Variation in Feedstock Wood Chemistry Strongly Influences Biochar Liming Potential
Abstract
1. Introduction
2. Materials and Methods
2.1. Feedstock and Biochar Preparation
2.2. pH Measurement of Feedstock Biochar and Soil–Biochar Incubation
2.3. Determination of Oxygen-Containing Surface Functional Groups by Boehm Titration
2.4. Spectroscopy
2.5. Statistical Analysis
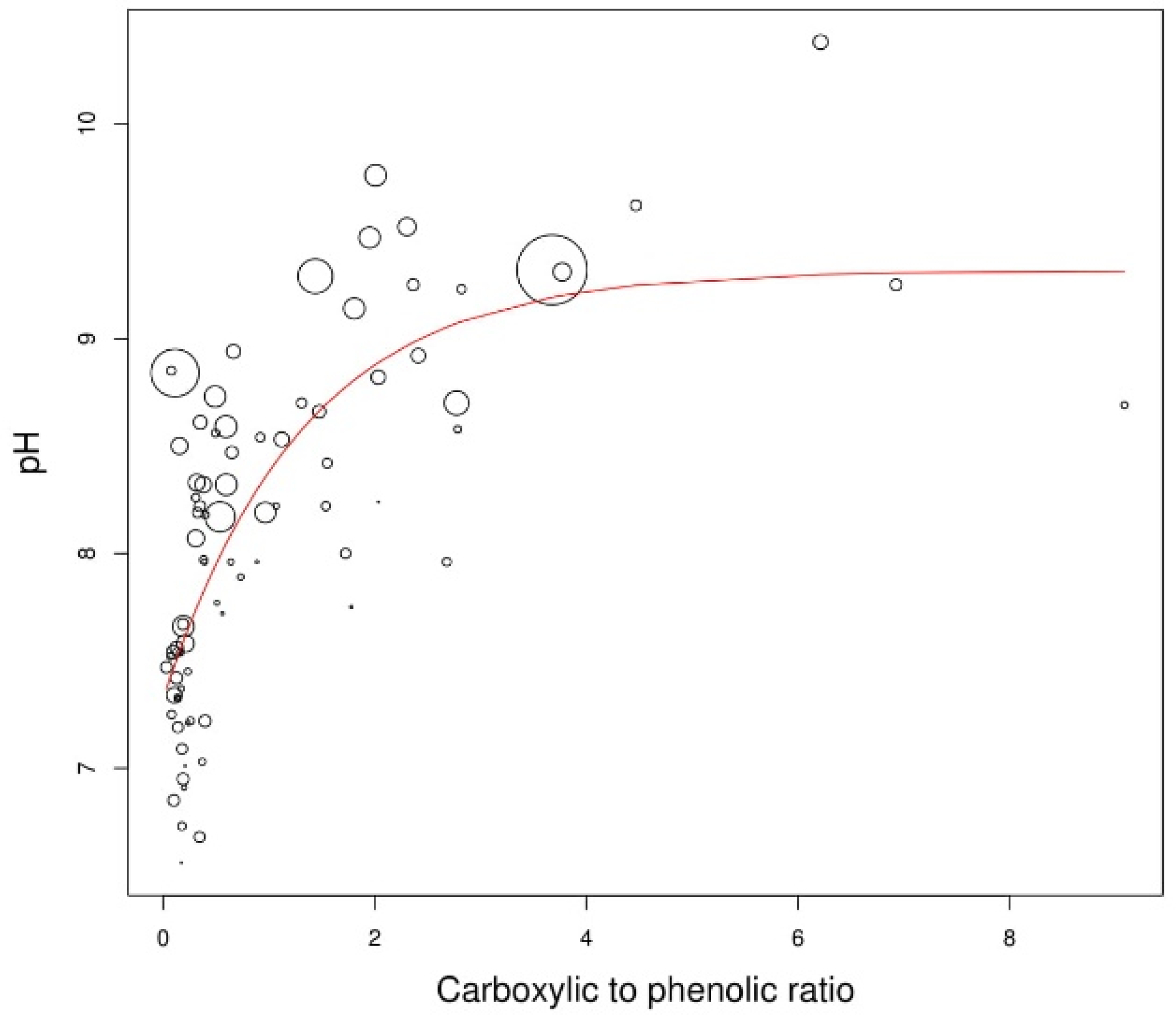
3. Results
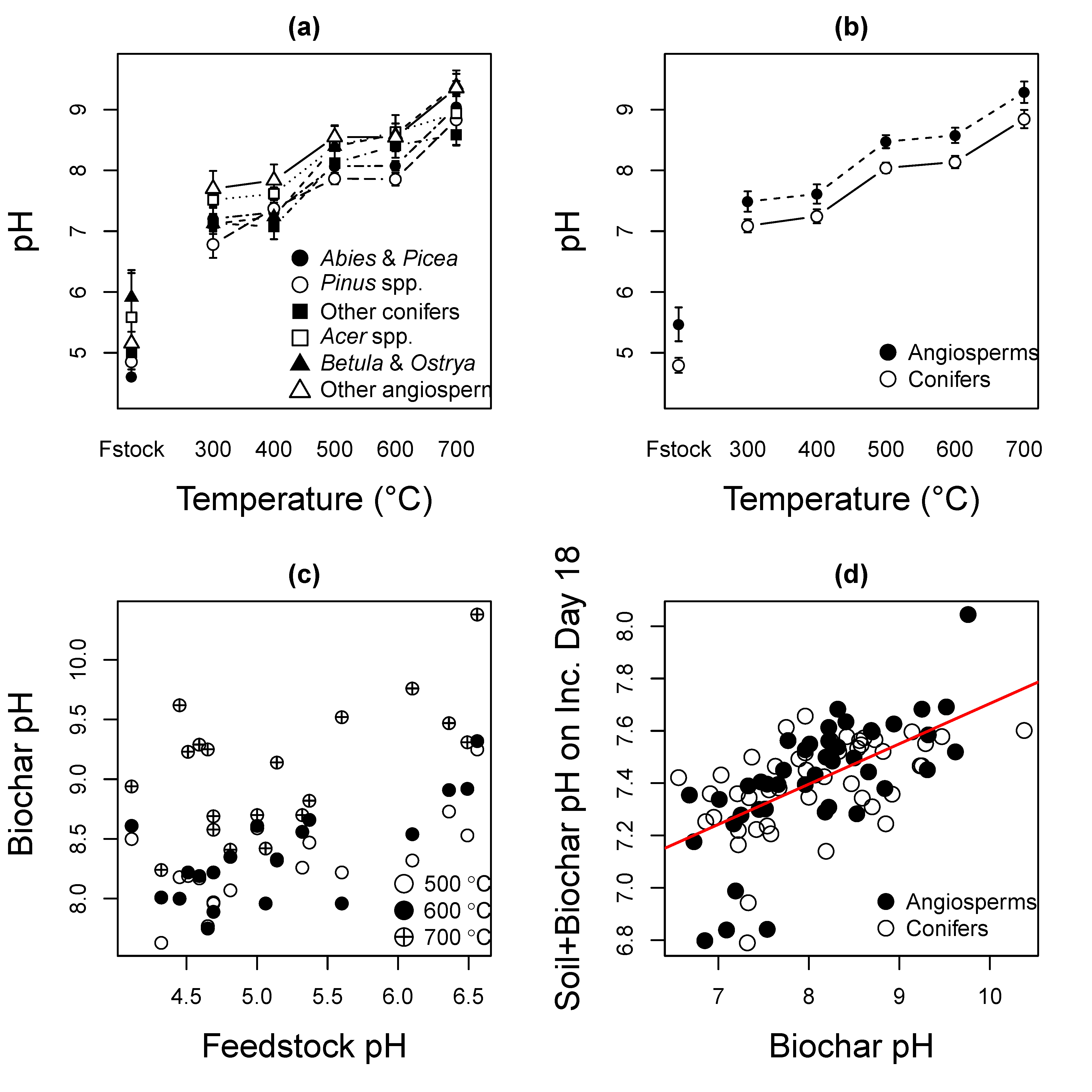
3.1. Effects of Pyrolysis Temperature and Species on Biochar pH
3.2. Feedstock and Biochar pH
3.3. Liming Potential of Biochar
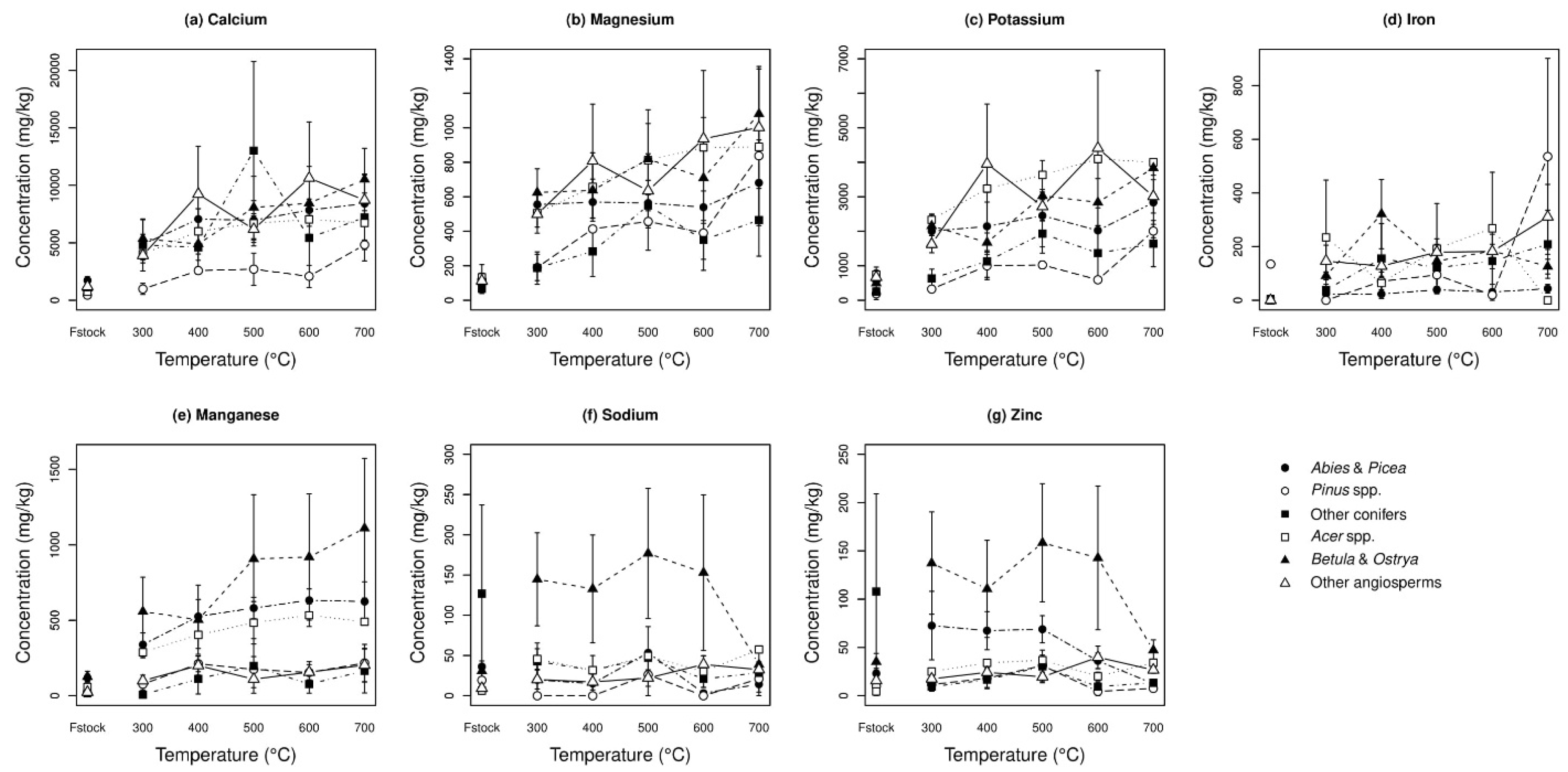
3.4. Elemental Analysis
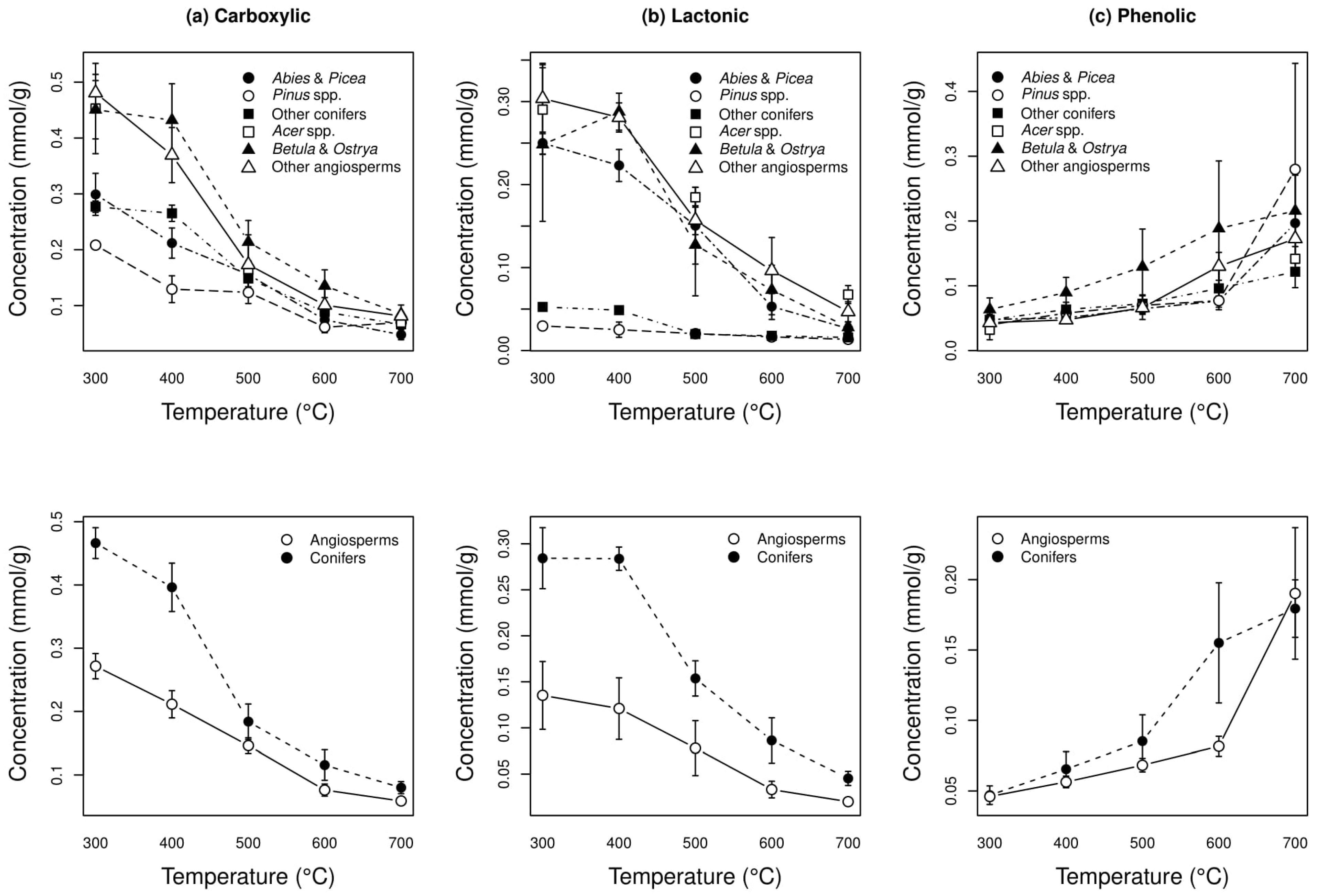
3.5. Surface Group Analysis
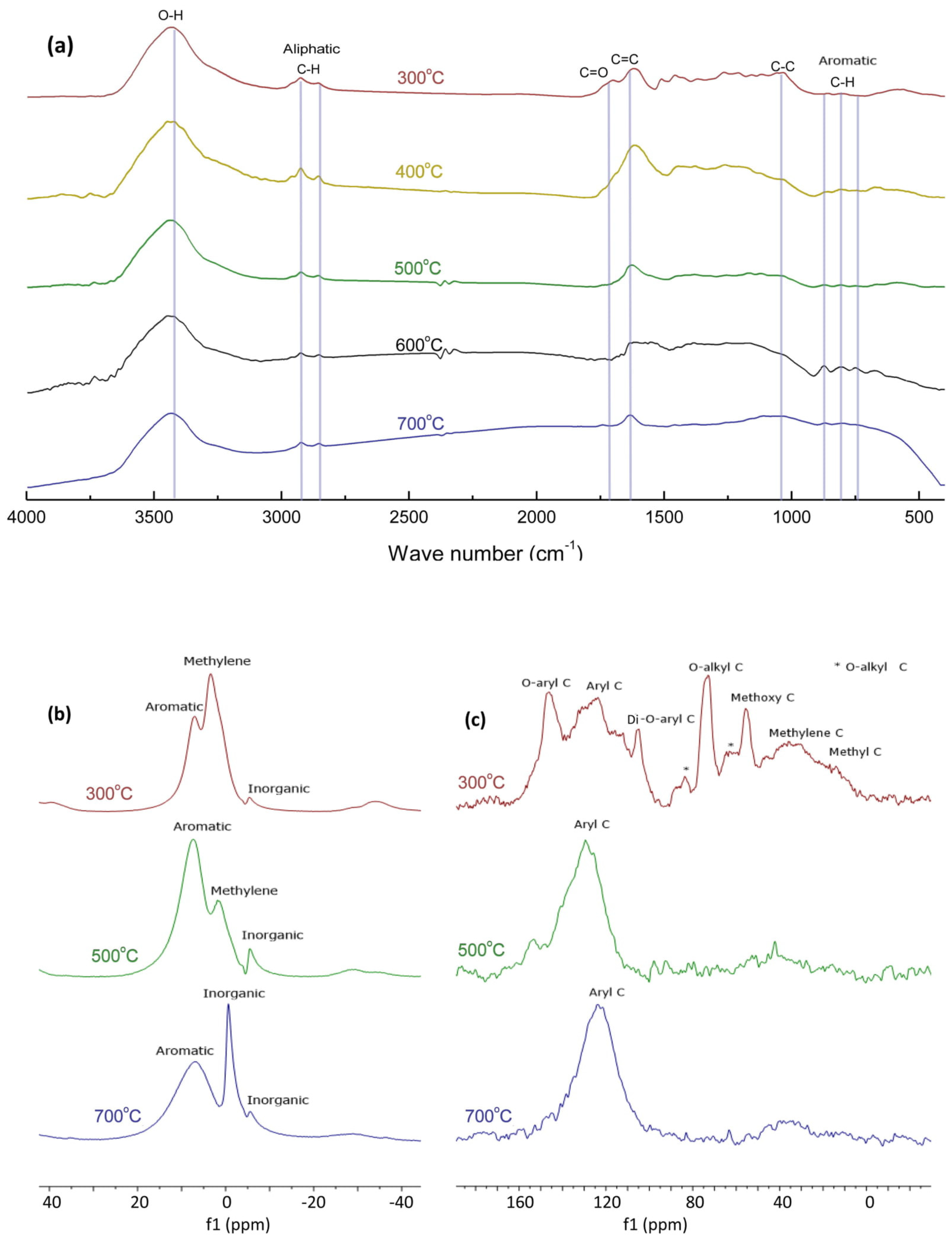
3.6. FTIR and NMR Spectroscopy
3.7. Modeling the Impact of Element and Surface Group Concentrations on Biochar pH
4. Discussion
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Lehmann, J.; Joseph, S. Biochar for Environmental Management: Science, Technology and Implementation, 2nd ed.; Earthscan: London, UK, 2015; pp. 3–71. [Google Scholar]
- Spokas, K.A. Review of the stability of biochar in soils: Predictability of O:C molar ratios. Carbon Manag. 2010, 1, 289–303. [Google Scholar] [CrossRef]
- Lehmann, J.; da Silva, J.P., Jr.; Steiner, C.; Nehls, T.; Zech, W.; Glaser, B. Nutrient availability and leaching in an archaeological Anthrosol and a Ferralsol of the Central Amazon basin: Fertilizer, manure and charcoal amendments. Plant Soil 2010, 249, 343–357. [Google Scholar] [CrossRef]
- Inguanzo, M.; Domínguez, A.; Menéndez, J.A.; Blanco, C.G.; Pis, J.J. On the pyrolysis of sewage sludge: The influence of pyrolysis conditions on solid, liquid and gas fractions. J. Anal. Appl. Pyrol. 2002, 63, 209–222. [Google Scholar] [CrossRef]
- Cantrell, K.; Ro, K.; Mahajan, D.; Anjom, M.; Hunt, P.G. Role of thermochemical conversion in livestock waste-to-energy treatments: Obstacles and opportunities. Ind. Eng. Chem. Res. 2007, 46, 8918–8927. [Google Scholar] [CrossRef]
- Spokas, K.A.; Cantrell, K.B.; Novak, J.M.; Archer, D.W.; Ippolito, J.A.; Collins, H.P.; Boateng, A.A.; Lima, I.M.; Lamb, M.C.; McAloon, A.J.; Lentz, R.D.; Nichols, K.A. Biochar: A synthesis of its agronomic impact beyond carbon sequestration. J. Env. Qual. 2012, 41, 973–989. [Google Scholar] [CrossRef] [PubMed]
- Ippolito, J.A.; Laird, D.A.; Busscher, W.J. Environmental benefits of biochar. J. Env. Qual. 2012, 41, 967–972. [Google Scholar] [CrossRef]
- Ekpete, O.A.; Horsfall, M.J.N.R. Preparation and characterization of activated carbon derived from fluted pumpkin stem waste (Telfairia occidentalis Hook F). Res. J. Chem. Sci. 2011, 1, 10–17. [Google Scholar]
- Qadeer, R.; Hanif, J.; Saleem, M.; Afzal, M. Characterization of activated charcoal. J. Chem. Soc. Pak. 1994, 16, 229–235. [Google Scholar]
- Liu, Y.; He, Z.; Uchimiya, M. Comparison of biochar formation from various agricultural by-products using FTIR spectroscopy. Mod. Appl. Sci. 2014, 9, 246–253. [Google Scholar] [CrossRef]
- Emmerich, F.G.; de Sousa, J.C.; Torriani, I.L.; Luengo, C.A. Applications of a granular model and percolation theory to the electrical resistivity of heat treated endocarp of babassu nut. Carbon 1987, 25, 417–424. [Google Scholar] [CrossRef]
- Chia, C.H.; Downie, A.; Munroe, P. Characteristics of biochar: Physical and structural properties. In Biochar for Environmental Management: Science, Technology and Implementation, 2nd ed.; Lehmann, J., Joseph, S., Eds.; Earthscan: London, UK, 2015; pp. 89–109. [Google Scholar]
- Vassilev, N.; Martos, E.; Mendes, G.; Martos, V.; Vassileva, M. Biochar of animal origin: A sustainable solution to the global problem of high-grade rock phosphate scarcity? J. Sci. Food Agric. 2013, 93, 1799–1804. [Google Scholar] [CrossRef]
- Kloss, S.; Zehetner, F.; Dellantonio, A.; Hamid, R.; Ottner, F.; Liedtke, V.; Schwanninger, M.; Gerzabek, M.H.; Soja, G. Characterization of slow pyrolysis biochars: Effects of feedstocks and pyrolysis temperature on biochar properties. J. Env. Qual. 2012, 41, 990. [Google Scholar] [CrossRef] [PubMed]
- Lehmann, J. Bio-energy in the black. Front. Ecol. Env. 2007, 5, 381–387. [Google Scholar] [CrossRef]
- Peng, X.Y.; Ye, L.L.; Wang, C.H.; Zhou, H.; Sun, B. Temperature- and duration-dependent rice straw-derived biochar: Characteristics and its effects on soil properties of an ultisol in southern China. Soil Tillage Res. 2011, 112, 159–166. [Google Scholar] [CrossRef]
- Lua, A.C.; Yang, T.; Guo, J. Effects of pyrolysis conditions on the properties of activated carbons prepared from pistachio-nut shells. J. Anal. Appl. Pyrol. 2004, 72, 279–287. [Google Scholar] [CrossRef]
- Basu, P. Biomass Gasification and Pyrolysis; Academic Press: Burlington, MA, USA, 2010. [Google Scholar]
- Demirbas, A. Effects of temperature and particle size on bio-char yield from pyrolysis of agricultural residues. J. Anal. Appl. Pyrol. 2004, 72, 243–248. [Google Scholar] [CrossRef]
- Beesley, L.; Moreno-Jiménez, E.; Gomez-Eyles, J.L.; Harris, E.; Robinson, B.; Sizmur, T. A review of biochars’ potential role in the remediation, revegetation and restoration of contaminated soils. Env. Pollut. 2011, 159, 3269–3282. [Google Scholar] [CrossRef] [PubMed]
- Wang, T.; Camps-Arbestain, M.; Hedley, M.; Singh, B.P.; Calvero-Pereira, R.; Wang, C. Determination of carbonate-C in biochars. Soil Res. 2014, 52, 495–504. [Google Scholar] [CrossRef]
- Liu, X.; Zhang, A.; Ji, C.; Joseph, S.; Bian, R.; Li, L.; Pan, G.; Paz-Ferreiro, J. Biochar’s effect on crop productivity and the dependence on experimental conditions—a meta-analysis of literature data. Plant Soil 2013, 373, 583–594. [Google Scholar] [CrossRef]
- Gomez-Eyles, J.L.; Sizmur, T.; Collins, C.D.; Hodson, M.E. Effects of biochar and the earthworm Eisenia fetida on the bioavailability of polycyclic aromatic hydrocarbons and potentially toxic elements. Env. Pollut. 2011, 159, 616–622. [Google Scholar] [CrossRef]
- Rees, F.; Simonnot, M.O.; Morel, J.L. Short-term effects of biochar on soil heavy metal mobility are controlled by intra-particle diffusion and soil pH increase. Eur. J. Soil Sci. 2014, 65, 149–161. [Google Scholar] [CrossRef]
- Mukherjee, A.; Zimmerman, A.R.; Harris, W. Surface chemistry variations among a series of laboratory-produced biochars. Geoderma 2011, 163, 247–255. [Google Scholar] [CrossRef]
- Van Zwieten, L.; Kimber, S.; Morris, S.; Chan, K.Y.; Downie, A.; Rust, J.; Joseph, S.; Cowie, A. Effects of biochar from slow pyrolysis of papermill waste on agronomic performance and soil fertility. Plant Soil 2009, 327, 235–246. [Google Scholar] [CrossRef]
- Wei, L.; Xu, G.; Shao, H.; Sun, J.; Chang, S.X. Regulating environmental factors of nutrients release from wheat straw biochar for sustainable agriculture. Clean Soil Air Water 2013, 41, 697–701. [Google Scholar] [CrossRef]
- Lai, W.; Lai, C.-M.; Ke, G.-R.; Chung, R.-S.; Chen, C.-T.; Cheng, C.-H.; Pai, C.-W.; Chen, S.-Y.; Chen, C.-C. The effects of woodchip biochar application on crop yield, carbon sequestration and greenhouse gas emissions from soils planted with rice or leaf beet. J. Taiwan Inst. Chem. Eng. 2013, 44, 1039–1044. [Google Scholar] [CrossRef]
- Sackett, T.E.; Basiliko, N.; Noyce, G.L.; Winsborough, C.; Schurman, J.; Ikeda, C.; Thomas, S.C. Soil and greenhouse gas responses to biochar additions in a temperate hardwood forest. GCB Bioenergy 2015, 7, 1062–1074. [Google Scholar] [CrossRef]
- Thomas, S.C.; Gale, N. Biochar and forest restoration: A review and meta-analysis of tree growth responses. New For. 2015, 46, 931–946. [Google Scholar] [CrossRef]
- Glaser, B.; Lehmann, J.; Zech, W. Ameliorating physical and chemical properties of highly weathered soils in the tropics with charcoal—A review. Biol. Fertil. Soils 2002, 35, 219–230. [Google Scholar] [CrossRef]
- Cheng, C.-H.; Lehmann, J.; Thies, J.E.; Burton, S.D.; Engelhard, M.H. Oxidation of black carbon by biotic and abiotic processes. Org. Geochem. 2006, 37, 1477–1488. [Google Scholar] [CrossRef]
- Pettersen, R.C. The chemical composition of wood. In The Chemistry of Solid Wood; Rowell, R., Ed.; American Chemical Society: Washington, DC, USA, 1984; Volume 207, pp. 57–126. [Google Scholar]
- Rowell, R.M. Handbook of Wood Chemistry and Wood Composites; CRC Press: Boca Raton, FL, USA, 2005; pp. 34–48. [Google Scholar]
- Meerts, P. Mineral nutrient concentrations in sapwood and heartwood: A literature review. Ann. Sci. 2002, 59, 713–722. [Google Scholar] [CrossRef]
- Pluchon, N.; Casetou, S.C.; Kardol, P.; Gundale, M.J.; Nilsson, M.-C.; Wardle, D.A. Influence of species identity and charring conditions on fire-derived charcoal traits. Can. J. Res. 2015, 45, 1669–1675. [Google Scholar] [CrossRef]
- Abdel-Fattah, T.M.; Mahmoud, M.E.; Ahmed, S.B.; Huff, M.D.; Lee, J.W.; Kumar, S. Biochar from woody biomass for removing metal contaminants and carbon sequestration. J. Korean Ind. Eng. Chem. 2015, 22, 103–109. [Google Scholar] [CrossRef]
- Xie, X.; Goodell, B.; Zhang, D.; Nagle, D.C.; Qian, Y.; Peterson, M.L.; Jellison, J. Characterization of carbons derived from cellulose and lignin and their oxidative behavior. Bioresour. Technol. 2009, 100, 1797–1802. [Google Scholar] [CrossRef] [PubMed]
- Müller-Hagedorn, M.; Bockhorn, H.; Krebs, L.; Müller, U. A comparative kinetic study on the pyrolysis of three different wood species. J. Anal. Appl. Pyrol. 2003, 68–69, 231–249. [Google Scholar] [CrossRef]
- Mohan, D.; Pittman, C.U.; Steele, P.H. Pyrolysis of wood/biomass for bio-oil: A critical review. Energy Fuels 2006, 20, 848–889. [Google Scholar] [CrossRef]
- Afifi, A.I.; Chornet, E.; Overend, R.P.; Hindermann, J.P. The upgrading of lignin-derived compounds: Case studies on model compounds. In Research in Thermochemical Biomass Conversion; Bridgwater, A., Kuester, J., Eds.; Springer: Dordrecht, The Netherlands, 1988; pp. 439–451. [Google Scholar]
- Thomas, S.C. Biochar and its potential in Canadian forestry. Silvic. Mag. 2013, 4–6. [Google Scholar]
- Mansfield, S.D.; Weineisen, H. Wood fiber quality and kraft pulping efficiencies of trembling aspen (Populus tremuloides Michx) clones. J. Wood Chem. Technol. 2007, 27, 135–151. [Google Scholar] [CrossRef]
- Heschel, H.; Klose, E. On the suitability of agricultural by-products for the manufacture of granular activated carbon. Fuel 1995, 74, 1786–1791. [Google Scholar] [CrossRef]
- Fidel, R.B.; Laird, D.A.; Thompson, M.L. Evaluation of modified Boehm titration methods for use with biochars. J. Env. Qual. 2013, 42, 1771–1778. [Google Scholar] [CrossRef]
- Goertzen, S.L.; Thériault, K.D.; Oickle, A.M.; Tarasuk, A.C.; Andreas, H.A. Standardization of the Boehm titration: Part I. CO2 expulsion and endpoint determination. Carbon 2010, 48, 1252–1261. [Google Scholar] [CrossRef]
- Enders, A.; Lehmann, J. Comparison of wet-digestion and dry-ashing methods for total elemental analysis of biochar. Commun. Soil Sci. Plant Anal. 2012, 43, 1042–1052. [Google Scholar] [CrossRef]
- McDonald, J.H. Handbook of Biological Statistics, 3rd ed.; Sparky House: Baltimore, MA, USA, 2013; pp. 4–6. [Google Scholar]
- Burnham, K.P.; Anderson, D.R. Model Selection and Multimodel Inference: A Practical Information-Theoretic Approach, 2nd ed.; Springer-Verlag: New York, NY, USA, 2002. [Google Scholar]
- R Core Team. R: A Language and Environment for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria, 2014. [Google Scholar]
- Spokas, K.A.; Novak, J.M.; Stewart, C.E.; Cantrell, K.B.; Uchimiya, M.; DuSaire, M.G.; Ro, K.S. Qualitative analysis of volatile organic compounds on biochar. Chemosphere 2011, 85, 869–882. [Google Scholar] [CrossRef]
- Gale, N.V.; Sackett, T.E.; Thomas, S.C. Thermal treatment and leaching of biochar alleviates plant growth inhibition from mobile organic compounds. PeerJ 2016, 4, e2385. [Google Scholar] [CrossRef] [PubMed]
- Ronsse, F.; van Hecke, S.; Dickinson, D.; Prins, W. Production and characterization of slow pyrolysis biochar: Influence of feedstock type and pyrolysis conditions. GCB Bioenergy 2013, 5, 104–115. [Google Scholar] [CrossRef]
- Novak, J.M.; Lima, I.; Xing, B.; Gaskin, J.W.; Steiner, C.; Das, K.C.; Ahmedna, M.; Rehrah, D.; Watts, D.W.; Busscher, W.J.; Schomberg, H. Characterization of designer biochar produced at different temperatures and their effects on a loamy sand. Ann. Env. Sci. 2009, 3, 195–206. [Google Scholar]
- Novak, J.M.; Cantrell, K.B.; Watts, D.W. Compositional and thermal evaluation of lignocellulosic and poultry litter chars via high and low temperature pyrolysis. Bioenergy Res. 2012, 6, 114–130. [Google Scholar] [CrossRef]
- Rutherford, D.W.; Wershaw, R.L.; Reeves, J.B., III. Development of acid functional groups and lactones during the thermal degradation of wood and wood components. In US Geological Survey Scientific Investigations Report 2007–5013; U.S. Geological Survey: Reston, VA, USA, 2008. [Google Scholar]
- Enders, A.; Hanley, K.; Whitman, T.; Joseph, J.; Lehmann, J. Characterization of biochars to evaluate recalcitrance and agronomic performance. Bioresour. Technol. 2012, 114, 644–653. [Google Scholar] [CrossRef] [PubMed]
- Han, Y.; Boateng, A.A.; Qi, P.X.; Lima, I.M.; Chang, J. Heavy metal and phenol adsorptive properties of biochars from pyrolyzed switchgrass and woody biomass in correlation with surface properties. J. Env. Manag. 2013, 118, 196–204. [Google Scholar] [CrossRef]
- Reeves III, J.B. Mid-infrared spectroscopy of biochars and spectral similarities to coal and kerogens: What are the implications? Appl. Spectrosc. 2012, 66, 689–695. [Google Scholar] [CrossRef]
- Brewer, C.E.; Hu, Y.-Y.; Schmidt-Rohr, K.; Loynachan, T.E.; Laird, D.A.; Brown, R.C. Extent of pyrolysis impacts on fast pyrolysis biochar properties. J. Env. Qual. 2012, 41, 1115–1122. [Google Scholar] [CrossRef]
- Reid, W.T. The relation of mineral composition to slagging, fouling and erosion during and after combustion. Prog. Energy Combust. Sci. 1984, 10, 159–169. [Google Scholar] [CrossRef]
- Liaw, S.B.; Wu, H. Leaching characteristics of organic and inorganic matter from biomass by water: Differences between batch and semi-continuous operations. Ind. Eng. Chem. Res. 2013, 52, 4280–4289. [Google Scholar] [CrossRef]
- Weast, R.C.; Astle, M.J.; Beyer, W.H. CRC Handbook of Chemistry and Physics, 66th ed.; CRC Press: Boca Raton, FL, USA, 1986. [Google Scholar]
- Berek, A.K.; Hue, N.V. Characterization of biochars and their use as an amendment to acid soils. Soil Sci. 2016, 181, 412–426. [Google Scholar] [CrossRef]
- Singh, B.; Singh, B.P.; Cowie, A.L. Characterization and evaluation of biochars for their application as a soil amendment. Aust. J. Soil Res. 2010, 48, 516–525. [Google Scholar] [CrossRef]
- Karberg, N.J.; Pregitzer, K.S.; King, J.S.; Friend, A.L.; Wood, J.R. Soil carbon dioxide partial pressure and dissolved inorganic carbonate chemistry under elevated carbon dioxide and ozone. Oecologia 2005, 142, 296–306. [Google Scholar] [CrossRef] [PubMed]
- Sawyer, J. Soil pH and Liming. Presented at the Soil Fertility and Nutrient Management Short Course, Iowa State University, Ames, IA, USA, 2004. Available online: http://www.agronext.iastate.edu/soilfertility/info/SoilpHandLiming2017.pdf (accessed on 1 April 2019).
- Basso, A.S. Effect of fast pyrolysis biochar on physical and chemical properties of a sandy soil. Master’s Thesis, Iowa State University, Ames, IA, USA, 2012. [Google Scholar] [CrossRef]
- Novak, J.M.; Busscher, W.J.; Laird, D.; Ahmedna, M.; Watts, D.W.; Niandou, M.A.S. Impact of biochar amendment on fertility of a southeastern coastal plain soil. Soil Sci. 2009, 174, 105–112. [Google Scholar] [CrossRef]
- Obia, A.; Cornelissen, G.; Mulder, J.; Dörsch, P. Effect of soil pH increase by biochar on NO, N2O and N2 production during denitrification in acid soils. PloS ONE 2015, 10, e0138781. [Google Scholar] [CrossRef]
- Keown, D.M.; Favas, G.; Hayashi, J.; Li, C.Z. Volatilization of alkali and alkaline earth metallic species during the pyrolysis of biomass: Differences between sugar cane bagasse and cane trash. Bioresour. Technol. 2005, 96, 1570–1577. [Google Scholar] [CrossRef]
- Wu, H.; Yip, K.; Kong, Z.; Li, C.Z.; Liu, D.; Yu, Y.; Gao, X. Removal and recycling of inherent inorganic nutrient species in Mallee biomass and derived biochars by water leaching. Ind. Eng. Chem. Res. 2011, 50, 12143–12151. [Google Scholar] [CrossRef]
- Lievens, C.; Mourant, D.; Gunawan, R.; Hu, X.; Wang, Y. Organic compounds leached from fast pyrolysis mallee leaf and bark biochars. Chemosphere 2015, 139, 659–664. [Google Scholar] [CrossRef]
- Ubando, A.T.; Culaba, A.B.; Aviso, K.B.; Ng, D.K.S.; Tan, R.R. Fuzzy mixed-integer linear programming model for optimizing a multi-functional bioenergy system with biochar production for negative carbon emissions. Clean Technol. Env. Policy 2014, 16, 1537–1549. [Google Scholar] [CrossRef]
- Lukin, V.V.; Epplin, F.M. Optimal frequency and quantity of agricultural lime applications. Agr. Syst. 2003, 76, 949–967. [Google Scholar] [CrossRef]
- Lima, H.N.; Schaefer, C.E.R.; Mello, J.W.V.; Gilkes, R.J.; Ker, J.C. Pedogenesis and pre-Colombian land use of “Terra Preta Anthrosols” (“Indian black earth”) of Western Amazonia. Geoderma 2002, 110, 1–17. [Google Scholar] [CrossRef]
| Analyte | Species Effect | Temperature Effect | Conifers (mg/Kg) | Angiosperms (mg/Kg) | p-Value | ||
|---|---|---|---|---|---|---|---|
| F-Value | F-Value | Mean | SE | Mean | SE | ||
| Ca | 4.9 18, 73 *** | 8.2 1, 90 ** | 5888.1 | 596.1 | 7294.8 | 724.4 | 0.143 |
| K | 4.6 18, 73 *** | 5.1 1, 90 * | 1680.1 | 202.1 | 3038.8 | 289.0 | <0.001 |
| Mg | 6.5 18, 73 *** | 8.1 1, 90 ** | 481.2 | 40.3 | 766.9 | 69.8 | <0.001 |
| Mn | 17.0 18, 73 *** | 4.0 1, 90 * | 317.1 | 40.2 | 407.1 | 64.4 | 0.239 |
| Fe | 2.4 18, 73 ** | 4.5 1, 90 * | 90.4 | 27.7 | 181.1 | 22.6 | <0.05 |
| Na | 7.2 18, 73 *** | 0.3 1, 90 † | 22.4 | 4.7 | 61.4 | 11.4 | <0.01 |
| Zn | 8.2 18, 73 *** | 1.5 1, 90 † | 31.5 | 5.3 | 55.6 | 9.6 | 0.030 |
| Carboxylic ‡ | 0.8 18, 70 † | 150.0 1, 90 *** | 0.153 ‡ | 0.014 | 0.247 ‡ | 0.026 | 0.002 |
| Lactonic ‡ | 2.4 18, 70 ** | 56.4 1, 87 *** | 0.077 ‡ | 0.013 | 0.169 ‡ | 0.018 | <0.05 |
| Phenolic ‡ | 1.4 18, 69 † | 48.0 1, 86 *** | 0.089 ‡ | 0.012 | 0.106 ‡ | 0.012 | 0.321 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gezahegn, S.; Sain, M.; Thomas, S.C. Variation in Feedstock Wood Chemistry Strongly Influences Biochar Liming Potential. Soil Syst. 2019, 3, 26. https://doi.org/10.3390/soilsystems3020026
Gezahegn S, Sain M, Thomas SC. Variation in Feedstock Wood Chemistry Strongly Influences Biochar Liming Potential. Soil Systems. 2019; 3(2):26. https://doi.org/10.3390/soilsystems3020026
Chicago/Turabian StyleGezahegn, Sossina, Mohini Sain, and Sean C. Thomas. 2019. "Variation in Feedstock Wood Chemistry Strongly Influences Biochar Liming Potential" Soil Systems 3, no. 2: 26. https://doi.org/10.3390/soilsystems3020026
APA StyleGezahegn, S., Sain, M., & Thomas, S. C. (2019). Variation in Feedstock Wood Chemistry Strongly Influences Biochar Liming Potential. Soil Systems, 3(2), 26. https://doi.org/10.3390/soilsystems3020026






