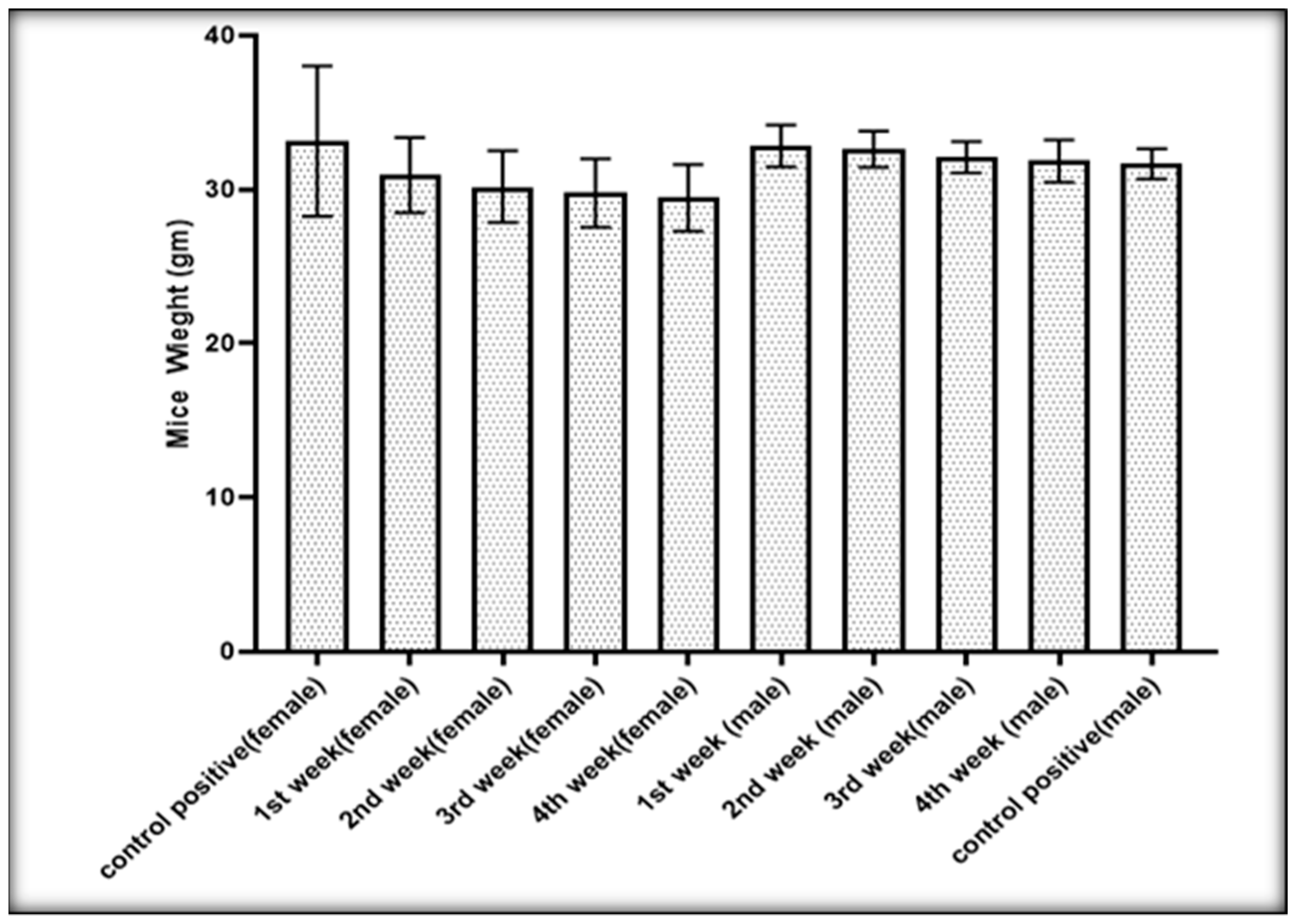
Effect of AgNPs on Mice Weight and Hydatid Cysts Size
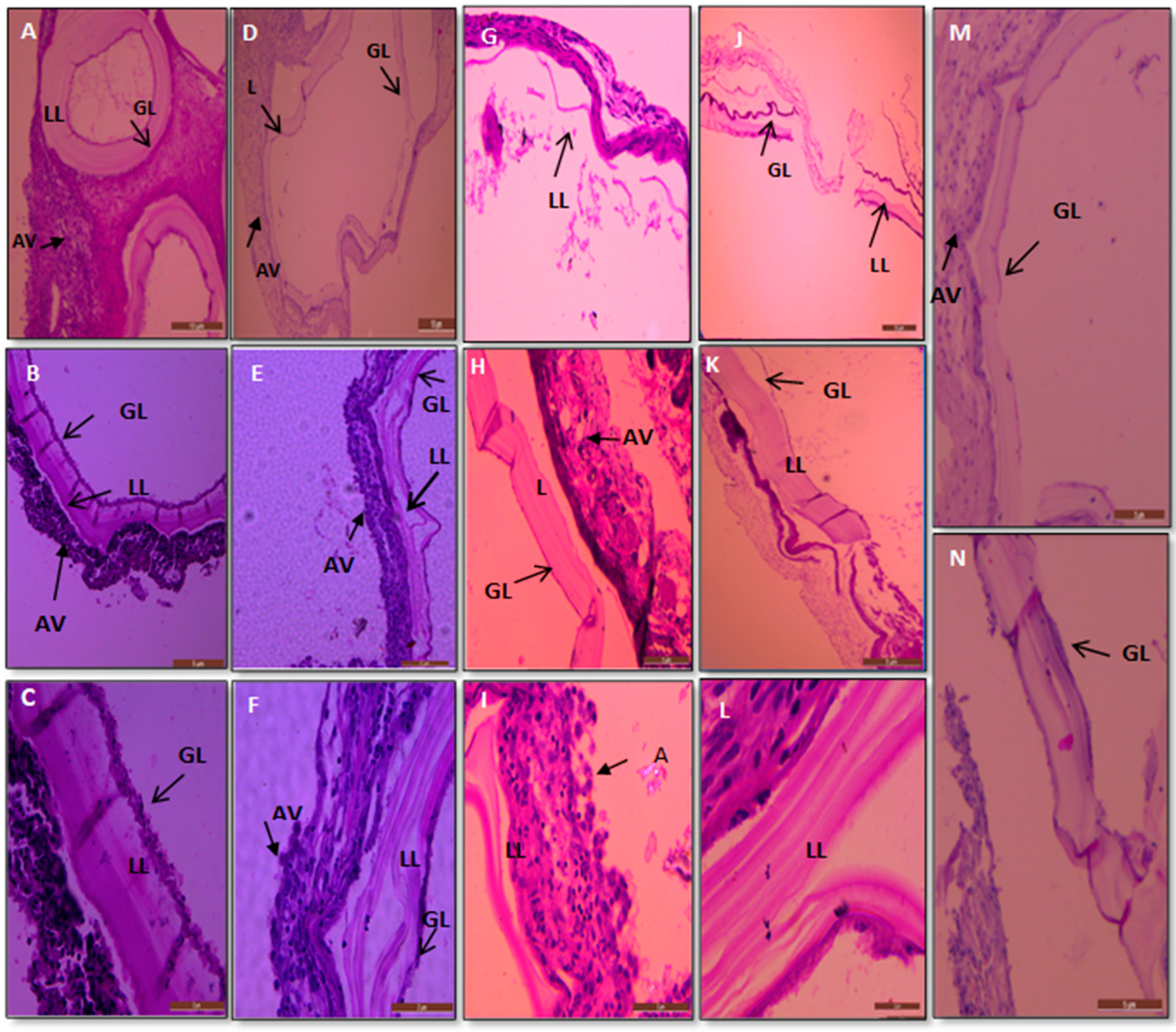
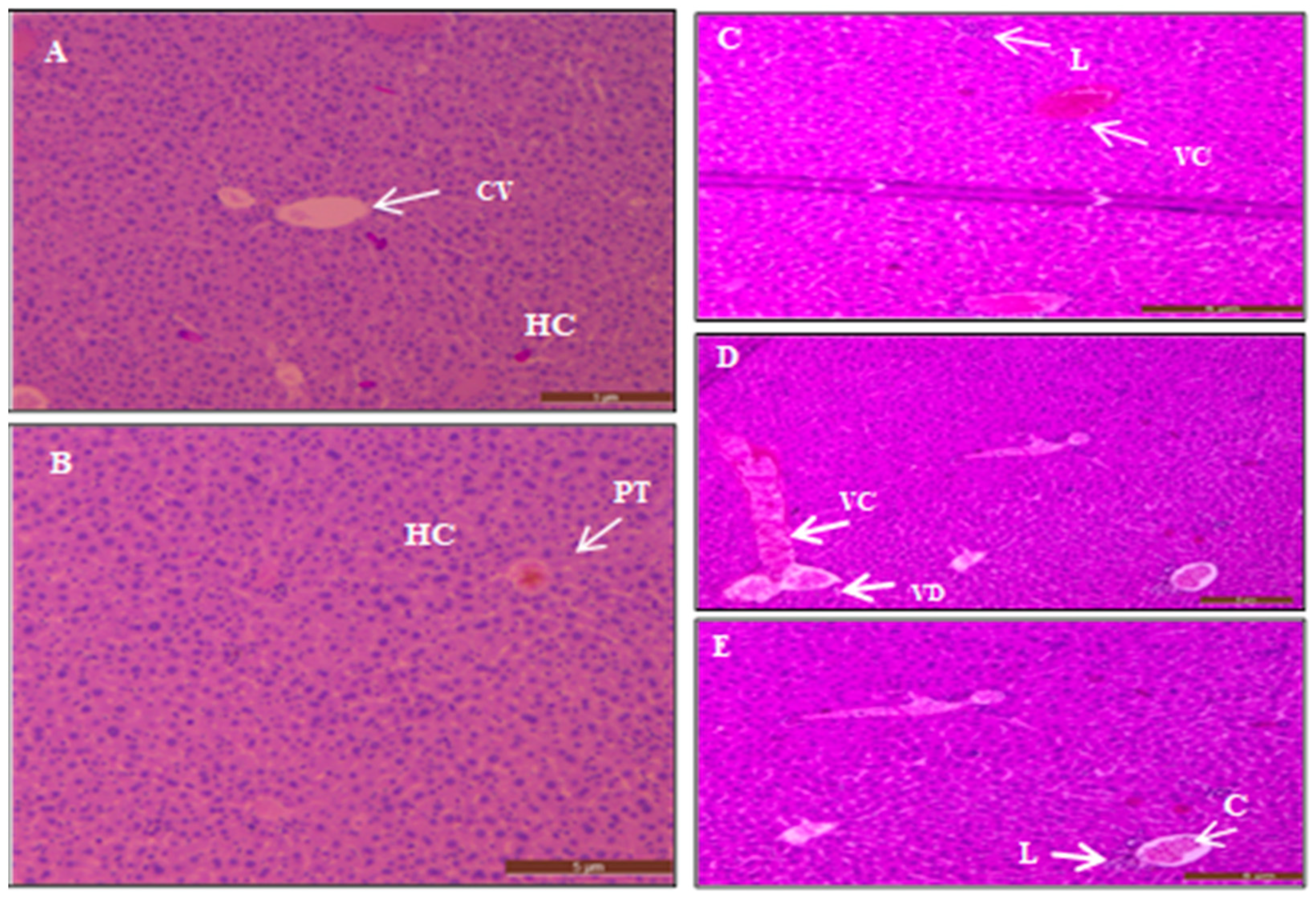
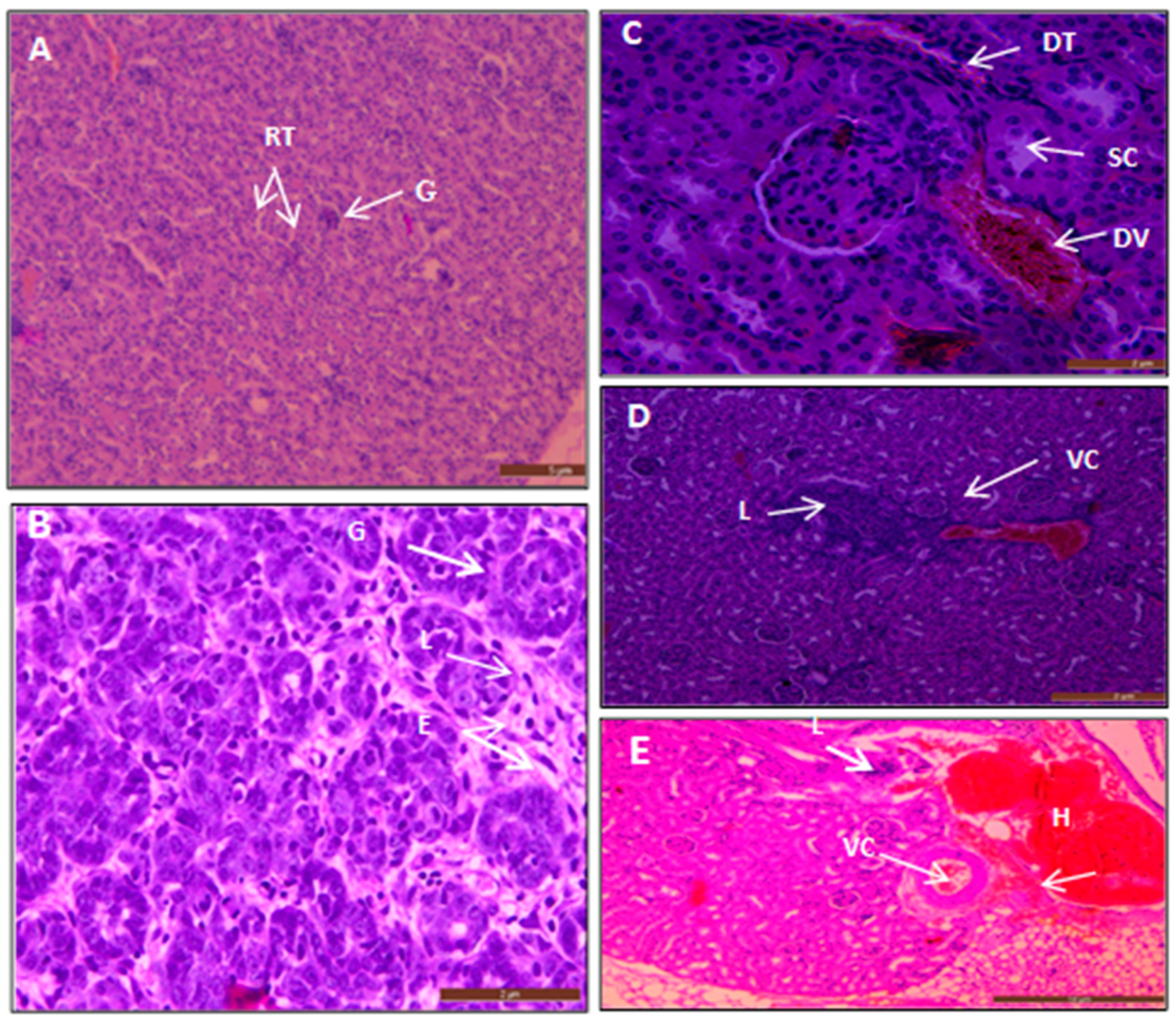
3.5. Histopathological Examinations
3.5.1. Hydatid Cysts from Treated and Untreated Infected Mice
3.5.2. The Histopathological Study of Acute Short-Term Toxicity of AgNPs
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Kern, P.; Menezes da Silva, A.; Akhan, O.; Müllhaupt, B.; Vizcaychipi, K.A.; Budke, C.; Vuitton, D.A. The Echinococcoses: Diagnosis, Clinical Management and Burden of Disease. Adv. Parasitol. 2017, 96, 259–369. [Google Scholar] [CrossRef]
- World Health Organization. Working to Overcome the Global Impact of Neglected Tropical Diseases: First WHO Report on Neglected Tropical Diseases: Summary; World Health Organization: Geneva, Switzerland, 2010; Available online: https://apps.who.int/iris/handle/10665/70503 (accessed on 9 November 2021).
- Benyan, A.K.; Mahdi, N.K.; Abdul-Amir, F.; Ubaid, O. Second reported case of multilocular hydatid disease in Iraq. Qatar Med. J. 2013, 2013, 28–29. [Google Scholar] [CrossRef]
- Hassan, Z.I.; Meerkhan, A.A.; Boufana, B.; Hama, A.A.; Ahmed, B.D.; Mero, W.M.S.; Orsten, S.; Interisano, M.; Pozio, E.; Casulli, A. Two haplotype clusters of Echinococcus granulosus sensu stricto in northern Iraq (Kurdistan region) support the hypothesis of a parasite cradle in the Middle East. Acta Trop. 2017, 172, 201–207. [Google Scholar] [CrossRef]
- Shi, H.; Lei, Y.; Wang, B.; Xing, G.; Lv, H.; Jiang, Y. Protoscolicidal effects of chenodeoxycholic acid on protoscoleces of Echinococcus granulosus. Exp. Parasitol. 2016, 167, 76–82. [Google Scholar] [CrossRef]
- Albi, A.; Baudin, F.; Matmar, M.; Archambeau, D.; Ozier, Y. Severe hypernatremia after hypertonic saline irrigation of hydatid cysts. Anesth. Analg. 2002, 95, 1806–1808. [Google Scholar] [CrossRef]
- Siles-Lucas, M.; Casulli, A.; Cirilli, R.; Carmena, D. Progress in the pharmacological treatment of human cystic and alveolar echinococcosis: Compounds and therapeutic targets. PLoS Negl. Trop. Dis. 2018, 12, e0006422. [Google Scholar] [CrossRef]
- Rai, M.; Ingle, A.P.; Paralikar, P.; Gupta, I.; Medici, S.; Santos, C.A. Recent advances in use of silver nanoparticles as antimalarial agents. Int. J. Pharm. 2017, 526, 254–270. [Google Scholar] [CrossRef]
- Burdușel, A.C.; Gherasim, O.; Grumezescu, A.M.; Mogoantă, L.; Ficai, A.; Andronescu, E. Biomedical Applications of Silver Nanoparticles: An Up-to-Date Overview. Nanomaterials 2018, 8, 681. [Google Scholar] [CrossRef]
- Barabadi, H.; Honary, S.; Ali Mohammadi, M.; Ahmadpour, E.; Rahimi, M.T.; Alizadeh, A.; Naghibi, F.; Saravanan, M. Green chemical synthesis of gold nanoparticles by using Penicillium aculeatum and their scolicidal activity against hydatid cyst protoscolices of Echinococcus granulosus. Environ. Sci. Pollut. Res. Int. 2017, 24, 5800–5810. [Google Scholar] [CrossRef]
- Rahimi, M.T.; Ahmadpour, E.; Rahimi Esboei, B.; Spotin, A.; Koshki, M.H.K.; Alizadeh, A.; Honary, S.; Barabadi, H.; Mohammadi, M.A. Scolicidal activity of biosynthesized silver nanoparticles against Echinococcus granulosus protoscolices. Int. J. Surg. 2015, 19, 128–133. [Google Scholar] [CrossRef]
- Salih, T.A.; Hassan, K.T.; Majeed, S.R.; Ibraheem, I.J.; Hassan, O.M.; Obaid, A. In vitro scolicidal activity of synthesised silver nanoparticles from aqueous plant extract against Echinococcus granulosus. Biotechnol. Rep. 2020, 28, e00545. [Google Scholar] [CrossRef] [PubMed]
- Nassef, N.E.; Saad, A.E.; Harba, N.M.; Beshay, E.V.N.; Gouda, M.A.; Shendi, S.S.; Mohamed, A.S.E. Evaluation of the therapeutic efficacy of albendazole-loaded silver nanoparticles against Echinococcus granulosus infection in experimental mice. J. Parasit. Dis. 2019, 43, 658–671. [Google Scholar] [CrossRef] [PubMed]
- Shnawa, B.H.; Al-Ali, S.J.; Swar, S.O. Nanoparticles as a new approach for treating hydatid cyst disease. In Veterinary Pathobiology and Public Health; Unique Scientific Publishers: Faisalabad, Pakistan, 2021; pp. 180–189. [Google Scholar] [CrossRef]
- Halwani, E.M. Rapid biosynthesis method and characterization of silver nanoparticles using Zizyphus spina christi leaf extract and their antibacterial efficacy in therapeutic application. J. Biomater. Nanobiotechnol. 2016, 8, 22–35. [Google Scholar] [CrossRef]
- Oluwaniyi, O.O.; Adegoke, H.I.; Adesuji, E.T.; Alabi, A.B.; Bodede, S.O.; Labulo, A.H.; Oseghale, C.O. Biosynthesis of silver nanoparticles using aqueous leaf extract of Thevetia peruviana Juss and its antimicrobial activities. Appl. Nanosci. 2016, 6, 903–912. [Google Scholar] [CrossRef]
- Smyth, J.D.; Barrett, N.J. Procedures for testing the viability of human hydatid cysts following surgical removal, especially after chemotherapy. Trans. R. Soc. Trop. Med. Hyg. 1980, 74, 649–652. [Google Scholar] [CrossRef]
- Shnawa, B.H.; Gorony, S.h.M.; Khalid, K.M. Efficacy of cyperus rotundus rhizomes-tubers extracts against protoscoleces of Echinococcus granulosus. World J. Pharm. Res. 2017, 6, 157–179. [Google Scholar] [CrossRef][Green Version]
- Ismail, E.; Saad, M. In Vivo and In Vitro Survival Rates of Protoscoleces Kept at Different Constant Temperature. Open J. Epidemiol. 2017, 7, 124–130. [Google Scholar] [CrossRef][Green Version]
- Klassen, C.D.; Casarett, L.J.; Amdur, M.O. Toxicology: The Basic Science of Poisons; Macmillan: Basingstoke, UK, 1986; ISBN 9780023646508. Available online: https://www.abebooks.co.uk/servlet/SearchResults?isbn=9780023646508 (accessed on 9 November 2021).
- Maneewattanapinyo, P.; Banlunara, W.; Thammacharoen, C.; Ekgasit, S.; Kaewamatawong, T. An evaluation of acute toxicity of colloidal silver nanoparticles. J. Vet. Med. Sci. 2011, 73, 1417–1423. [Google Scholar] [CrossRef]
- Adeyemi, O.S.; Dewumi, I. Biochemical evaluation of silver nanoparticles in Wistar rats. Int. Sch. Res. Not. 2014, 2014, 196091. [Google Scholar] [CrossRef]
- Pérez-Serrano, J.; Denegri, G.; Casado, N.; Rodriguez-Caabiero, F. In vivo effect of oral albendazole and albendazole sulphoxide on development of secondary echinococcosis in mice. Int. J. Parasitol. 1997, 27, 1341–1345. [Google Scholar] [CrossRef]
- Ahmadnia, S.; Moazeni, M.; Mohammadi-Samani, S.; Oryan, A. In vivo evaluation of the efficacy of albendazole sulfoxide and albendazole sulfoxide loaded solid lipid nanoparticles against hydatid cyst. Exp. Parasitol. 2013, 135, 314–319. [Google Scholar] [CrossRef]
- Slaoui, M.; Fiette, L. Histopathology procedures: From tissue sampling to histopathological evaluation. Methods Mol. Biol. 2011, 691, 69–82. [Google Scholar] [CrossRef]
- Iravani, S. Green synthesis of metal nanoparticles using plants. Green Chem. 2011, 13, 2638–2650. [Google Scholar] [CrossRef]
- Soltani, S.; Rafiei, A.; Ramezani, Z.; Abbaspour, M.R.; Jelowdar, A.; Kahvaz, M.S. Evaluation of the Hydatid Cyst Membrane Permeability of Albendazole and Albendazole Sulfoxide-Loaded Solid Lipid Nanoparticles. Jundishapur J. Nat. Pharm. Prod. 2017, 12, e34723. [Google Scholar] [CrossRef]
- Choi, J.; Reipa, V.; Hitchins, V.M.; Goering, P.L.; Malinauskas, R.A. Physicochemical characterization and in V itro hemolysis evaluation of silver nanoparticles. Toxicol. Sci. 2011, 123, 133–143. [Google Scholar] [CrossRef] [PubMed]
- Mittal, A.K.; Chisti, Y.; Banerjee, U.C. Synthesis of metallic nanoparticles using plant extracts. Biotechnol. Adv. 2013, 31, 346–356. [Google Scholar] [CrossRef]
- Shnawa, B.H. Advances in the use of nanoparticles as anti-cystic echinococcosis agents: A review article. J. Pharm. Res. Int. 2018, 24, 1–14. [Google Scholar] [CrossRef]
- Albalawi, A.E.; Alanazi, A.D.; Baharvand, P.; Sepahvand, M.; Mahmoudvand, H. High Potency of Organic and Inorganic Nanoparticles to Treat Cystic Echinococcosis: An Evidence-Based Review. Nanomaterials 2020, 10, 2538. [Google Scholar] [CrossRef] [PubMed]
- Jalil, P.J.; Shnawa, B.H.; Hamad, S.M. Silver Nanoparticles: Green Synthesis, Characterization, Blood Compatibility and Protoscolicidal Efficacy against Echinococcus granulosus. Pak. Vet. J. 2021, 41, 393–399. [Google Scholar] [CrossRef]
- Shnawa, B.H.; Hamad, S.M.; Barzinjy, A.A.; Kareem, P.A.; Ahmed, M.H. Scolicidal activity of biosynthesized zinc oxide nanoparticles by Mentha longifolia L. leaves against Echinococcus granulosus protoscolices. Emergent Mater. 2021, 1–11. [Google Scholar] [CrossRef]
- Abalaka, M.; Daniyan, S.; Mann, A. Evaluation of the antimicrobial activities of two Ziziphus species (Ziziphus mauritiana L. and Ziziphus spinachristi L.) on some microbial pathogens. Afr. J. Pharm. Pharmacol. 2010, 4, 135–139. [Google Scholar] [CrossRef]
- Khaleel, S.M.J.; Jaran, A.S.; Haddadin, M.S.Y. Evaluation of Total Phenolic Content and Antioxidant Activity of Three Leaf Extracts of Ziziphus spina-christi (Sedr) Grown in Jordan. J. Adv. Med. Med. Res. 2016, 14, 1–8. [Google Scholar] [CrossRef]
- Mervat, E.-H.; Mohamed, A.A.; Salem, M.Z.; Abdelkareem, M.S.; Ali, H.M. Chemical composition, antioxidant capacity and antibacterial activity against some potato bacterial pathogens of fruit extracts from Phytolacca dioica and Ziziphus spina-christi grown in Egypt. Sci. Hortic. 2018, 233, 225–232. [Google Scholar] [CrossRef]
- Ibrahim, H.M.M. Green synthesis and characterization of silver nanoparticles using banana peel extract and their antimicrobial activity against representative microorganisms. J. Radiat. Res. Appl. Sci. 2015, 8, 265–275. [Google Scholar] [CrossRef]
- Oves, M.; Aslam, M.; Rauf, M.A.; Qayyum, S.; Qari, H.A.; Khan, M.S.; Alam, M.Z.; Tabrez, S.; Pugazhendhi, A.; Ismail, I.M.I. Antimicrobial and anticancer activities of silver nanoparticles synthesized from the root hair extract of Phoenix dactylifera. Mater. Sci. Eng. C 2018, 89, 429–443. [Google Scholar] [CrossRef] [PubMed]
- Arreche, R.A.; de Oca-Vásquez, G.M.; Vega-Baudrit, J.R.; Vázquez, P.G. Synthesis of Silver Nanoparticles Using Extracts from Yerba Mate (Ilex paraguariensis) Wastes. Waste Biomass Valorization 2020, 11, 245–253. [Google Scholar] [CrossRef]
- Faried, M.; Shameli, K.; Miyake, M.; Hajalilou, A.; Zamanian, A.; Zakaria, Z.; Abouzari-Lotf, E.; Hara, H.; Khairudinet, N.B.B.A.; Nordi, M.F.B.M. A green approach for the synthesis of silver nanoparticles using ultrasonic radiation’s times in sodium alginate media: Characterization and antibacterial evaluation. J. Nanomater. 2016, 2016, 4941231. [Google Scholar] [CrossRef]
- Dos Santos, C.A.; Seckler, M.M.; Ingle, A.P.; Gupta, I.; Galdiero, S.; Galdiero, M.; Gade, A.; Rai, M. Silver nanoparticles: Therapeutical uses, toxicity, and safety issues. J. Pharm. Sci. 2014, 103, 1931–1944. [Google Scholar] [CrossRef]
- Naseri, M.; Akbarzadeh, A.; Spotin, A.; Akbari, N.A.; Mahami-Oskouei, M.; Ahmadpour, E. Scolicidal and apoptotic activities of albendazole sulfoxide and albendazole sulfoxide-loaded PLGA-PEG as a novel nanopolymeric particle against Echinococcus granulosus protoscoleces. Parasitol. Res. 2016, 115, 4595–4603. [Google Scholar] [CrossRef]
- Sardari, R.R.R.; Zarchi, S.R.; Talebi, A.; Nasri, S.; Imani, S.; Khordamehr, A.; Sheshde, S.A.R. Toxicological effects of silver nanoparticles in rats. Afr. J. Microbiol. Res. 2012, 6, 5587–5593. [Google Scholar] [CrossRef]
- Elkhawass, E.A.; Mohallal, M.E.; Soliman, M.F.M. Acute toxicity of different sizes of silver nanoparticles intraperitoneally injected in BALB/c mice using two toxicological methods. Int. J. Pharm. Pharm. Sci. 2015, 7, 94–99. [Google Scholar]
- AshaRani, P.V.; Low Kah Mun, G.; Hande, M.P.; Valiyaveettil, S. Cytotoxicity and genotoxicity of silver nanoparticles in human cells. ACS Nano 2009, 3, 279–290. [Google Scholar] [CrossRef] [PubMed]
- Huang, H.; Lai, W.; Cui, M.; Liang, L.; Lin, Y.; Fang, Q.; Liu, Y.; Xie, L. An Evaluation of Blood Compatibility of Silver Nanoparticles. Sci. Rep. 2016, 6, 25518. [Google Scholar] [CrossRef]
- Bhol, K.C.; Schechter, P.J. Effects of nanocrystalline silver (NPI 32101) in a rat model of ulcerative colitis. Dig. Dis. Sci. 2007, 52, 2732–2742. [Google Scholar] [CrossRef]
- Said, D.E.; Elsamad, L.M.; Gohar, Y.M. Validity of silver, chitosan, and curcumin nanoparticles as anti-Giardia agents. Parasitol. Res. 2012, 111, 545–554. [Google Scholar] [CrossRef]
- Kim, Y.S.; Kim, J.S.; Cho, H.S.; Rha, D.S.; Kim, J.M.; Park, J.D.; Choi, B.S.; Lim, R.; Chang, H.K.; Chung, Y.H.; et al. Twenty-eight-day oral toxicity, genotoxicity, and gender-related tissue distribution of silver nanoparticles in Sprague-Dawley rats. Inhal. Toxicol. 2008, 20, 575–583. [Google Scholar] [CrossRef]
- Xue, Y.; Zhang, S.; Huang, Y.; Zhang, T.; Liu, X.; Hu, Y.; Zhang, Z.; Tang, M. Acute toxic effects and gender-related biokinetics of silver nanoparticles following an intravenous injection in mice. J. Appl. Toxicol. 2012, 32, 890–899. [Google Scholar] [CrossRef]
- Wen, H.; Dan, M.; Yang, Y.; Lyu, J.; Shao, A.; Cheng, X.; Chen, L.; Xu, L. Acute toxicity and genotoxicity of silver nanoparticle in rats. PLoS ONE 2017, 12, e0185554. [Google Scholar] [CrossRef]
- Durán, N.; Silveira, C.P.; Durán, M.; Martinez, D.S.T. Silver nanoparticle protein corona and toxicity: A mini-review. J. Nanobiotechnol. 2015, 13, 55. [Google Scholar] [CrossRef]
- Zhang, W.-B.; Jones, M.K.; Li, J.; Mcmanus, D.P. Echinococcus granulosus: Pre-culture of protoscoleces in vitro significantly increases development and viability of secondary hydatid cysts in mice. Exp. Parasitol. 2005, 110, 88–90. [Google Scholar] [CrossRef]
- Ahmadnia, S.; Moazeni, M.; Mohammadi-Samani, S.; Alavi, A.M. Hydatid cyst formation in male BALB/c mice following the intraperitoneal injection of live protoscoleces and activated oncospheres: A comparative study. J. Parasit. Dis. 2014, 38, 77–80. [Google Scholar] [CrossRef]
- Farhadi, M.; Haniloo, A.; Rostamizadeh, K.; Faghihzadeh, S. Efficiency of flubendazole-loaded mPEG-PCL nanoparticles: A promising formulation against the protoscoleces and cysts of Echinococcus granulosus. Acta Trop. 2018, 187, 190–200. [Google Scholar] [CrossRef]
- Aminpour, S.; Rafiei, A.; Jelowdar, A.; Kouchak, M. Evaluation of the Protoscolicidal Effects of Albendazole and Albendazole Loaded Solid Lipid Nanoparticles. Iran J. Parasitol. 2019, 14, 127–135. [Google Scholar] [CrossRef] [PubMed]
- Navvabi, A.; Homaei, A.; Khademvatan, S.; Khadem Ansari, M.H.; Keshavarz, M. Combination of TiO2 nanoparticles and Echinometra mathaeis gonad extracts: In vitro and in vivo scolicidal activity against hydatid cysts. Biocatal. Agric. Biotechnol. 2019, 22, 101432. [Google Scholar] [CrossRef]
- Jelowdar, A.; Rafiel, A.; Abbaspour, M.R.; Rashidi, I.; Rahdar, M. Efficacy of combined albendazol and praziquntel and their loaded solid lipid nanoparticles components in chemoprophylaxis of experimental hydatidosis. Asian Pac. J. Trop. Biomed. 2017, 7, 549–554. [Google Scholar] [CrossRef]
- Darvishi, M.M.; Moazeni, M.; Alizadeh, M.; Abedi, M.; Tamaddon, A.M. Evaluation of the efficacy of albendazole sulfoxide (ABZ-SO)-loaded chitosan-PLGA nanoparticles in the treatment of cystic echinococcosis in laboratory mice. Parasitol. Res. 2020, 119, 4233–4241. [Google Scholar] [CrossRef]
- Aryamand, S.; Khademvatan, S.; Hazrati Tappeh, K.; Heshmatian, B.; Jelodar, A. In Vitro and in Vivo Scolicidal Activities of Holothuria leucospilota Extract and CeO2 Nanoparticles against Hydatid Cyst. Iran J. Parasitol. 2019, 14, 269–279. [Google Scholar] [PubMed]
- Ismail, E.; Saad, M.; Elsadig, A. Effect of Albendazole and Mebendazole on Hydatid Cyst of Mice. Open J. Epidemiol. 2017, 7, 307–316. [Google Scholar] [CrossRef]
- Torabi, N.; Dobakhti, F.; Faghihzadeh, S.; Haniloo, A. In vitro and in vivo effects of chitosan-praziquantel and chitosan-albendazole nanoparticles on Echinococcus granulosus Metacestodes. Parasitol. Res. 2018, 117, 2015–2023. [Google Scholar] [CrossRef] [PubMed]
- Taylor, D.H.; Richards, K.S.; Morris, D.L. Rapid recovery of Echinococcus granulosus following ‘successful’ albendazole therapy in a gerbil model. J. Helminthol. 1989, 63, 349–352. [Google Scholar] [CrossRef]
- Pérez-Serrano, J.; Casado, N.; Guillermo Denegri Rodriguez-Caabeiro, F. The effects of albendazole and albendazole sulphoxide combination-therapy on Echinococcus granulosus in vitro. Int. J. Parasitol. 1994, 24, 219–224. [Google Scholar] [CrossRef]
- Moazeni, M.; Larki, S.; Saharkhiz, M.J.; Oryan, A.; Ansary Lari, M.; Mootabi Alavi, A. In vivo study of the efficacy of the aromatic water of Zataria multiflora on hydatid cysts. Antimicrob. Agents Chemother. 2014, 58, 6003–6008. [Google Scholar] [CrossRef] [PubMed]
- Lv, H.; Jiang, Y.; Liao, M.; Sun, H.; Zhang, S.; Peng, X. In vitro and in vivo treatments of Echinococcus granulosus with Huaier aqueous extract and albendazole liposome. Parasitol. Res. 2013, 112, 193–198. [Google Scholar] [CrossRef] [PubMed]



Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hamad, S.M.; Shnawa, B.H.; Jalil, P.J.; Ahmed, M.H. Assessment of the Therapeutic Efficacy of Silver Nanoparticles against Secondary Cystic Echinococcosis in BALB/c Mice. Surfaces 2022, 5, 91-112. https://doi.org/10.3390/surfaces5010004
Hamad SM, Shnawa BH, Jalil PJ, Ahmed MH. Assessment of the Therapeutic Efficacy of Silver Nanoparticles against Secondary Cystic Echinococcosis in BALB/c Mice. Surfaces. 2022; 5(1):91-112. https://doi.org/10.3390/surfaces5010004
Chicago/Turabian StyleHamad, Samir Mustafa, Bushra Hussain Shnawa, Parwin Jalal Jalil, and Mukhtar H. Ahmed. 2022. "Assessment of the Therapeutic Efficacy of Silver Nanoparticles against Secondary Cystic Echinococcosis in BALB/c Mice" Surfaces 5, no. 1: 91-112. https://doi.org/10.3390/surfaces5010004
APA StyleHamad, S. M., Shnawa, B. H., Jalil, P. J., & Ahmed, M. H. (2022). Assessment of the Therapeutic Efficacy of Silver Nanoparticles against Secondary Cystic Echinococcosis in BALB/c Mice. Surfaces, 5(1), 91-112. https://doi.org/10.3390/surfaces5010004







