Liquid Nanoclay: Synthesis and Applications to Transform an Arid Desert into Fertile Land
Abstract
1. Introduction
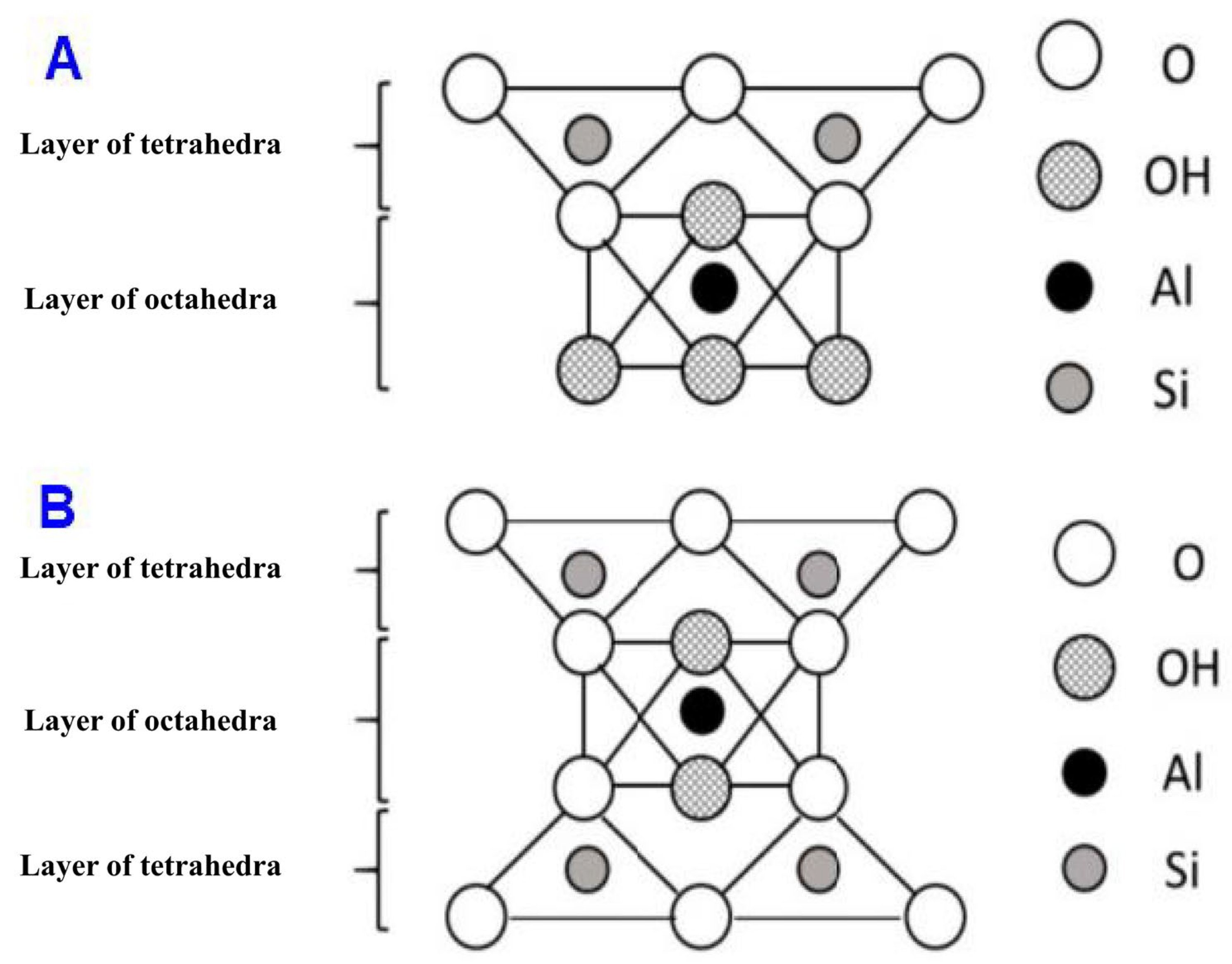
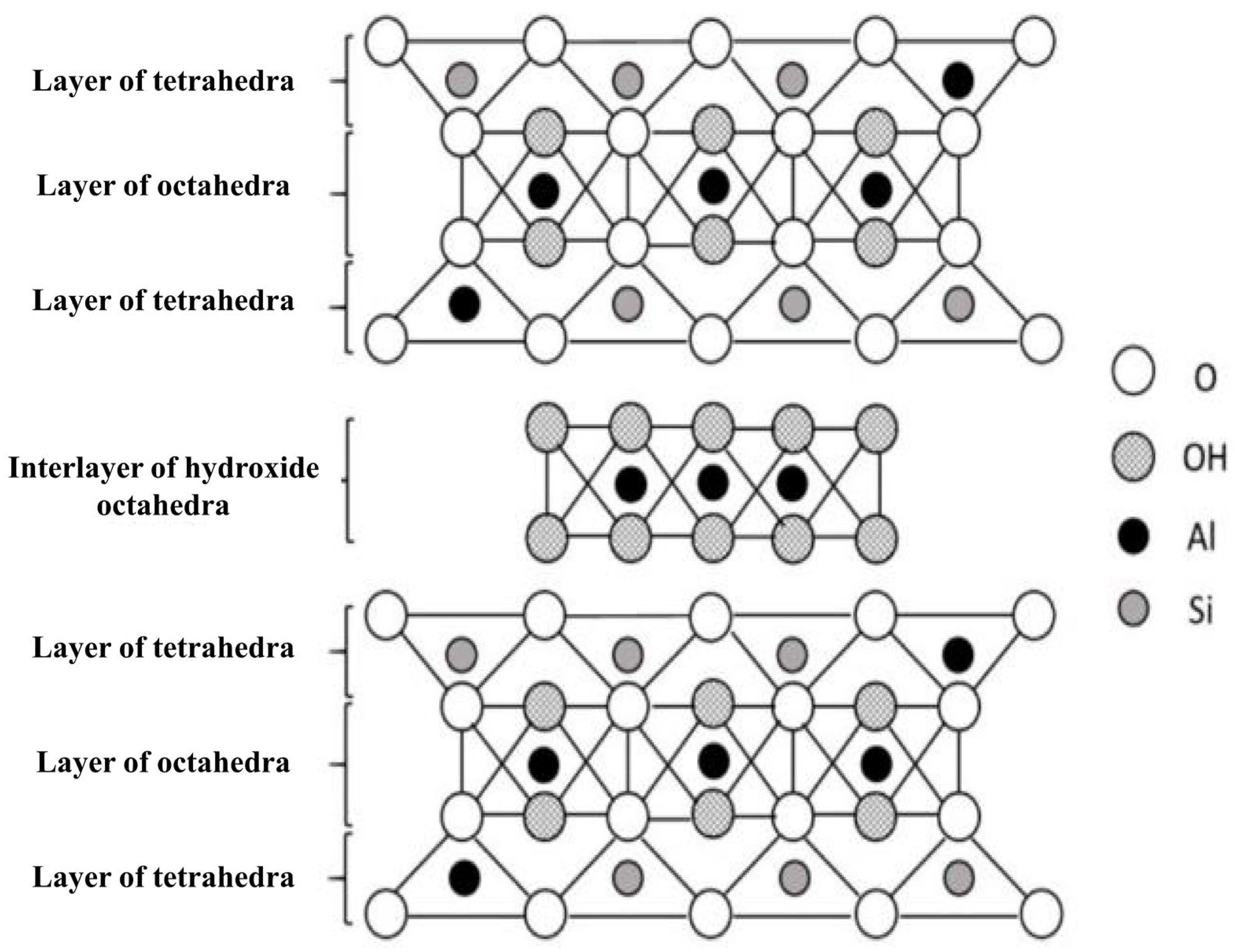
2. Properties and Structure of Nanoclay and Clay
Structure of Nanoclay
3. Characteristics of Cement-Based Materials Utilizing Calcined Nanoclay and Nanoclay
3.1. Workability
3.2. Mechanical Properties
3.2.1. Strength Enhancement
3.2.2. Elastic Modulus
3.2.3. Toughness Improvement
3.2.4. Resistance to Chloride Penetration
3.2.5. Frost Resistance
3.2.6. Acid and Sulfate Resistance
3.2.7. High-Temperature Resistance
3.2.8. Structure and Crystallization
4. Properties of Nanoclay Composite
4.1. Nanoclay Chemical Structure and Modification Methods
4.2. Synthesis of Liquid Nanoclay
4.3. Synthesis of Nanoclay
4.3.1. Solution-Blending Method
4.3.2. Melt-Blending Method
4.3.3. In Situ Polymerization Method
4.3.4. Use of Ammonium Salts in Nanoclay Synthesis
5. Nanoclay Application as a Soil Modifier
5.1. Nanoclay as a Soil Properties Modifier
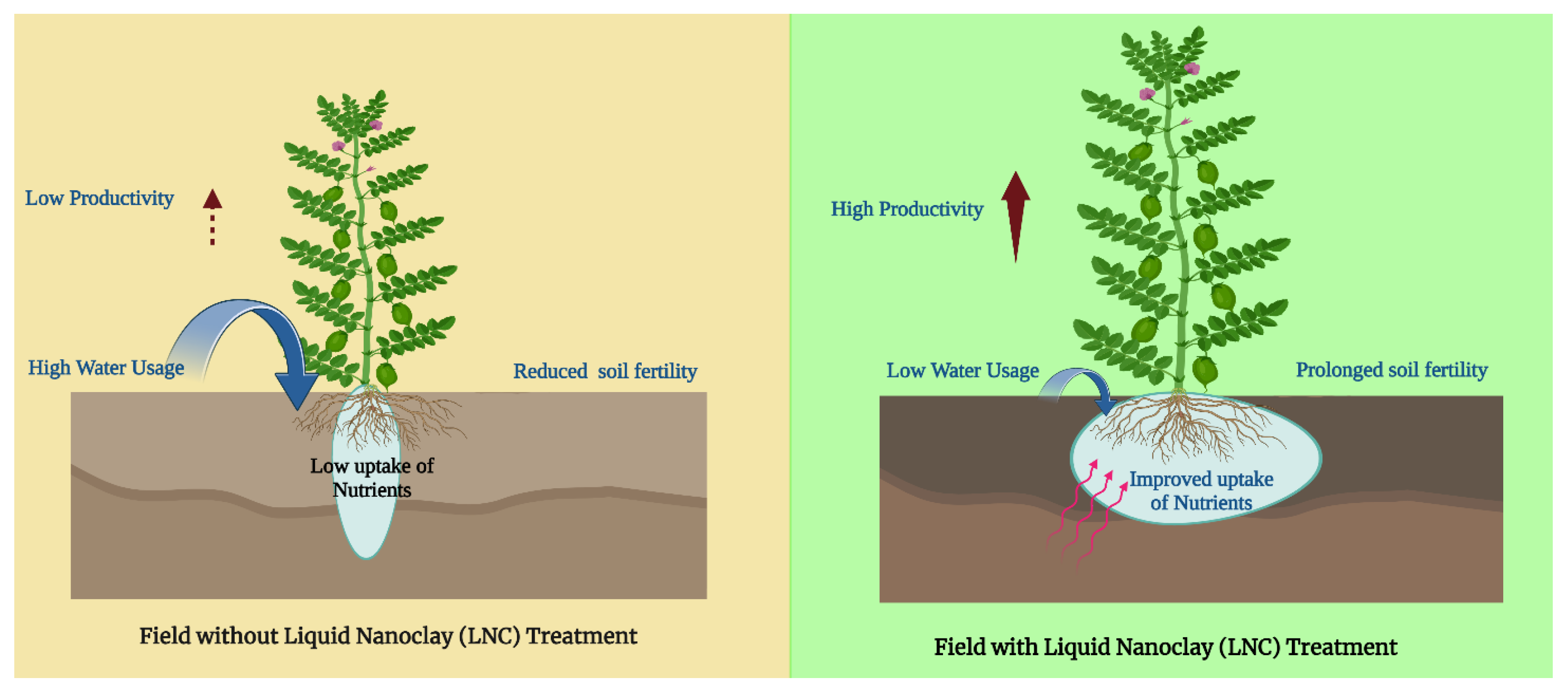
5.1.1. Improved Water Retention and Drainage
5.1.2. Soil Stabilization
5.1.3. Reduced Erosion
5.1.4. Enhanced Seed Germination
5.1.5. Nanoclays as Fertilizers for Sandy Soils
5.1.6. Using Nanotechnology for the Efficient Delivery of Fertilizers, Chemicals, Herbicides, Pesticides, and Plant Growth Regulators
5.1.7. Impact of Different Zinc Carriers on Zinc Fractions
5.1.8. Improving Nutrient Use Efficiency with Nano-Fertilizers
- The diminutive size of nano-fertilizers creates sites for plant nutrient metabolism, while their extensive surface area amplifies their impact. This synergy leads to heightened plant growth while requiring less essential nutrient consumption.
- With particle sizes below 100 nm, nano-fertilizers exhibit an accelerated penetration rate within the plant system.
- Nano-fertilizer particles, due to their smaller size and greater surface area compared to plant leaves and roots, facilitate superior penetration into plants from applied surfaces. This leads to increased utilization and bioavailability of the nano-fertilizers.
- Reduced particle size leads to increased surface area and a higher particle count per volume. This advantage is harnessed by those applying the chemicals, enhancing efficacy.
- Integrating micro-particles with fertilizers enhances the absorption and delivery of nutrients to crops [120].
5.1.9. Nutrients and Fertilizers
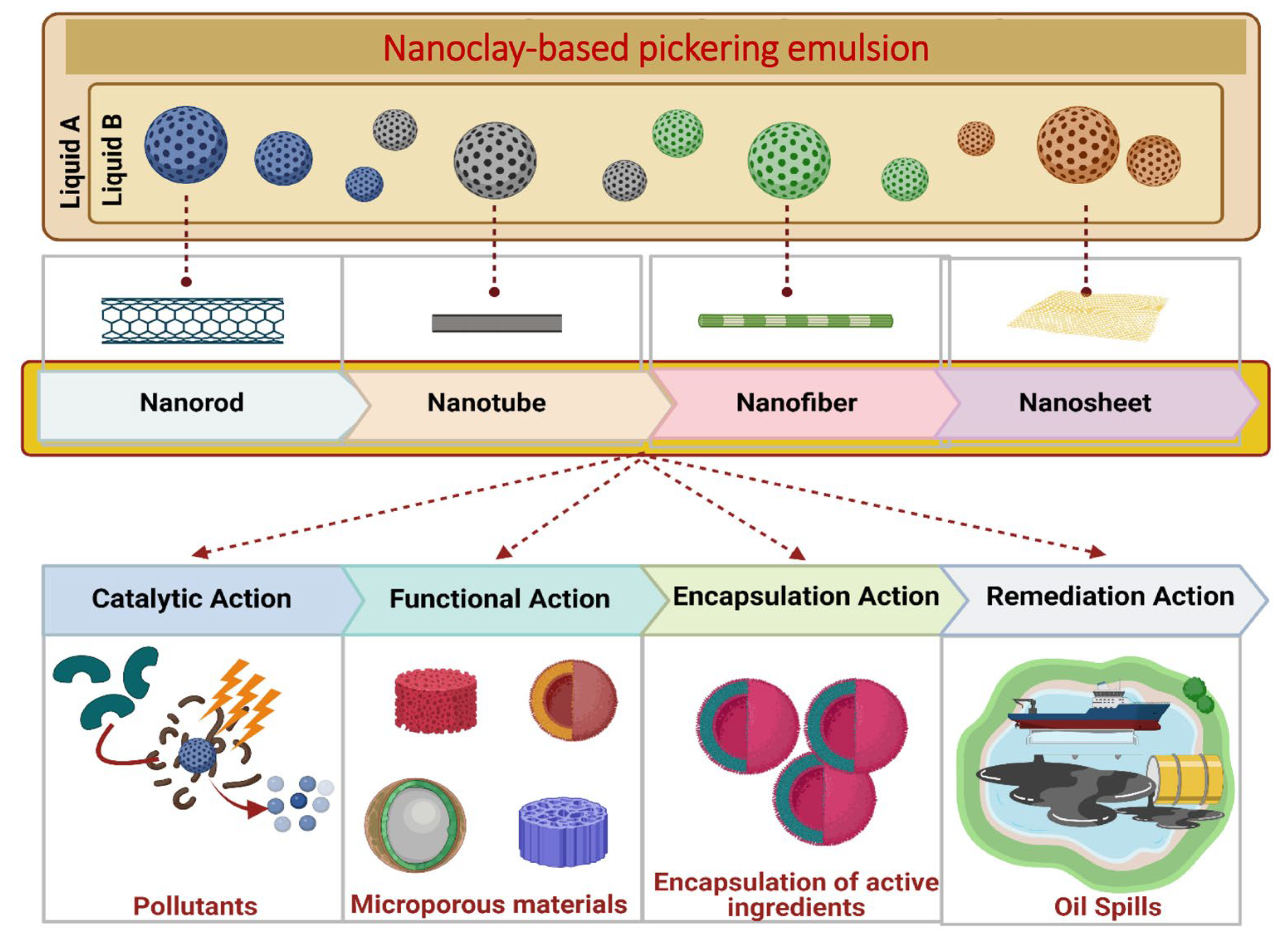
5.1.10. Nanoclay as a Carriers and Delivery System for Agrochemicals and Bioactive Molecules
6. Nanoclay for Improving Plant Performance
6.1. Nanoclays and Their Expanding Horizons
6.2. Toxicity and Environmental Impacts
7. Global Climate Change Mitigation
Carbon Dioxide (CO2) Capture and Storage
8. Limitation of Nanoclay in Desert Agriculture
8.1. Limited Availability
8.2. Environmental Concerns
8.3. Economic Feasibility
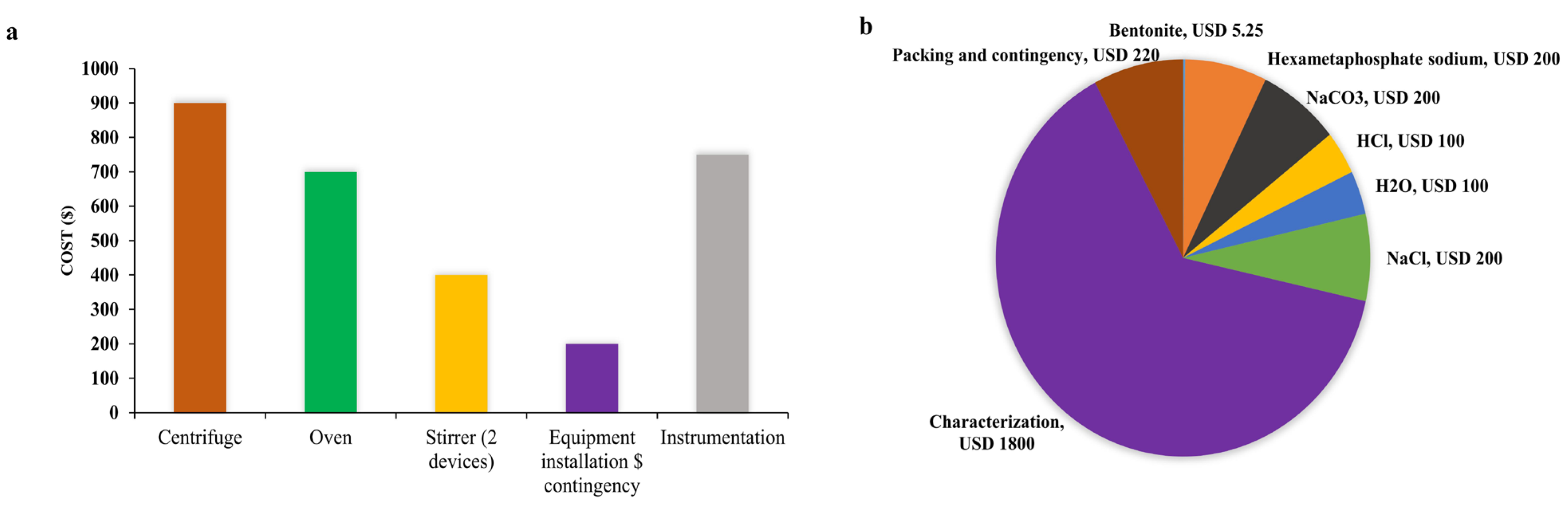
8.4. Cost Analysis
8.5. Long-Term Effect of Liquid Nanoclay on Soil Health and Fertility
9. Future Directions and Recommendations
9.1. Discussion of the Potential for Further Research and Development of Liquid Nanoclay
9.2. Recommendations for Liquid Nanoclay Implementation in Arid Regions
9.3. Advanced Formulations
9.4. Precision Application Systems and Technological Advancements
10. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Nazir, M.S.; Mohamad Kassim, M.H.; Mohapatra, L.; Gilani, M.A.; Raza, M.R.; Majeed, K. Characteristic properties of nanoclays and characterization of nanoparticulates and nanocomposites. Nanoclay Reinf. Polym. Compos. Nanocompos. Bionanocompos. 2016, 35–55. [Google Scholar]
- Müller, K.; Bugnicourt, E.; Latorre, M.; Jorda, M.; Echegoyen Sanz, Y.; Lagaron, J.M.; Miesbauer, O.; Bianchin, A.; Hankin, S.; Bölz, U. Review on the processing and properties of polymer nanocomposites and nanocoatings and their applications in the packaging, automotive and solar energy fields. Nanomaterials 2017, 7, 74. [Google Scholar] [CrossRef]
- Rytwo, G. Clay minerals as an ancient nanotechnology: Historical uses of clay organic interactions, and future possible perspectives. Macla 2008, 9, 15–17. [Google Scholar]
- Lee, S.M.; Tiwari, D. Organo and inorgano-organo-modified clays in the remediation of aqueous solutions: An overview. Appl. Clay Sci. 2012, 59, 84–102. [Google Scholar] [CrossRef]
- Uddin, M.K. A review on the adsorption of heavy metals by clay minerals, with special focus on the past decade. Chem. Eng. J. 2017, 308, 438–462. [Google Scholar] [CrossRef]
- Weaver, C.E.; Pollard, L.D. Developments in Sedimentology. In The Chemistry of Clay Minerals; Elsevier: Amsterdam, The Netherlands, 1973; Volume 15, pp. 1–213. [Google Scholar]
- Jawaid, M.; Qaiss, A.; Bouhfid, R. Nanoclay Reinforced Polymer Composites; Springer: Singapore, 2016. [Google Scholar]
- Savic, I.; Stojiljkovic, S.; Savic, I.; Gajic, D. Industrial application of clays and clay minerals. In Clays and Clay Minerals: Geological Origin, Mechanical Properties and Industrial Applications; Wesley, L.R., Ed.; Nova Science Publishers: Hauppauge, NY, USA, 2014; pp. 379–402. [Google Scholar]
- Barton, C.; Karathanasis, A.D. Clay minerals. In Encyclopedia of Soil Science-Two-Volume Set; CRC Press: Boca Raton, FL, USA, 2005; pp. 276–280. [Google Scholar]
- Majeed, K.; Jawaid, M.; Hassan, A.; Bakar, A.A.; Khalil, H.A.; Salema, A.; Inuwa, I. Potential materials for food packaging from nanoclay/natural fibres filled hybrid composites. Mater. Des. 2013, 46, 391–410. [Google Scholar] [CrossRef]
- Wei, J.; Meyer, C. Sisal fiber-reinforced cement composite with Portland cement substitution by a combination of metakaolin and nanoclay. J. Mater. Sci. 2014, 49, 7604–7619. [Google Scholar] [CrossRef]
- Kausar, A.; Ahmad, I.; Maaza, M.; Eisa, M. State-of-the-Art Nanoclay Reinforcement in Green Polymeric Nanocomposite: From Design to New Opportunities. Minerals 2022, 12, 1495. [Google Scholar] [CrossRef]
- Abulyazied, D.E.; Ene, A. An Investigative Study on the Progress of Nanoclay-Reinforced Polymers: Preparation, Properties, and Applications: A Review. Polymers 2021, 13, 4401. [Google Scholar] [CrossRef]
- Perera, M.; Jayarathna, L.; Yakandawala, D.; Seneweera, S. Nanoclay Composites as Agrochemical Carriers. In Clay Composites: Environmental Applications; Springer: Berlin/Heidelberg, Germany, 2023; pp. 543–557. [Google Scholar] [CrossRef]
- Kristian OLESEN Liquid Nano-Clay. Available online: http://www.rexresearch.com/olesennanoclay/olesen.html (accessed on 30 January 2024).
- Shakil, U.A.; Hassan, S.B.A.; Yahya, M.Y.; Nurhadiyanto, D. A review of properties and fabrication techniques of fiber reinforced polymer nanocomposites subjected to simulated accidental ballistic impact. Thin-Walled Struct. 2021, 158, 107150. [Google Scholar] [CrossRef]
- Manna, S.; Das, P.; Basak, P.; Sharma, A.K.; Singh, V.K.; Patel, R.K.; Pandey, J.K.; Ashokkumar, V.; Pugazhendhi, A. Separation of pollutants from aqueous solution using nanoclay and its nanocomposites: A review. Chemosphere 2021, 280, 130961. [Google Scholar] [CrossRef]
- Ditta, A. Role of Nanoclay Polymers in Agriculture: Applications and perspectves. In Nanohybrids in Environmental and Biomedical Applications; Sharma, S.K., Ed.; CRC Press: Boca Raton, FL, USA, 2019; pp. 323–334. ISBN 978-1-351-25684-1. [Google Scholar] [CrossRef]
- Mukhopadhyay, R.; De, N. Nano clay polymer composite: Synthesis, characterization, properties and application in rainfed agriculture. Glob. J. Bio-Sci. Biotechnol. 2014, 3, 133–138. [Google Scholar]
- Fathi, R.; Elyasi, M.; Khazaei, J. Investigating and studying the effect of Montmorillonite Nanoclay on consolidation and strength behavior of loose and soft fine-grained soil (Case study: Fine-grained soil of Kermanshah Faculty of Agriculture). Amirkabir J. Civ. Eng. 2023, 55, 1402. [Google Scholar] [CrossRef]
- Floody, M.C.; Theng, B.; Reyes, P.; Mora, M. Natural nanoclays: Applications and future trends—A Chilean perspective. Clay Minerals 2009, 44, 161–176. [Google Scholar] [CrossRef]
- Jacquet, A.; Geatches, D.L.; Clark, S.J.; Greenwell, H.C. Understanding cationic polymer adsorption on mineral surfaces: Kaolinite in cement aggregates. Minerals 2018, 8, 130. [Google Scholar] [CrossRef]
- Bergaya, F.; Lagaly, G. Introduction to clay science: Techniques and applications. In Developments in Clay Science; Elsevier: Amsterdam, The Netherlands, 2013; Volume 5, pp. 1–7. [Google Scholar] [CrossRef]
- Kango, S.; Kalia, S.; Celli, A.; Njuguna, J.; Habibi, Y.; Kumar, R. Surface modification of inorganic nanoparticles for development of organic–inorganic nanocomposites—A review. Prog. Polym. Sci. 2013, 38, 1232–1261. [Google Scholar] [CrossRef]
- Tullio, S.; Chalcraft, D. Converting natural nanoclay into modified nanoclay augments the toxic effect of natural nanoclay on aquatic invertebrates. Ecotoxicol. Environ. Saf. 2020, 197, 110602. [Google Scholar] [CrossRef]
- Mousavi, M.; Fini, E.H.; Hung, A.M. Underlying molecular interactions between sodium montmorillonite clay and acidic bitumen. J. Phys. Chem. C 2019, 123, 15513–15522. [Google Scholar] [CrossRef]
- Li, A.; Wang, A.-Q.; Chen, J.-M. Preparation and Properties of Poly (acrylic acid-potassium acrylate)/Attapulgite Superabsorbent Composite. J. Funct. Polym. 2004, 17, 200–206. [Google Scholar]
- Mazloomi, F.; Jalali, M. Effects of vermiculite, nanoclay and zeolite on ammonium transport through saturated sandy loam soil: Column experiments and modeling approaches. Catena 2019, 176, 170–180. [Google Scholar] [CrossRef]
- Adebajo, M.O.; Frost, R.L.; Kloprogge, J.T.; Carmody, O.; Kokot, S. Porous materials for oil spill cleanup: A review of synthesis and absorbing properties. J. Porous Mater. 2003, 10, 159–170. [Google Scholar] [CrossRef]
- Srithammaraj, K. Electrical Property-Investigation of Porous Clay Heterostructures Derived from Naturally-Occurring Clay Minerals for Smart Packaging. Ph.D. Thesis, Chulalongkorn University, Bangkok, Thailand, 2017. [Google Scholar] [CrossRef]
- Ma, C.; Eggleton, R.A. Cation exchange capacity of kaolinite. Clays Clay Miner. 1999, 47, 174–180. [Google Scholar] [CrossRef]
- Taylor, R.; Smith, T. The engineering geology of clay minerals: Swelling, shrinking and mudrock breakdown. Clay Miner. 1986, 21, 235–260. [Google Scholar] [CrossRef]
- Wypych, F.; Bergaya, F.; Schoonheydt, R.A. From polymers to clay polymer nanocomposites. In Developments in Clay Science; Elsevier: Amsterdam, The Netherlands, 2018; Volume 9, pp. 331–359. [Google Scholar] [CrossRef]
- Rafiee, R.; Shahzadi, R. Mechanical properties of nanoclay and nanoclay reinforced polymers: A review. Polym. Compos. 2019, 40, 431–445. [Google Scholar] [CrossRef]
- Dejaeghere, I.; Sonebi, M.; De Schutter, G. Influence of nano-clay on rheology, fresh properties, heat of hydration and strength of cement-based mortars. Constr. Build. Mater. 2019, 222, 73–85. [Google Scholar] [CrossRef]
- Farzadnia, N.; Ali, A.A.A.; Demirboga, R.; Anwar, M.P. Effect of halloysite nanoclay on mechanical properties, thermal behavior and microstructure of cement mortars. Cem. Concr. Res. 2013, 48, 97–104. [Google Scholar] [CrossRef]
- Mohd Faizal, M.; Hamidah, M.; Muhd Norhasri, M. Strength and chloride content of nanoclayed ultra-high performance concrete. In Proceedings of the Structure, Materials and Construction Engineering Conference (CONS ENG’14), Istanbul, Turkey, 20–22 November 2014; Dakam: Schiller Park, IL, USA, 2014; pp. 99–111. [Google Scholar]
- Fan, Y.; Zhang, S.; Kawashima, S.; Shah, S.P. Influence of kaolinite clay on the chloride diffusion property of cement-based materials. Cem. Concr. Compos. 2014, 45, 117–124. [Google Scholar] [CrossRef]
- Heikal, M.; Abdel-Gawwad, H.A.; Ababneh, F.A. Positive impact performance of hybrid effect of nano-clay and silica nano-particles on composite cements. Constr. Build. Mater. 2018, 190, 508–516. [Google Scholar] [CrossRef]
- Fadzil, A.M.; Muhd Norhasri, M.; Hamidah, M.; Zaidi, M.; Mohd Faizal, J. Alteration of nano metakaolin for ultra high performance concrete. In Proceedings of the CIEC 2013: International Civil and Infrastructure Engineering Conference 2013, Kuching, Malaysia, 22–25 September 2013; Springer: Singapore, 2014; pp. 887–894. [Google Scholar] [CrossRef]
- Muhd Norhasri, M.S.; Fadzil, A.M. Inclusion of nano metaclayed; as additive in ultra high performance concrete (UHPC). Constr. Build. Mater. 2019, 201, 590–598. [Google Scholar] [CrossRef]
- Alsallami, Z.H.A. Effect of sulfate in sand on some mechanical properties of nano metakaolin normal concrete. J. Babylon Univ. Eng. Sci. 2016, 24, 107–116. [Google Scholar]
- Alsallami, Z. The Effect of Sulfate in Sand on Some Mechanical Properties of Nano Metakaolin Self Compacting Concrete. Master’s Thesis, Babylon University, Babylon, Iraq, 2017. [Google Scholar]
- Aly, M.; Hashmi, M.S.J.; Olabi, A.G.; Messeiry, M.; Hussain, A.I. Effect of nano clay particles on mechanical, thermal and physical behaviours of waste-glass cement mortars. Mater. Sci. Eng. A 2011, 528, 7991–7998. [Google Scholar] [CrossRef]
- Hakamy, A.; Shaikh, F.U.A.; Low, I.M. Thermal and mechanical properties of hemp fabric-reinforced nanoclay–cement nanocomposites. J. Mater. Sci. 2014, 49, 1684–1694. [Google Scholar] [CrossRef]
- Langaroudi, M.A.M.; Mohammadi, Y. Effect of nano-clay on workability, mechanical, and durability properties of self-consolidating concrete containing mineral admixtures. Constr. Build. Mater. 2018, 191, 619–634. [Google Scholar] [CrossRef]
- Niu, X.-J.; Li, Q.-B.; Hu, Y.; Tan, Y.-S.; Liu, C.-F. Properties of cement-based materials incorporating nano-clay and calcined nano-clay: A review. Constr. Build. Mater. 2021, 284, 122820. [Google Scholar] [CrossRef]
- Girgin, Z.C. Effect of slag, nano clay and metakaolin on mechanical performance of basalt fibre cementitious composites. Constr. Build. Mater. 2018, 192, 70–84. [Google Scholar] [CrossRef]
- Fan, Y.; Zhang, S.; Wang, Q.; Shah, S.P. The effects of nano-calcined kaolinite clay on cement mortar exposed to acid deposits. Constr. Build. Mater. 2016, 102, 486–495. [Google Scholar] [CrossRef]
- Diab, A.M.; Abd Elmoaty, M.; Elyamany, H.; Sreh, M. Effect of nanomaterials additives on performance of concrete resistance against magnesium sulfate and acids. Constr. Build. Mater. 2019, 210, 210–231. [Google Scholar] [CrossRef]
- Morsy, M.S.; Al-Salloum, Y.A.; Abbas, H.; Alsayed, S.H. Behavior of blended cement mortars containing nano-metakaolin at elevated temperatures. Constr. Build. Mater. 2012, 35, 900–905. [Google Scholar] [CrossRef]
- Wang, W.C. Compressive strength and thermal conductivity of concrete with nanoclay under Various High-Temperatures. Constr. Build. Mater. 2017, 147, 305–311. [Google Scholar] [CrossRef]
- Shalby, O.B.; Elkady, H.M.; Nasr, E.A.R.; Kohail, M. Assessment of mechanical and fire resistance for hybrid nano-clay and steel fibres concrete at different curing ages. J. Struct. Fire Eng. 2019, 11, 189–203. [Google Scholar] [CrossRef]
- Yuan, Q.; Chen, J.; Yang, Y.; Misra, R.D. Nanoparticle interface driven microstructural evolution and crystalline phases of polypropylene: The effect of nanoclay content on structure and physical properties. Mater. Sci. Eng. A 2010, 527, 6002–6011. [Google Scholar] [CrossRef]
- Arora, A.; Choudhary, V.; Sharma, D.K. Effect of clay content and clay/surfactant on the mechanical, thermal and barrier properties of polystyrene/organoclay nanocomposites. J. Polym. Res. 2011, 18, 843–857. [Google Scholar] [CrossRef]
- Kusmono; Ishak, Z.M.; Chow, W.S.; Takeichi, T.; Rochmadi. Effect of clay modification on the morphological, mechanical, and thermal properties of polyamide 6/polypropylene/montmorillonite nanocomposites. Polym. Compos. 2010, 31, 1156–1167. [Google Scholar] [CrossRef]
- Ngo, T.-D.; Ton-That, M.T.; Hoa, S.V.; Cole, K.C. Effect of temperature, duration and speed of pre-mixing on the dispersion of clay/epoxy nanocomposites. Compos. Sci. Technol. 2009, 69, 1831–1840. [Google Scholar] [CrossRef]
- Albdiry, M.; Yousif, B.; Ku, H.; Lau, K. A critical review on the manufacturing processes in relation to the properties of nanoclay/polymer composites. J. Compos. Mater. 2013, 47, 1093–1115. [Google Scholar] [CrossRef]
- Jatav, G.; Nirmal, D. Application of nanotechnology in soil-plant system. An. As. J. Soil Sci. 2013, 8, 176–184. [Google Scholar]
- Uddin, F. Clays, nanoclays, and montmorillonite minerals. Metall. Mater. Trans. A 2008, 39, 2804–2814. [Google Scholar] [CrossRef]
- Nadziakiewicza, M.; Kehoe, S.; Micek, P. Physico-Chemical Properties of Clay Minerals and Their Use as a Health Promoting Feed Additive. Animals 2019, 9, 714. [Google Scholar] [CrossRef]
- Fawzi, N.I.; Qurani, I.Z. Can We Practice Sustainable Agriculture on Suboptimal Land? In Suboptimal Land Series-Part 2; Tay Juhana Foundation: Jakarta, Indonesia, 2020. [Google Scholar]
- Lazzara, G.; Massaro, M.; Riela, S. Current status of nanoclay phytotoxicity. Phytotoxicity Nanoparticles 2018, 151–174. [Google Scholar] [CrossRef]
- Jayrajsinh, S.; Shankar, G.; Agrawal, Y.K.; Bakre, L. Montmorillonite nanoclay as a multifaceted drug-delivery carrier: A review. J. Drug Deliv. Sci. Technol. 2017, 39, 200–209. [Google Scholar] [CrossRef]
- Abdollahi, M.; Alboofetileh, M.; Rezaei, M.; Behrooz, R. Comparing physico-mechanical and thermal properties of alginate nanocomposite films reinforced with organic and/or inorganic nanofillers. Food Hydrocoll. 2013, 32, 416–424. [Google Scholar] [CrossRef]
- Ray, S.S.; Okamoto, M. Polymer/layered silicate nanocomposites: A review from preparation to processing. Prog. Polym. Sci. 2003, 28, 1539–1641. [Google Scholar] [CrossRef]
- Ho, M.-W.; Lam, C.-K.; Lau, K.-t.; Ng, D.H.; Hui, D. Mechanical properties of epoxy-based composites using nanoclays. Compos. Struct. 2006, 75, 415–421. [Google Scholar] [CrossRef]
- Bauli, C.R.; Lima, G.F.; de Souza, A.G.; Ferreira, R.R.; Rosa, D.S. Eco-friendly carboxymethyl cellulose hydrogels filled with nanocellulose or nanoclays for agriculture applications as soil conditioning and nutrient carrier and their impact on cucumber growing. Colloids Surf. A Physicochem. Eng. Asp. 2021, 623, 126771. [Google Scholar] [CrossRef]
- Alamri, H.; Low, I.M. Effect of water absorption on the mechanical properties of nanoclay filled recycled cellulose fibre reinforced epoxy hybrid nanocomposites. Compos. Part A Appl. Sci. Manuf. 2013, 44, 23–31. [Google Scholar] [CrossRef]
- Lu, T.; Gou, H.; Rao, H.; Zhao, G. Recent progress in nanoclay-based Pickering emulsion and applications. J. Environ. Chem. Eng. 2021, 9, 105941. [Google Scholar] [CrossRef]
- Lynsey Hart 2020. How to Make Liquid Nanoclay. Available online: https://nanografi.com/blog/how-to-make-liquid-nanoclay/ (accessed on 2 February 2024).
- Mohapi, M.; Sefadi, J.S.; Mochane, M.J.; Magagula, S.I.; Lebelo, K. Effect of LDHs and Other Clays on Polymer Composite in Adsorptive Removal of Contaminants: A Review. Crystals 2020, 10, 957. [Google Scholar] [CrossRef]
- Debnath, D.; Dhibar, A.K.; Khatua, B. Studies on the morphology and properties of PMMA-organoclay; nanocomposites with reference to the manufacturing techniques. Polym. Plast. Technol. Eng. 2010, 49, 1087–1094. [Google Scholar] [CrossRef]
- Ports, B.F.; Weiss, R.A. One-step melt extrusion process for preparing polyolefin/clay nanocomposites using natural montmorillonite. Ind. Eng. Chem. Res. 2010, 49, 11896–11905. [Google Scholar] [CrossRef]
- Jollands, M.; Gupta, R.K. Effect of mixing conditions on mechanical properties of; polylactide/montmorillonite clay nanocomposites. J. Appl. Polym. Sci. 2010, 118, 1489–1493. [Google Scholar] [CrossRef]
- Yarahmadi, N.; Jakubowicz, I.; Hjertberg, T. Development of poly(vinyl chloride)/montmorillonite; nanocomposites using chelating agents. Polym. Degrad. Stab. 2010, 95, 132–137. [Google Scholar] [CrossRef]
- Quigley, J.P.; Baird, D.G. Improved mechanical properties of organoclay/nylon 6 nanocomposites prepared via a super-critical carbon dioxide-aided. Polym. Compos. 2015, 36, 527–537. [Google Scholar] [CrossRef]
- Guo, F.; Aryana, S.; Han, Y.; Jiao, Y. A review of the synthesis and applications of polymer–nanoclay composites. Appl. Sci. 2018, 8, 1696. [Google Scholar] [CrossRef]
- Sarkar, S.; Datta, S.C.; Biswas, D.R. Synthesis and characterization of nanoclay–polymer composites from soil clay with respect to their water-holding capacities and nutrient-release behavior. J. Appl. Polym. Sci. 2014, 131, 1–8. [Google Scholar] [CrossRef]
- Patel, H.A. Synthesis and Characterization of Nanoclays for Polymeric Nanocomposites, Paints and Adsorption Applications. 2011. Available online: http://hdl.handle.net/10603/89251 (accessed on 2 September 2023).
- Verma, M.K.; Pandey, P.; De, N. Characterization of water retention and release capacity of innovative nano clay polymer composite superabsorbent. J. Pharmacogn. Phytochem. 2017, 6, 42–48. [Google Scholar]
- Liu, M.; Guo, T. Preparation and swelling properties of crosslinked sodium polyacrylate. J. Appl. Polym. Sci. 2001, 82, 1515–1520. [Google Scholar] [CrossRef]
- Nge, T.T.; Hori, N.; Takemura, A.; Ono, H. Swelling behavior of chitosan/poly (acrylic acid) complex. J. Appl. Polym. Sci. 2004, 92, 2930–2940. [Google Scholar] [CrossRef]
- Lokhande, H.; Varadarajan, P. A new Guargum-based superabsorbent polymer synthesised using gamma radiation as a soil additive. Bioresour. Technol. 1992, 42, 119–122. [Google Scholar] [CrossRef]
- Mortland, M. Clay-organic complexes and interactions. Adv. Agron. 1970, 22, 75–117. [Google Scholar]
- Ghazi, H.; Baziar, M.; Mirkazemi, S. The effects of nano-material additives on the basic properties of soil. In Proceedings of the 14th Asian Regional Conference of Geotechnic, Hong Kong, China, 23–27 May 2011. [Google Scholar]
- Coo, J.L.; So, Z.P.; Ng, C.W. Effect of nanoparticles on the shrinkage properties of clay. Eng. Geol. 2016, 213, 84–88. [Google Scholar] [CrossRef]
- Zhang, G. Soil nanoparticles and their influence on engineering properties of soils. In Advances in Measurement and Modeling of Soil Behavior; ASCE: Reston, VA, USA, 2007; pp. 1–13. [Google Scholar] [CrossRef]
- Dane, J.H.; Hopmans, J.W.; Topp, G. Pressure plate extractor. In Methods of Soil Analysis; Soil Science Society of America: Madison, WI, USA, 2002; Volume 4, pp. 688–690. [Google Scholar]
- Dartevelle, S. Numerical and Granulometric Approaches to Geophysical Granular Flows. Ph.D. Thesis, Michigan Technological University, Houghton, MI, USA, 2003. [Google Scholar]
- Holister, P.; Weener, J.-W.; Roman, C.; Harper, T. Nanoparticles. Technol. White Pap. 2003, 3, 1–11. [Google Scholar]
- Khalid, N.; Mukri, M.; Kamarudin, F.; Abdul Ghani, A.H.; Arshad, M.F.; Sidek, N.; Ahmad Jalani, A.Z.; Bilong, B. Effect of nanoclay in soft soil stabilization. In InCIEC 2014: Proceedings of the International Civil and Infrastructure Engineering Conference 2014; Springer: Singapore, 2015; pp. 905–914. [Google Scholar]
- Rafahi, H.G. Wind Erosion and Conservation, 4th ed.; University of Tehran: Tehran, Iran, 2006; p. 320. (In Persian) [Google Scholar]
- Ekhtesasi, M.; Sepehr, A. Investigation of wind erosion process for estimation, prevention, and control of DSS in Yazd–Ardakan plain. Environ. Monit. Assess. 2009, 159, 267–280. [Google Scholar] [CrossRef] [PubMed]
- Rosario-Diaz, J.; Haro-De Rosario, A.; Canero-Leon, R. Contingent valuation of erosion externalities: The case of the hydrographic basin of the Alto Almanzora in Sierra de Filabres, Spain. J. Environ. Prot. Ecol. 2013, 14, 1185–1194. [Google Scholar]
- Olesen, K. Turning Sandy Soil to Farmland: 66% Water Saved in Sandy Soil Treated with NanoClay; Desert Control Institute Inc.: Yuma, AZ, USA, 2010; Volume 1, pp. 1–10. [Google Scholar]
- Padidar, M.; Jalalian, A.; Abdouss, M.; Najafi, P.; Honarjoo, N.; Fallahzade, J. Effects of nanoclay on some physical properties of sandy soil and wind erosion. Int. J. Soil Sci. 2016, 11, 9–13. [Google Scholar] [CrossRef]
- He, J.-J.; Cai, Q.-G.; Tang, Z.-J. Wind tunnel experimental study on the effect of PAM on soil wind erosion control. Environ. Monit. Assess. 2008, 145, 185–193. [Google Scholar] [CrossRef] [PubMed]
- Hazirei, F.; Zare Ernani, M. Investigation of effect of clay-lime mulch for sand dunes fixation. Water Soil 2013, 27, 373–380. [Google Scholar]
- Shelar, A.; Nile, S.H.; Singh, A.V.; Rothenstein, D.; Bill, J.; Xiao, J.; Chaskar, M.; Kai, G.; Patil, R. Recent advances in nano-enabled seed treatment strategies for sustainable agriculture: Challenges, risk assessment, and future perspectives. Nano-Micro Letters 2023, 15, 54. [Google Scholar] [CrossRef]
- Neme, K.; Nafady, A.; Uddin, S.; Tola, Y.B. Application of nanotechnology in agriculture, postharvest loss reduction and food processing: Food security implication and challenges. Heliyon 2021, 7, e08539. [Google Scholar] [CrossRef] [PubMed]
- Pulizzi, F. Nano in the future of crops. Nat. Nanotechnol. 2019, 14, 507. [Google Scholar] [CrossRef]
- Pramanik, P.; Krishnan, P.; Maity, A.; Mridha, N.; Mukherjee, A.; Rai, V. Application of nanotechnology in agriculture. Environ. Nanotechnol. 2020, 4, 317–348. [Google Scholar]
- Jiang, M.; Song, Y.; Kanwar, M.K.; Ahammed, G.J.; Shao, S.; Zhou, J. Phytonanotechnology applications in modern agriculture. J. Nanobiotechnology 2021, 19, 1–20. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Dimkpa, C.; Deng, C.; Elmer, W.H.; Gardea-Torresdey, J.; White, J.C. Impact of engineered nanomaterials on rice (Oryza sativa L.): A critical review of current knowledge. Environ. Pollut. 2022, 297, 118738. [Google Scholar] [CrossRef] [PubMed]
- Szőllősi, R.; Molnár, Á.; Kondak, S.; Kolbert, Z. Dual effect of nanomaterials on germination and seedling growth: Stimulation vs. phytotoxicity. Plants 2020, 9, 1745. [Google Scholar] [CrossRef] [PubMed]
- Chandrasekaran, U.; Luo, X.; Wang, Q.; Shu, K. Are there unidentified factors involved in the germination of nanoprimed seeds? Front. Plant Sci. 2020, 11, 832. [Google Scholar] [CrossRef] [PubMed]
- Nile, S.H.; Thiruvengadam, M.; Wang, Y.; Samynathan, R.; Shariati, M.A.; Rebezov, M.; Nile, A.; Sun, M.; Venkidasamy, B.; Xiao, J. Nano-priming as emerging seed priming technology for sustainable agriculture—Recent developments and future perspectives. J. Nanobiotechnology 2022, 20, 1–31. [Google Scholar]
- Shao, C.; Zhao, H.; Wang, P. Recent development in functional nanomaterials for sustainable and smart agricultural chemical technologies. Nano Converg. 2022, 9, 1–17. [Google Scholar]
- Everaert, M.; Warrinnier, R.; Baken, S.; Gustafsson, J.-P.; De Vos, D.; Smolders, E. Phosphate-exchanged Mg–Al layered double hydroxides: A new slow release phosphate fertilizer. ACS Sustain. Chem. Eng. 2016, 4, 4280–4287. [Google Scholar] [CrossRef]
- Songkhum, P.; Wuttikhun, T.; Chanlek, N.; Khemthong, P.; Laohhasurayotin, K. Controlled release studies of boron and zinc from layered double hydroxides as the micronutrient hosts for agricultural application. Appl. Clay Sci. 2018, 152, 311–322. [Google Scholar] [CrossRef]
- Indira, M.; Krupanidhi, S.; Venkateswarulu, T.; Pallavi, G.; Peele, K.A. Current Aspects of Nanotechnology: Applications in Agriculture. In Biobased Nanotechnology for Green Applications; Springer: Singapore, 2021; pp. 73–99. [Google Scholar]
- Sorrentino, A.; Gorrasi, G.; Tortora, M.; Vittoria, V. Barrier properties of polymer/clay nanocomposites. Polym. Nanocomposites 2006, 17, 273–292. [Google Scholar]
- Mandal, N.; Datta, S.; Manjaiah, K. Synthesis, characterization and controlled release study of Zn from Zincated nanoclay polymer composites (ZNCPCs) in relation to equilibrium water absorbency under Zn deficient Typic Haplustept. Ann. Plant Soil Res. 2015, 17, 187–195. [Google Scholar]
- Gao, X.; Zhang, F.; Hoffland, E. Malate Exudation by Six Aerobic Rice Genotypes Varying in Zinc Uptake Efficiency. J. Environ. Qual. 2009, 38, 2315–2321. [Google Scholar] [CrossRef]
- Impa, S.; Johnson-Beebout, S.E. Mitigating zinc deficiency and achieving high grain Zn in rice through integration of soil chemistry and plant physiology research. Plant Soil 2012, 361, 3–41. [Google Scholar] [CrossRef]
- Adhikary, S.; Mandal, N.; Rakshit, R.; Das, A.; Kumar, V.; Kumari, N.; Choudhary, S.K.; Homa, F. Field evaluation of Zincated nanoclay polymer composite (ZNCPC): Impact on DTPA-extractable Zn, sequential Zn fractions and apparent Zn recovery under rice rhizosphere. Soil Tillage Res. 2020, 201, 104607. [Google Scholar] [CrossRef]
- Wisawapipat, W.; Janlaksana, Y.; Christl, I. Zinc solubility in tropical paddy soils: A multi-chemical extraction technique study. Geoderma 2017, 301, 1–10. [Google Scholar] [CrossRef]
- Scheinost, A.C.; Kretzschmar, R.; Pfister, S.; Roberts, D.R. Combining Selective Sequential Extractions, X-ray Absorption Spectroscopy, and Principal Component Analysis for Quantitative Zinc Speciation in Soil. Environ. Sci. Technol. 2002, 36, 5021–5028. [Google Scholar] [CrossRef] [PubMed]
- Al-Juthery, H.W.; Lahmod, N.R.; Al-Taee, R.A. Intelligent, nano-fertilizers: A new technology for improvement nutrient use efficiency (article review). IOP Conf. Ser. Earth Environ. Sci. 2021, 735, 012086. [Google Scholar] [CrossRef]
- Kopittke, P.M.; Lombi, E.; Wang, P.; Schjoerring, J.K.; Husted, S. Nanomaterials as fertilizers for improving plant mineral nutrition and environmental outcomes. Environ. Sci. Nano 2019, 6, 3513–3524. [Google Scholar] [CrossRef]
- Manjaiah, K.M.; Mukhopadhyay, R.; Paul, R.; Datta, S.C.; Kumararaja, P.; Sarkar, B. Clay minerals and zeolites for environmentally sustainable agriculture. In Modified Clay and Zeolite Nanocomposite Materials; Elsevier: Amsterdam, The Netherlands, 2019; pp. 309–329. [Google Scholar]
- Shafigh, M.; Hamidpour, M.; Abbaszadeh-Dahaji, P.; Mozafari, V.; Furrer, G. Bioavailability of Zn from layered double hydroxides: The effects of plant growth-promoting rhizobacteria (PGPR). Appl. Clay Sci. 2019, 182, 105283. [Google Scholar] [CrossRef]
- Yuan, G. An organoclay formula for the slow release of soluble compounds. Appl. Clay Sci. 2014, 100, 84–87. [Google Scholar] [CrossRef]
- Bhardwaj, D.; Sharma, M.; Sharma, P.; Tomar, R. Synthesis and surfactant modification of clinoptilolite and montmorillonite for the removal of nitrate and preparation of slow release nitrogen fertilizer. J. Hazard. Mater. 2012, 227, 292–300. [Google Scholar] [CrossRef]
- Madusanka, N.; Sandaruwan, C.; Kottegoda, N.; Sirisena, D.; Munaweera, I.; De Alwis, A.; Karunaratne, V.; Amaratunga, G.A. Urea–hydroxyapatite-montmorillonite nanohybrid composites as slow release nitrogen compositions. Appl. Clay Sci. 2017, 150, 303–308. [Google Scholar] [CrossRef]
- Said, A.; Zhang, Q.; Qu, J.; Liu, Y.; Lei, Z.; Hu, H.; Xu, Z. Mechanochemical activation of phlogopite to directly produce slow-release potassium fertilizer. Appl. Clay Sci. 2018, 165, 77–81. [Google Scholar] [CrossRef]
- Pulimi, M.; Subramanian, S. Nanomaterials for soil fertilisation and contaminant removal. In Nanoscience in Food and Agriculture 1; Springer: Singapore, 2016; pp. 229–246. [Google Scholar]
- Singh, S.; Sangwan, S.; Sharma, P.; Devi, P.; Moond, M. Nanotechnology for sustainable agriculture: An emerging perspective. J. Nanosci. Nanotechnol. 2021, 21, 3453–3465. [Google Scholar] [CrossRef] [PubMed]
- Ashraf, S.A.; Siddiqui, A.J.; Abd Elmoneim, O.E.; Khan, M.I.; Patel, M.; Alreshidi, M.; Moin, A.; Singh, R.; Snoussi, M.; Adnan, M. Innovations in nanoscience for the sustainable development of food and agriculture with implications on health and environment. Sci. Total Environ. 2021, 768, 144990. [Google Scholar] [CrossRef] [PubMed]
- Sastry, R.K.; Rashmi, H.; Rao, N. Nanotechnology for enhancing food security in India. Food Policy 2011, 36, 391–400. [Google Scholar] [CrossRef]
- Gogos, A.; Knauer, K.; Bucheli, T.D. Nanomaterials in plant protection and fertilization: Current state, foreseen applications, and research priorities. J. Agric. Food Chem. 2012, 60, 9781–9792. [Google Scholar] [CrossRef] [PubMed]
- Kahru, A.; Savolainen, K. Potential hazard of nanoparticles: From properties to biological and environmental effects. Toxicology 2010, 2, 89–91. [Google Scholar] [CrossRef]
- Handy, R.D.; Shaw, B.J. Toxic effects of nanoparticles and nanomaterials: Implications for public health, risk assessment and the public perception of nanotechnology. Health Risk Soc. 2007, 9, 125–144. [Google Scholar] [CrossRef]
- Yadav, T.; Mungray, A.A.; Mungray, A.K. Fabricated nanoparticles: Current status and potential phytotoxic threats. In Reviews of Environmental Contamination and Toxicology Volume: With Cumulative and Comprehensive Index; Springer: Singapore, 2014; pp. 83–110. [Google Scholar]
- Mintova, S.; Jaber, M.; Valtchev, V. Nanosized microporous crystals: Emerging applications. Chem. Soc. Rev. 2015, 44, 7207–7233. [Google Scholar] [CrossRef]
- Chen, J.; Lü, S.; Zhang, Z.; Zhao, X.; Li, X.; Ning, P.; Liu, M. Environmentally friendly fertilizers: A review of materials used and their effects on the environment. Sci. Total Environ. 2018, 613, 829–839. [Google Scholar] [CrossRef]
- Nadiminti, P.P.; Sharma, H.; Kada, S.R.; Pfeffer, F.M.; O’Dell, L.A.; Cahill, D.M. Use of Mg–Al nanoclay as an efficient vehicle for the delivery of the herbicide 2, 4-dichlorophenoxyacetic acid. ACS Sustain. Chem. Eng. 2019, 7, 10962–10970. [Google Scholar] [CrossRef]
- Padil, V.V.; Kumar, K.A.; Murugesan, S.; Torres-Mendieta, R.; Wacławek, S.; Cheong, J.Y.; Černík, M.; Varma, R.S. Sustainable and safer nanoclay composites for multifaceted applications. Green Chem. 2022, 24, 3081–3114. [Google Scholar] [CrossRef]
- Hamid, Y.; Tang, L.; Hussain, B.; Usman, M.; Liu, L.; Ulhassan, Z.; He, Z.; Yang, X. Sepiolite clay: A review of its applications to immobilize toxic metals in contaminated soils and its implications in soil–plant system. Environ. Technol. Innov. 2021, 23, 101598. [Google Scholar] [CrossRef]
- Vejan, P.; Khadiran, T.; Abdullah, R.; Ahmad, N. Controlled release fertilizer: A review on developments, applications and potential in agriculture. J. Control. Release 2021, 339, 321–334. [Google Scholar] [CrossRef] [PubMed]
- Yendluri, R. Nanoclays: A new avenue for drug delivery. EC Pharmacol. Toxicol. ECO 2019, 2, 20–22. [Google Scholar]
- Sunil, B.H.; Pushpalatha, M.; Basavaprasad, V.; Huvanna, T. Modified nano-clay formulation and their application. Int. J. Chem. Stud. 2018, 6, 705–710. [Google Scholar]
- Gupta, R.K.; Agarwal, S.; Roth, E. Nanoclay-Based Solid Sorbents for Carbon Dioxide Capture. U.S. Patent No. 10,507.453, 17 December 2019. [Google Scholar]
- Khajeh, M.; Ghaemi, A. Strontium hydroxide-modified nanoclay montmorillonite for CO2 capture: Response surface methodology and adsorption mechanism. Int. J. Environ. Anal. Chem. 2021, 103, 5311–5336. [Google Scholar] [CrossRef]
- Roth, E.A.; Agarwal, S.; Gupta, R.K. Nanoclay-based solid sorbents for CO2 capture. Energy Fuels 2013, 27, 4129–4136. [Google Scholar] [CrossRef]
- Rao; Pochan, J.M. Mechanics of polymer−clay nanocomposites. Macromolecules 2007, 40, 290–296. [Google Scholar] [CrossRef]
- Schöny, G.; Dietrich, F.; Fuchs, J.; Hofbauer, H. Experimental study of the adsorber performance in a multi-stage fluidized bed system for continuous CO2 capture by means of temperature swing adsorption. Fuel Process. Technol. 2018, 173, 103–111. [Google Scholar]
- Wibbelmann, M.; Schmutz, U.; Wright, J.; Udall, D.; Rayns, F.; Kneafsey, M.; Trenchard, L.; Bennett, J.; Turner, M.L. Mainstreaming Agroecology: Implications for Global Food and Farming Systems; Coventry University: Coventry, UK, 2013. [Google Scholar]
- Chaudhary, A.K.; Prakash, H.P.; Hitaishi, S.K.; Kumar, A. Plant breeding for drought resistance. In Modern Approaches in Crop Improvement; Singh, J., Nigam, R., Kumar, A., Kapoor, N., Tiwari, P., Eds.; Weser Books: Zittau, Germany, 2019; pp. 43–48. [Google Scholar]
- Damalas, C.A.; Eleftherohorinos, I.G. Pesticide exposure, safety issues, and risk assessment indicators. Int. J. Environ. Res. Public Health 2011, 8, 1402–1419. [Google Scholar] [CrossRef] [PubMed]
- Krug, H.F. Nanosafety research--are we on the right track? Angew. Chem. Int Ed Engl. 2014, 53, 12304–12319. [Google Scholar] [CrossRef] [PubMed]
- Iavicoli, I.; Leso, V.; Beezhold, D.H.; Shvedova, A.A. Nanotechnology in agriculture: Opportunities, toxicological implications, and occupational risks. Toxicol. Appl. Pharmacol. 2017, 329, 96–111. [Google Scholar] [CrossRef] [PubMed]
- Klaine, S.J.; Alvarez, P.J.; Batley, G.E.; Fernandes, T.F.; Handy, R.D.; Lyon, D.Y.; Mahendra, S.; McLaughlin, M.J.; Lead, J.R. Nanomaterials in the environment: Behavior, fate, bioavailability, and effects. Environ. Toxicol. Chem. Int. J. 2008, 27, 1825–1851. [Google Scholar] [CrossRef] [PubMed]
- Pérez, S.; la Farré, M.; Barceló, D. Analysis, behavior and ecotoxicity of carbon-based nanomaterials in the aquatic environment. TrAC Trends Anal. Chem. 2009, 28, 820–832. [Google Scholar] [CrossRef]
- Scown, T.M.; Santos, E.M.; Johnston, B.D.; Gaiser, B.; Baalousha, M.; Mitov, S.; Lead, J.R.; Stone, V.; Fernandes, T.F.; Jepson, M. Effects of aqueous exposure to silver nanoparticles of different sizes in rainbow trout. Toxicol. Sci. 2010, 115, 521–534. [Google Scholar] [CrossRef] [PubMed]
- Huang, S.; Wang, L.; Liu, L.; Hou, Y.; Li, L. Nanotechnology in agriculture, livestock, and aquaculture in China. A review. Agron. Sustain. Dev. 2015, 35, 369–400. [Google Scholar] [CrossRef]
- Kah, M. Nanopesticides and nanofertilizers: Emerging contaminants or opportunities for risk mitigation? Front. Chem. 2015, 3, 64. [Google Scholar] [CrossRef] [PubMed]
- Biglari Quchan Atigh, Z.; Heidari, A.; Karimi, A.; Pezhman, M.A.; Asgari Lajayer, B.; Lima, E.C. Purification and economic analysis of nanoclay from bentonite. Environ. Sci. Pollut. Res. 2021, 28, 13690–13696. [Google Scholar] [CrossRef]
- Kantesaria, N.; Sharma, S. Exfoliation and extraction of nanoclay from montmorillonite mineral rich bentonite soil. In Proceedings of the Advances in Computer Methods and Geomechanics: IACMAG Symposium 2019; Springer: Singapore, 2020; Volume 2, pp. 1–12. [Google Scholar] [CrossRef]
- Lakitan, B.; Hadi, B.; Herlinda, S.; Siaga, E.; Widuri, L.I.; Kartika, K.; Lindiana, L.; Yunindyawati, Y.; Meihana, M. Recognizing farmers’ practices and constraints for intensifying rice production at Riparian Wetlands in Indonesia. NJAS-Wagening. J. Life Sci. 2018, 85, 10–20. [Google Scholar] [CrossRef]
- Sen, J.; Prakash, P.; De, N. Nano-clay composite and phyto-nanotechnology: A new horizon to food security issue in Indian agriculture. J. Global Biosci. 2015, 4, 2187–2198. [Google Scholar]
- Perera, K.Y.; Hopkins, M.; Jaiswal, A.K.; Jaiswal, S. Nanoclays-containing bio-based packaging materials: Properties, applications, safety, and regulatory issues. J. Nanostructure Chem. 2023, 14, 71–93. [Google Scholar] [CrossRef] [PubMed]
- Liu, C. November 12. Liquid Nanoclay: Transforming Soil to Shape the Future of Farming. Earth.Org. Available online: https://earth.org/liquid-nanoclay/ (accessed on 4 September 2023).


| Clay | Nutrient/Fertilizer | Refs. |
|---|---|---|
| Layered double hydroxides | Zinc | [123] |
| Layered double hydroxides | Boron (Na2B4O7•10H2O) and Zinc (Zn(NO3)2•6H2O) | [111] |
| Montmorillonite | CuSO4 | [124] |
| Montmorillonite | NH4-N | [28] |
| Montmorillonite | Nitrate | [125] |
| Montmorillonite-hydroxyapatite nanohybrid | Urea | [126] |
| Phlogopite | K | [127] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Abd-Elsalam, K.A.; Mehmood, M.A.; Ashfaq, M.; Abdelkhalek, T.E.; Hassan, R.K.; Ravichandran, M. Liquid Nanoclay: Synthesis and Applications to Transform an Arid Desert into Fertile Land. Soil Syst. 2024, 8, 73. https://doi.org/10.3390/soilsystems8030073
Abd-Elsalam KA, Mehmood MA, Ashfaq M, Abdelkhalek TE, Hassan RK, Ravichandran M. Liquid Nanoclay: Synthesis and Applications to Transform an Arid Desert into Fertile Land. Soil Systems. 2024; 8(3):73. https://doi.org/10.3390/soilsystems8030073
Chicago/Turabian StyleAbd-Elsalam, Kamel A., Mirza Abid Mehmood, Muhammad Ashfaq, Toka E. Abdelkhalek, Rawan K. Hassan, and Mythili Ravichandran. 2024. "Liquid Nanoclay: Synthesis and Applications to Transform an Arid Desert into Fertile Land" Soil Systems 8, no. 3: 73. https://doi.org/10.3390/soilsystems8030073
APA StyleAbd-Elsalam, K. A., Mehmood, M. A., Ashfaq, M., Abdelkhalek, T. E., Hassan, R. K., & Ravichandran, M. (2024). Liquid Nanoclay: Synthesis and Applications to Transform an Arid Desert into Fertile Land. Soil Systems, 8(3), 73. https://doi.org/10.3390/soilsystems8030073








