Biotic Interactions in Experimental Antarctic Soil Microcosms Vary with Abiotic Stress
Abstract
1. Introduction
2. Materials and Methods
3. Results
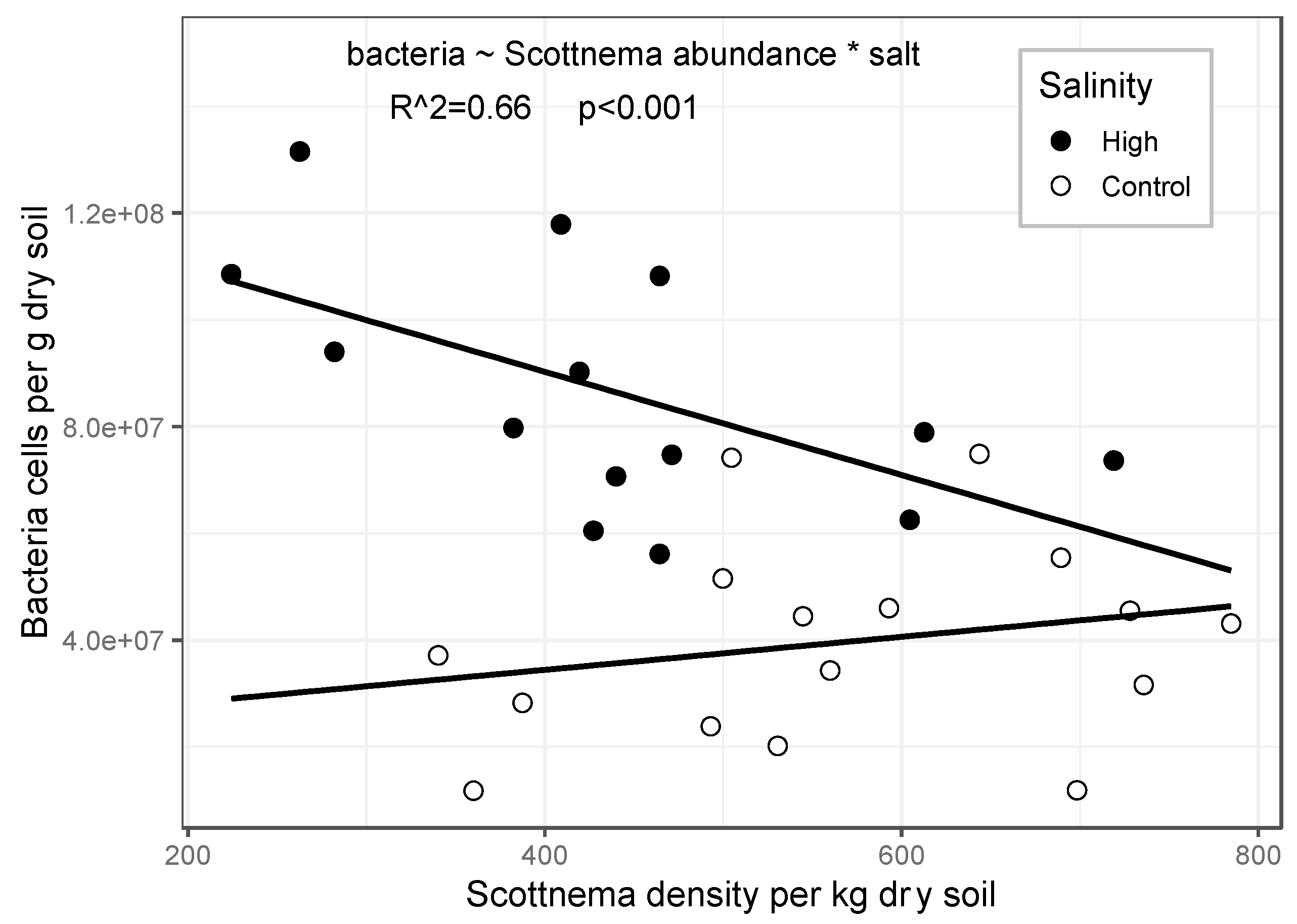
3.1. Effect of Treatments on Bacteria
3.2. Effect of Treatments on Nematodes
3.3. Biotic Interactions
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Maestre, F.T.; Bowker, M.A.; Escolar, C.; Puche, M.D.; Soliveres, S.; Maltez-Mouro, S.; Garcia-Palacios, P.; Castillo-Monroy, A.P.; Martínez, I.; Escudero, A. Do biotic interactions modulate ecosystem functioning along stress gradients? Insights from semi-arid plant and biological soil crust communities. Philos. Trans. R. Soc. B Biol. Sci. 2010, 365, 2057–2070. [Google Scholar] [CrossRef] [PubMed]
- Kolar, C.S.; Rahel, F.J. Interaction of a biotic factor (predator presence) and an abiotic factor (low oxygen) as an influence on benthic invertebrate communities. Oecologia 1993, 95, 210–219. [Google Scholar] [CrossRef] [PubMed]
- Heikkinen, R.K.; Luoto, M.; Virkkala, R.; Pearson, R.G.; Körber, J.H. Biotic interactions improve prediction of boreal bird distributions at macro-scales. Glob. Ecol. Biogeogr. 2007, 16, 754–763. [Google Scholar] [CrossRef]
- Coleman, D.C.; Anderson, R.V.; Cole, C.V.; Elliott, E.T.; Woods, L.; Campion, M.K. Trophic interactions in soils as they affect energy and nutrient dynamics. IV. Flows of metabolic and biomass carbon. Microb. Ecol. 1977, 4, 373–380. [Google Scholar] [CrossRef] [PubMed]
- Wall, D.H.; Moore, J.C. Interactions underground: Soil biodiversity, mutualism, and ecosystem processes. BioScience 1999, 49, 109–117. [Google Scholar] [CrossRef]
- Fountain, A.G.; Lyons, W.B.; Burkins, M.B.; Dana, G.L.; Doran, P.T.; Lewis, K.J.; McKnight, D.M.; Moorhead, D.L.; Parsons, A.N.; Priscu, J.C.; et al. Physical Controls on the Taylor Valley Ecosystem, Antarctica. Bioscience 1999, 49, 961–971. [Google Scholar] [CrossRef]
- Freckman, D.W.; Virginia, R.A. Low-diversity Antarctic soil nematode communities: Distribution and response to disturbance. Ecology 1997, 78, 363–369. [Google Scholar] [CrossRef]
- Treonis, A.M.; Wall, D.H.; Virginia, R.A. Invertebrate Biodiversity in Antarctic Dry Valley Soils and Sediments. Ecosystems 1999, 2, 482–492. [Google Scholar] [CrossRef]
- Wall, D.H. Global change tipping points: Above- and below-ground biotic interactions in a low diversity ecosystem. Philos. Trans. R. Soc. B Biol. Sci. 2007, 362, 2291–2306. [Google Scholar] [CrossRef]
- Adams, B.J.; Wall, D.H.; Virginia, R.A.; Broos, E.; Knox, M.A. Ecological Biogeography of the Terrestrial Nematodes of Victoria Land, Antarctica. ZooKeys 2014, 419, 29–71. [Google Scholar] [CrossRef]
- Ayres, E.; Wall, D.H.; Adams, B.J.; Barrett, J.E.; Virginia, R.A. Unique similarity of faunal communities across aquatic–terrestrial interfaces in a polar desert ecosystem: Soil–sediment boundaries and faunal community. Ecosystems 2007, 10, 523–535. [Google Scholar] [CrossRef]
- Wall, D.H.; Virginia, R.A. Controls on soil biodiversity: Insights from extreme environments. Appl. Soil Ecol. 1999, 13, 137–150. [Google Scholar] [CrossRef]
- Courtright, E.M.; Wall, D.H.; Virginia, R.A. Determining habitat suitability for soil invertebrates in an extreme environment: The McMurdo Dry Valleys, Antarctica. Antarct. Sci. 2001, 13, 9–17. [Google Scholar] [CrossRef]
- Poage, M.A.; Barrett, J.E.; Virginia, R.A.; Wall, D.H. The Influence of Soil Geochemistry on Nematode Distribution, Mcmurdo Dry Valleys, Antarctica. Arct. Antarct. Alp. Res. 2008, 40, 119–128. [Google Scholar] [CrossRef]
- Fountain, A.G.; Levy, J.S.; Gooseff, M.N.; Van Horn, D. The McMurdo Dry Valleys: A landscape on the threshold of change. Geomorphology 2014, 225, 25–35. [Google Scholar] [CrossRef]
- Fountain, A.G.; Saba, G.; Adams, B.; Doran, P.; Fraser, W.; Gooseff, M.; Obryk, M.; Priscu, J.C.; Stammerjohn, S.; Virginia, R.A. the Impact of a Large-Scale Climate Event on Antarctic Ecosystem Processes. Bioscience 2016, 66, 848–863. [Google Scholar] [CrossRef]
- Ball, B.A.; Barrett, J.; Gooseff, M.N.; Virginia, R.A.; Wall, D.H.; Becky, A. Implications of meltwater pulse events for soil biology and biogeochemical cycling in a polar desert. Polar Res. 2011, 30, 14555. [Google Scholar] [CrossRef]
- Ball, B.A.; Levy, J. The role of water tracks in altering biotic and abiotic soil properties and processes in a polar desert in Antarctica. J. Geophys. Res. Biogeosci. 2015, 120, 270–279. [Google Scholar] [CrossRef]
- Ball, B.A.; Virginia, R.A. Meltwater seep patches increase heterogeneity of soil geochemistry and therefore habitat suitability. Geoderma 2012, 189, 652–660. [Google Scholar] [CrossRef]
- Gooseff, M.N.; Wlostowski, A.; McKnight, D.M.; Jaros, C. Hydrologic connectivity and implications for ecosystem processes—Lessons from naked watersheds. Geomorphology 2017, 277, 63–71. [Google Scholar] [CrossRef]
- Barrett, J.; Virginia, R.; Wall, D.; Cary, S.; Adams, B.; Hacker, A.; Aislabie, J. Co-variation in soil biodiversity and biogeochemistry in northern and southern Victoria Land, Antarctica. Antarct. Sci. 2006, 18, 535–548. [Google Scholar] [CrossRef]
- Nielsen, U.N.; Wall, D.H.; Adams, B.J.; Virginia, R.A.; Ball, B.A.; Gooseff, M.N.; McKnight, D.M. The ecology of pulse events: Insights from an extreme climatic event in a polar desert ecosystem. Ecosphere 2012, 3, 1–15. [Google Scholar] [CrossRef]
- Andriuzzi, W.S.; Adams, B.J.; Barrett, J.E.; Virginia, R.A.; Wall, D.H. Observed trends of soil fauna in the Antarctic Dry Valleys: Early signs of shifts predicted under climate change. Ecology 2018, 99, 312–321. [Google Scholar] [CrossRef]
- Gooseff, M.N.; Barrett, J.E.; Adams, B.J.; Doran, P.T.; Fountain, A.G.; Lyons, W.B.; McKnight, D.M.; Priscu, J.C.; Sokol, E.R.; Takacs-Vesbach, C.; et al. Decadal ecosystem response to an anomalous melt season in a polar desert in Antarctica. Nat. Ecol. Evol. 2017, 1, 1334–1338. [Google Scholar] [CrossRef]
- Wynn-Williams, D.D. Cyanobacteria in Deserts. In The Ecology of Cyanobacteria; Whitton, B.A., Potts, M., Eds.; Kluwer Academic Publishers: Dordrecht, The Netherlands, 2000; pp. 341–366. [Google Scholar]
- Nkem, J.N.; Virginia, R.A.; Barrett, J.E.; Wall, D.H.; Li, G. Salt tolerance and survival thresholds for two species of Antarctic soil nematodes. Polar Biol. 2006, 29, 643–651. [Google Scholar] [CrossRef]
- Treonis, A.M.; Wall, D.H. Soil Nematodes and Desiccation Survival in the Extreme Arid Environment of the Antarctic Dry Valleys. Integr. Comp. Biol. 2005, 45, 741–750. [Google Scholar] [CrossRef]
- Crowe, J.H.; Madin, K.A.C. Anhydrobiosis in nematodes: Evaporative water loss and survival. J. Exp. Zool. 1975, 193, 323–333. [Google Scholar] [CrossRef]
- Freckman, D.W.; Womersley, C. Physiological Adaptations of Nematodes in Chihuahuan Desert Soil. In New Trends in Soil Biology; Lebrun, P., André, H.M., de Medts, A., Grégoire-Wibo, C., Wathy, G.., Eds.; Dieu-Brichard Publishers: Louvain-LaNeuve, Belgium, 1983; pp. 396–404. [Google Scholar]
- Van Horn, D.J.; Okie, J.G.; Buelow, H.N.; Gooseff, M.N.; Barrett, J.E.; Takacs-Vesbach, C.D. Soil Microbial Responses to Increased Moisture and Organic Resources along a Salinity Gradient in a Polar Desert. Appl. Environ. Microbiol. 2014, 80, 3034–3043. [Google Scholar] [CrossRef]
- Lozupone, C.A.; Knight, R. Global patterns in bacterial diversity. Proc. Natl. Acad. Sci. USA 2007, 104, 11436–11440. [Google Scholar] [CrossRef]
- Wang, J.; Yang, D.; Zhang, Y.; Shen, J.; Van Der Gast, C.; Hahn, M.W.; Wu, Q. Do Patterns of Bacterial Diversity along Salinity Gradients Differ from Those Observed for Macroorganisms? PLoS ONE 2011, 6, e27597. [Google Scholar] [CrossRef]
- Zahran, H.H. Diversity, adaptation and activity of the bacterial flora in saline environments. Biol. Fertil. Soils 1997, 25, 211–223. [Google Scholar] [CrossRef]
- Hogg, I.D.; Cary, S.C.; Convey, P.; Newsham, K.K.; O’Donnell, A.G.; Adams, B.J.; Aislabie, J.; Frati, F.; Stevens, M.I.; Wall, D.H. Biotic interactions in Antarctic terrestrial ecosystems: Are they a factor? Soil Biol. Biochem. 2006, 38, 3035–3040. [Google Scholar] [CrossRef]
- Shaw, E.A.; Adams, B.J.; Barrett, J.E.; Lyons, W.B.; Virginia, R.A.; Wall, D.H. Stable C and N isotope ratios reveal soil food web structure and identify the nematode Eudorylaimus antarcticus as an omnivore–predator in Taylor Valley, Antarctica. Polar Biol. 2018, 41, 1013–1018. [Google Scholar] [CrossRef]
- Magalhães, C.M.; Machado, A.; Frank-Fahle, B.; Lee, C.K.; Cary, S.C. The ecological dichotomy of ammonia-oxidizing archaea and bacteria in the hyper-arid soils of the Antarctic Dry Valleys. Front. Microbiol. 2014, 5, 515. [Google Scholar] [CrossRef]
- Arenz, B.; Blanchette, R. Distribution and abundance of soil fungi in Antarctica at sites on the Peninsula, Ross Sea Region and McMurdo Dry Valleys. Soil Biol. Biochem. 2011, 43, 308–315. [Google Scholar] [CrossRef]
- Powers, L.E.; Ho, M.; Freckman, D.W.; Virginia, R.A. Distribution, Community Structure, and Microhabitats of Soil Invertebrates along an Elevational Gradient in Taylor Valley, Antarctica. Arct. Alp. Res. 1998, 30, 133–141. [Google Scholar] [CrossRef]
- Takacs-Vesbach, C.; Zeglin, L.; Barrett, J.; Gooseff, M.N.; Priscu, J.C. Factors Promoting Microbial Diversity in the McMurdo Dry Valleys, Antarctica. In Life in Antarctic Deserts and Other Cold Dry Environments; Cambridge University Press (CUP): Cambridge, UK, 2010; pp. 221–257. [Google Scholar]
- Lee, C.K.; Barbier, B.A.; Bottos, E.M.; McDonald, I.R.; Cary, S.C. The Inter-Valley Soil Comparative Survey: The ecology of Dry Valley edaphic microbial communities. ISME J. 2011, 6, 1046–1057. [Google Scholar] [CrossRef]
- Okie, J.G.; Van Horn, D.J.; Storch, D.; Barrett, J.E.; Gooseff, M.N.; Kopsova, L.; Takacs-Vesbach, C.D. Niche and metabolic principles explain patterns of diversity and distribution: Theory and a case study with soil bacterial communities. Proc. R. Soc. B Biol. Sci. 2015, 282, 20142630. [Google Scholar] [CrossRef]
- Altrichter, A.E.; Takacs-Vesbach, C.D.; Van Horn, D.J.; Barrett, J.E.; Geyer, K.M.; Gooseff, M.N. Bacterial community composition of divergent soil habitats in a polar desert. FEMS Microbiol. Ecol. 2014, 89, 490–494. [Google Scholar]
- Nielsen, U.N.; Wall, D.H.; Adams, B.J.; Virginia, R.A. Antarctic nematode communities: Observed and predicted responses to climate change. Polar Biol. 2011, 34, 1701–1711. [Google Scholar] [CrossRef]
- Tamppari, L.; Anderson, R.; Archer, P.; Douglas, S.; Kounaves, S.; McKay, C.; Ming, D.; Moore, Q.; Quinn, J.; Smith, P.; et al. Effects of extreme cold and aridity on soils and habitability: McMurdo Dry Valleys as an analogue for the Mars Phoenix landing site. Antarct. Sci. 2012, 24, 211–228. [Google Scholar] [CrossRef]
- Ball, B.A.; Adams, B.J.; Barrett, J.; Wall, D.H.; Virginia, R.A. Soil biological responses to C, N and P fertilization in a polar desert of Antarctica. Soil Biol. Biochem. 2018, 122, 7–18. [Google Scholar] [CrossRef]
- Zeglin, L.H.; Sinsabaugh, R.L.; Barrett, J.E.; Gooseff, M.N.; Takacs-Vesbach, C.D. Landscape Distribution of Microbial Activity in the McMurdo Dry Valleys: Linked Biotic Processes, Hydrology, and Geochemistry in a Cold Desert Ecosystem. Ecosystems 2009, 12, 562–573. [Google Scholar] [CrossRef]
- Smith, T.; Wall, D.; Hogg, I.D.; Adams, B.; Nielsen, U.N.; Virginia, R. Thawing permafrost alters nematode populations and soil habitat characteristics in an Antarctic polar desert ecosystem. Pedobiologia 2012, 55, 75–81. [Google Scholar] [CrossRef]
- Franco, A.L.; Knox, M.A.; Andriuzzi, W.S.; De Tomasel, C.M.; Sala, O.E.; Wall, D.H. Nematode exclusion and recovery in experimental soil microcosms. Soil Biol. Biochem. 2017, 108, 78–83. [Google Scholar] [CrossRef]
- Bouwman, L.A.; Bloem, J.; Van den Boogert, P.H.; Bremer, F.; Hoenderboom, G.H.; De Ruiter, P.C. Short-term and long-term effects of bacterivorous nematodes and nematophagous fungi on carbon and nitrogen mineralization in microcosms. Biol. Fertil. Soils 1994, 17, 249–256. [Google Scholar] [CrossRef]
- Freckman, D.; Virginia, R. Extraction of nematodes from Dry Valley Antarctic soils. Polar Biol. 1993, 13, 483–487. [Google Scholar] [CrossRef]
- Frey, S.; Elliott, E.; Paustian, K. Bacterial and fungal abundance and biomass in conventional and no-tillage agroecosystems along two climatic gradients. Soil Biol. Biochem. 1999, 31, 573–585. [Google Scholar] [CrossRef]
- Sistla, S.A.; Moore, J.C.; Simpson, R.T.; Gough, L.; Shaver, G.R.; Schimel, J.P. Long-term warming restructures Arctic tundra without changing net soil carbon storage. Nature 2013, 497, 615–618. [Google Scholar] [CrossRef]
- Shaw, E.A.; Wall, D.H. MDV Lab Microcosm Data; Mendeley Data; Elsevier: Amsterdam, The Netherlands, 2018. [Google Scholar]
- Overhoff, A.; Freckman, D.W.; Virginia, R.A. Life cycle of the microbivorous Antarctic Dry Valley nematode Scottnema lindsayae (Timm 1971). Polar Biol. 1993, 13, 151–156. [Google Scholar] [CrossRef]
- Porazinska, D.L.; Wall, D.H.; Virginia, R.A. Population Age Structure of Nematodes in the Antarctic Dry Valleys: Perspectives on Time, Space, and Habitat Suitability. Arctic, Antarct. Alp. Res. 2002, 34, 159–168. [Google Scholar] [CrossRef]
- Knox, M.A.; Wall, D.H.; Virginia, R.A.; Vandegehuchte, M.L.; Gil, I.S.; Adams, B.J. Impact of diurnal freeze–thaw cycles on the soil nematode Scottnema lindsayae in Taylor Valley, Antarctica. Polar Biol. 2015, 39, 583–592. [Google Scholar] [CrossRef]
- Ingham, R.E.; Trofymow, J.; Ingham, E.R.; Coleman, D.C. Interactions of bacteria, fungi, and their nematode grazers: Effects on nutrient cycling and plant growth. Ecol. Monogr. 1985, 55, 119–140. [Google Scholar] [CrossRef]
- Anderson, R.V.; Elliott, E.T.; McClellan, J.F.; Coleman, D.C.; Cole, C.V.; Hunt, H.W. Trophic interactions in soils as they affect energy and nutrient dynamics. III. Biotic interactions of bacteria, amoebae and nematodes. Microb. Ecol. 1978, 4, 361–371. [Google Scholar] [CrossRef]
- Burkins, M.B.; Virginia, R.A.; Chamberlain, C.P.; Wall, D.H. Origin and Distribution of Soil Organic Matter in Taylor Valley, Antarctica. Ecology 2000, 81, 2377–2391. [Google Scholar] [CrossRef]
- Knox, M.A.; Andriuzzi, W.S.; Buelow, H.N.; Takacs-Vesbach, C.; Wall, D.H.; Adams, B.J. Decoupled responses of soil bacteria and their invertebrate consumer to warming, but not freeze-thaw cycles, in the Antarctic Dry Valleys. Ecol. Lett. 2017, 20, 1242–1249. [Google Scholar] [CrossRef]


| Taxa | Effects | Citations |
|---|---|---|
| Populations | ||
| Microbes | -Gene expression of AOA or AOB changes in more saline, drier valleys 1 -Moisture was positively correlated to fungi abundance while salinity was negatively correlated 2 | 1 Magalhães, et al. [36] 2 Arenz and Blanchette [37] |
| Invertebrates | -Scottnema and Plectus are both negatively affected by salt, but type and concentration matter 3 -Populations of Scottnema, Eudorylaimus, and Plectus are negatively related to salinity 4 | 3 Nkem, Virginia, Barrett, Wall and Li [26] 4 Powers, et al. [38] |
| Communities | ||
| Microbes | -Composition shifts with elevated salinity from Actinobacteria to Firmicutes dominated 1,5 -Greater community diversity in drier soils 2 -Salinity drives community composition in 4 valleys 3 -Alpha diversity of communities declines with salinity 4 -Soil moisture is a significant predictor of bacterial community diversity at genus level 5 | 1 Van Horn, et al. [30] 2 Takacs-Vesbach, et al. [39] 3 Lee, et al. [40] 4 Okie, et al. [41] 5 Geyer, et al. [42] |
| Invertebrates | -Greater community diversity in less saline soils 6,7,8 -Greater community diversity in wetter soils 6,7,8 -Nematodes Plectus and Eudorylaimus are associated with wetter soils, Scottnema with drier 6,7,8,9 | 6 Nielsen, et al. [43] 7 Ayres, et al. [11] 8 Treonis, et al. [8] 9.Powers, et al. [38] |
| Ecosystem | ||
| Microbes | -Water tracks alter respiration rates, depending on soil chemistry 1 -Lower microbial biomass in saltier, drier valleys 2 -Moisture addition did not affect microbial biomass in field experiment 3 -Along with pH and organic C, salinity was a predictor of microbial activity in lake and stream margins 4 | 1 Ball and Virginia [19] 2 Tamppari, et al. [44] 3 Ball, et al. [45] 4 Zeglin, et al. [46] |
| Invertebrates | -Water tracks affect soil invertebrate habitats, via soil chemistry changes 5, and have lower invertebrate abundance, associated with higher salinity 6 -Salinity and moisture are drivers of habitat suitability for invertebrates, S. lindsayae found in saltier, drier soils than other nematodes 7 | 5 Ball and Virginia [19] 6 Smith, et al. [47] 7 Courtright, et al. [13] |
| Effect | d.f. | F | p | Effect | d.f. | F | p |
|---|---|---|---|---|---|---|---|
| Eudorylaimus total abundance | Scottnema total abundance | ||||||
| C | 1.32 | 19.89 | <0.0001 | C | 1.31 | 2.02 | 0.165 |
| M | 1.32 | 0.29 | 0.597 | M | 1.31 | 1.57 | 0.220 |
| S | 1.32 | 0.10 | 0.759 | S | 1.31 | 6.25 | 0.018 |
| C*M | 1.32 | 0.033 | 0.858 | C*M | 1.31 | 0.62 | 0.437 |
| M*S | 1.32 | 1.252 | 0.271 | M*S | 1.31 | 0.48 | 0.494 |
| C*S | 1.32 | 0.164 | 0.688 | C*S | 1.31 | 0.10 | 0.758 |
| C*M*S | 1.32 | 3.362 | 0.076 | C*M*S | 1.31 | 3.21 | 0.083 |
| Scottnema adults | Scottnema juveniles | ||||||
| C | 1.35 | 0.82 | 0.371 | C | 1.31 | 1.46 | 0.237 |
| M | 1.35 | 4.82 | 0.035 | M | 1.31 | 0.38 | 0.544 |
| S | 1.35 | 8.47 | 0.007 | S | 1.31 | 4.24 | 0.048 |
| C*M | 1.35 | 0.195 | 0.662 | C*M | 1.31 | 0.38 | 0.541 |
| M*S | 1.35 | 0.49 | 0.489 | M*S | 1.31 | 0.039 | 0.844 |
| C*S | 1.35 | 0.11 | 0.73 | C*S | 1.31 | 0.226 | 0.638 |
| C*M*S | 1.35 | 5.75 | 0.023 | C*M*S | 1.31 | 1.675 | 0.205 |
| Scottnema females | Scottnema males | ||||||
| C | 1.31 | 3.60 | 0.067 | C | 1.31 | 0.35 | 0.5574 |
| M | 1.31 | 2.01 | 0.167 | M | 1.31 | 3.239 | 0.0817 |
| S | 1.31 | 9.81 | 0.004 | S | 1.31 | 2.023 | 0.1649 |
| C*M | 1.31 | 0.72 | 0.403 | C*M | 1.31 | 0.448 | 0.5085 |
| M*S | 1.31 | 1.49 | 0.232 | M*S | 1.31 | 0.677 | 0.4169 |
| C*S | 1.31 | 0.37 | 0.548 | C*S | 1.31 | 0.599 | 0.4450 |
| C*M*S | 1.31 | 3.38 | 0.076 | C*M*S | 1.31 | 3.436 | 0.0733 |
| Bacteria cells | |||||||
| C | 3.53 | 7.34 | 0.0003 | ||||
| M | 1.53 | 0.35 | 0.557 | ||||
| S | 1.53 | 3.51 | 0.067 | ||||
| C*M | 3.53 | 0.75 | 0.527 | ||||
| M*S | 1.53 | 0.03 | 0.870 | ||||
| C*S | 3.53 | 16.41 | <0.0001 | ||||
| C*M*S | 3.53 | 0.68 | 0.568 | ||||
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Shaw, E.A.; Wall, D.H. Biotic Interactions in Experimental Antarctic Soil Microcosms Vary with Abiotic Stress. Soil Syst. 2019, 3, 57. https://doi.org/10.3390/soilsystems3030057
Shaw EA, Wall DH. Biotic Interactions in Experimental Antarctic Soil Microcosms Vary with Abiotic Stress. Soil Systems. 2019; 3(3):57. https://doi.org/10.3390/soilsystems3030057
Chicago/Turabian StyleShaw, E. Ashley, and Diana H. Wall. 2019. "Biotic Interactions in Experimental Antarctic Soil Microcosms Vary with Abiotic Stress" Soil Systems 3, no. 3: 57. https://doi.org/10.3390/soilsystems3030057
APA StyleShaw, E. A., & Wall, D. H. (2019). Biotic Interactions in Experimental Antarctic Soil Microcosms Vary with Abiotic Stress. Soil Systems, 3(3), 57. https://doi.org/10.3390/soilsystems3030057





