Pyrolysis Temperature Effects on Biochar–Water Interactions and Application for Improved Water Holding Capacity in Vineyard Soils
Abstract
1. Introduction
2. Materials and Methods
2.1. Sample Collection
2.2. Feedstock Preparation
2.3. Thermo-Gravimetric Analysis
2.4. Biochar Production
2.5. Characterization of Feedstocks and Biochars
2.6. Hydrostatic Water Retention
3. Results and Discussion
3.1. Thermo-Gravimetric Analysis
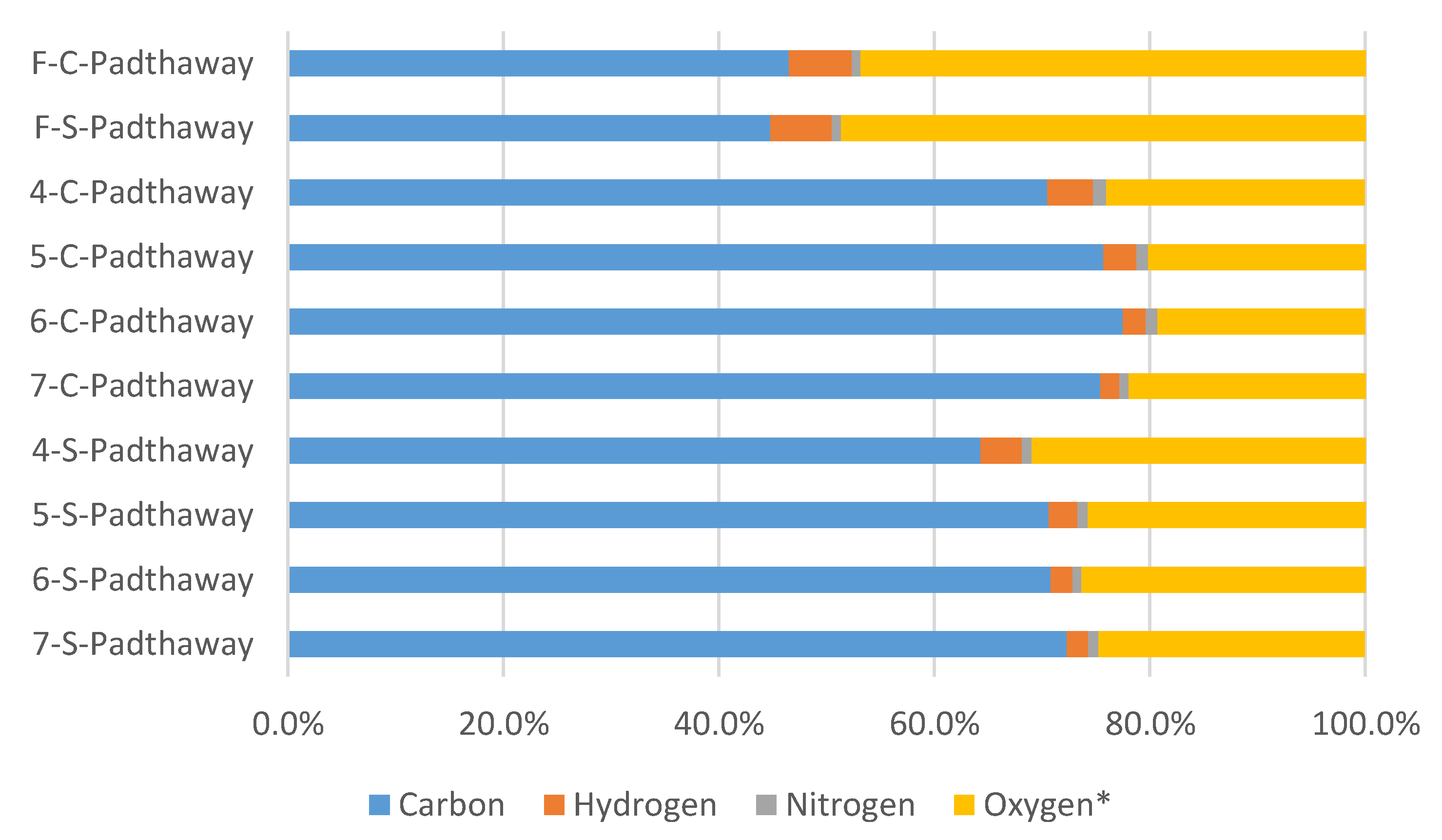
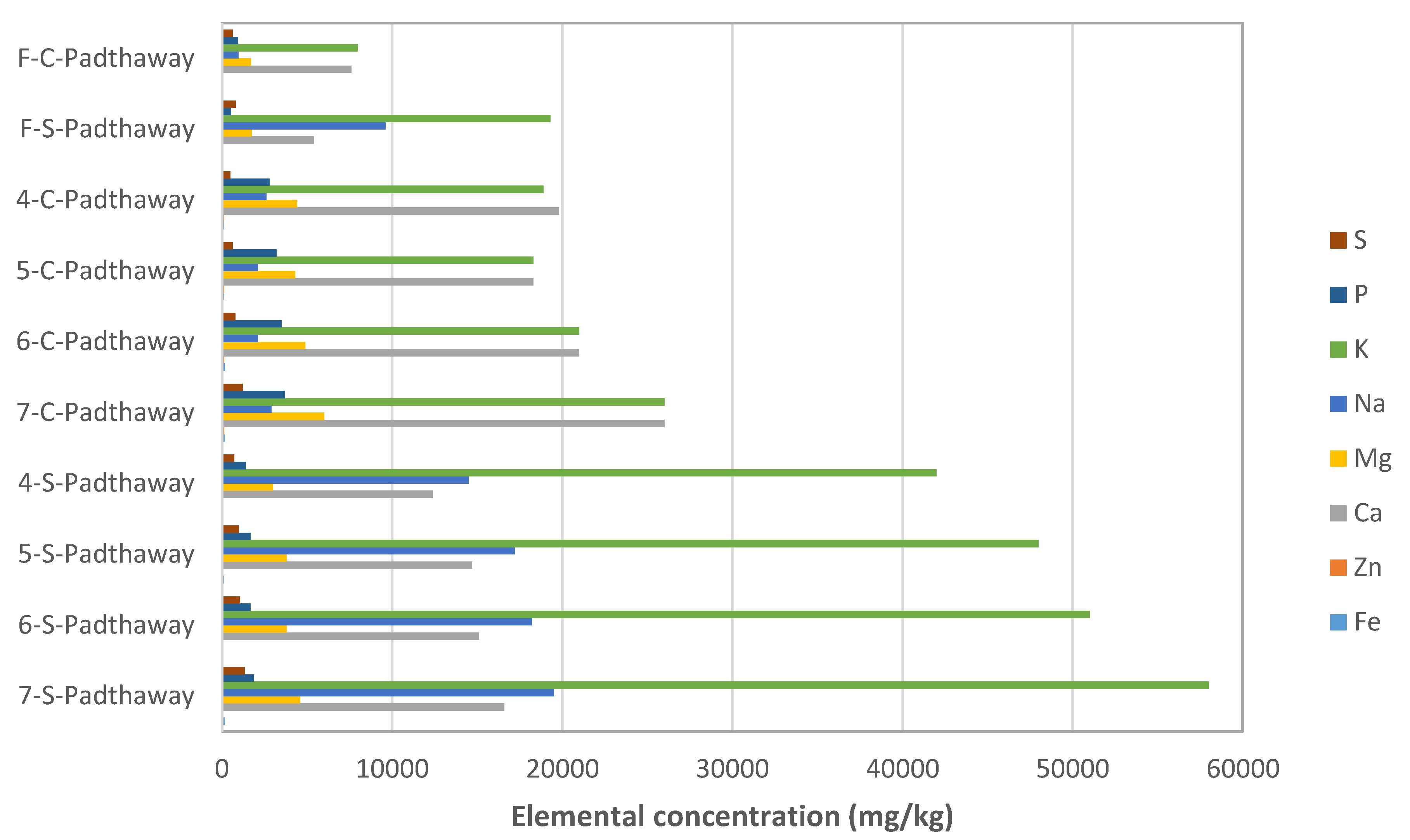
3.2. Biochar Physico-Chemical Characterisation
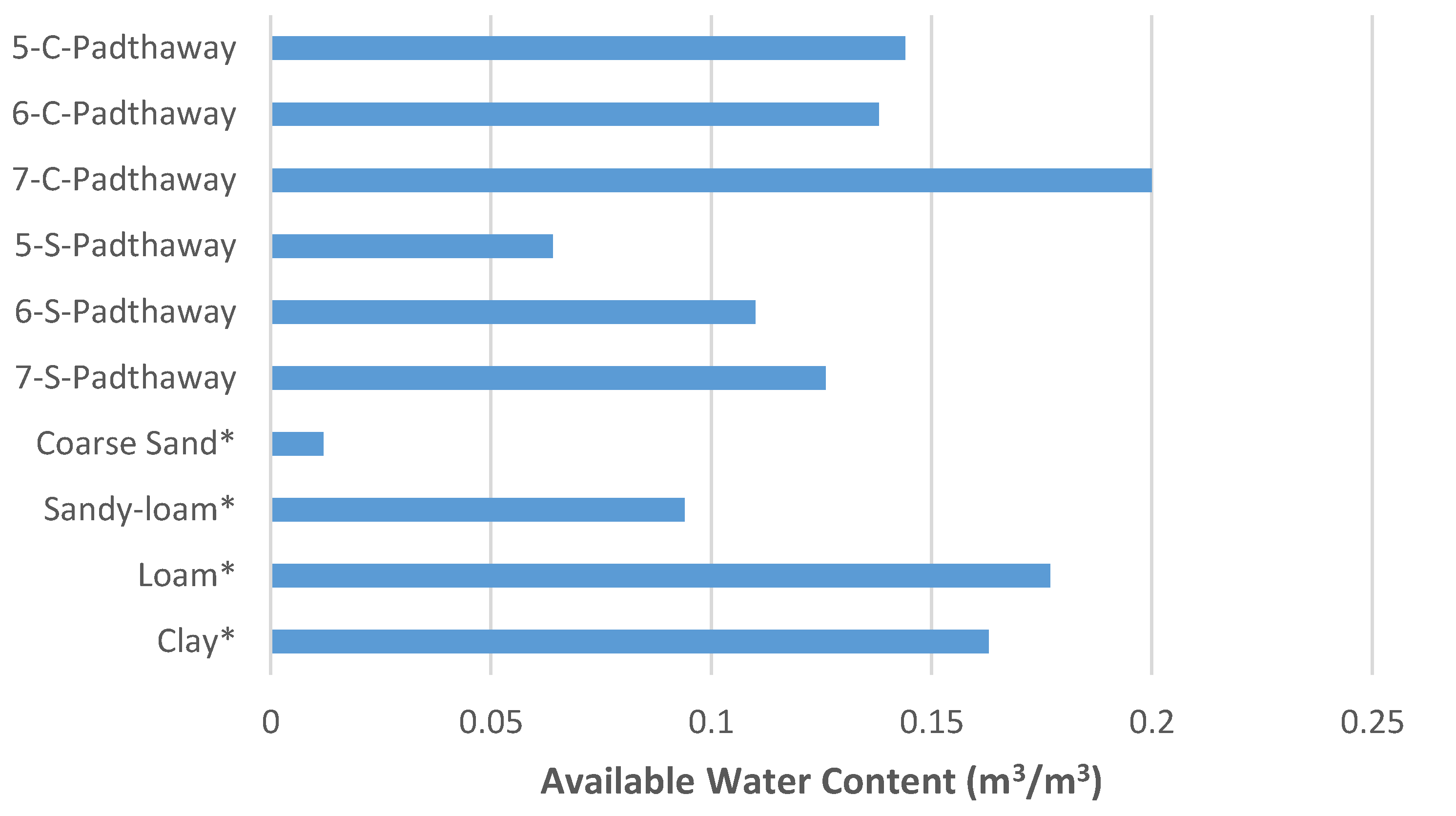
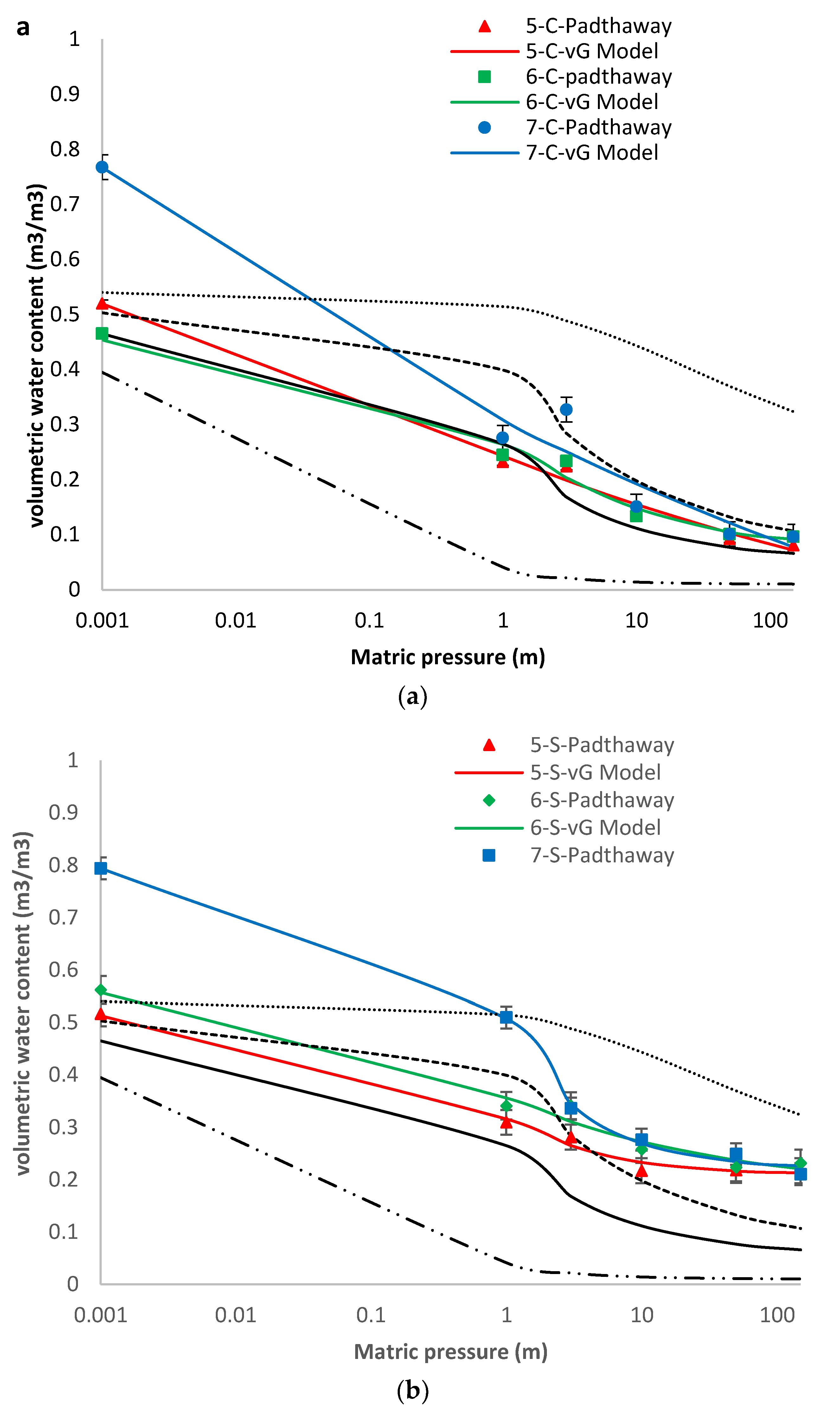
3.3. Hydrostatic Water Retention
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Masulili, A.; Utomo, W.; Syechfani, M. Rice husk biochar for rice based cropping system in acid soil 1. The characteristics of rice husk biochar and its influence on the properties of acid sulphate soils and rice growth in West Kalimantan, Indonesia. J. Agric. Sci. 2010, 2, 39–47. [Google Scholar] [CrossRef]
- Abel, S.; Peters, A.; Trinks, S.; Schonsky, H.; Facklam, M.; Wessolek, G. Impact of biochar and hydrochar addition on water retention and water repellency of sandy soils. Geoderma 2013, 202, 183–191. [Google Scholar] [CrossRef]
- Basso, A.; Miguez, F.; Laird, D.; Horton, R.; Westgate, M. Assessing potential of biochar for increasing water-holding capacity of sandy soils. Gcb Bioenergy 2013, 5, 132–143. [Google Scholar] [CrossRef]
- Sun, F.; Lu, S. Biochars improve aggregate stability, water retention, and pore-space properties of clayey soil. J. Plant Nutr. Soil Sci. 2014, 177, 26–33. [Google Scholar] [CrossRef]
- Blanco-Canqui, H. Biochar and soil physical properties. Soil Sci. Soc. Am. J. 2017, 81, 687–711. [Google Scholar] [CrossRef]
- Jeffery, S.; Meinders, M.; Stoof, C.; Bezemer, M.; van de Voorde, T.; Mommer, L.; van Groenigen., J. Biochar application does not improve the soil hydrological function of a sandy soil. Geoderma 2015, 251, 47–54. [Google Scholar] [CrossRef]
- Hardie, M.; Clotherier, B.; Bound, S.; Oliver, G.; Close, D. Does biochar influence soil physical properties and soil water availability? Plant Soil 2014, 376, 347–361. [Google Scholar] [CrossRef]
- Major, J.; Rondon, M.; Molina, D.; Riha, S.; Lehmann, J. Nutrient leaching in a Colombian savanna oxisol amended with biochar. J. Environ. Qual. 2012, 41, 1076–1086. [Google Scholar] [CrossRef]
- Kinney, T.; Masiello, C.; Dugan, B.; Hockaday, W.; Dean, M.; Zygourakis, K.; Barnes, R. Hydrologic properties of biochars produced at different temperatures. Biomass Bioenergy 2012, 41, 34–43. [Google Scholar] [CrossRef]
- Das, O.; Sarmah, A. The love-hate relationship of pyrolysis biochar and water: A perspective. Sci. Total Environ. 2015, 512, 682–685. [Google Scholar] [CrossRef] [PubMed]
- Dunnigan, L.; Ashman, P.; Zhang, X.; Kwong, C. Production of biochar from rice husk: Particulate emissions from the combustion of raw pyrolysis volatiles. J. Clean. Prod. 2018, 172, 1639–1645. [Google Scholar] [CrossRef]
- Gray, M.; Johnson, M.; Dragila, M.; Kleber, M. Water uptake in biochars: The roles of porosity and hydrophobicity. Plant Soil 2014, 61, 196–205. [Google Scholar] [CrossRef]
- Muhlack, R.; Portumathi, R.; Jeffery, D. Sustainable wineries through waste valorisation: A review of grape marc utilisation for value-added products. Waste Manag. 2018, 72, 99–118. [Google Scholar] [CrossRef]
- Smart, R.; Coombe, B. Water relations of grapevines. In Water Deficits and Plant Growth; Kozlowzki, T.T., Ed.; Academic Press: New York, NY, USA, 1983; Volume VII, pp. 137–196. [Google Scholar]
- McCarthy, M.; Jones, L.; Due, G. Irrigation—Principals and Practices. In Viticulture Practices; Coombe, B.G., Dry, P.R., Eds.; Winetitles: Adelaide, Australia, 1992; Volume 2, pp. 104–128. ISBN 1875130012. [Google Scholar]
- Medrano, H.; Tomas, M.; Martorell, S.; Escalona, J.; Pou, A.; Fuentes, S.; Flexas, J.; Bota, J. Improving water use efficiency of vineyards in semi-arid regions. A review. Agron. Sustain. Dev. 2015, 35, 499–517. [Google Scholar] [CrossRef]
- Roberts, K.; Gloy, B.; Joseph, S.; Scott, N.; Lehmann, J. Life cycle assessment of biochar systems: Estimating the energetic, economic and climate change potential. Environ. Sci. Technol. 2010, 44, 827–833. [Google Scholar] [CrossRef]
- Wheal, M.; Fowles, T.; Palmer, L. A cost-effective acid digestion method using closed polypropylene tubes for inductively coupled plasma optical emission spectrometry (ICP-OES) analysis of plant essential elements. Anal. Methods 2011, 3, 2854–2863. [Google Scholar] [CrossRef]
- Lastoskie, C.; Gubbins, K.; Quirke, N. Pore size distribution analysis of microporous carbons: A density functional theory approach. J. Phys. Chem. 1993, 97, 4786–4796. [Google Scholar] [CrossRef]
- Kameyama, K.; Miyamoto, T.; Shiono, T.; Shinogi, T. Influence of Sugarcane Bagasse-derived Biochar Application on Nitrate Leachin in Calcaric Dark Red Soil. J. Environ. Qual. 2012, 41, 1131–1137. [Google Scholar] [CrossRef]
- Cresswell, H.; Green, T.; McKenzie, N. The Adequacy of Pressure Plate Apparatus for Determining Soil Water Retention. Soil Sci. Soc. Am. J. 2008, 72, 41–49. [Google Scholar] [CrossRef]
- Bittelli, M.; Flury, M. Errors in Water Retention Curves Determined with Pressure Plates. Soil Sci. Soc. Am. J. 2009, 73, 1453–1460. [Google Scholar] [CrossRef]
- Van Genuchten, M.T. A closed-form equation for predicting the hydraulic conductivity of unsaturated soils. Soil Sci. Soc. Am. J. 1980, 44, 892–898. [Google Scholar] [CrossRef]
- Grant, C.; Groenevelt, P.; Robinson, N. Application of the Groenevelt-Grant soil water retention model to predict the hydraulic conductivity. Aust. J. Soil Res. 2010, 48, 447–458. [Google Scholar] [CrossRef]
- Dunnigan, L.; Morton, B.J.; Hall, P.A.; Kwong, C.W. Production of biochar and bioenergy from rice husk: Influence of feedstock drying on particulate matter and the associated polycyclic aromatic hydrocarbon emissions. Atmos. Environ. 2018, 190, 218–225. [Google Scholar] [CrossRef]
- Atkinson, C.; Fitzgerald, J.; Hipps, N. Potential mechanisms for achieving agricultural benefits from biochar application to temperate soils: A review. Plant Soil 2010, 337, 1–18. [Google Scholar] [CrossRef]
- Yao, Y.; Gao, B.; Inyang, M.; Zimmerman, A.; Cao, X.; Prullammanappallil, P.; Yang, L. Biochar derived from anaerobically digested sugar beet tailings: Characterization and phosphate removal potential. Bioresour. Technol. 2011, 102, 6273–6278. [Google Scholar] [CrossRef]
- Jung, K.W.; Hwang, M.J.; Ahn, K.H.; Ok, Y.S. Kinetic study on phosphate removal from aqueous solution by biochar derived from peanut shell as renewable adsorptive media. Int. J. Environ. Sci. Technol. 2015, 12, 3363–3372. [Google Scholar] [CrossRef]
- Cheng, C.; Lehmann, J.; Engelhard, M. Natural oxidation of black carbon in soils: Changes in molecular form and surface charge along a climosequence. Geochim. Cosmochim. Acta 2008, 72, 1598–1610. [Google Scholar] [CrossRef]
- Pattiya, A. Bio-oil production via fast pyrolysis of biomass residues from cassava plants in a fluidised-bed reactor. Bioresour. Technol. 2011, 102, 1959–1967. [Google Scholar] [CrossRef]
- Kasozi, G.N.; Zimmerman, A.R.; Nkedi-Kizza, P.; Gao, B. Catechol and humic acid sorption onto a range of laboratory-produced black carbons (biochars). Environ. Sci. Technol. 2010, 44, 6189–6195. [Google Scholar] [CrossRef]
- Marshall, J.A.; Morton, B.J.; Muhlack, R.; Chittleborough, D.; Kwong, C.W. Recovery of phosphate from calcium-containing aqueous solution resulting from biochar-induced calcium phosphate precipitation. J. Clean. Prod. 2017, 165, 27–35. [Google Scholar] [CrossRef]
- Keiluweit, M.; Nico, P.; Johnson, M.; Kleber, M. Dynamic molecular structure of plant biomass-derived black carbon (biochar). Environ. Sci. Technol. 2010, 44, 1247–1253. [Google Scholar] [CrossRef]
- Chen, B.; Zhou, D.; Zhu, L. Transitional adsorption and partition of nonpolar and polar aromatic contaminants by biochars of pine needles with different pyrolytic temperatures. Environ. Sci. Technol. 2008, 42, 5137–5143. [Google Scholar] [CrossRef]
- Raykov, T.; Marcoulides, G.A. An Introduction to Applied Multivariate Analysis; Routledge Taylor and Francis Group: New York, NY, USA, 2008; pp. 211–240. [Google Scholar]
- Manly, B.F.J. Multivariate Statisctical Methods: A Primer; Chapman and Hall/CRC: Boca Raton, FL, USA, 2005; pp. 75–90. [Google Scholar]
- Karhu, K.; Mattila, T.; Bergstrom, I.; Regina, K. Biochar addition to agricultural soil increased CH4 uptake and water holding capacity—Results from a short-term pilot field study. Agric. Ecosyst. Environ. 2011, 140, 309–313. [Google Scholar] [CrossRef]
- Novak, J.M.; Busscher, W.J.; Watts, D.W.; Amonette, J.E.; Ippolito, J.A.; Lima, I.M.; Gaskin, J.; Das, K.C.; Steiner, C.; Ahmedna, M.; et al. Biochars Impact on Soil-Moisture Storage in an Ultisol and Two Aridisols. Soil Sci. 2012, 177, 310–320. [Google Scholar] [CrossRef]
- Blackwell, P.; Krull, E.; Butler, G.; Herbert, A.; Solaiman, Z. Effect of banded biochar on dryland wheat production and fertiliser use in south-western Australia: An agronomic and economic perspective. Aust. J. Soil Res. 2010, 48, 531–545. [Google Scholar] [CrossRef]
Sample Availability: Not available. |


| Sample Identification | Zeta Potential (mV ± SE *) | pH (±SE *) | Surface Area (m2/g) | Pore Volume (mL/g) |
|---|---|---|---|---|
| 4-C-Padthaway | −33.4 ± 0.13 | 8.17 ± 0.066 | 183 | 0.053 |
| 5-C-Padthaway | −32.5 ± 0.34 | 10.03 ± 0.023 | 281 | 0.077 |
| 6-C-Padthaway | −33.2 ± 0.29 | 9.35 ± 0.006 | 399 | 0.106 |
| 7-C-Padthaway | −29.0 ± 0.44 | 9.82 ± 0.024 | 516 | 0.135 |
| 4-S-Padthaway | −41.2 ± 0.95 | 9.17 ± 0.143 | 200 | 0.057 |
| 5-S-Padthaway | −43.5 ± 1.02 | 10.31 ± 0.025 | 302 | 0.082 |
| 6-S-Padthaway | −41.1 ± 0.88 | 9.95 ± 0.031 | 445 | 0.114 |
| 7-S-Padthaway | −41.2 ± 0.58 | 10.69 ± 0.012 | 560 | 0.147 |
| Sample Identification | Van Genuchten Parameters | Groenevelt-Grant Model Parameters | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| θres | α | N | M | R2 | SSE | K0 | K1 | n | R2 | SSE | |
| 5-C-Padthaway | −0.240 | 178.832 | 2.324 | 0.038 | 0.992 | 1.1 × 10−3 | 0.140 | 0.510 | 0.298 | 0.982 | 2.3 × 10−3 |
| 6-C-Padthaway | 0.820 | 0.198 | 0.494 | 2.008 | 0.982 | 1.7 × 10−3 | 0.394 | 0.426 | 0.358 | 0.982 | 1.7 × 10−3 |
| 7-C-Padthaway | −0.485 | 768.4 | 10.263 | 0.007 | 0.971 | 9.4 × 10−3 | 0.213 | 0.673 | 0.521 | 0.960 | 1.3 × 10−2 |
| 5-S-Padthaway | 0.211 | 1.419 | 0.707 | 1.297 | 0.991 | 5.9 × 10−4 | 0.265 | 0.310 | 0.648 | 0.991 | 6.4 × 10−4 |
| 6-S-Padthaway | 0.177 | 10.982 | 0.750 | 0.394 | 0.980 | 1.6 × 10−3 | 0.232 | 0.387 | 0.325 | 0.980 | 1.6 × 10−3 |
| 7-S-Padthaway | 0.220 | 2.388 | 3.017 | 0.257 | 0.998 | 6.0 × 10−4 | 0.674 | 0.569 | 0.958 | 0.997 | 7.4 × 10−4 |
| Coarse Sand * | 0.010 | 16.746 | 1.897 | 0.470 | - | - | 0.074 | 0.386 | 0.921 | - | - |
| Sandy-loam * | 0.053 | 3.244 | 2.95 | 0.190 | - | - | 0.674 | 0.398 | 0.847 | - | - |
| Loam * | 0.061 | 1.782 | 2.542 | 0.160 | - | - | 1.57 | 0.413 | 0.666 | - | - |
| Clay * | 0.178 | 0.142 | 0.774 | 0.371 | - | - | 51.8 | 0.460 | 0.269 | - | - |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Marshall, J.; Muhlack, R.; Morton, B.J.; Dunnigan, L.; Chittleborough, D.; Kwong, C.W. Pyrolysis Temperature Effects on Biochar–Water Interactions and Application for Improved Water Holding Capacity in Vineyard Soils. Soil Syst. 2019, 3, 27. https://doi.org/10.3390/soilsystems3020027
Marshall J, Muhlack R, Morton BJ, Dunnigan L, Chittleborough D, Kwong CW. Pyrolysis Temperature Effects on Biochar–Water Interactions and Application for Improved Water Holding Capacity in Vineyard Soils. Soil Systems. 2019; 3(2):27. https://doi.org/10.3390/soilsystems3020027
Chicago/Turabian StyleMarshall, Jon, Richard Muhlack, Benjamin J. Morton, Lewis Dunnigan, David Chittleborough, and Chi Wai Kwong. 2019. "Pyrolysis Temperature Effects on Biochar–Water Interactions and Application for Improved Water Holding Capacity in Vineyard Soils" Soil Systems 3, no. 2: 27. https://doi.org/10.3390/soilsystems3020027
APA StyleMarshall, J., Muhlack, R., Morton, B. J., Dunnigan, L., Chittleborough, D., & Kwong, C. W. (2019). Pyrolysis Temperature Effects on Biochar–Water Interactions and Application for Improved Water Holding Capacity in Vineyard Soils. Soil Systems, 3(2), 27. https://doi.org/10.3390/soilsystems3020027





