Lomefloxacin—Occurrence in the German River Erft, Its Photo-Induced Elimination, and Assessment of Ecotoxicity
Abstract
:1. Introduction
2. Experiments
2.1. Chemicals and Reagents
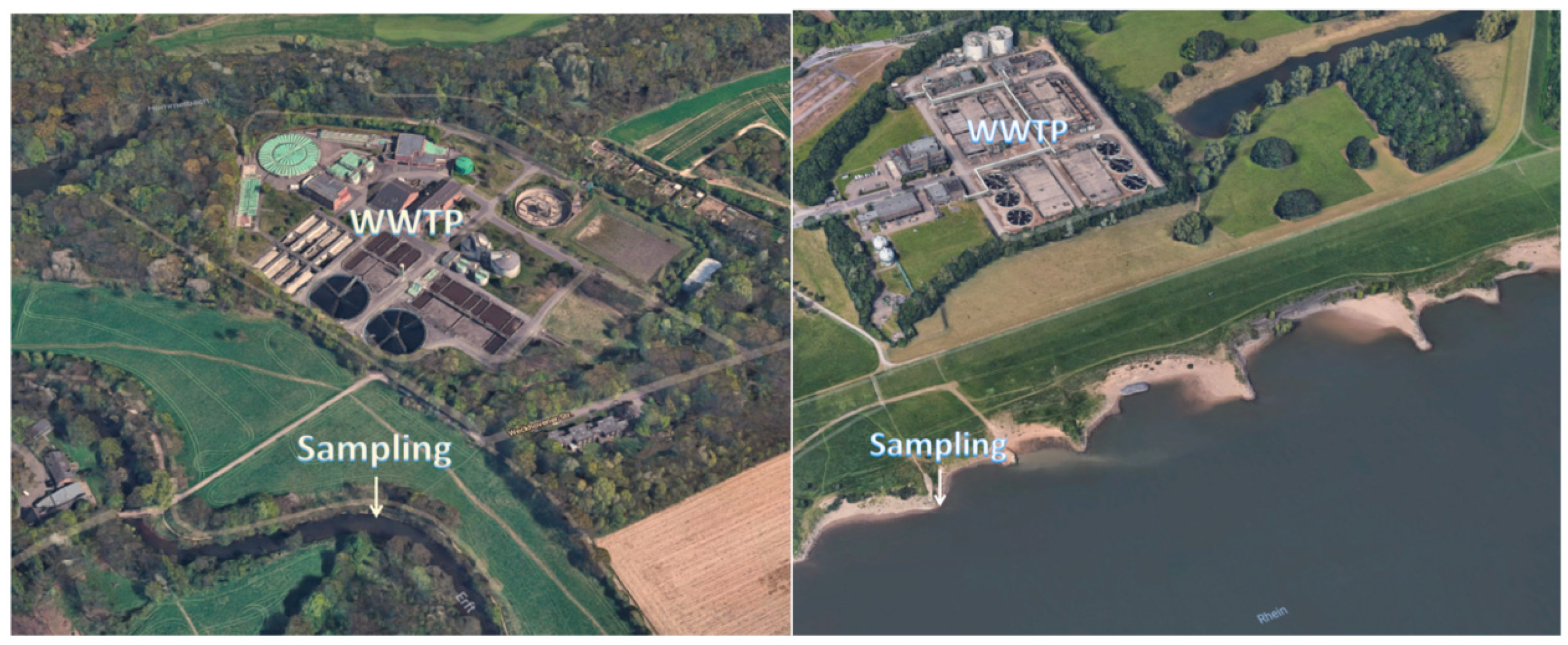
2.2. Solid-Phase Extraction of River Water Samples
2.3. Photodegradation Experiments
2.4. HPLC-ESI-Q-TOF-MS and HPLC-ESI-IT-MS
2.5. TOC
2.6. Kinetic Analysis and Determination of Quantum Efficiency of the Photodegradation
2.7. Determination of Quantum Efficiency
2.8. Assessment of Ecotoxicology
3. Results and Discussion
3.1. Occurrence of Lomefloxacin in Surface Water
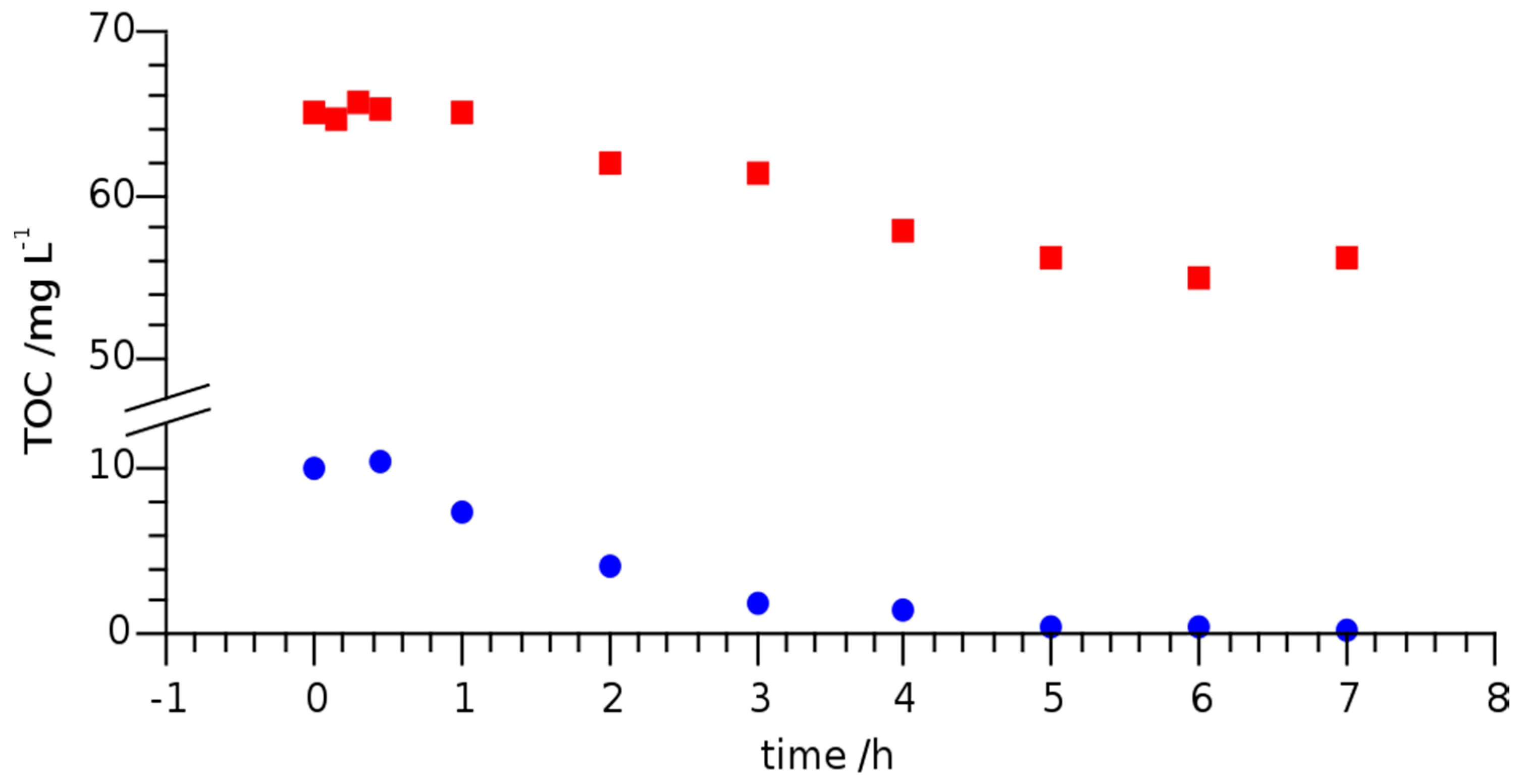
3.2. Photoinduced Degradation of Lomefloxacin—Kinetic of Degradation
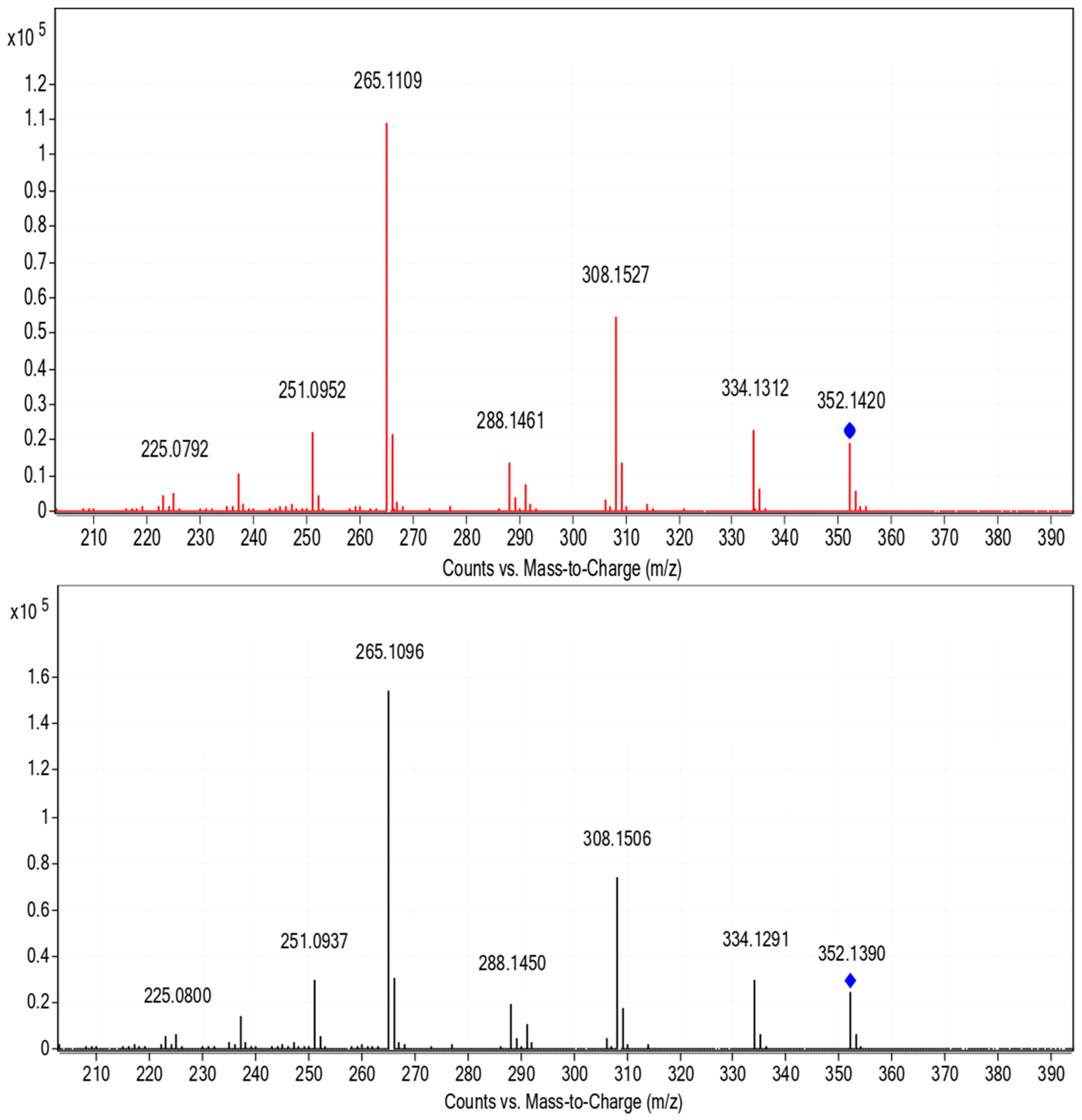
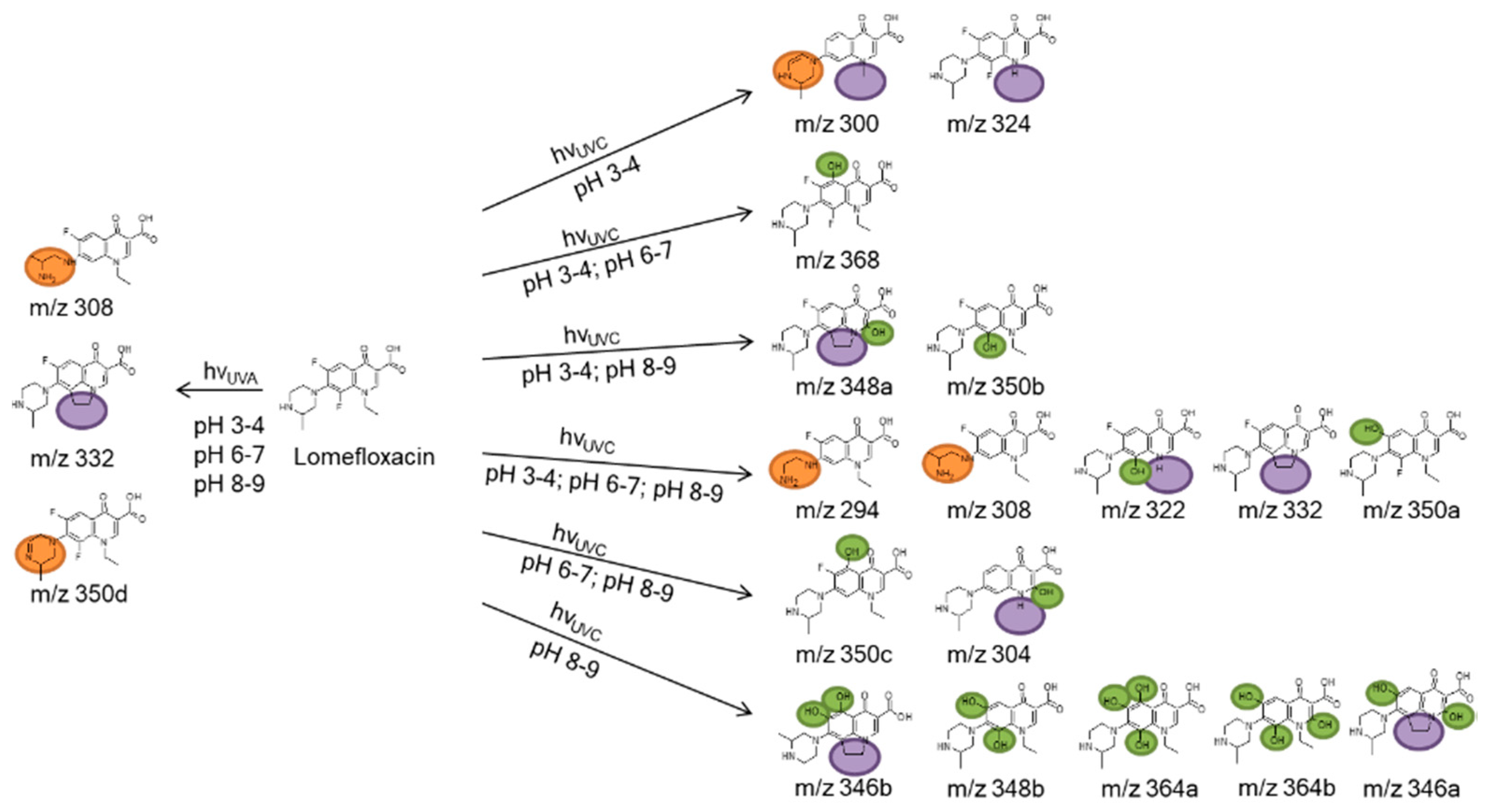
3.3. Photoinduced Degradation Products of Lomefloxacin
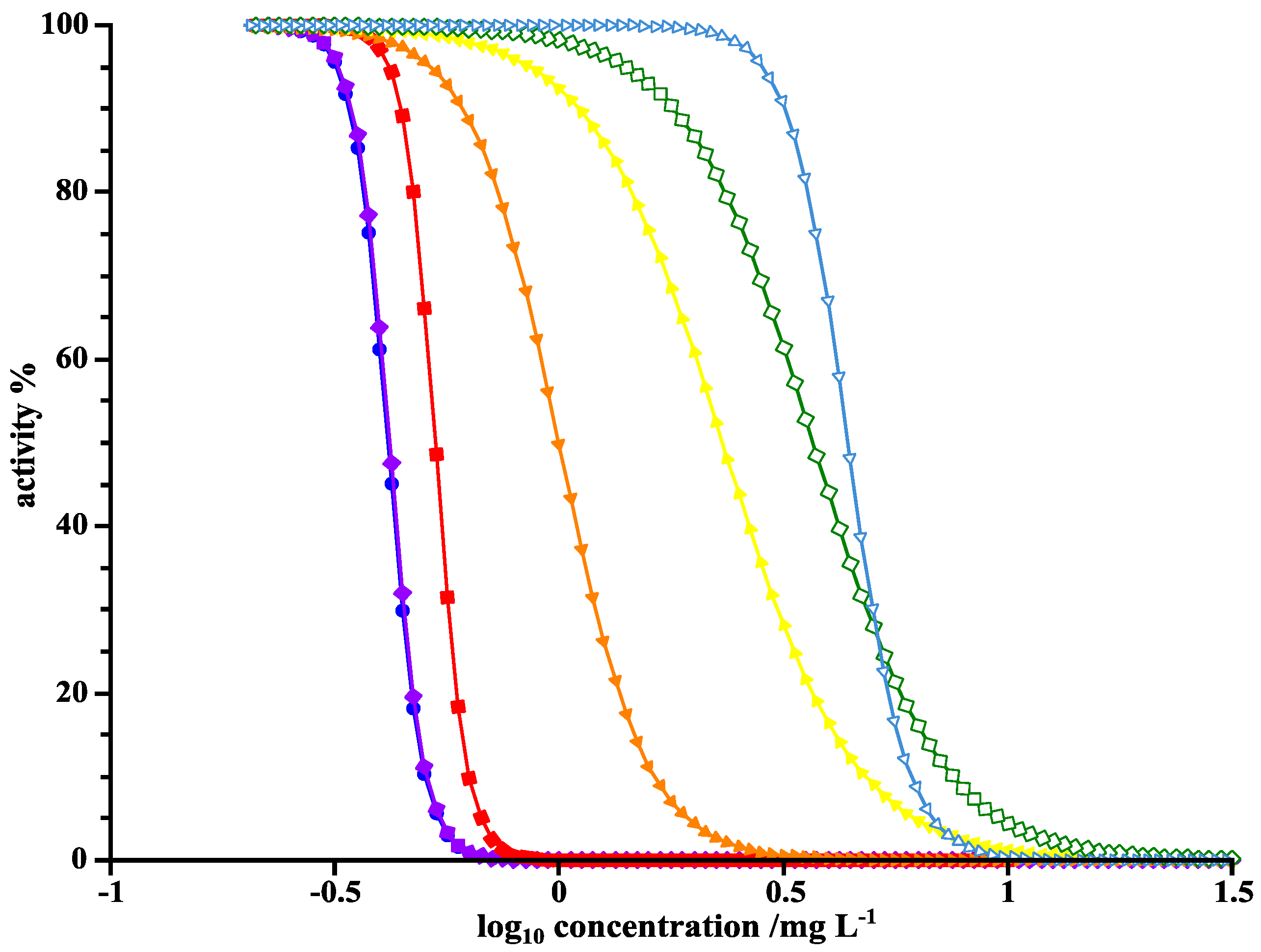
3.4. Assessment of Ecotoxicity
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Zhang, P.; Zhou, H.; Li, K.; Zhao, X.; Liu, Q.; Li, D.; Zhao, G. Occurrence of pharmaceuticals and personal care products, and their associated environmental risks in a large shallow lake in north China. Environ. Geochem. Health 2018, 40, 1525–1539. [Google Scholar] [CrossRef]
- Sousa, J.C.G.; Ribeiro, A.R.; Barbosa, M.O.; Pereira, M.F.R.; Silva, A.M.T. A review on environmental monitoring of water organic pollutants identified by EU guidelines. J. Hazard. Mater. 2018, 344, 146–162. [Google Scholar] [CrossRef]
- Gogoi, A.; Mazumder, P.; Kumar, V.; Chaminda, G.G.T.; Kyoungjin, A.; Kumar, M. Groundwater for Sustainable Development Occurrence and fate of emerging contaminants in water environment: A review. Groundw. Sustain. Dev. 2018, 6, 169–180. [Google Scholar] [CrossRef]
- Hamad, B. The antibiotics market. Nat. Rev. Drug Discov. 2010, 9, 675–676. [Google Scholar] [CrossRef] [PubMed]
- Kümmerer, K. Antibiotics in the aquatic environment—A review—Part I. Chemosphere 2009, 75, 417–434. [Google Scholar] [CrossRef] [PubMed]
- Nakata, H.; Kannan, K.; Jones, P.D.; Giesy, J.P. Determination of fluoroquinolone antibiotics in wastewater effluents by liquid chromatography-mass spectrometry and fluorescence detection. Chemosphere 2005, 58, 759–766. [Google Scholar] [CrossRef] [PubMed]
- Parsons, S. Advanced Oxidation Processes for Water and Wastewater Treatment; IWA Publishing: London, UK, 2004. [Google Scholar]
- Yuan, F.; Hu, C.; Hu, X.; Qu, J.; Yang, M. Degradation of selected pharmaceuticals in aqueous solution with UV and UV/H2O2. Water Res. 2009, 43, 1766–1774. [Google Scholar] [CrossRef] [PubMed]
- Hain, E.; Wammer, K.H.; Blaney, L. Comment on Photodegradation of sulfathiazole under simulated sunlight: Kinetics, photo-induced structural rearrangement, and antimicrobial activities of photoproducts by Niu et al. Water Research 124 2017 576 e 583. Water Res. 2018, 131, 205–207. [Google Scholar] [CrossRef] [PubMed]
- Niu, X.; Glady-croué, J.; Croué, J. Photodegradation of sulfathiazole under simulated sunlight: Kinetics, photo-induced structural rearrangement, and antimicrobial activities of photoproducts. Water Res. 2017, 124, 576–583. [Google Scholar] [CrossRef]
- Voigt, M.; Bartels, I.; Nickisch-Hartfiel, A.; Jaeger, M. Determination of minimum inhibitory concentration and half maximal inhibitory concentration of antibiotics and their degradation products to assess the eco-toxicological potential. Toxicol. Environ. Chem. 2019, 101, 315–338. [Google Scholar] [CrossRef]
- Dan, A.; Zhang, X.; Dai, Y.; Chen, C.; Yang, Y. Occurrence and removal of quinolone, tetracycline, and macrolide antibiotics from urban wastewater in constructed wetlands. J. Clean. Prod. 2020, 252, 119677. [Google Scholar] [CrossRef]
- Gothwal, R. Shashidhar Occurrence of high levels of fluoroquinolones in aquatic environment due to effluent discharges from bulk drug manufacturers. J. Hazard. Toxic Radioact. Waste 2017, 21, 05016003. [Google Scholar] [CrossRef]
- Zhang, Z.; Xie, X.; Yu, Z.; Cheng, H. Influence of chemical speciation on photochemical transformation of three fluoroquinolones (FQs) in water: Kinetics, mechanism, and toxicity of photolysis products. Water Res. 2019, 148, 19–29. [Google Scholar] [CrossRef]
- Tammam, M.H. Photostability studies on gemifloxacin and lomefloxacin in bulk powder and dosage forms. Eur. J. Chem. 2014, 5, 73–80. [Google Scholar] [CrossRef] [Green Version]
- Miranda, A.C.; Lepretti, M.; Rizzo, L.; Caputo, I.; Vaiano, V.; Sacco, O.; Lopes, W.S.; Sannino, D. Surface water disinfection by chlorination and advanced oxidation processes: Inactivation of an antibiotic resistant E. coli strain and cytotoxicity evaluation. Sci. Total Environ. 2016, 554–555, 1–6. [Google Scholar] [CrossRef]
- Guo, C.; Wang, K.; Hou, S.; Wan, L.; Lv, J.; Zhang, Y.; Qu, X.; Chen, S.; Xu, J. H2O2and/or TiO2photocatalysis under UV irradiation for the removal of antibiotic resistant bacteria and their antibiotic resistance genes. J. Hazard. Mater. 2017, 323, 710–718. [Google Scholar] [CrossRef] [Green Version]
- Vracko, M. Mathematical (Structural) Descriptors in QSAR: Applications in Drug Design and Environmental Toxicology; Elsevier: Amsterdam, The Netherlands, 2016; Volume 1, ISBN 9781681081977. [Google Scholar]
- Zhu, H.; Shen, Z.; Tang, Q.; Ji, W.; Jia, L. Degradation mechanism study of organic pollutants in ozonation process by QSAR analysis. Chem. Eng. J. 2014, 255, 431–436. [Google Scholar] [CrossRef]
- Kruhlak, N.L.; Contrera, J.F.; Benz, R.D.; Matthews, E.J. Progress in QSAR toxicity screening of pharmaceutical impurities and other FDA regulated products. Adv. Drug Deliv. Rev. 2007, 59, 43–55. [Google Scholar] [CrossRef]
- Abdel-Aziz, A.A.M.; Asiri, Y.A.; Al-Agamy, M.H.M. Design, synthesis and antibacterial activity of fluoroquinolones containing bulky arenesulfonyl fragment: 2D-QSAR and docking study. Eur. J. Med. Chem. 2011, 46, 5487–5497. [Google Scholar] [CrossRef]
- Anquetin, G.; Greiner, J.; Mahmoudi, N.; Santillana-Hayat, M.; Gozalbes, R.; Farhati, K.; Derouin, F.; Aubry, A.; Cambau, E.; Vierling, P. Design, synthesis and activity against Toxoplasma gondii, Plasmodium spp., and Mycobacterium tuberculosis of new 6-fluoroquinolones. Eur. J. Med. Chem. 2006, 41, 1478–1493. [Google Scholar] [CrossRef]
- Toolaram, A.P.; Haddad, T.; Leder, C.; Kümmerer, K. Initial hazard screening for genotoxicity of photo-transformation products of ciprofloxacin by applying a combination of experimental and in-silico testing. Environ. Pollut. 2016, 211, 148–156. [Google Scholar] [CrossRef]
- Voigt, M.; Bartels, I.; Nickisch-Hartfiel, A.; Jaeger, M. Elimination of macrolides in water bodies using photochemical oxidation. AIMS Environ. Sci. 2018, 5, 372–388. [Google Scholar] [CrossRef]
- Voigt, M.; Bartels, I.; Nickisch-Hartfiel, A.; Jaeger, M. Photoinduced degradation of sulfonamides, kinetic, and structural characterization of transformation products and assessment of environmental toxicity. Toxicol. Environ. Chem. 2017, 99, 1304–1327. [Google Scholar] [CrossRef]
- Voigt, M.; Jaeger, M. On the photodegradation of azithromycin, erythromycin and tylosin and their transformation products—A kinetic study. Sustain. Chem. Pharm. 2017, 5, 131–140. [Google Scholar] [CrossRef]
- Kuhn, H.; Braslavsky, S.E.; Schmidt, R. Chemical Actinometry. IUPAC Tech. Rep. 2004, 76, 2105–2146. [Google Scholar] [CrossRef]
- Hatchard, C.G.; Parker, C. A New Sensitive Chemical Actinometer. II. Potassium Ferrioxalate as a Standard Chemical Actinometer. Proc. R. Soc. A Math. Phys. Eng. Sci. 1956, 235, 518–536. [Google Scholar]
- Sun, L.; Bolton, J.R. Determination of the Quantum Yield for the Photochemical Generation of Hydroxyl Radicals in TiO2 Suspensions. J. Phys. Chem. 1996, 100, 4127–4134. [Google Scholar] [CrossRef]
- Kochany, J.; Bolton, J.R. Mechanism of photodegradation of aqueous organic pollutants. 2. Measurement of the primary rate constants for reaction of hydroxyl radicals with benzene and some halobenzenes using an EPR spin-trapping method following the photolysis of hydrogen peroxide. Environ. Sci. Technol. 1992, 26, 262–265. [Google Scholar] [CrossRef]
- Kochany, J.; Bolton, J.R. Of aqueous organic pollutants. 1. EPR spin-trapping technique for the determination of hydroxyl radical rate constants in the photooxidation of chlorophenols following. J. Phys. Chem. 1991, 95, 5116–5120. [Google Scholar] [CrossRef]
- Chan, P.Y.; Gamal El-Din, M.; Bolton, J.R. A solar-driven UV/Chlorine advanced oxidation process. Water Res. 2012, 46, 5672–5682. [Google Scholar] [CrossRef]
- Li, K.; Zhang, P.; Ge, L.; Ren, H.; Yu, C.; Chen, X.; Zhao, Y. Concentration-dependent photodegradation kinetics and hydroxyl-radical oxidation of phenicol antibiotics. Chemosphere 2014, 111, 278–282. [Google Scholar] [CrossRef] [PubMed]
- Fernández, R.L.; McDonald, J.A.; Khan, S.J.; Le-Clech, P. Removal of pharmaceuticals and endocrine disrupting chemicals by a submerged membrane photocatalysis reactor (MPR). Sep. Purif. Technol. 2014, 127, 131–139. [Google Scholar] [CrossRef]
- Jung, Y.J.; Kim, W.G.; Yoon, Y.; Kang, J.-W.; Hong, Y.M.; Kim, H.W. Removal of amoxicillin by UV and UV/H2O2 processes. Sci. Total Environ. 2012, 420, 160–167. [Google Scholar] [CrossRef] [PubMed]
- Voigt, M.; Savelsberg, C.; Jaeger, M. Photodegradation of the antibiotic spiramycin studied by high-performance liquid chromatography-electrospray ionization-quadrupole time-of-flight mass spectrometry. Toxicol. Environ. Chem. 2017, 99, 624–640. [Google Scholar] [CrossRef]
- Mauser, H. Formale Kinetik; Experimentelle Methoden der Physik und der Chemie; Bertelsmann-Universitätsverlag: Düsseldorf, Germany, 1974; ISBN 9783571191889. [Google Scholar]
- Mazellier, P.; Méité, L.; De Laat, J. Photodegradation of the steroid hormones 17beta-estradiol (E2) and 17alpha-ethinylestradiol (EE2) in dilute aqueous solution. Chemosphere 2008, 73, 1216–1223. [Google Scholar] [CrossRef] [PubMed]
- Wiegand, I.; Hilpert, K.; Hancock, R.E.W. Agar and broth dilution methods to determine the minimal inhibitory concentration (MIC) of antimicrobial substances. Nat. Protoc. 2008, 3, 163–175. [Google Scholar] [CrossRef]
- EUCAST. ISO 20776-1:2007; IOS: Geneva, Switzerland, 2007. [Google Scholar]
- Martin, T.; Harten, P.; Venkatapathy, R.; Young, D. TEST (Toxicity Estimation Software Tool) Ver 4.1.; U.S. Environmental Protection Agency: Washington, DC, USA, 2016. [Google Scholar]
- QSAR Toolbox. Laboratory of Mathematical Chemistry; University Bourgas: Bourgas, Bulgaria, 2012. [Google Scholar]
- Gao, L.; Shi, Y.; Li, W.; Niu, H.; Liu, J.; Cai, Y. Occurrence of antibiotics in eight sewage treatment plants in Beijing, China. Chemosphere 2012, 86, 665–671. [Google Scholar] [CrossRef]
- Li, W.; Shi, Y.; Gao, L.; Liu, J.; Cai, Y. Occurrence, distribution and potential affecting factors of antibiotics in sewage sludge of wastewater treatment plants in China. Sci. Total Environ. 2013, 445–446, 306–313. [Google Scholar] [CrossRef]
- Rosa, J.M.; Tambourgi, E.B.; Vanalle, R.M.; Carbajal Gamarra, F.M.; Curvelo Santana, J.C.; Araújo, M.C. Application of continuous H2O2/UV advanced oxidative process as an option to reduce the consumption of inputs, costs and environmental impacts of textile effluents. J. Clean. Prod. 2019, 246, 119012. [Google Scholar] [CrossRef]
- Al-Mamun, M.R.; Kader, S.; Islam, M.S.; Khan, M.Z.H. Photocatalytic activity improvement and application of UV-TiO2 photocatalysis in textile wastewater treatment: A review. J. Environ. Chem. Eng. 2019, 7, 103248. [Google Scholar] [CrossRef]
- Oppenländer, T. Photochemical Purification of Water and Air: Advanced Oxidation Processes (AOPs): Principles, Reaction Mechanisms, Reactor Concepts (Chemistry); WILEY-VCH Verlag: Weinheim, Germany, 2003; ISBN 3-527-30463-7. [Google Scholar]
- Homem, V.; Santos, L. Degradation and removal methods of antibiotics from aqueous matrices--a review. J. Environ. Manag. 2011, 92, 2304–2347. [Google Scholar] [CrossRef] [PubMed]
- Calza, P.; Minella, M.; Demarchis, L.; Sordello, F.; Minero, C. Photocatalytic rate dependence on light absorption properties of different TiO2 specimens. Catal. Today 2020, 340, 12–18. [Google Scholar] [CrossRef] [Green Version]
- Fasani, E.; Mella, M.; Caccia, D.; Tassi, S.; Fagnoni, M.; Albini, A. The photochemistry of lomefloxacin. An aromatic carbene as the key intermediate in photodecomposition. Chem. Commun. 1997, 1329–1330. [Google Scholar] [CrossRef]
- Budai, M.; Gróf, P.; Zimmer, A.; Pápai, K.; Klebovich, I.; Ludányi, K. UV light induced photodegradation of liposome encapsulated fluoroquinolones: An MS study. J. Photochem. Photobiol. A Chem. 2008, 198, 268–273. [Google Scholar] [CrossRef]
- Fasani, E.; Albini, A.; Mella, M.; Rampi, M.; Negra, F.B. Light and drugs: The photochemistry of fluoroquinolone antibiotics. Int. J. Photoenergy 1999, 1, 7–11. [Google Scholar] [CrossRef]
- Fasani, E.; Monti, S.; Manet, I.; Tilocca, F.; Pretali, L.; Mella, M.; Albini, A. Inter- and intramolecular photochemical reactions of fleroxacin. Org. Lett. 2009, 11, 1875–1878. [Google Scholar] [CrossRef]
- Liu, C.; Nanaboina, V.; Korshin, G.V.; Jiang, W. Spectroscopic study of degradation products of ciprofloxacin, norfloxacin and lomefloxacin formed in ozonated wastewater. Water Res. 2012, 46, 5235–5246. [Google Scholar] [CrossRef]

| Fluoroquinolone | Lomefloxacin |
|---|---|
| Chemical Structure |  |
| Molecular Formula | C17H19F2N3O3 |
| CAS Reg. No. | 98079-51-7 |
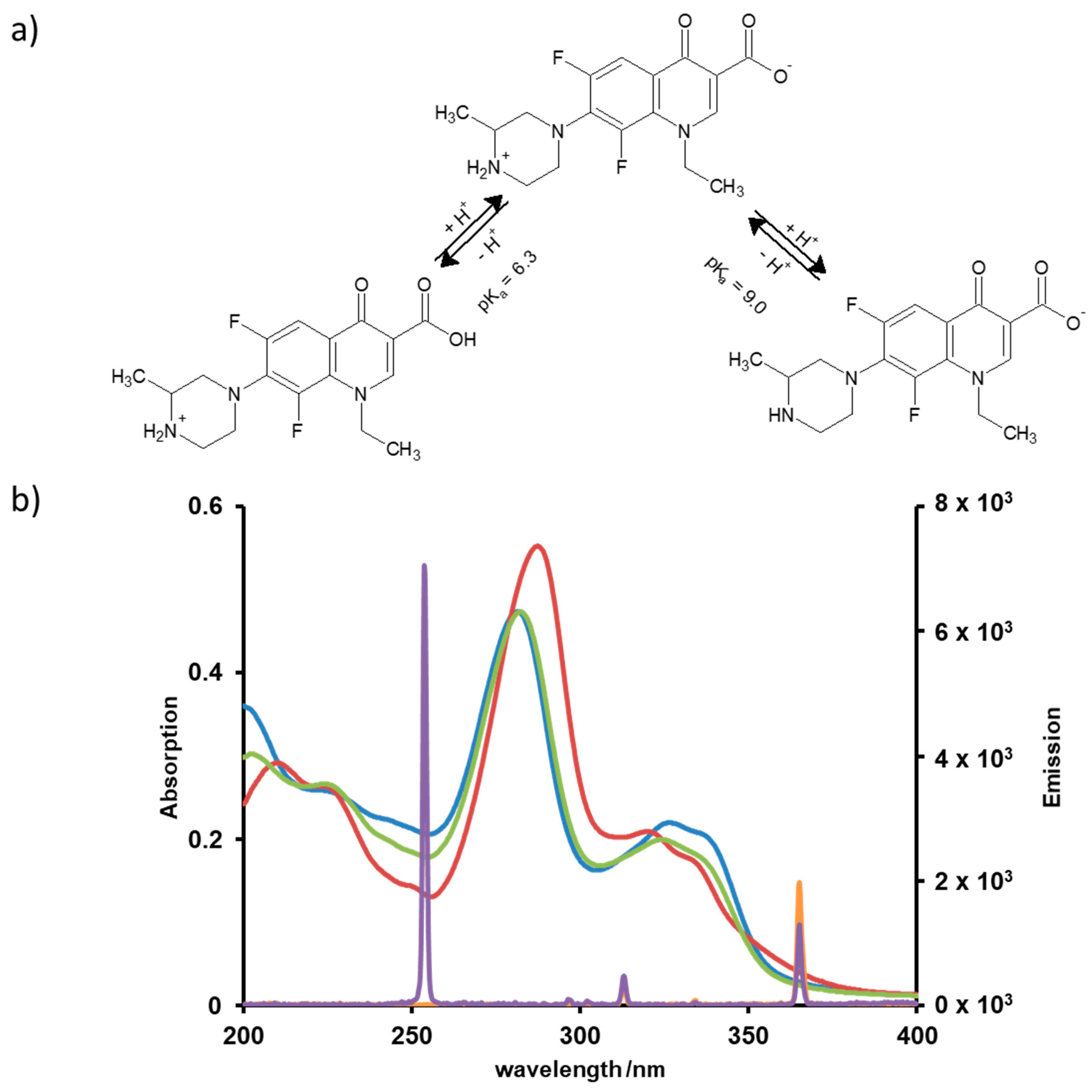
| pKa (1) | 6.3; 9.0 |
| [M+H]+ (exact) | 352.147 |
| [M+H]+ (accurate) (2) | 352.147 |
| MS/MS (3) | 308 |
| MS/MS/MS (3) | 265; 288 |
| λ (εmax)/nm at pH 3 | 287 |
| εmax/L·mol−1 at pH 3 | 37624 |
| λ (εmax)/nm at pH 7 | 282 |
| εmax/L·mol−1 at pH 7 | 32548 |
| λ (εmax)/nm at pH 9 | 282 |
| εmax/L·mol−1 at pH 9 | 32181 |
| Radiation Source | Water Matrix | pH | k1/min−1 | t1/2/min | Φ254 nm | Φ313 nm | Φ365 nm |
|---|---|---|---|---|---|---|---|
| UVA | MilliQ | 3–4 | 0.96 | 0.72 | - | 0.63 | 0.29 |
| MilliQ | 4–6 | 0.82 | 0.85 | - | 0.52 | 0.32 | |
| MilliQ | 6–7 | 1.22 | 0.57 | - | 0.73 | 0.45 | |
| MilliQ | 8–9 | 1.99 | 0.35 | - | 1.30 | 0.79 | |
| UVC | MilliQ | 3–4 | 0.57 | 1.21 | 0.02 | 0.13 | 0.39 |
| MilliQ | 4–6 | 0.80 | 0.87 | 0.02 | 0.15 | 0.62 | |
| MilliQ | 6–7 | 2.83 | 0.24 | 0.09 | 0.57 | 2.40 | |
| MilliQ | 8–9 | 2.04 | 0.34 | 0.06 | 0.45 | 1.90 | |
| River Erft | 6–7 | 1.49 | 0.47 | ||||
| Effluent WWTP | 8 | 1.47 | 0.47 |
| pH | H2O2 | TiO2 | UVA | UVC | ||
|---|---|---|---|---|---|---|
| /mg·L−1 | P25/mg·L−1 | k/min−1 | t1/2/min | k/min−1 | t1/2/min | |
| 3–4 | 0 | 0 | 0.96 | 0.72 | 0.57 | 1.21 |
| 0 | 50 | 1.71 | 0.41 | |||
| 0 | 100 | 0.79 | 0.88 | |||
| 10 | 0 | 0.93 | 0.75 | |||
| 30 | 0 | 1.03 | 0.68 | |||
| 6–7 | 0 | 0 | 1.22 | 0.57 | 2.83 | 0.24 |
| 0 | 50 | 1.07 | 0.65 | |||
| 0 | 100 | 1.42 | 0.49 | |||
| 10 | 0 | 2.10 | 0.33 | |||
| 30 | 0 | 1.84 | 0.38 | |||
| 8–9 | 0 | 0 | 1.99 | 0.35 | 2.04 | 0.34 |
| 0 | 50 | 0.80 | 0.87 | |||
| 0 | 100 | 1.05 | 0.66 | |||
| 10 | 0 | 1.30 | 0.53 | |||
| 30 | 0 | 2.34 | 0.30 | |||
| [M + H]+ (Theoretical) | [M + H]+ (Observed) | MS2 | MS3 | Proposed Structure | Reference |
|---|---|---|---|---|---|
| 352.147 Lomefloxacin | 352.147 | 308 | 265 |  | |
| 294.125 | 294.123 | 276 | 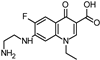 | this study | |
| 300.134 | 300.148 | 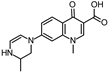 | this study | ||
| 304.129 | 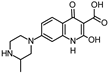 | this study | |||
| 308.140 | 308.140 | 290 | 270 | 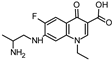 | [50,51] |
| 322.120 |  | this study | |||
| 324.115 | 324.1468 | 281 | 253 | 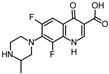 | this study |
| 332.140 | 332.136 | 288 | 245 | 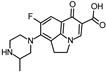 | [50,51,52,53] |
| 346.140 | 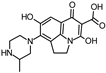 | this study | |||
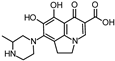 | |||||
| 348.135 | 348.135 | 330 | 304 | 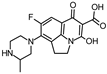 | [51] |
| 348.135 | 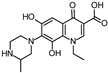 | ||||
| 350.151 | 350.144 |  | [51] | ||
| 350.151 | 306 | 263 | 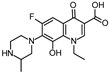 | ||
| 350.151 | 332 | 267 |  | ||
| 364.150 | 346 | 195 |  | this study | |
| 364.150 | 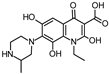 | ||||
| 368.142 | 368.111 | 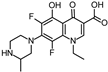 | [54] |
| Irradiation Time/min | P. fluorescens/µg·mL−1 | B. subtilis/µg·mL−1 | ||||||
|---|---|---|---|---|---|---|---|---|
| Irradiation Source | UVA | UVC | UVA | UVC | ||||
| MIC | IC50 (a) | MIC | IC50 (a) | MIC | IC50 (a) | MIC | IC50 (a) | |
| 0 | <0.55 | 0.41 | <0.55 | 0.56 | 0.28 | 0.14 | 0.28 | 0.14 |
| 1 | <0.55 | 0.42 | 1.11 | 0.80 | 0.28 | 0.17 | 0.55 | 0.22 |
| 2.5 | 1.10 | 0.53 | 4.44 | 2.34 | 0.55 | 0.13 | 2.22 | 0.51 |
| 3.5 | 2.21 | 1.00 | 8.88 | 0.95 | 1.10 | 0.26 | >2.22 | 0.21 |
| 5 | 4.42 | 2.31 | >8.88 | 2.59 | 2.21 | 0.44 | >2.22 | 0.60 |
| 7 | 8.83 | 3.68 | >8.88 | 1.76 | >2.21 | 0.18 | >2.22 | 0.45 |
| 30 | >8.83 | 4.41 | >8.88 | 1.27 | >2.21 | 0.65 | >2.22 | 0.22 |
| Substance | Daphnia magna LC50 (48 h) mg/L a | Fathead Minnow LC50 (96 h) mg/L a | Fathead Minnow LC50 (96 h) mg/L b,c | Actinopterygii LC50 (96 h) mg/L b,d | Branchiopoda LC50 (48 h) mg/L b,d |
|---|---|---|---|---|---|
| Lomefloxacin | 7.87 | 0.42 | 3.15 × 102 | 2.51 × 106 | 8.52 × 102 |
| 294 | 9.62 | 1.95 | 2.77 × 103 | 3.69 × 108 | 9.94 × 103 |
| 300 | 63.58 | 2.10 | 1.76 × 103 | 1.12 × 108 | 3.37 × 103 |
| 304 | 24.82 | 3.16 | 7.80 × 103 | 3.83 × 109 | 1.88 × 104 |
| 308 | 9.80 | 1.50 | 1.54 × 103 | 1.15 × 108 | 3.46 × 103 |
| 322 | 15.04 | 1.39 | 2.97 × 103 | 7.20 × 108 | 8.61 × 103 |
| 324 | 4.28 | 0.40 | 1.09 × 103 | 1.02 × 108 | 3.35 × 103 |
| 332 | 16.38 | 0.70 | 5.00 × 102 | 1.07 × 107 | 1.14 × 103 |
| 346a | 17.20 | 0.63 | 4.73 × 103 | 1.03 × 109 | 1.06 × 104 |
| 346b | 13.67 | 0.22 | 9.50 × 102 | 3.44 × 107 | 2.05 × 103 |
| 348a | 12.18 | 0.44 | 1.44 × 103 | 1.46 × 108 | 4.14 × 103 |
| 348b | 17.07 | 0.90 | 2.62 × 103 | 2.76 × 108 | 5.63 × 103 |
| 350a | 13.05 | 0.70 | 8.78 × 102 | 3.91 × 107 | 2.19 × 103 |
| 350b | 15.62 | 0.36 | 9.06 × 102 | 3.91 × 107 | 2.19 × 103 |
| 350c | 15.95 | 0.69 | 5.21 × 102 | 1.13 × 107 | 1.20 × 103 |
| 350d | 9.22 | 0.09 | 1.99 × 101 | 7.95 × 102 | 1.36 × 102 |
| 364a | 9.46 | 0.67 | 1.51 × 103 | 1.22 × 108 | 3.87 × 103 |
| 364b | 15.67 | 0.78 | 8.52 × 103 | 3.64 × 109 | 2.01 × 104 |
| 368 | 10.75 | 0.42 | 3.73 × 102 | 6.69 × 106 | 9.55 × 102 |
| pH | water | takt (P. fluorescens)/min | takt (B. subtilis)/min |
|---|---|---|---|
| 3–4 | MilliQ | 5.40 | 8.36 |
| 6–7 | MilliQ | 1.09 | 1.68 |
| 8–9 | MilliQ | 1.22 | 1.89 |
| 8 | Effluent | 2.09 | 3.24 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Voigt, M.; Hentschel, B.; Theiss, N.; Savelsberg, C.; Bartels, I.; Nickisch-Hartfiel, A.; Jaeger, M. Lomefloxacin—Occurrence in the German River Erft, Its Photo-Induced Elimination, and Assessment of Ecotoxicity. Clean Technol. 2020, 2, 74-90. https://doi.org/10.3390/cleantechnol2010006
Voigt M, Hentschel B, Theiss N, Savelsberg C, Bartels I, Nickisch-Hartfiel A, Jaeger M. Lomefloxacin—Occurrence in the German River Erft, Its Photo-Induced Elimination, and Assessment of Ecotoxicity. Clean Technologies. 2020; 2(1):74-90. https://doi.org/10.3390/cleantechnol2010006
Chicago/Turabian StyleVoigt, Melanie, Benjamin Hentschel, Nina Theiss, Christina Savelsberg, Indra Bartels, Anna Nickisch-Hartfiel, and Martin Jaeger. 2020. "Lomefloxacin—Occurrence in the German River Erft, Its Photo-Induced Elimination, and Assessment of Ecotoxicity" Clean Technologies 2, no. 1: 74-90. https://doi.org/10.3390/cleantechnol2010006
APA StyleVoigt, M., Hentschel, B., Theiss, N., Savelsberg, C., Bartels, I., Nickisch-Hartfiel, A., & Jaeger, M. (2020). Lomefloxacin—Occurrence in the German River Erft, Its Photo-Induced Elimination, and Assessment of Ecotoxicity. Clean Technologies, 2(1), 74-90. https://doi.org/10.3390/cleantechnol2010006





