Archaeometric Identification of a Perfume from Roman Times
Abstract
1. Introduction
2. Materials and Methods
2.1. XRD Analysis
2.2. Scanning Electron Microscopy—Energy Dispersive Spectroscopy
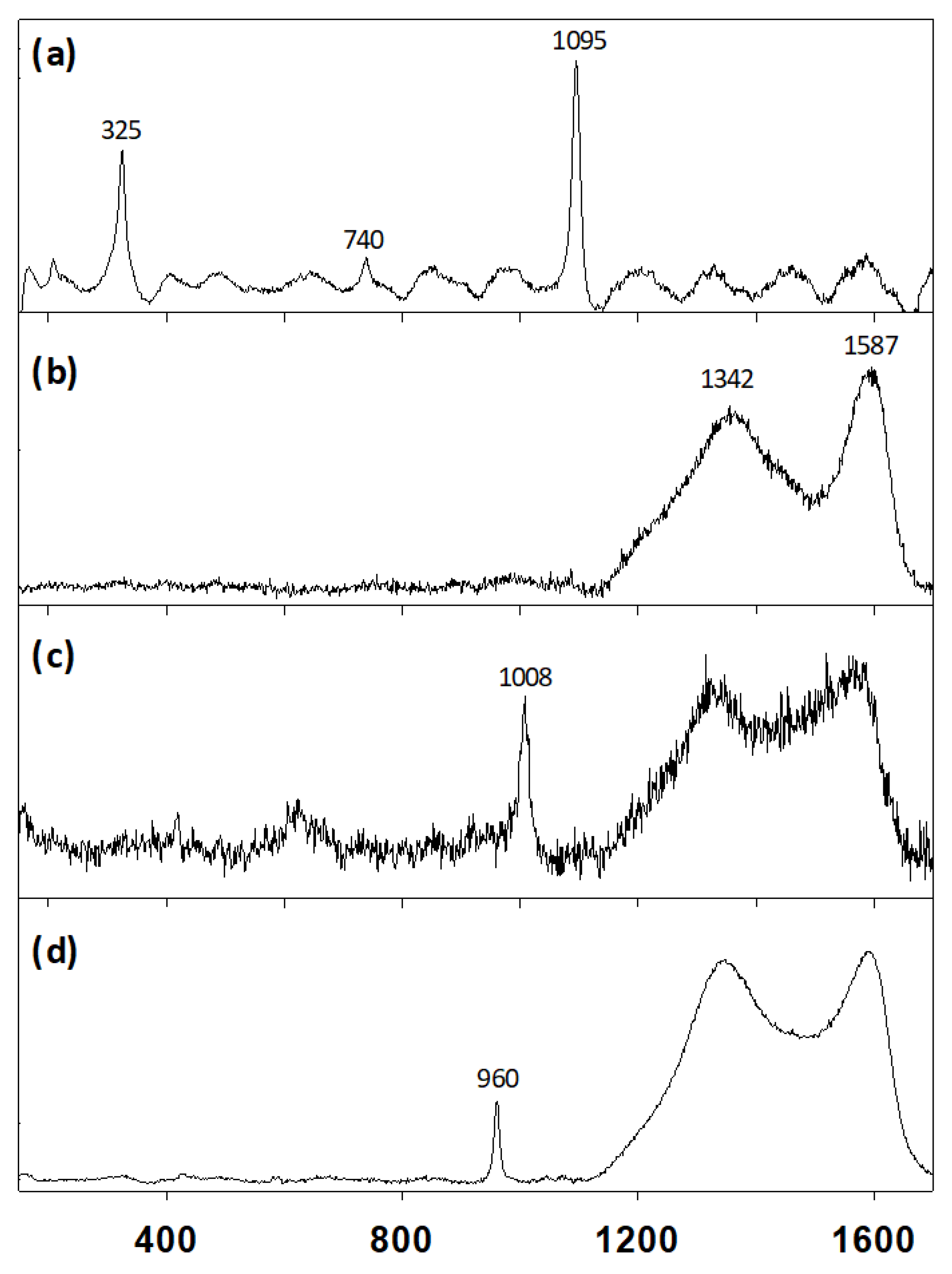
2.3. Micro-Raman Spectroscopy
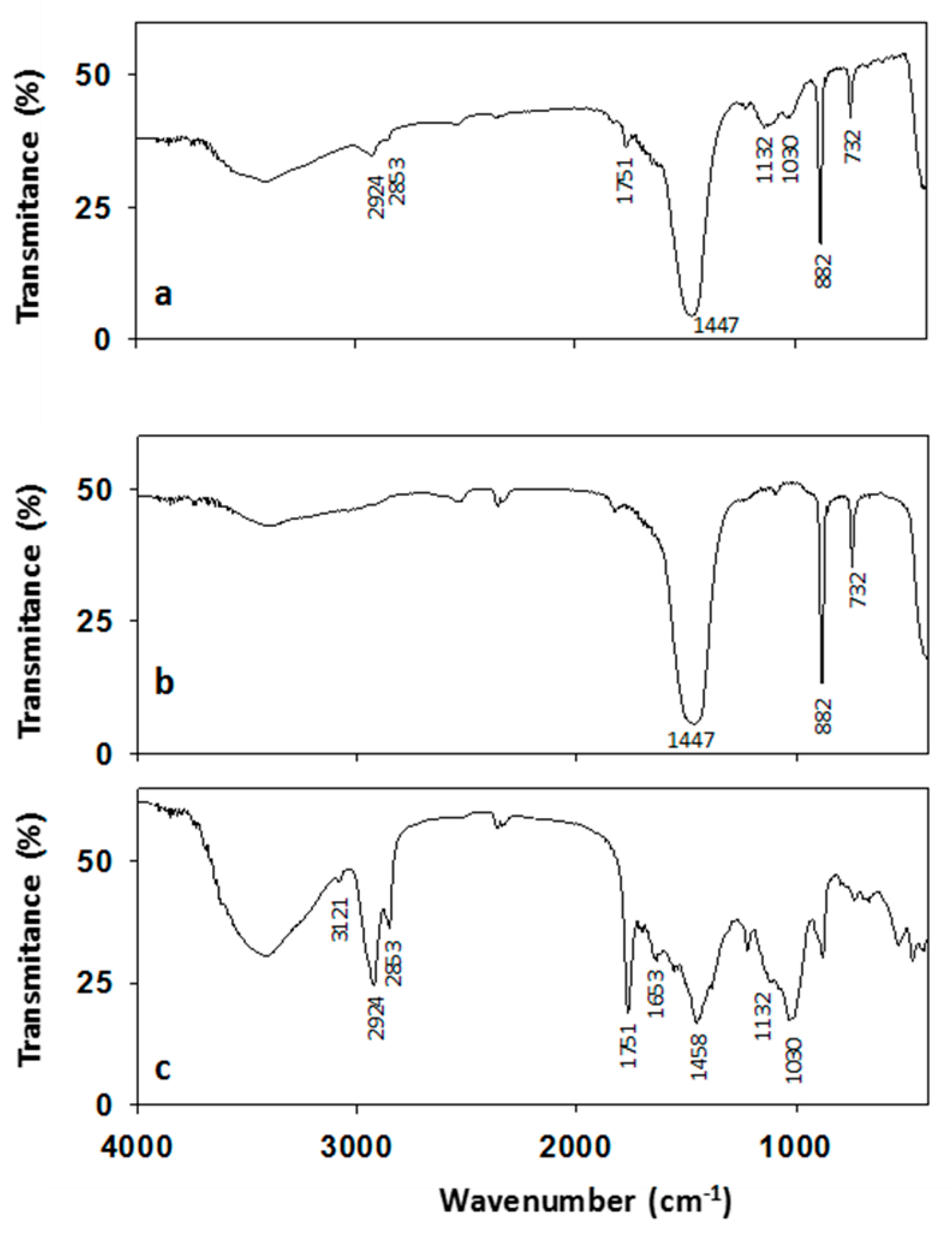
2.4. Fourier Transform Infrared Spectroscopy
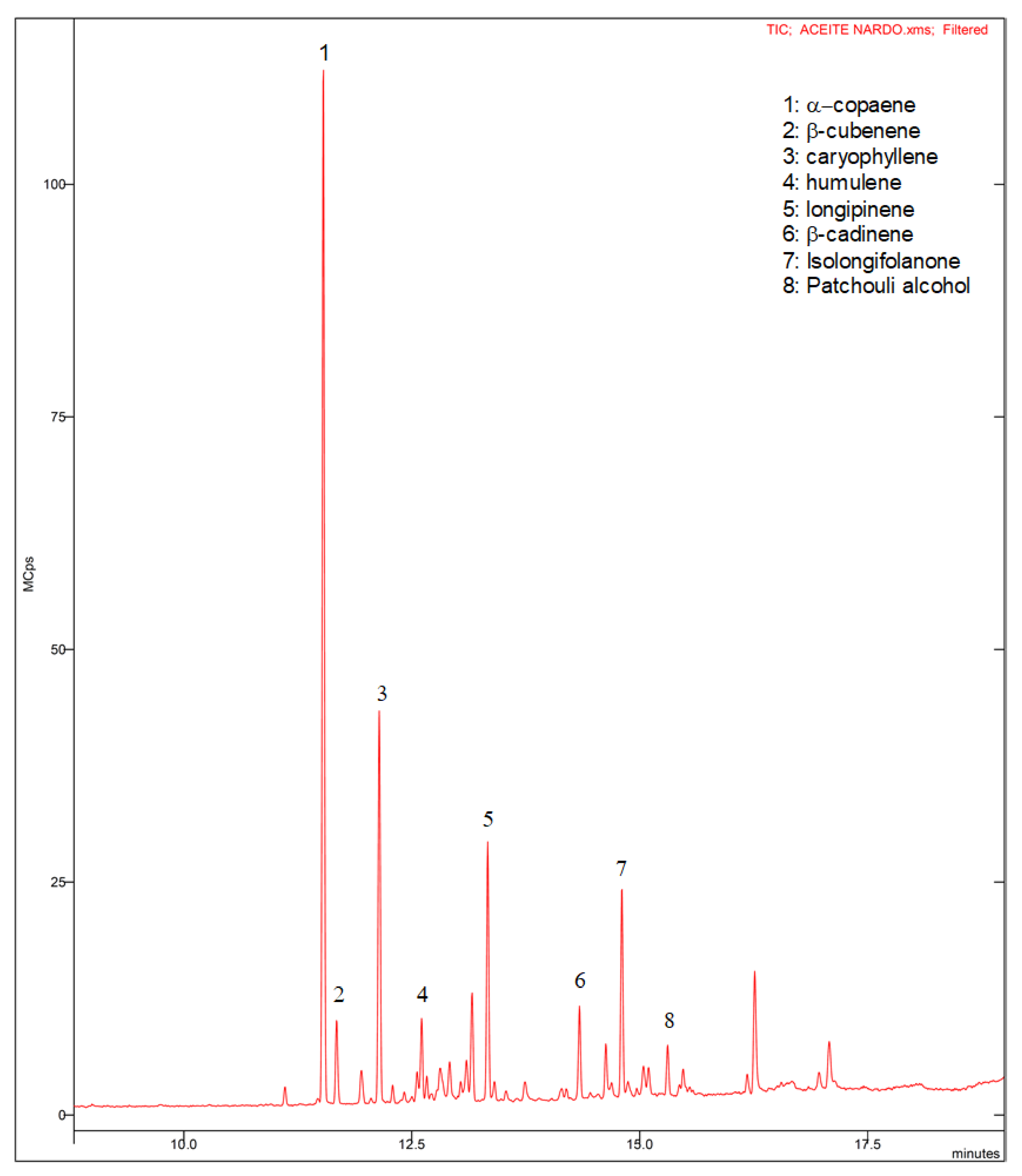
2.5. Gas Chromatography–Mass Spectrometry
3. Results and Discussion
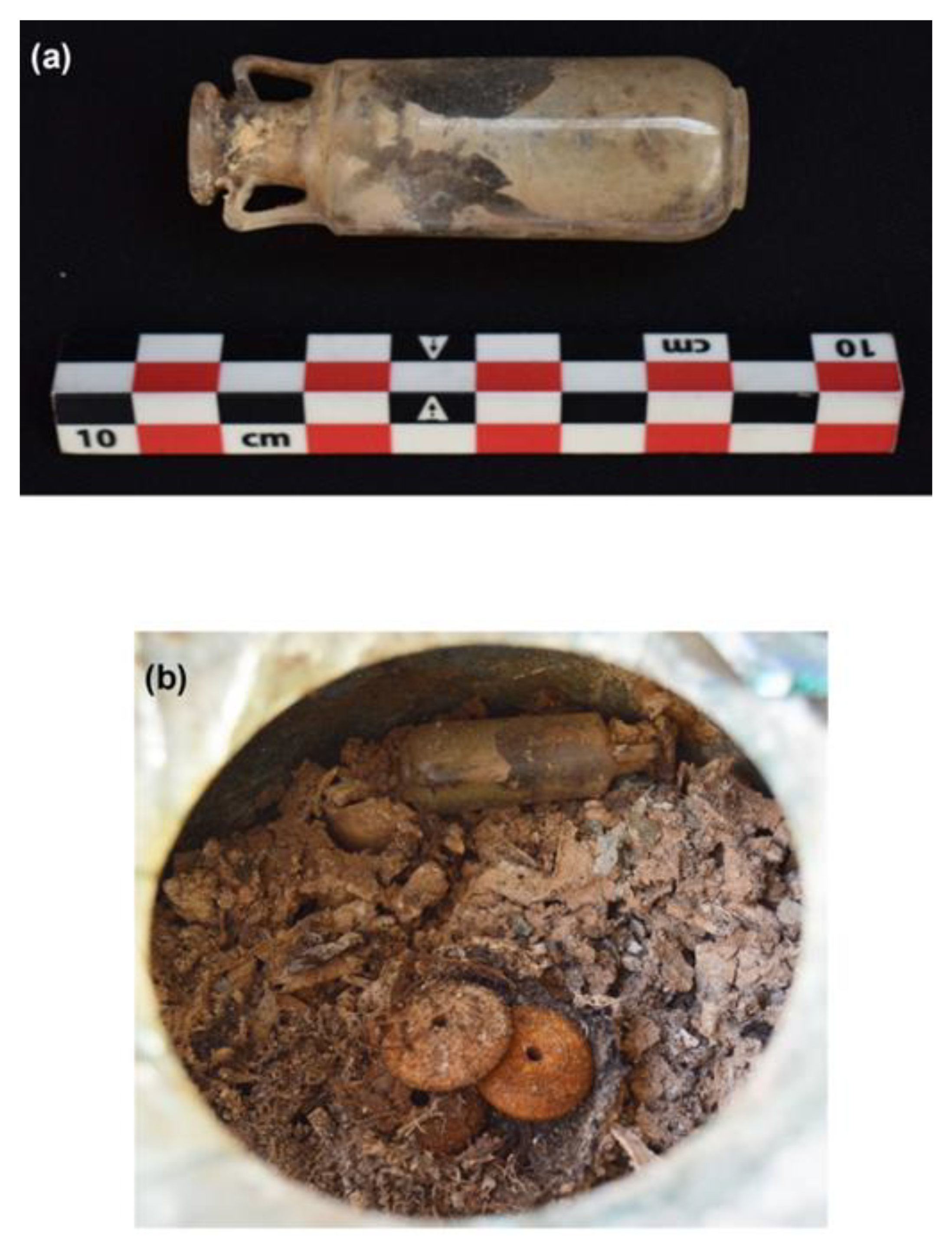
3.1. Analysis of the Unguentarium Stopper
3.2. Analysis of the Unguentarium Contents
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Román Rodríguez, J.M.; Santos Allely, A.; Vázquez Paz, J.A. A New Hypogeal Tomb from Roman Times in Carmona (Seville). Romvla 2019, 18, 203–209. [Google Scholar]
- Gómez Fernández, V.; Sibón Olano, J.F. La Necrópolis Altoimperial: Nuevos Datos a Partir de Los Resultados de La Excavación Arqueológica Realizada En La Avenida de Andalucía, 35 (Cádiz). In Las Necrópolis de Cádiz: Apuntes de Arqueología Gaditana; SPAL: Ferrara, Italy, 2010; Volume 19, pp. 219–221. Available online: https://dialnet.unirioja.es/servlet/articulo?codigo=3636153 (accessed on 20 April 2023).
- Evershed, R.P.; Berstan, R.; Grew, F.; Copley, M.S.; Charmant, A.J.H.; Barham, E.; Mottram, H.R.; Brown, G. Formulation of a Roman Cosmetic. Nature 2004, 432, 35–36. [Google Scholar] [CrossRef] [PubMed]
- Koupadi, K.; Boyatzis, S.C.; Roumpou, M.; Kalogeropoulos, N.; Kotzamani, D. Organic Remains in Early Christian Egyptian Metal Vessels: Investigation with Fourier Transform Infrared Spectroscopy and Gas Chromatography–Mass Spectrometry. Heritage 2021, 4, 3611–3629. [Google Scholar] [CrossRef]
- Ribechini, E.; Modugno, F.; Colombini, M.P.; Evershed, R.P. Gas Chromatographic and Mass Spectrometric Investigations of Organic Residues from Roman Glass Unguentaria. J. Chromatogr. A 2008, 1183, 158–169. [Google Scholar] [CrossRef] [PubMed]
- Agozzino, P.; Avellone, G.; Donato, I.D.; Filizzola, F. Identification of Organic Compounds in Fictile Unguentaria from Two Sicilian Necropolis of Greek Age (5th Century, b.C.) by GC-MS Analysis. Ann. Chim. 2007, 97, 859–865. [Google Scholar] [CrossRef] [PubMed]
- Walter, P.; Martinetto, P.; Tsoucaris, G.; Bréniaux, R.; Lefebvre, M.A.; Richard, G.; Talabot, J.; Dooryhee, E. Making Make-up in Ancient Egypt. Nature 1999, 397, 483–484. [Google Scholar] [CrossRef]
- Belgiorno, M.R. Profumi Di Afrodite e Il Segreto Dell’olio: Scoperte Archeologiche a Cipro; Gangemi Editore: Rome, Italy, 2007; pp. 1–255. [Google Scholar]
- Brun, J.-P. The Production of Perfumes in Antiquity: The Cases of Delos and Paestum. Am. J. Archaeol. 2000, 104, 277–308. [Google Scholar] [CrossRef]
- Plinio. Historia Natural. Libros XII-XVI; Manzanero Cano, F.; García Arribas, I.; Arribas Hernáez, M.L.; Moure Casas, A.M.; Sancho Bermejo, J.L., Translators; Biblioteca Clásica Gredos: Madrid, Spain, 2016. [Google Scholar]
- Castel, C.; Fernandez, X.; Filippi, J.-J.; Brun, J.-P. Perfumes in Mediterranean Antiquity. Flavour Fragr. J. 2009, 24, 326–334. [Google Scholar] [CrossRef]
- Vaquerizo Gil, D. Necrópolis Urbanas En Baetica; Secretariado de Publicaciones de la Universidad de Sevilla: Sevilla, Spain, 2010. [Google Scholar]
- Colombini, M.P.; Andreotti, A.; Bonaduce, I.; Modugno, F.; Ribechini, E. Analytical Strategies for Characterizing Organic Paint Media Using Gas Chromatography/Mass Spectrometry. Acc. Chem. Res. 2010, 43, 715–727. [Google Scholar] [CrossRef]
- Domenech-Carbo, M.T. Novel Analytical Methods for Characterising Binding Media and Protective Coatings in Artworks. Anal. Chim. Acta 2008, 621, 109–139. [Google Scholar] [CrossRef]
- Manzano, E.; Rodriguez-Simón, L.R.; Navas, N.; Checa-Moreno, R.; Romero-Gámez, M.; Capitan-Vallvey, L.F. Study of the GC–MS Determination of the Palmitic–Stearic Acid Ratio for the Characterisation of Drying Oil in Painting: La Encarnación by Alonso Cano as a Case Study. Talanta 2011, 84, 1148–1154. [Google Scholar] [CrossRef] [PubMed]
- Gebremariam, K.F.; Kvittingen, L.; Banica, F. Physico-Chemical Characterization of Pigments and Binders of Murals in a Church in Ethiopia. Archaeometry 2016, 58, 271–283. [Google Scholar] [CrossRef]
- Colin, J. Luxe Oriental et Parfums Masculins Dans La Rome Alexandrine (d’après Cicéron et Lucrèce). Rev. Belg. De Philol. Et D’histoire 1955, 33, 5–19. [Google Scholar] [CrossRef]
- Hayes, J.W. A New Type of Early Christian Ampulla. Annu. Br. Sch. Athens 1971, 66, 243–248. [Google Scholar] [CrossRef]
- Mayer i Olivé, M. Opercula, Los Tapones de Ánfora: Un Indicador Económico Controvertido. In Instrumenta Inscripta Latina II; Verlag des Geschichtsverein für Kärnten: Klagenfurt, Austria, 2008; pp. 223–239. [Google Scholar]
- Chassouant, L.; Olmer, F.; Delpino, C.; Celant, A.; Vieillescazes, C.; Magri, D.; Mathe, C. Protocol Comparison for Organic Residue Analyses from Waterproofing Materials and Shards of Roman Archaeological Amphorae. Crystals 2021, 11, 1300. [Google Scholar] [CrossRef]
- Hesnard, A.; Gianfrotta, P.A. Les Bouchons d’amphore En Pouzzolane. In Amphores Romaines et Histoire Économique: Dix ans de Recherche; Actes du Colloque in Sienne: Rome, Italy, 1989; pp. 393–441. [Google Scholar]
- Mayer, M. ¿Qué función y significación pudieron tener los símbolos y letreros presentes en los operculos anfóricos? In Quaderni Friulani di Archeologia XXII/XXIII-2012/2013; pp. 17–25. Available online: https://opac.rivistefriulane.it/ricerca/dettaglio/que-funcion-y-que-significacion-pudieron-tener-los-simbolos-y-letreros-presentes/19053 (accessed on 20 April 2023).
- Benoit, F. L’archéologie Sous Marine En Provence. Riv. Di Studi Liguri 1952, 18, 238–307. [Google Scholar]
- Gunasekaran, S.; Anbalagan, G.; Pandi, S. Raman and Infrared Spectra of Carbonates of Calcite Structure. J. Raman Spectrosc. 2006, 37, 892–899. [Google Scholar] [CrossRef]
- Galvez, M.E.; Beyssac, O.; Martinez, I.; Benzerara, K.; Chaduteau, C.; Malvoisin, B.; Malavieille, J. Graphite Formation by Carbonate Reduction during Subduction. Nat. Geosci. 2013, 6, 473–477. [Google Scholar] [CrossRef]
- Cerrato, E.J.; Íñiguez, L.; Cosano, D.; Guiral, C.; Ruiz, J.R. Multi-Analytical Identification of a Painting Workshop at the Roman Archaeological Site of Bilbilis (Saragossa, Spain). J. Archaeol. Sci. Rep. 2021, 38, 103108. [Google Scholar] [CrossRef]
- Bertoluzza, A.; Brasili, P.; Castrì, L.; Facchini, F.; Fagnano, C.; Tinti, A. Preliminary Results in Dating Human Skeletal Remains by Raman Spectroscopy. J. Raman Spectrosc. 1997, 28, 185–188. [Google Scholar] [CrossRef]
- Connan, J. Use and Trade of Bitumen in Antiquity and Prehistory: Molecular Archaeology Reveals Secrets of Past Civilizations. Philos. Trans. R. Soc. B Biol. Sci. 1999, 354, 33–50. [Google Scholar] [CrossRef]
- Formenti, F.; Duthel, J.M. The Analysis of Wine and Other Organics inside Amphoras of the Roman Period. In The Origins and Ancient History of Wine: Food and Nutrition in History and Anthropology; McGovern, P.E., Fleming, S.T., Katz, S.H., Eds.; Routledge: Oxfordshire, UK, 2003; pp. 79–85. ISBN 0203392833. [Google Scholar]
- Jehlička, J.; Urban, O.; Pokorný, J. Raman Spectroscopy of Carbon and Solid Bitumens in Sedimentary and Metamorphic Rocks. Spectrochim. Acta A Mol. Biomol. Spectrosc. 2003, 59, 2341–2352. [Google Scholar] [CrossRef] [PubMed]
- Khatibi, S.; Abarghani, A.; Liu, K.; Guedes, A.; Valentim, B.; Ostadhassan, M. Backtracking to Parent Maceral from Produced Bitumen with Raman Spectroscopy. Minerals 2020, 10, 679. [Google Scholar] [CrossRef]
- Toboła, T.; Botor, D. Raman Spectroscopy of Organic Matter and Rare Minerals in the Kłodawa Salt Dome (Central Poland) Cap-Rock and Triassic Cover–Indicators of Hydrothermal Solution Migration. Spectrochim Acta A Mol. Biomol. Spectrosc. 2020, 231, 118121. [Google Scholar] [CrossRef]
- Cârciumaru, M.; Ion, R.-M.; Niţu, E.-C.; Ştefănescu, R. New Evidence of Adhesive as Hafting Material on Middle and Upper Palaeolithic Artefacts from Gura Cheii-Râşnov Cave (Romania). J. Archaeol. Sci. 2012, 39, 1942–1950. [Google Scholar] [CrossRef]
- Marković, M.; Mezzatesta, E.; Porcier, S.; Vieillescazes, C.; Mathe, C. Rethinking the Process of Animal Mummification in Ancient Egypt: Molecular Characterization of Embalming Material and the Use of Brassicaceae Seed Oil in the Mummification of Gazelle Mummies from Kom Mereh, Egypt. Molecules 2022, 27, 1532. [Google Scholar] [CrossRef]
- Luemba, M.; Chen, Z.; Ntibahanana, J. Molecular Markers of Neoproterozoic-Lower Paleozoic Petroleum Systems and Their Geological Significance: A Case Study of the Cratonic Basins in Western China. J. Pet. Sci. Eng. 2021, 204, 108707. [Google Scholar] [CrossRef]
- Thienpont, J.R.; Yang, Z.; Hall, R.I.; Wolfe, B.B.; Hollebone, B.P.; Blais, J.M. Tracking Petrogenic Hydrocarbons in Lakes of the Peace-Athabasca Delta in Alberta, Canada Using Petroleum Biomarkers. Environ. Pollut. 2021, 286, 117286. [Google Scholar] [CrossRef]
- Wu, J.; Fang, P.; Wang, X.-C.; Li, B.; Liu, K.; Ma, X.; Li, S.; Li, M. The Potential Occurrence Modes of Hydrocarbons in Asphaltene Matrix and Its Geochemical Implications. Fuel 2020, 278, 118233. [Google Scholar] [CrossRef]
- Simoneit, B.R.T.; Lonsdale, P.F. Hydrothermal Petroleum in Mineralized Mounds at the Seabed of Guaymas Basin. Nature 1982, 295, 198–202. [Google Scholar] [CrossRef]
- Olivares, M.; Irazola, M.; Murelaga, X.; Baceta, J.I.; Tarriño, A.; Castro, K.; Etxebarria, N. Sourcing Sedimentary Cherts with Archaeological Use through the Combination of Chromatographic and Spectroscopic Techniques. Appl. Geochem. 2013, 33, 252–259. [Google Scholar] [CrossRef]
- Rullkötter, J.; Nissenbaum, A. Dead Sea Asphalt in Egyptian Mummies: Molecular Evidence. Naturwissenschaften 1988, 75, 618–621. [Google Scholar] [CrossRef] [PubMed]
- Spiro, B.; Welte, D.H.; Rullkoetter, J.; Schaefer, R.G. Asphalts, Oils, and Bituminous Rocks from the Dead Sea Area—A Geochemical Correlation Study. AAPG Bull. 1983, 67, 1163–1175. [Google Scholar]
- Landis, M.S.; Studabaker, W.B.; Pancras, J.P.; Graney, J.R.; Puckett, K.; White, E.M.; Edgerton, E.S. Source Apportionment of an Epiphytic Lichen Biomonitor to Elucidate the Sources and Spatial Distribution of Polycyclic Aromatic Hydrocarbons in the Athabasca Oil Sands Region, Alberta, Canada. Sci. Total Environ. 2019, 654, 1241–1257. [Google Scholar] [CrossRef] [PubMed]
- Marynowski, L.; Gawe, A.; Poprawa, P.; Zywiecki, M.M.; Ke, B.; Merta, H. Origin of Organic Matter from Tectonic Zones in the Western Tatra Mountains Crystalline Basement, Poland: An Example of Bitumen—Source Rock Correlation. Mar. Pet. Geol. 2006, 23, 261–279. [Google Scholar] [CrossRef]
- Charrié-Duhaut, A.; Connan, J.; Darnell, M.; Spangenberg, J.; Szymczyk, E.; Bissada, A.; Albrecht, P. Molecular and Isotopic Characterization of Organic Samples from the Wreck of the Saint-Etienne Merchant Ship (XVIIIth Century): Identification of Pitch, Fat, Hair and Sulfur. Org. Geochem. 2009, 40, 647–665. [Google Scholar] [CrossRef]
- Hamm, S.; Bleton, J.; Tchapla, A. Headspace Solid Phase Microextraction for Screening for the Presence of Resins in Egyptian Archaeological Samples. J. Sep. Sci. 2004, 27, 235–243. [Google Scholar] [CrossRef]
- Łucejko, J.; Connan, J.; Orsini, S.; Ribechini, E.; Modugno, F. Chemical Analyses of Egyptian Mummification Balms and Organic Residues from Storage Jars Dated from the Old Kingdom to the Copto-Byzantine Period. J. Archaeol. Sci. 2017, 85, 1–12. [Google Scholar] [CrossRef]
- Strausz, O.P.; Lown, E.M.; Morales-Izquierdo, A.; Kazmi, N.; Montgomery, D.S.; Payzant, J.D.; Murgich, J. Chemical Composition of Athabasca Bitumen: The Distillable Aromatic Fraction. Energy Fuels 2011, 25, 4552–4579. [Google Scholar] [CrossRef]
- Hauck, T.C.; Connan, J.; Charrié-Duhaut, A.; Le Tensorer, J.-M.; Al Sakhel, H. Molecular Evidence of Bitumen in the Mousterian Lithic Assemblage of Hummal (Central Syria). J. Archaeol. Sci. 2013, 40, 3252–3262. [Google Scholar] [CrossRef]
- Connan, J.; Nissenbaum, A. Conifer Tar on the Keel and Hull Planking of the Ma’agan Mikhael Ship (Israel, 5th Century BC): Identification and Comparison with Natural Products and Artefacts Employed in Boat Construction. J. Archaeol. Sci. 2003, 30, 709–719. [Google Scholar] [CrossRef]
- Rossiter, K.J. Structure—Odor Relationships. Chem. Rev. 1996, 96, 3201–3240. [Google Scholar] [CrossRef] [PubMed]
- Hale, S.E.; Endo, S.; Arp, H.P.H.; Zimmerman, A.R.; Cornelissen, G. Sorption of the Monoterpenes α-Pinene and Limonene to Carbonaceous Geosorbents Including Biochar. Chemosphere 2015, 119, 881–888. [Google Scholar] [CrossRef] [PubMed]
- Van Roon, A.; Parsons, J.R.; Te Kloeze, A.M.; Govers, H.A.J. Fate and Transport of Monoterpenes through Soils. Part I. Prediction of Temperature Dependent Soil Fate Model Input-Parameters. Chemosphere 2005, 61, 599–609. [Google Scholar] [CrossRef] [PubMed]
- André, J. Les Noms de Plantes Dans La Rome Antique: 47 (Eudes Anciennes); Belles Let.: Oklahoma City, OK, USA, 2010. [Google Scholar]
- Dioscórides De Materia Medica. 1563. Available online: https://www.cervantesvirtual.com/obra/de-materia-medica-espanol/ (accessed on 20 April 2023).
- Van Beek, T.A.; Joulain, D. The Essential Oil of Patchouli, Pogostemon Cablin: A Review. Flavour Fragr. J. 2018, 33, 6–51. [Google Scholar] [CrossRef]
- Chauhan, R.S.; Nautiyal, M.C.; Figueredo, G.; Rana, V.S. Effect of Post Harvest Drying Methods on the Essential Oil Composition of Nardostachys Jatamansi DC. J. Essent. Oil Bear. Plants 2017, 20, 1090–1096. [Google Scholar] [CrossRef]
- Cornara, L.; Ambu, G.; Trombetta, D.; Denaro, M.; Alloisio, S.; Frigerio, J.; Labra, M.; Ghimire, G.; Valussi, M.; Smeriglio, A. Comparative and Functional Screening of Three Species Traditionally Used as Antidepressants: Valeriana Officinalis, L., Valeriana Jatamansi Jones Ex Roxb. and Nardostachys Jatamansi (D.Don) DC. Plants 2020, 9, 994. [Google Scholar] [CrossRef]
- Satyal, P.; Chhetri, B.K.; Dosoky, N.S.; Poudel, A.; Setzer, W.N. Chemical Composition of Nardostachys Grandiflora Rhizome Oil from Nepal. A Contribution to the Chemotaxonomy and Bioactivity of Nardostachys. Nat. Prod. Commun. 2015, 10, 1067–1070. [Google Scholar] [CrossRef]
- Pérez González, J. Elaboración y comercialización de perfumes y ungüentos en Roma Los unguentarii. Rev. Estud. Filosóficos Históricos Antig. 2017, 31, 81–110. [Google Scholar]
- Azara, P. La Bellesa: Gràcia i Desgràcia. In Històries de Tocador. Cosmètica i Bellesa a l’Antiguitat; Barcelona, Spain. 2013. Available online: https://futur.upc.edu/11233972 (accessed on 20 April 2023).
- Evershed, R.P. Biomolecular Archaeology and Lipids. World Archaeol. 1993, 25, 74–93. [Google Scholar] [CrossRef]
- Manhita, A.; Martins, S.; da Silva, M.G.; Lopes, M.d.C.; Dias, C.B. Transporting Olive Oil in Roman Times: Chromatographic Analysis of Dressel 20 Amphorae from Pax Julia Civitas, Lusitania. Chromatographia 2020, 83, 1055–1064. [Google Scholar] [CrossRef]
- Garnier, N.; Frère, D. Parfums de L’antiquité: La Rose et L’encens En Méditerranée; Musée Royal de Mariemont: Morlanwelz, Belgique, 2008; Volume 61. [Google Scholar]
- Biers, W.R.; Gerhardt, K.O.; Braniff, R.A. Lost Scents: Investigations of Corinthian Plastic Vases by Gas Chromatography-Mass Spectrometry. MASCA Res. Pap. Sci. Archaeol. 1994, 1, 11. [Google Scholar]
- Scatozza Höricht, L.A.; Chianese, L.; Piccioli, C.; Sacchi, R. Ercolano 1738–1988. In 250 Anni di Ricerca Archeologica; Soprintendenza Archeologica di Pompei: Rome, Italy, 1993; p. 551. [Google Scholar]
- Garnier, N.; Silvino, T.; Tokarksi, C.; Rolando, C. Parfums et Odeurs Dans L’ANTIQUITÉ; Presses Universitaires de Rennes: Rennes, France, 2008; Volume 71. [Google Scholar]
- Bouwmeester, H.J. Engineering the Essence of Plants. Nat. Biotechnol. 2006, 24, 1359–1361. [Google Scholar] [CrossRef] [PubMed]







| Element | Inner Side (%) | Outer Side (%) |
|---|---|---|
| C | 21.7 | 40.8 |
| O | 52.9 | 48.7 |
| Ca | 10.8 | 3.7 |
| Mg | 13.8 | 4.4 |
| Na | - | 1.2 |
| Si | - | 0.1 |
| P | - | 0.9 |
| S | - | 0.2 |
| Peak | Compound | tR (min) | Peak | Compound | tR (min) |
|---|---|---|---|---|---|
| From bitumen | From the essence | ||||
| 1 | Decane | 6.88 | 12 | α-Copaen-8-ol | 11.29 |
| 3 | 4,5-Dimethylnonane | 7.55 | 13 | β-Patchoulene | 11.74 |
| 4 | Undecane | 7.65 | 15 | Caryophyllene | 12.19 |
| 5 | 6-Ethyl-2-methylnonane | 7.76 | 16 | Thujopsene | 12.26 |
| 6 | Dodecane | 9.71 | 17 | α-Guaiene | 12.35 |
| 7 | Tridecane | 10.11 | 18 | Alloaromadendrene | 12.42 |
| 8 | Tetradecane | 10.65 | 19 | Seychellene | 12.63 |
| 9 | Pentadecane | 10.79 | 20 | α-Bulnesene | 12.78 |
| 10 | Hexadecane | 10.91 | 21 | Aromadendrene | 12.82 |
| 11 | Methylpentadecane | 11.02 | 22 | α-Patchoulene | 12.95 |
| 14 | Heptadecane | 11.82 | 23 | Acyphyllene | 13.13 |
| 25 | Octadecane | 13.54 | 24 | Isoaromadendrene | 13.22 |
| 28 | Nonadecane | 13.76 | 26 | α-Cubenene | 13.66 |
| 30 | Retene | 20.85 | 27 | Azulol | 15.34 |
| 34 | Dimethylretenes | 15–20 | 29 | Patchoulenol | 15.43 |
| 35 | Methylretenes | 18–23 | From the fat | ||
| 36 | Staranes | 17–19 | 31 | Squalene | 25.42 |
| 37 | Terpanes | 15–20 | 32 | β-Sitosterol | 26.22 |
| 33 | Stigmasterol | 26.75 | |||
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Cosano, D.; Román, J.M.; Lafont, F.; Ruiz Arrebola, J.R. Archaeometric Identification of a Perfume from Roman Times. Heritage 2023, 6, 4472-4491. https://doi.org/10.3390/heritage6060236
Cosano D, Román JM, Lafont F, Ruiz Arrebola JR. Archaeometric Identification of a Perfume from Roman Times. Heritage. 2023; 6(6):4472-4491. https://doi.org/10.3390/heritage6060236
Chicago/Turabian StyleCosano, Daniel, Juan Manuel Román, Fernando Lafont, and José Rafael Ruiz Arrebola. 2023. "Archaeometric Identification of a Perfume from Roman Times" Heritage 6, no. 6: 4472-4491. https://doi.org/10.3390/heritage6060236
APA StyleCosano, D., Román, J. M., Lafont, F., & Ruiz Arrebola, J. R. (2023). Archaeometric Identification of a Perfume from Roman Times. Heritage, 6(6), 4472-4491. https://doi.org/10.3390/heritage6060236







