The Extent of Insulin Resistance in Patients That Cleared Viral Hepatitis C Infection and the Role of Pre-Existent Type 2 Diabetes Mellitus: A Meta-Analysis
Abstract
1. Introduction
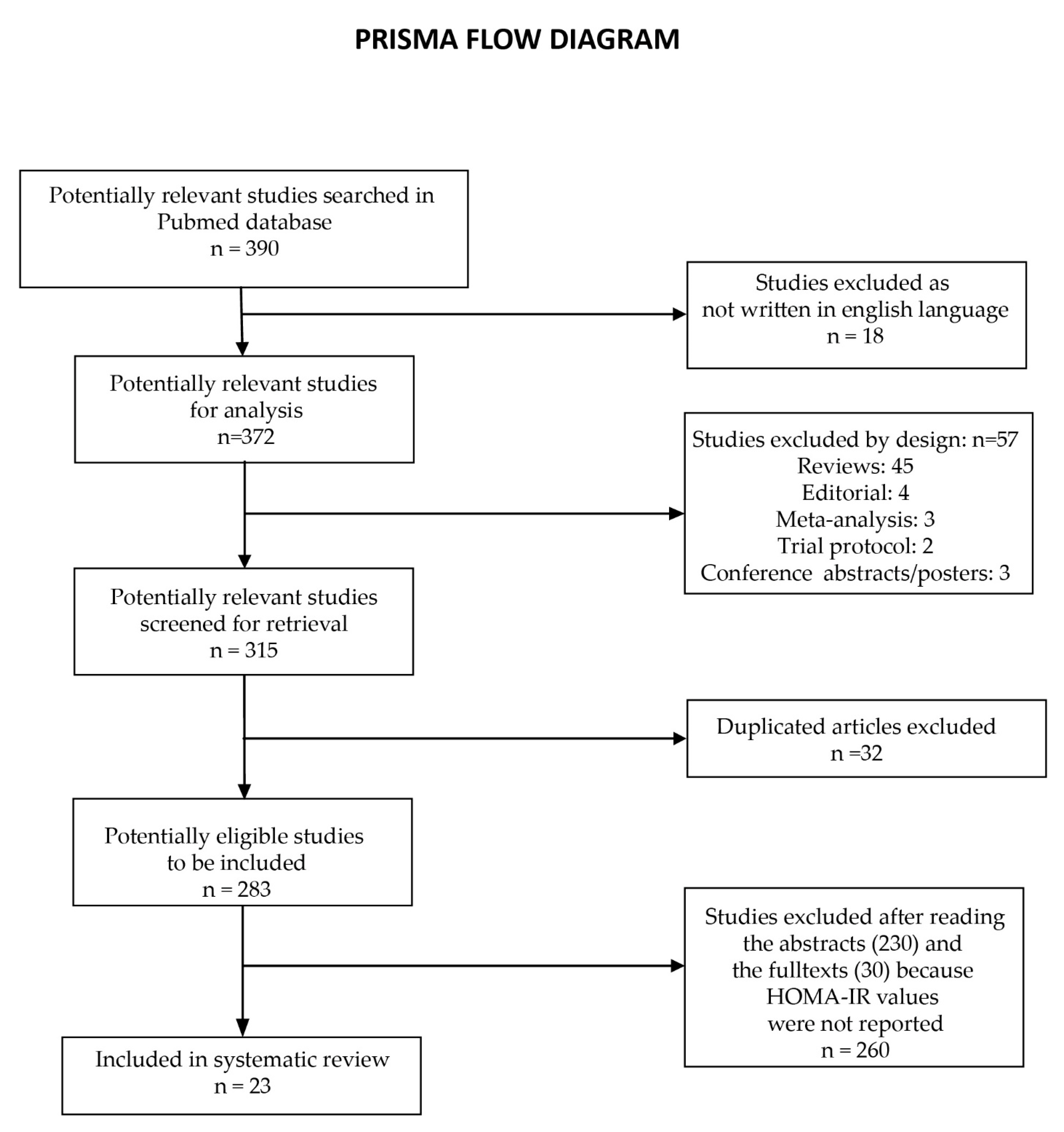
2. Materials and Methods
2.1. Quality Assessment
2.2. Data Extraction
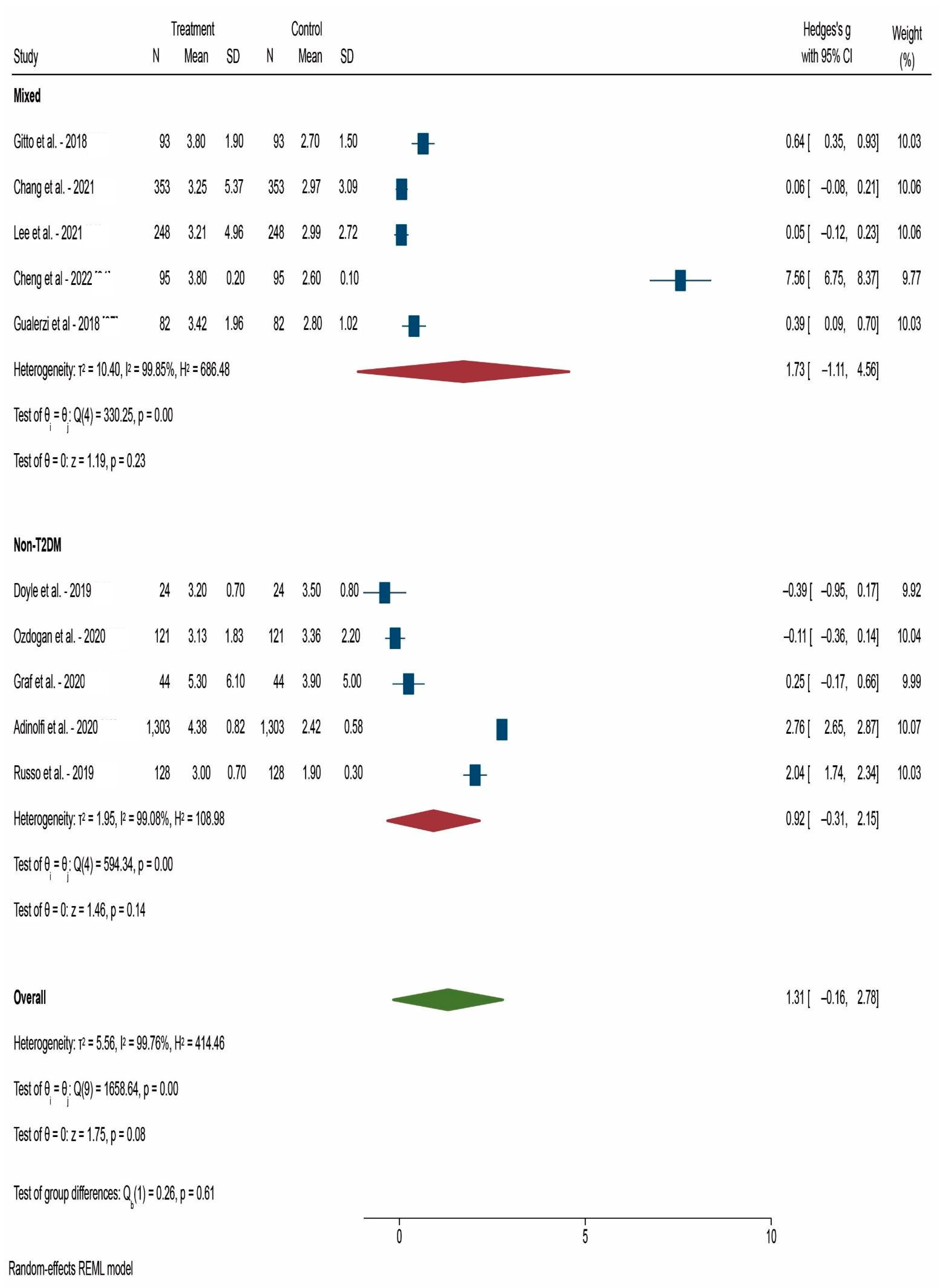
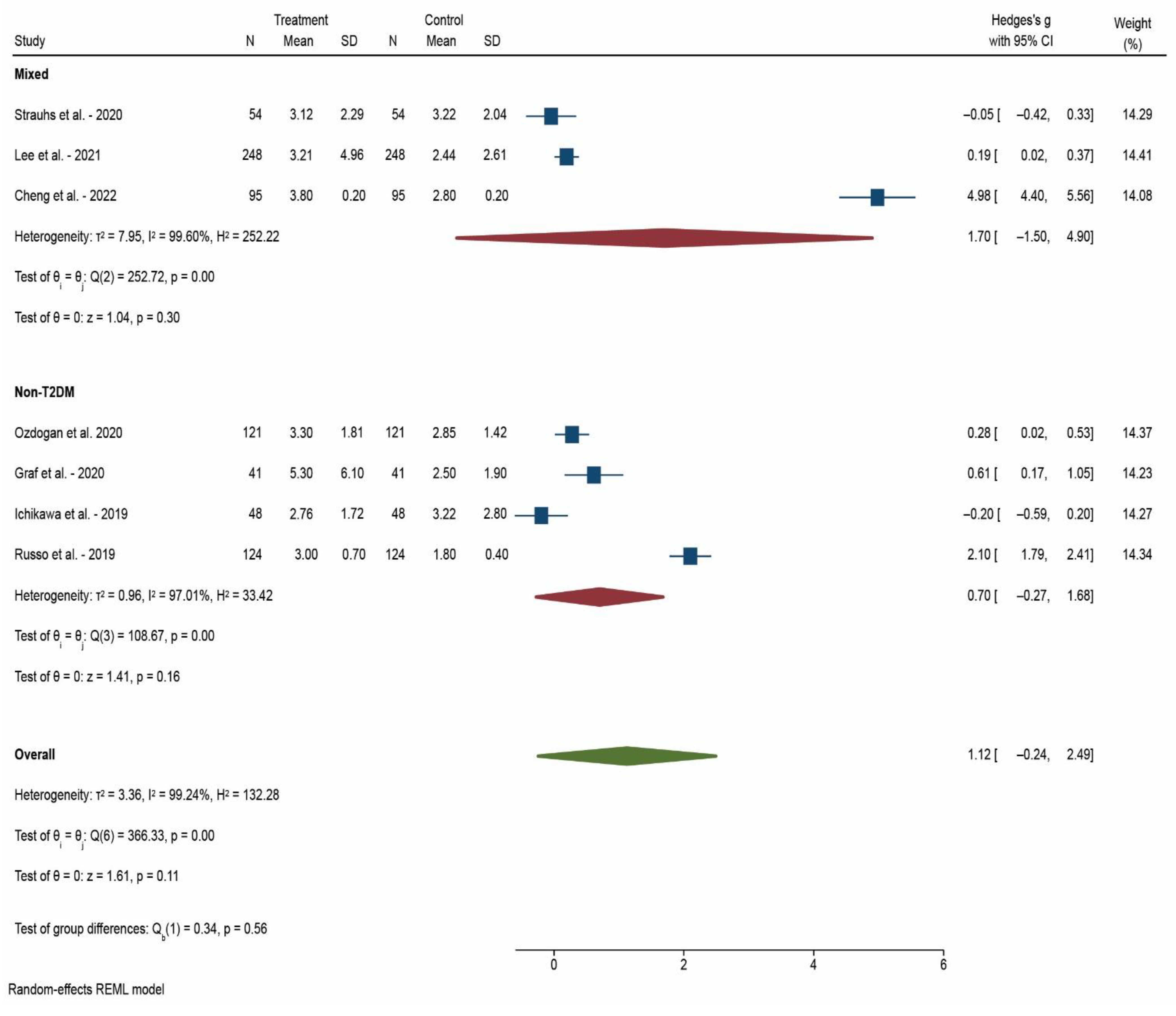
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Lemieux, I.; Després, J.P. Metabolic Syndrome: Past, Present and Future. Nutrients 2020, 12, 3507. [Google Scholar] [CrossRef] [PubMed]
- Bence, K.K.; Birnbaum, M.J. Metabolic drivers of non-alcoholic fatty liver disease. Mol. Metab. 2021, 50, 101143. [Google Scholar] [CrossRef] [PubMed]
- Ibrahim, M.; Parveen, B.; Zahiruddin, S.; Parveen, R.; Khan, M.A.; Gupta, A.; Ahmad, S. Uplc/Ms Based Phytochemical Screening and Antidiabetic Properties of Picrorhiza Kurroa in Mitigating Glucose-Induced Metabolic Dysregulation and Oxidative Stress. Farmacia 2021, 69, 749–755. [Google Scholar] [CrossRef]
- Santoleri, D.; Titchenell, P.M. Resolving the Paradox of Hepatic Insulin Resistance. Cell Mol. Gastroenterol. Hepatol. 2019, 7, 447–456. [Google Scholar] [CrossRef]
- Katz, A.; Nambi, S.S.; Mather, K.; Baron, A.D.; Follmann, D.A.; Sullivan, G.; Quon, M.J. Quantitative insulin sensitivity check index: A simple, accurate method for assessing insulin sensitivity in humans. J. Clin. Endocrinol. Metab. 2000, 85, 2402–2410. [Google Scholar] [CrossRef] [PubMed]
- Farshadpour, F.; Taherkhani, R.; Ravanbod, M.R.; Eghbali, S.S. Prevalence and Genotype Distribution of Hepatitis C Virus Infection among Patients with Type 2 Diabetes Mellitus. Med. Princ. Pract. 2018, 27, 308–316. [Google Scholar] [CrossRef] [PubMed]
- Kralj, D.; Virović Jukić, L.; Stojsavljević, S.; Duvnjak, M.; Smolić, M.; Čurčić, I.B. Hepatitis C Virus, Insulin Resistance, and Steatosis. J. Clin. Transl. Hepatol. 2016, 4, 66–75. [Google Scholar] [CrossRef] [PubMed]
- Ansaldi, F.; Orsi, A.; Sticchi, L.; Bruzzone, B.; Icardi, G. Hepatitis C virus in the new era: Perspectives in epidemiology, prevention, diagnostics and predictors of response to therapy. World J. Gastroenterol. 2014, 20, 9633–9652. [Google Scholar] [CrossRef]
- Narne, P. Impaired insulin exocytosis in chronic hepatitis C infection: Contributory role of p38δ MAPK-protein kinase D-golgi complex axis. Clin. Sci. 2020, 134, 1449–1456. [Google Scholar] [CrossRef]
- Himoto, T.; Nomura, T.; Tani, J.; Miyoshi, H.; Morishita, A.; Yoneyama, H.; Haba, R.; Masugata, H.; Masaki, T. Exacerbation of insulin resistance and hepatic steatosis deriving from zinc deficiency in patients with HCV-related chronic liver disease. Biol. Trace Elem. Res. 2015, 163, 81–88. [Google Scholar] [CrossRef]
- Abenavoli, L.; Masarone, M.; Peta, V.; Milic, N.; Kobyliak, N.; Rouabhia, S.; Persico, M. Insulin resistance and liver steatosis in chronic hepatitis C infection genotype 3. World J. Gastroenterol. 2014, 20, 15233–15240. [Google Scholar] [CrossRef] [PubMed]
- Wójcik, K.; Jabłonowska, E.; Omulecka, A.; Piekarska, A. Insulin resistance, adipokine profile and hepatic expression of SOCS-3 gene in chronic hepatitis C. World J. Gastroenterol. 2014, 20, 10449–10456. [Google Scholar] [CrossRef]
- Chien, C.H.; Lin, C.L.; Hu, C.C.; Chang, J.J.; Chien, R.N. Clearance of Hepatitis C Virus Improves Insulin Resistance During and After Peginterferon and Ribavirin Therapy. J. Interferon. Cytokine Res. 2015, 35, 981–989. [Google Scholar] [CrossRef] [PubMed]
- Adinolfi, L.E.; Nevola, R.; Guerrera, B.; D’Alterio, G.; Marrone, A.; Giordano, M.; Rinaldi, L. Hepatitis C virus clearance by direct-acting antiviral treatments and impact on insulin resistance in chronic hepatitis C patients. J. Gastroenterol. Hepatol. 2018, 33, 1379–1382. [Google Scholar] [CrossRef] [PubMed]
- Wallace, T.M.; Levy, J.C.; Matthews, D.R. Use and abuse of HOMA modeling. Diabetes Care 2004, 27, 1487–1495. [Google Scholar] [CrossRef]
- Stasi, C.; Sadalla, S.; Carradori, E.; Monti, M.; Petraccia, L.; Madia, F.; Gragnani, L.; Zignego, A.L. Longitudinal evaluation of liver stiffness and outcomes in patients with chronic hepatitis C before and after short- and long-term IFN-free antiviral treatment. Curr. Med. Res. Opin. 2020, 36, 245–249. [Google Scholar] [CrossRef]
- Stasi, C.; Triboli, E.; Arena, U.; Urraro, T.; Petrarca, A.; Gragnani, L.; Laffi, G.; Zignego, A.L. Assessment of liver stiffness in patients with HCV and mixed cryoglobulinemia undergoing rituximab treatment. J. Transl. Med. 2014, 12, 21. [Google Scholar] [CrossRef]
- Adinolfi, L.E.; Petta, S.; Fracanzani, A.L.; Nevola, R.; Coppola, C.; Narciso, V.; Rinaldi, L.; Calvaruso, V.; Pafundi, P.C.; Lombardi, R.; et al. Reduced incidence of type 2 diabetes in patients with chronic hepatitis C virus infection cleared by direct-acting antiviral therapy: A prospective study. Diabetes Obes. Metab. 2020, 22, 2408–2416. [Google Scholar] [CrossRef]
- Cheng, P.N.; Chen, J.Y.; Chiu, Y.C.; Chiu, H.C.; Tsai, L.M. Augmenting central arterial stiffness following eradication of HCV by direct acting antivirals in advanced fibrosis patients. Sci. Rep. 2019, 9, 1426. [Google Scholar] [CrossRef]
- Ichikawa, T.; Miyaaki, H.; Miuma, S.; Motoyoshi, Y.; Narita, S.; Toda, S.; Takahashi, Y.; Honda, T.; Yajima, H.; Uehara, R.; et al. Carotid Intima-media Thickness and Small Dense Low-density Lipoprotein Cholesterol Increase after One Year of Treatment with Direct-acting Antivirals in Patients with Hepatitis C Virus Infection. Intern. Med. 2019, 58, 1209–1215. [Google Scholar] [CrossRef]
- Strauhs-Nitsch, L.; Campiolo, M.F.; Morsoletto, D.B.G.; Pissaia Junior, A.; Ivantes, C.A.P. Curing hepatitis c with the new direct acting antivirals did not improve insulin resistance after one year. Arq. Gastroenterol. 2020, 57, 267–271. [Google Scholar] [CrossRef] [PubMed]
- Elhelbawy, M.; Abdel-Razek, W.; Alsebaey, A.; Hashim, M.; Elshenawy, H.; Waked, I. Insulin resistance does not impair response of chronic hepatitis C virus to direct-acting antivirals, and improves with the treatment. Eur. J. Gastroenterol. Hepatol. 2019, 31, 16–23. [Google Scholar] [CrossRef]
- Özdoğan, O.; Yaraş, S.; Ateş, F.; Üçbilek, E.; Sezgin, O.; Altıntaş, E. The impact of direct-acting antiviral treatment on lipid metabolism and insulin resistance in chronic hepatitis C patients: Temporary? Permanent? Turk. J. Gastroenterol. 2020, 31, 384–392. [Google Scholar] [CrossRef] [PubMed]
- Graf, C.; Welzel, T.; Bogdanou, D.; Vermehren, J.; Beckel, A.; Bojunga, J.; Friedrich-Rust, M.; Dietz, J.; Kubesch, A.; Mondorf, A.; et al. Hepatitis C Clearance by Direct-Acting Antivirals Impacts Glucose and Lipid Homeostasis. J. Clin. Med. 2020, 9, 2702. [Google Scholar] [CrossRef]
- Gitto, S.; Cicero, A.F.G.; Loggi, E.; Giovannini, M.; Conti, F.; Grandini, E.; Guarneri, V.; Scuteri, A.; Vitale, G.; Cursaro, C.; et al. Worsening of Serum Lipid Profile after Direct Acting Antiviral Treatment. Ann. Hepatol. 2018, 17, 64–75. [Google Scholar] [CrossRef]
- Doyle, M.-A.; Galanakis, C.; Mulvihill, E.; Crawley, A.; Cooper, C.L. Hepatitis C Direct Acting Antivirals and Ribavirin Modify Lipid but not Glucose Parameters. Cells 2019, 8, 252. [Google Scholar] [CrossRef] [PubMed]
- Chang, M.-L.; Hu, J.-H.; Pao, L.-H.; Lin, M.-S.; Kuo, C.-J.; Chen, S.-C.; Fan, C.-M.; Chang, M.-Y.; Chien, R.-N. Critical role of triglycerides for adiponectin levels in hepatitis C: A joint study of human and HCV core transgenic mice. BMC Immunol. 2021, 22, 54. [Google Scholar] [CrossRef]
- Lee, H.; Chien, R.N.; Pao, L.H.; Kuo, C.J.; Huang, P.H.; Chang, M.L. Decoupled Glucose and Lipid Metabolic Recovery after Viral Clearance in Direct-Acting Antiviral-Treated HCV Patients: A 3-Year Prospective Cohort Study. Cells 2021, 10, 2934. [Google Scholar] [CrossRef]
- Alsebaey, A.; Elhelbawy, M.; Abdel-Razek, W.; Hashim, M.; Elshenawy, H.; Waked, I. HCV treatment with direct acting antivirals improves the insulin sensitivity. Expert Rev. Anti. Infect. Ther. 2019, 17, 749–754. [Google Scholar] [CrossRef]
- Abdel Moneim, A.; Suleiman, H.A.; Mahmoud, B.; Mabrouk, D.; Zaky, M.Y.; Mahmoud, B. Viral clearance ameliorates hematological and inflammatory markers among diabetic patients infected with hepatitis C genotype 4. Clin. Exp. Med. 2020, 20, 231–240. [Google Scholar] [CrossRef]
- Cheng, P.-N.; Sun, H.-Y.; Feng, I.C.; Chiu, Y.-C.; Wang, S.-T.; Tan, D.C.; Chiu, H.-C.; Chien, S.-C.; Young, K.-C. Interdependence of glycemic and lipid modulation in cured chronic hepatitis C patients by direct-acting antiviral agents. J. Microbiol. Immunol. Infect. 2022. [Google Scholar] [CrossRef] [PubMed]
- Ciancio, A.; Bosio, R.; Bo, S.; Pellegrini, M.; Sacco, M.; Vogliotti, E.; Fassio, G.; Bianco Mauthe Degerfeld, A.G.F.; Gallo, M.; Giordanino, C.; et al. Significant improvement of glycemic control in diabetic patients with HCV infection responding to direct-acting antiviral agents. J. Med. Virol. 2018, 90, 320–327. [Google Scholar] [CrossRef]
- Russo, F.P.; Zanetto, A.; Gambato, M.; Bortoluzzi, I.; Al Zoairy, R.; Franceschet, E.; De Marchi, F.; Marzi, L.; Lynch, E.N.; Floreani, A.; et al. Hepatitis C virus eradication with direct-acting antiviral improves insulin resistance. J. Viral. Hepat. 2020, 27, 188–194. [Google Scholar] [CrossRef] [PubMed]
- Lin, C.H.; Chen, J.J.; Lee, P.L.; Tung, H.D.; Cheng, C.T.; Kao, H.J.; Wu, Y.H.; Pang, M.G.; Chuang, T.W. Lipid profile changes after direct acting antiviral treatment in different genotypes of chronic hepatitis C virus-infected patients. Adv. Dig. Med. 2021, 8, 139–145. [Google Scholar] [CrossRef]
- Alzahaby, A.A.A.E.-H.; Abdel-Halim, M.M.; Hussien, A.A.E.-s. Effect of direct acting anti-viral agents on insulin resistance in chronic HCV patients. Egypt. J. Hosp. Med. 2018, 72, 4413–4419. [Google Scholar] [CrossRef]
- Nevola, R.; Rinaldi, L.; Zeni, L.; Sasso, F.C.; Pafundi, P.C.; Guerrera, B.; Marrone, A.; Giordano, M.; Adinolfi, L.E. Metabolic and renal changes in patients with chronic hepatitis C infection after hepatitis C virus clearance by direct-acting antivirals. JGH Open 2020, 4, 713–721. [Google Scholar] [CrossRef] [PubMed]
- Gualerzi, A.; Bellan, M.; Smirne, C.; Tran Minh, M.; Rigamonti, C.; Burlone, M.E.; Bonometti, R.; Bianco, S.; Re, A.; Favretto, S. Improvement of insulin sensitivity in diabetic and non diabetic patients with chronic hepatitis C treated with direct antiviral agents. PLoS ONE 2018, 13, e0209216. [Google Scholar] [CrossRef] [PubMed]
- Hashim, A.E.; Kandeel, H.T.; Hendy, O.M.; El-Mola, K.; El-Raey, F.M.; Attia, M.S. Effect of new direct-acting antiviral drugs on insulin resistance and glycemic control after treatment of chronic hepatitis C virus infection in type 2 diabetic patients. Al-Azhar Assiut Med. J. 2017, 15, 187. [Google Scholar]
- Salomone, F.; Catania, M.; Montineri, A.; Bertino, G.; Godos, J.; Rizzo, L.; Magrì, G.; Li Volti, G. Hepatitis C virus eradication by direct antiviral agents improves glucose tolerance and reduces post-load insulin resistance in nondiabetic patients with genotype 1. Liver Int. 2018, 38, 1206–1211. [Google Scholar] [CrossRef]
- Yosef, T.; Ibrahim, W.A.; El-Ghandour, A.; Attia, S.; El-Nakeep, S. Effect of different direct-acting antiviral regimens for treatment of nondiabetic hepatitis C virus–infected Egyptian patients on insulin resistance and sensitivity. Egypt. J. Intern. Med. 2021, 33, 45. [Google Scholar] [CrossRef]
- Lonardo, A.; Ballestri, S.; Guaraldi, G.; Nascimbeni, F.; Romagnoli, D.; Zona, S.; Targher, G. Fatty liver is associated with an increased risk of diabetes and cardiovascular disease - Evidence from three different disease models: NAFLD, HCV and HIV. World J. Gastroenterol. 2016, 22, 9674–9693. [Google Scholar] [CrossRef] [PubMed]
- Dawood, A.A.; Nooh, M.Z.; Elgamal, A.A. Factors Associated with Improved Glycemic Control by Direct-Acting Antiviral Agent Treatment in Egyptian Type 2 Diabetes Mellitus Patients with Chronic Hepatitis C Genotype 4. Diabetes Metab. J. 2017, 41, 316–321. [Google Scholar] [CrossRef] [PubMed]
- Chang, M.-L.; Kuo, C.-J.; Pao, L.-H.; Hsu, C.-M.; Chiu, C.-T. The evolving relationship between adiponectin and insulin sensitivity in hepatitis C patients during viral clearance. Virulence 2017, 8, 1255–1264. [Google Scholar] [CrossRef] [PubMed]
- Chang, M.-L. Metabolic alterations and hepatitis C: From bench to bedside. World J. Gastroenterol. 2016, 22, 1461. [Google Scholar] [CrossRef]
- Knobler, H.; Malnick, S. Hepatitis C and insulin action: An intimate relationship. World J. Hepatol. 2016, 8, 131–138. [Google Scholar] [CrossRef] [PubMed]
- Brenachot, X.; Ramadori, G.; Ioris, R.M.; Veyrat-Durebex, C.; Altirriba, J.; Aras, E.; Ljubicic, S.; Kohno, D.; Fabbiano, S.; Clement, S.; et al. Hepatic protein tyrosine phosphatase receptor gamma links obesity-induced inflammation to insulin resistance. Nat. Commun. 2017, 8, 1820. [Google Scholar] [CrossRef]
- Pasc, P.; Berdea, D.E.; Dobjanschi, L.; Judea-Pusta, C.T.; Popescu, M.I. Descriptive Analysis of Real-World Medication Patterns and One-Year Outcomes of Acute Coronary Syndrome Patients with Metabolic Syndrome in a Tertiary Care Hospital. Farmacia 2021, 69, 498–508. [Google Scholar] [CrossRef]
- Yaribeygi, H.; Atkin, S.L.; Simental-Mendía, L.E.; Sahebkar, A. Molecular mechanisms by which aerobic exercise induces insulin sensitivity. J. Cell Physiol. 2019, 234, 12385–12392. [Google Scholar] [CrossRef]
- He, X.; Wu, D.; Hu, C.; Xu, T.; Liu, Y.; Liu, C.; Xu, B.; Tang, W. Role of Metformin in the Treatment of Patients with Thyroid Nodules and Insulin Resistance: A Systematic Review and Meta-Analysis. Thyroid 2019, 29, 359–367. [Google Scholar] [CrossRef]
- Kujawska-Luczak, M.; Szulinska, M.; Skrypnik, D.; Musialik, K.; Swora-Cwynar, E.; Kregielska-Narozna, M.; Markuszewski, L.; Grzymislawska, M.; Bogdanski, P. The influence of orlistat, metformin and diet on serum levels of insulin-like growth factor-1 in obeses women with and without insulin resistance. J. Physiol. Pharmacol. 2018, 69, 737–745. [Google Scholar] [CrossRef]
- Gower, B.A.; Fernández, J.R.; Beasley, T.M.; Shriver, M.D.; Goran, M.I. Using genetic admixture to explain racial differences in insulin-related phenotypes. Diabetes 2003, 52, 1047–1051. [Google Scholar] [CrossRef] [PubMed]

| No. | Name | Year | No. of Patients | Weeks | Diabetes | HOMA-IR Pre-Treatment | HOMA-IR Post-Treatment |
|---|---|---|---|---|---|---|---|
| 1. | Adinolfi et al. [18] | 2020 | 1303 | 24 | No | 4.38 ± 0.82 | 2.42 ± 0.58 |
| 2. | Cheng et al. [19] | 2019 | 102 | 12 | Mixed | 0.20 ± 0.09 | 0.19 ± 0.10 |
| 3. | Ichikawa et al. [20] | 2019 | 48 | 52 | No | 2.76 ± 1.72 | 3.22 ± 2.8 |
| 4. | Strauhs Nitsch et al. [21] | 2020 | 54 | 52 | Mixed | 3.12 ± 2.29 | 3.22 ± 2.04 |
| 5. | Elhelbawy et al. [22] | 2018 | 511 | 12 | Mixed | 3.21 ± 2.36 | 1.90 ± 1.58 |
| 6. | Ozdogan et al. [23] | 2020 | 121 | 24 | No | 3.13 ± 1.83 | 3.36 ± 2.20 |
| 7. | Ozdogan et al. [23] | 2020 | 121 | 52 | No | 3.13 ± 1.83 | 2.85 ± 1.420 |
| 8. | Graf et al. [24] | 2020 | 46 | 12 | No | 5.30 ± 6.10 | 3.90 ± 5.00 |
| 9. | Graf et al. [24] | 2020 | 44 | 24 | No | 5.30 ± 6.10 | 3.90 ± 5.00 |
| 10. | Graf et al. [24] | 2020 | 41 | 52 | No | 5.30 ± 6.10 | 2.50 ± 1.90 |
| 11. | Gitto et al. [25] | 2018 | 93 | 24 | Mixed | 3.80 ± 1.90 | 2.70 ± 1.50 |
| 12. | Doyle et al. [26] | 2019 | 24 | 12 | No | 2.70 ± 0.60 | 3.50 ± 0.80 |
| 13. | Doyle et al. [26] | 2019 | 24 | 24 | No | 3.20 ± 0.70 | 3.50 ± 0.80 |
| 14. | Chang et al. [27] | 2021 | 353 | 24 | Mixed | 3.25 ± 5.37 | 2.97 ± 3.09 |
| 15. | Lee et al. [28] | 2021 | 248 | 12 | Mixed | 3.21 ± 4.96 | 3.32 ± 5.23 |
| 16. | Lee et al. [28] | 2021 | 248 | 24 | Mixed | 3.21 ± 4.96 | 2.99 ± 2.72 |
| 17. | Lee et al. [28] | 2021 | 248 | 52 | Mixed | 3.21 ± 4.96 | 2.44 ± 2.61 |
| 18. | Alsebaey et al. [29] | 2019 | 385 | 12 | No | 2.78 ± 1.8 | 1.82 ± 1.46 |
| 19. | Alsebaey et al. [29] | 2019 | 126 | 12 | Yes | 5.16 ± 3.28 | 2.88 ± 2.68 |
| 20. | Moneim et al. [30] | 2019 | 30 | 12 | Yes | 2.89 ± 0.25 | 2.51 ± 12.00 |
| 21. | Cheng et al. [31] | 2022 | 95 | 24 | Mixed | 3.8 ± 0.2 | 2.6 ± 0.1 |
| 22. | Cheng et al. [31] | 2022 | 95 | 52 | Mixed | 3.8 ± 0.2 | 2.8 ± 0.2 |
| 23. | Ciancio et al. [32] | 2017 | 101 | 12 | Yes | 5.2 ± 2.5 | 3.1 ± 1.6 |
| 24. | Russo et al. [33] | 2019 | 135 | 12 | No | 3.0 ± 0.7 | 2.4 ± 0.5 |
| 25. | Russo et al. [33] | 2019 | 128 | 24 | No | 3.0 ± 0.7 | 1.9 ± 0.3 |
| 26. | Russo et al. [33] | 2019 | 124 | 52 | No | 3.0 ± 0.7 | 1.8 ± 0.4 |
| 27. | Lin et al. [34] | 2020 | 394 | 12 | Mixed | 2.05 ± 1.28 | 1.92 ± 1.18 |
| 28. | Alzahaby et al. [35] | 2018 | 20 | 12 | No | 3.20 ± 0.70 | 2.50 ± 0.80 |
| 29. | Alzahaby et al. [35] | 2018 | 20 | 12 | Yes | 6.80 ± 2.60 | 4.90 ± 2.0 |
| 30. | Nevola et al. [36] | 2020 | 343 | 12 | Mixed | 2.85 ± 0.74 | 2.15 ± 0.52 |
| 31. | Gualerzi et al. [37] | 2018 | 82 | 24 | Mixed | 3.42 ± 1.96 | 2.80 ± 1.02 |
| 32. | Hashim et al. [38] | 2022 | 25 | 12 | No | 5.33 ± 0.91 | 4.65 ± 0.97 |
| 33. | Hashim et al. [38] | 2022 | 25 | 12 | Yes | 7.14 ± 1.43 | 6.12 ± 1.47 |
| 34. | Salomone et al. [39] | 2017 | 32 | 12 | Mixed | 3.72 ± 0.81 | 2.32 ± 0.73 |
| 35. | Yosef et al. (treatment A) [40] | 2021 | 25 | 12 | Mixed | 3.19 ± 1.35 | 2.17 ± 0.96 |
| 36. | Yosef et al. (treatment B) [40] | 2021 | 25 | 12 | Mixed | 3.33 ± 1.31 | 2.38 ± 0.9 |
| 37. | Yosef et al. (treatment C) [40] | 2021 | 25 | 12 | Mixed | 2.9 ± 1.22 | 2.04 ± 0.76 |
| 38. | Yosef et al. (treatment D) [40] | 2021 | 25 | 12 | Mixed | 3.55 ± 1.48 | 2.38 ± 1.09 |
| Group | No. of Studies | Hedges’s g | p-Value |
|---|---|---|---|
| Duration | |||
| After 12 weeks | 20 | 0.624 | <0.001 * |
| After 24 weeks | 10 | 1.311 | 0.080 |
| After 52 weeks | 7 | 1.123 | 0.107 |
| Diabetic status | |||
| Mixed patients | 18 | 1.093 | 0.017 * |
| Non-T2DM | 15 | 0.658 | 0.013 * |
| T2DM | 4 | 1.055 | <0.001 * |
| TOTAL | 37 | 0.907 | <0.001 * |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Popescu, M.-S.; Drocas, A.I.; Osman, A.; Firu, D.-M.; Pădureanu, V.; Mărginean, C.M.; Pîrvu, D.-C.; Mitruț, R.; Mărgăritescu, D.N.; Radu, A.; et al. The Extent of Insulin Resistance in Patients That Cleared Viral Hepatitis C Infection and the Role of Pre-Existent Type 2 Diabetes Mellitus: A Meta-Analysis. Reports 2022, 5, 42. https://doi.org/10.3390/reports5040042
Popescu M-S, Drocas AI, Osman A, Firu D-M, Pădureanu V, Mărginean CM, Pîrvu D-C, Mitruț R, Mărgăritescu DN, Radu A, et al. The Extent of Insulin Resistance in Patients That Cleared Viral Hepatitis C Infection and the Role of Pre-Existent Type 2 Diabetes Mellitus: A Meta-Analysis. Reports. 2022; 5(4):42. https://doi.org/10.3390/reports5040042
Chicago/Turabian StylePopescu, Marian-Sorin, Andrei Ioan Drocas, Andrei Osman, Dan-Mihai Firu, Vlad Pădureanu, Cristina Maria Mărginean, Daniel-Cristian Pîrvu, Radu Mitruț, Dragoș Nicolae Mărgăritescu, Antonia Radu, and et al. 2022. "The Extent of Insulin Resistance in Patients That Cleared Viral Hepatitis C Infection and the Role of Pre-Existent Type 2 Diabetes Mellitus: A Meta-Analysis" Reports 5, no. 4: 42. https://doi.org/10.3390/reports5040042
APA StylePopescu, M.-S., Drocas, A. I., Osman, A., Firu, D.-M., Pădureanu, V., Mărginean, C. M., Pîrvu, D.-C., Mitruț, R., Mărgăritescu, D. N., Radu, A., Calina, D., Docea, A. O., & Mitruț, P. (2022). The Extent of Insulin Resistance in Patients That Cleared Viral Hepatitis C Infection and the Role of Pre-Existent Type 2 Diabetes Mellitus: A Meta-Analysis. Reports, 5(4), 42. https://doi.org/10.3390/reports5040042









