Powders Based on Ca2P2O7-CaCO3-H2O System as Model Objects for the Development of Bioceramics
Abstract
:1. Introduction
2. Materials and Methods
2.1. Synthesis of Powders
2.2. Preparation of Ceramic Samples
2.3. Characterization Methods
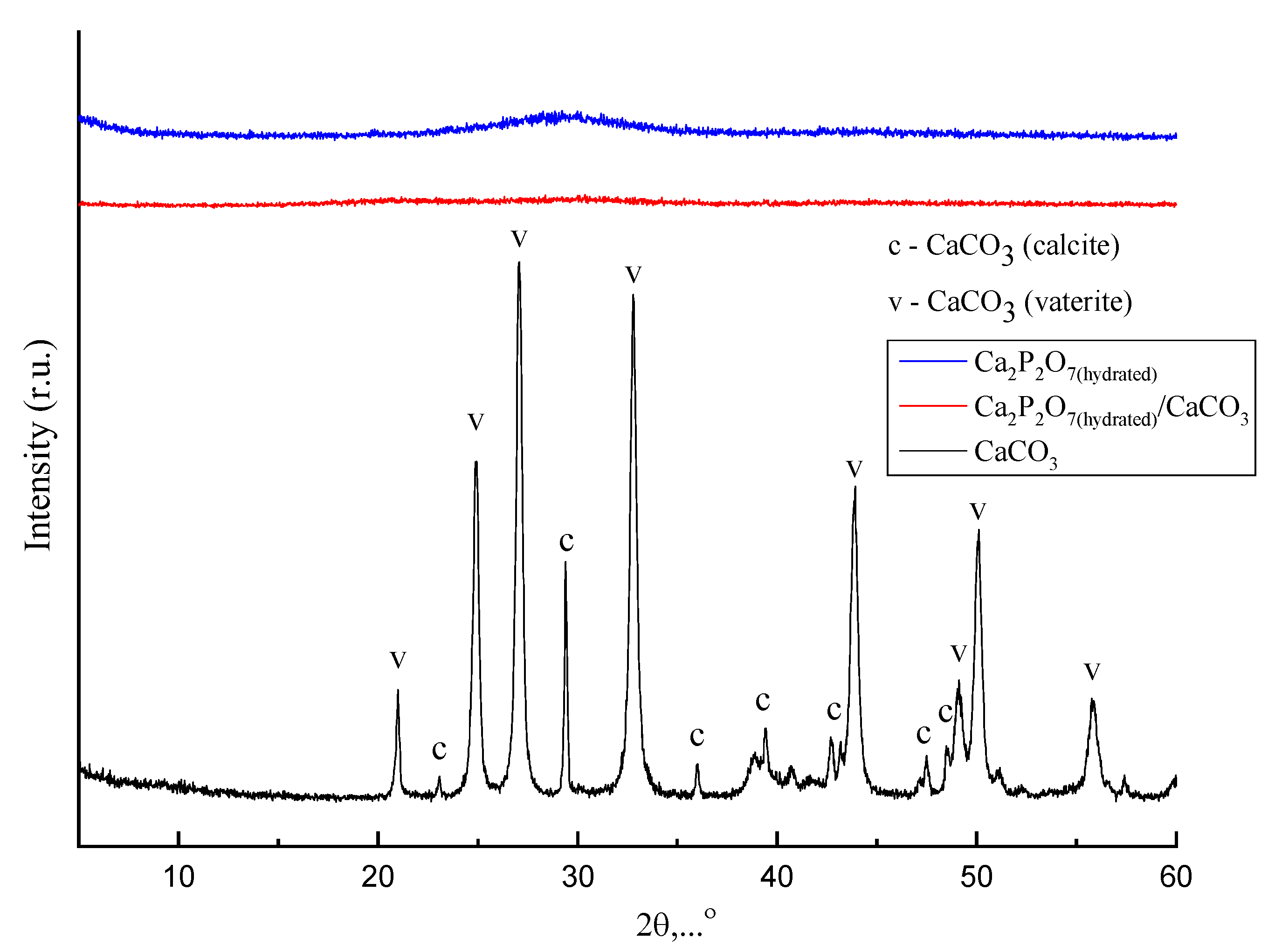
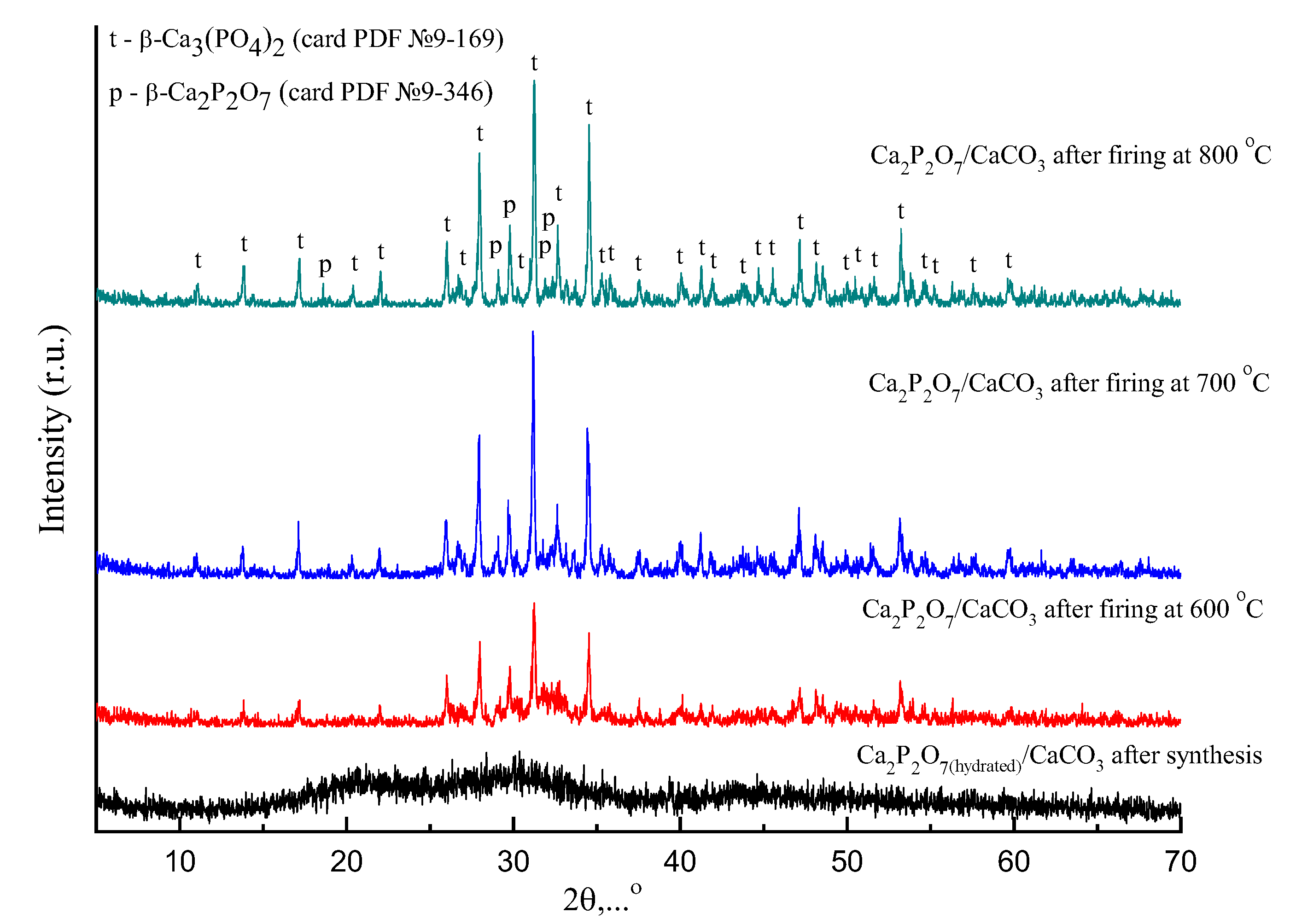
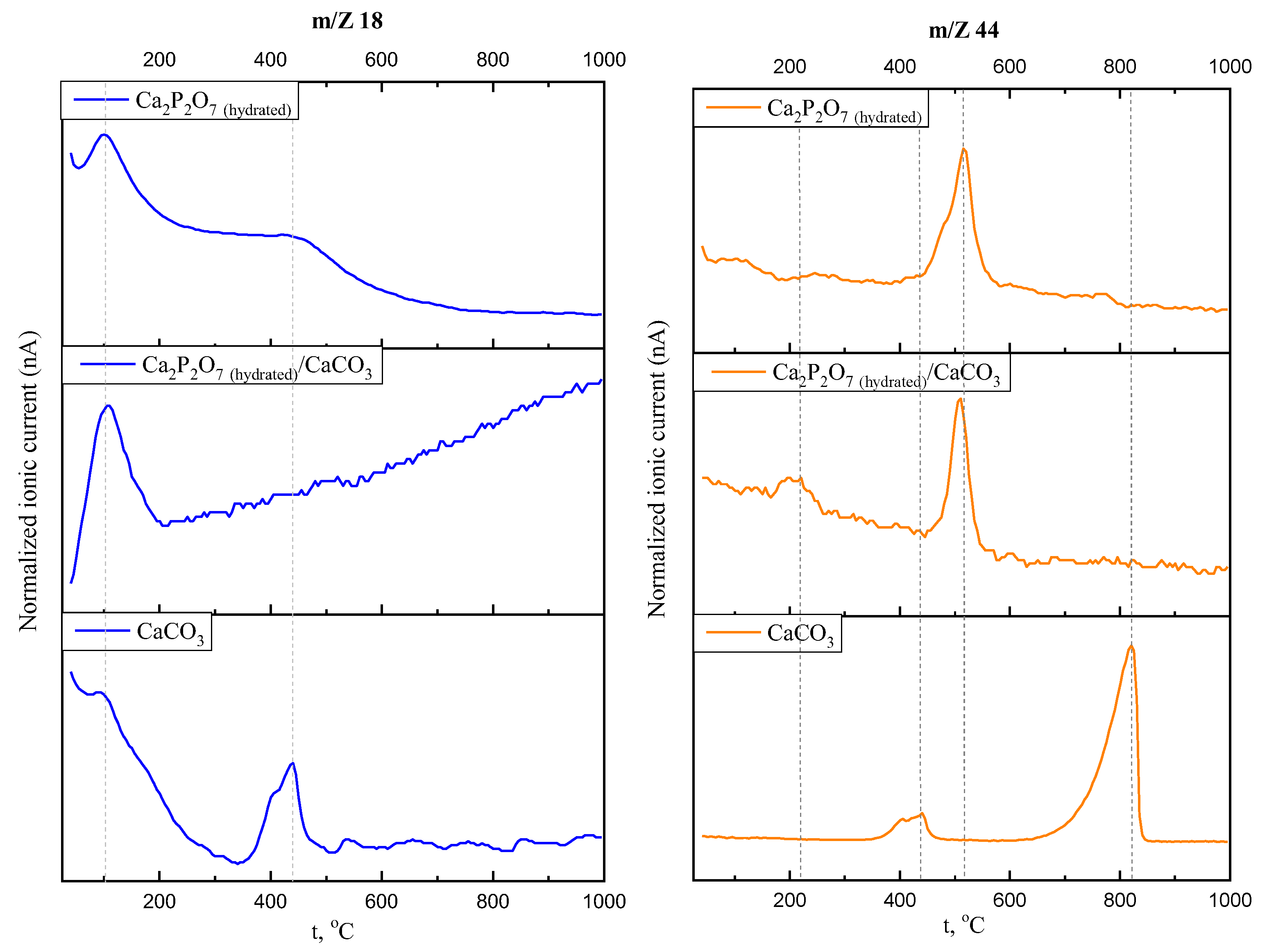
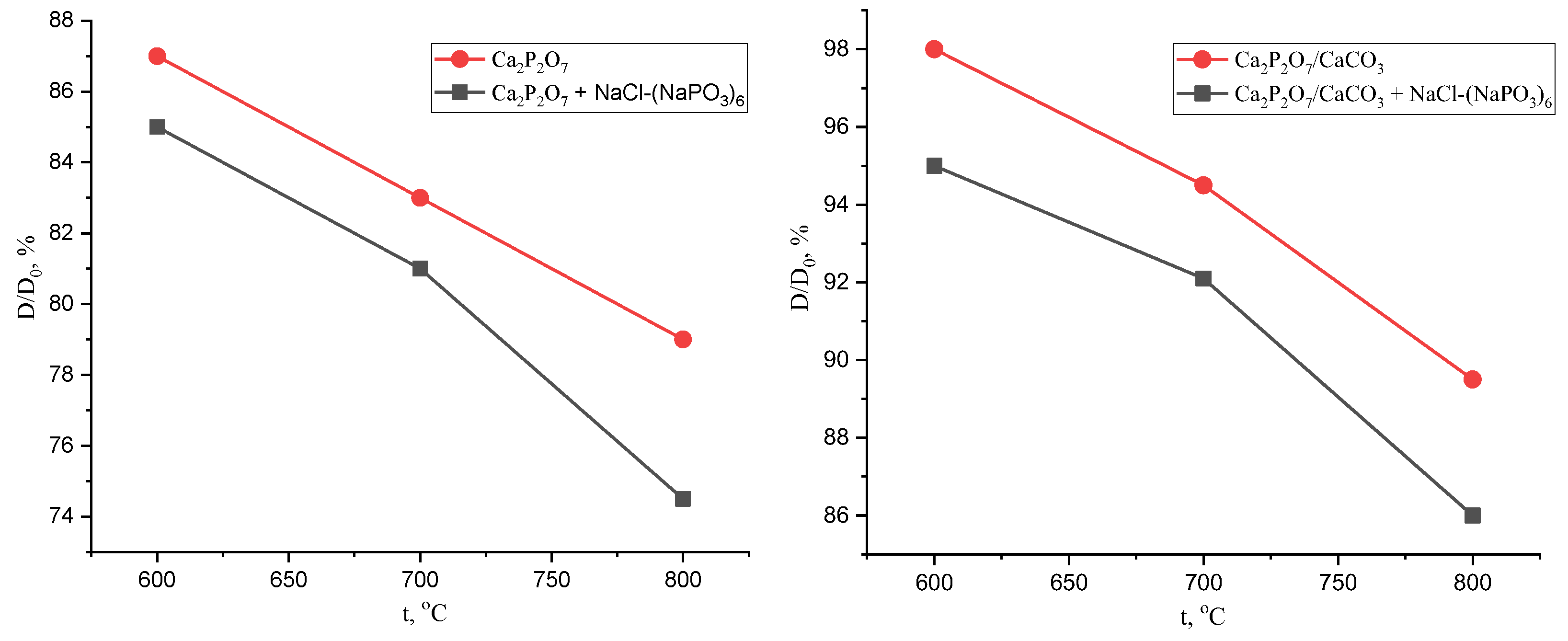
3. Results and Discussions
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Fiume, E.; Magnaterra, G.; Rahdar, A.; Verné, E.; Baino, F. Hydroxyapatite for Biomedical Applications: A Short Overview. Ceramics 2021, 4, 542. [Google Scholar] [CrossRef]
- Yao, Y.; Qin, W.; Xing, B.; Sha, N.; Jiao, T.; Zhao, Z. High performance hydroxyapatite ceramics and a triply periodic minimum surface structure fabricated by digital light processing 3D printing. J. Adv. Ceram. 2021, 10, 39–48. [Google Scholar] [CrossRef]
- Esslinger, S.; Grebhardt, A.; Jaeger, J.; Kern, F.; Killinger, A.; Bonten, C.; Gadow, R. Additive Manufacturing of β-Tricalcium Phosphate Components via Fused Deposition of Ceramics (FDC). Materials 2021, 14, 156. [Google Scholar] [CrossRef]
- Fadeeva, I.V.; Ryzhov, A.P.; Titov, D.D.; Filippov, Y.Y.; Tyut’Kova, Y.B.; Davydova, G.A.; Barinov, S.M. Barium-Substituted Tricalcium Phosphate Ceramics. Inorg. Mater. 2022, 58, 317–324. [Google Scholar] [CrossRef]
- Safronova, T.V.; Putlyaev, V.I.; Knot’Ko, A.V.; Krut’Ko, V.K.; Musskaya, O.N.; Ulasevich, S.A.; Vorob’Eva, N.A.; Telitsin, V.D. Calcium Phosphate Ceramic in the System Ca(PO3)2–Ca2P2O7 Based on Powder Mixtures Containing Calcium Hydrophosphate. Glass Ceram. 2018, 75, 279–286. [Google Scholar] [CrossRef]
- Moussa, H.; Jiang, W.; Alsheghri, A.; Mansour, A.; El Hadad, A.; Pan, H.; Tang, R.; Song, J.; Vargas, J.; McKee, M.D.; et al. High strength brushite bioceramics obtained by selective regulation of crystal growth with chiral biomolecules. Acta Biomater. 2020, 106, 351–359. [Google Scholar] [CrossRef]
- Boanini, E.; Pagani, S.; Tschon, M.; Rubini, K.; Fini, M.; Bigi, A. Monetite vs. Brushite: Different Influences on Bone Cell Response Modulated by Strontium Functionalization. J. Funct. Biomater. 2022, 13, 65. [Google Scholar] [CrossRef] [PubMed]
- Hu, J.; Zhang, R.; Chen, H.; Wu, Y.; Chen, L.; Zhang, Q.; Ren, H.; Yan, Y. The study on calcium polyphosphate/poly-amino acid composite for supportive bone substitute materials in vitro. New J. Chem. 2021, 45, 8072–8082. [Google Scholar] [CrossRef]
- Safronova, T.; Kiselev, A.; Selezneva, I.; Shatalova, T.; Lukina, Y.; Filippov, Y.; Toshev, O.; Tikhonova, S.; Antonova, O.; Knotko, A. Bioceramics Based on β-Calcium Pyrophosphate. Materials 2022, 15, 3105. [Google Scholar] [CrossRef]
- Lee, J.H.; Chang, B.-S.; Jeung, U.-O.; Park, K.-W.; Kim, M.-S.; Lee, C.-K. The first clinical trial of beta-calcium pyrophosphate as a novel bone graft extender in instrumented posterolateral lumbar fusion. Clin. Orthop. Surg. 2011, 3, 238–244. [Google Scholar] [CrossRef]
- Lobo, S.; Arinzeh, T. Biphasic Calcium Phosphate Ceramics for Bone Regeneration and Tissue Engineering Applications. Materials 2010, 3, 815. [Google Scholar] [CrossRef]
- Laskus, A.; Kolmas, J. Ionic Substitutions in Non-Apatitic Calcium Phosphates. Int. J. Mol. Sci. 2017, 18, 2542. [Google Scholar] [CrossRef] [PubMed]
- Elkhouly, H.; Mamdouh, W.; El-Korashy, D.I. Electrospun nano-fibrous bilayer scaffold prepared from polycaprolactone/gelatin and bioactive glass for bone tissue engineering. J. Mater. Sci. Mater. Med. 2021, 32, 111. [Google Scholar] [CrossRef] [PubMed]
- Peranidze, K.; Safronova, T.V.; Kildeeva, N.R. Fibrous Polymer-Based Composites Obtained by Electrospinning for Bone Tissue Engineering. Polymers 2021, 14, 96. [Google Scholar] [CrossRef]
- Zhang, X.; Meng, S.; Huang, Y.; Xu, M.; He, Y.; Lin, H.; Han, J.; Chai, Y.; Wei, Y.; Deng, X. Electrospun Gelatin/β-TCP Composite Nanofibers Enhance Osteogenic Differentiation of BMSCs and In Vivo Bone Formation by Activating Ca2+-Sensing Receptor Signaling. Stem Cells Int. 2015, 2015, 507154. [Google Scholar] [CrossRef] [PubMed]
- Pritzker, K.P. Crystal-associated arthropathies: What’s new in old joints. J. Am. Geriatr. Soc. 1980, 28, 439–445. [Google Scholar] [CrossRef]
- Mitrovic, D.R. Pathology of articular deposition of calcium salts and their relationship to osteoarthrosis. Ann. Rheum. Dis. 1983, 42, 19–26. [Google Scholar] [CrossRef]
- Filippov, Y.Y.; Murashko, A.M.; Evdokimov, P.V.; Safronova, T.V.; Putlayev, V.I. Stereolithography 3D printed calcium pyrophosphate microporous ceramics for bone grafting. Open Ceram. 2021, 8, 100185. [Google Scholar] [CrossRef]
- Winkler, T.; Sass, F.A.; Duda, G.N.; Schmidt-Bleek, K. A review of biomaterials in bone defect healing, remaining shortcomings and future opportunities for bone tissue engineering: The unsolved challenge. Bone Jt. Res. 2018, 7, 232–243. [Google Scholar] [CrossRef]
- Lin, F.H.; Liao, C.J.; Chen, K.S.; Sun, J.S.; Liu, H.C. Degradation behaviour of a new bioceramic: Ca2P2O7 with addition of Na4P2O7·10H2O. Biomaterials 1997, 18, 915–921. [Google Scholar] [CrossRef]
- Safronova, T.V.; Kuznetsov, A.; Korneychuk, S.; Putlyaev, V.; Shekhirev, M. Calcium phosphate powders synthesized from solutions with [Ca2+]/[PO43−] = 1 for bioresorbable ceramics. Cent. Eur. J. Chem. 2009, 7, 184–191. [Google Scholar] [CrossRef]
- Safronova, T.V.; Sadilov, I.S.; Chaikun, K.V.; Shatalova, T.B.; Filippov, Y.Y. Ceramics Based on a Powder Mixture of Calcium Hydroxyapatite, Monocalcium Phosphate Monohydrate, and Sodium Hydrogen Phosphate Homogenized under Mechanical Activation Conditions. Inorg. Mater. Appl. Res. 2020, 11, 879–885. [Google Scholar] [CrossRef]
- Safronova, T.V.; Putlyaev, V.I.; Filippov, Y.Y.; Shatalova, T.B.; Naberezhnyi, D.O.; Nasriddinov, A.F.; Larionov, D.S. Ceramics Based on Powder Mixtures Containing Calcium Hydrogen Phosphates and Sodium Salts (Na2CO3, Na4P2O7, and NaPO3). Inorg. Mater. 2018, 54, 724–735. [Google Scholar] [CrossRef]
- Safronova, T.V.; Putlyaev, V.I.; Shekhirev, M.A.; Tretyakov, Y.D.; Kuznetsov, A.V.; Belyakov, A.V. Densification additives for hydroxyapatite ceramics. J. Eur. Ceram. Soc. 2009, 29, 1925–1932. [Google Scholar] [CrossRef]
- Shiryaev, M.; Safronova, T.; Putlyaev, V. Calcium phosphate powders synthesized from calcium chloride and potassium hydrophosphate. J. Therm. Anal. Calorim. 2010, 101, 707–713. [Google Scholar] [CrossRef]
- Safronova, T.V.; Shatalova, T.B.; Filippov, Y.Y.; Krut’ko, V.K.; Musskaya, O.N.; Safronov, A.S.; Toshev, O.U. Ceramics in the Ca2P2O7–Ca(PO3)2 System Obtained by Annealing of the Samples Made from Hardening Mixtures Based on Calcium Citrate Tetrahydrate and Monocalcium Phosphate Monohydrate. Inorg. Mater. Appl. Res. 2020, 11, 777–786. [Google Scholar] [CrossRef]
- Safronova, T.F.; Putlyaev, V.I.; Filippov, Y.; Larionov, D.S.; Evdokimov, P.V.; Averina, A.E.; Klimashina, E.S.; Ivanov, V.K. Porous ceramics based on calium pyrophosphate. New Refract. 2015, 1, 46–51. [Google Scholar]
- Sudareva, N.N.; Popryadukhin, P.V.; Saprykina, N.N.; Suvorova, O.M.; Yukina, G.Y.; Galibin, O.V.; Vilesov, A.D. CaCO3 vaterites as components of target drug delivery systems. Cell Ther. Transplant. 2020, 9, 13–19. [Google Scholar] [CrossRef]
- Safronova, T.V.; Putlyaev, V.I.; Filippov, Y.Y.; Knot’Ko, A.V.; Klimashina, E.S.; Peranidze, K.K.; Evdokimov, P.V.; Vladimirova, S.A. Powders synthesized from calcium acetate and mixed-anionic solutions, containing orthophosphate and carbonate ions, for obtaining bioceramic. Glass Ceram. 2018, 75, 118–123. [Google Scholar] [CrossRef]
- Kumar, T.S.; Manjubala, I.; Gunasekaran, J. Synthesis of carbonated calcium phosphate ceramics using microwave irradiation. Biomaterials 2000, 21, 1623–1629. [Google Scholar] [CrossRef]
- Lardani, L.; Derchi, G.; Marchio, V.; Carli, E. One-Year Clinical Performance of Activa™ Bioactive-Restorative Composite in Primary Molars. Children 2022, 9, 433. [Google Scholar] [CrossRef] [PubMed]
- Safronova, T.V.; Kurbatova, S.A.; Shatalova, T.B.; Knotko, A.V.; Yevdokimov, P.V.; Putlyayev, V.I. Calcium Pyrophosphate Powder for Production of Bioceramics Synthesized from Pyrophosphoric Acid and Calcium Acetate. Inorg. Mater. Appl. Res. 2017, 8, 118–125. [Google Scholar] [CrossRef]
- He, F.; Yang, F.; Zhu, J.; Peng, Y.; Tian, X.; Chen, X. Fabrication of a Novel Calcium Carbonate Composite Ceramic as Bone Substitute. J. Am. Ceram. Soc. 2014, 98, 223–228. [Google Scholar] [CrossRef]



| Target Product | Labeling | Concentrations | ||
|---|---|---|---|---|
| (NH4)2CO3 | (NH4)2P2O7 Previously Obtained | (CH3COO)2Ca | ||
| CaCO3 | CC | 0.5 M | - | 0.5 M |
| Ca2P2O7·xH2O/CaCO3 | CPP/CC | 0.125 M | 0.125 M | 0.5 M |
| Ca2P2O7·xH2O | CPP | - | 0.25 M | 0.5 M |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Peranidze, K.; Safronova, T.V.; Filippov, Y.; Kazakova, G.; Shatalova, T.; Rau, J.V. Powders Based on Ca2P2O7-CaCO3-H2O System as Model Objects for the Development of Bioceramics. Ceramics 2022, 5, 423-434. https://doi.org/10.3390/ceramics5030032
Peranidze K, Safronova TV, Filippov Y, Kazakova G, Shatalova T, Rau JV. Powders Based on Ca2P2O7-CaCO3-H2O System as Model Objects for the Development of Bioceramics. Ceramics. 2022; 5(3):423-434. https://doi.org/10.3390/ceramics5030032
Chicago/Turabian StylePeranidze, Kristina, Tatiana V. Safronova, Yaroslav Filippov, Gilyana Kazakova, Tatiana Shatalova, and Julietta V. Rau. 2022. "Powders Based on Ca2P2O7-CaCO3-H2O System as Model Objects for the Development of Bioceramics" Ceramics 5, no. 3: 423-434. https://doi.org/10.3390/ceramics5030032
APA StylePeranidze, K., Safronova, T. V., Filippov, Y., Kazakova, G., Shatalova, T., & Rau, J. V. (2022). Powders Based on Ca2P2O7-CaCO3-H2O System as Model Objects for the Development of Bioceramics. Ceramics, 5(3), 423-434. https://doi.org/10.3390/ceramics5030032








