Abstract
A preliminary description is presented of the well-preserved frozen mummies of two cubs of the extinct cave lion Panthera spelaea (finds of 2017–2018, Semyuelyakh River, Yakutia, eastern Siberia, Russia). The fossil lion cubs were found in close proximity, but they do not belong to the same litter, since their radiocarbon ages differ: the female (named ‘Sparta’) was dated to 27,962 ± 109 uncal years BP, and the male (named ‘Boris’) was dated to 43,448 ± 389 uncal years BP. The lion cubs have similar individual ages, 1–2 months. The general tone of the colour of the fur coat of Sparta is greyish to light brown, whereas, in Boris, the fur is generally lighter, greyish yellowish. It is, therefore, possible that light colouration prevailed with age in cave lions and was adaptive for northern snow-covered landscapes. The article discusses the results of computed tomography of cubs of the cave lion, the possible reasons for their death, and the peculiarities of their existence in the Siberian Arctic.
1. Introduction
The Pleistocene Siberian Arctic zone was replete with large mammals and is astonishingly different from the Arctic zone of the present day. Along with mammoth (Mammuthus primigenius), woolly rhinoceros (Coelodonta antiquitatis), bison (Bison priscus), and saiga (Saiga tatarica), lived the cave lion (Panthera spelaea), a close relative of contemporary Afro-Asiatic lion P. leo L. It was on top of the trophic pyramid of the land polar ecosystem, being a part of the large Pleistocene carnivore guild alongside wolf Canis lupus L., wolverine Gulo gulo L. and brown bear Ursus arctos L. It is still unknown exactly how cave lions adapted to life in the harsh conditions of the high latitudes with their rapid season periodicity, strong winds, and cold and long winters with associated continuous nights.
Mummified remains have provided valuable information about how mammoths [1,2,3], rhinoceros [4], horses [5], and bison [6] have adapted to Pleistocene north Siberian conditions. Thus far, there are only four such finds of Panthera spelaea cubs, all in varying states of preservation. All four cubs were found between 2015 to 2018 in Yakutia, in the river basin of the Indigirka River. The first two were found in 2015 in the Uyandina River (Figure 1) and were only able to be examined quite superficially [7,8]. In this article, we will provide a preliminary description of two new mummified cave lion cubs from Yakutia.

Figure 1.
The locations of the cave lion cubs finds: on the Uyandina River (indicated by a red triangle) and on the Semyuelyakh River (indicated by a red asterisk).
2. Place of the Find and Geological Age
In 2017, Boris Berezhnev, a local resident and a licensed mammoth tusk collector, while searching for mammoth tusks, found a lion cub, named ‘Boris’ (Figure 1), on the Semyuelyakh River (a tributary stream of the Tirekhtyakh River, river basin of the Indigirka River). In 2018, only 15 m away from this find, the fossil of a second lion cub was found, a female cub that was named ‘Sparta’ (initially ‘Spartak’). The lion cubs were discovered in the shoreline denudation at a depth of 10 to 12 m from the top of the Yedoma Late Pleistocene deposits (glacial complex). Bone remains from members of the typical fauna of the Yakutian mammoth steppe were also found in the same location: wolf, brown bear, woolly mammoth, woolly rhinoceros, Lena horse, reindeer, and steppe bison.
A mummy of a bird was also discovered in the same location as these other finds. The bird was radiocarbon dated to 42,600 ± 1100 years BP (OxA-38572), which corresponds to a calibrated age between 44,163–48,752 cal years BP. The bird carcass was genetically identified as a female horned lark (Eremophila alpestris). This is a species that usually inhabits open habitat such as the steppe environment that existed in Siberia at the time [9].
3. Material and Methods
In November 2018, the frozen remains of Boris were delivered to Yakutsk and microbiologically tested in the sanitary-epidemiological station of Yakutsk for possible carriage of infectious diseases (including foot and mouth disease, anthrax, brucellosis, and rinderpest), all of which yielded negative results. The frozen remains of Sparta were delivered to Yakutsk in January 2019 and were also tested for infectious diseases; they also yielded negative results. The results of the present paper are based on the studies of the exterior morphology of the cave lion cubs’ mummies conducted in November 2018 and May 2019, as well as genetic-based sex determination.
The cave lion cub mummies were described and measured following the methods proposed for large, wild mammals by Gromov et al. [10]. Computed tomography was carried out on both cubs (Boris, December 2017; Sparta, December 2018) on a computed tomography scanner (Siemens Somatom Emotion 16 slice, Yakutsk city) to investigate the condition of their bones and soft tissue. Radiocarbon analyses of the lion cubs were carried out by the Institute for High-Dimensional Medical Imaging, Jikei University, Tokyo, Japan (code IAAA).
3.1. Sampling and DNA Lab Work
In order to molecularly determine sex, we extracted DNA, built double-stranded Illumina libraries, and shotgun-sequenced the two cave lion samples of hair and skin following the approach of Stanton et al. [11]. Briefly, the tissue was digested in a buffer optimised to digest keratin-rich tissues [12], before extracting DNA using the methodology of Yang et al. [13]. We built double-stranded Illumina libraries according to Meyer and Kircher [14], including 3U of USER enzyme (New England Biolabs) to excise uracil residues resulting from post-mortem damage. This library was then used as stock for 10 and 13 indexing PCR amplifications (for ‘Boris’ and ‘Sparta’, respectively) using double-unique p5–p7 indexed primers. Amplified libraries were then pooled in approximately equimolar amounts. Purification and size selection of the pooled libraries was performed using Agencourt AMPure XP beads (Beckman Coulter, Brea, CA, USA) to remove long and short fragments. We ran the libraries on a high-sensitivity DNA chip on a Bioanalyzer 2100 (Agilent, Santa Clara, CA, USA) and sequenced them on an Illumina NovaSeq6000 S4 (150 bp paired-end [PE] setup) at the SciLifeLab sequencing facility in Stockholm.
3.2. Data Processing and Molecular Sexing
Sequencing reads were trimmed and overlapping reads merged using SeqPrep v1.137 (https://github.com/jstjohn/SeqPrep (accessed on 5 February 2020)). with default settings but with a minor modification in the source code, allowing us to choose the best quality scores of bases in the merged region instead of aggregating the scores following Palkopoulou et al. [15]. We hard masked repeat regions using RepeatModeller v1.0.11 [16]. Merged reads were then mapped to this masked cat genome using BWA-ALN v0.7.13 [17], using the slightly modified default settings of deactivated seeding (−l 16,500), more substitutions (−n 0.01), and allowing up to two gaps (−o 2). DNA analysis and duplicates were removed (separately for each PCR reaction) using a custom perl script that removes reads with identical start and end positions, keeping the first observed such read. In total, 10.8 M and 18.1 M unique reads were mapped to the reference genome for ‘Boris’ and ‘Sparta’, respectively, and determined sex by calculating the coverage. The null hypothesis is that the coverage is not significantly lower for the X chromosome versus the autosomes, and the individual is, therefore, a female. If the coverage is significantly lower for the X chromosome, the alternative hypothesis of the individual being a male can be accepted.
3.3. Method for CT Inspection
We acquired the internal structure of each body as image data by X-ray CT examination. This allowed us to non-destructively obtain anatomical features of the body such as the skeletal shape and the residual state of soft tissues. In normal X-ray CT examination, cross-sectional images are obtained in the axial, sagittal, and coronal directions with respect to the body axis of the target organism. However, since remains found in the permafrost have been subjected to external forces for long periods of time, they display complicated aspects such as partial twisting of the body axis, local changes in posture, and deformation due to pressure. Therefore, in order to obtain the most anatomically accurate axial image possible, the tomographic image direction has to be slightly changed for each part of the body.
For each object, we acquired a continuous tomographic image with as fine a slice thickness and narrow tomographic interval as possible, with respect to the body axis at the time of CT imaging. We constructed a three-dimensional object with equal resolution in the X, Y, and Z directions by interpolating the voxel data. From these volume data, we interpreted the anatomical features of the hard and soft tissues of each individual. This was achieved by creating an appropriate cross-sectional image in the axial, sagittal, and coronal directions, taking into account the twist of the body axis and the change of posture of the body part being analysed. The volume data were converted into a three-dimensional image using a volume rendering method whereby the original anatomical form was displayed as a three-dimensional stereoscopic image, and the image was read while changing the window value and the level value. These three-dimensional images were then image-cut on a surface in an arbitrary direction to observe the interior of specific organs and measure the distance, volume, etc.
3.4. Radiocarbon Dating Method
Body tissue samples (hair, skin, and muscle) were treated with an HCl solution (0.2 M) in order to chemically remove any contaminant, and collagens were extracted from bone samples. These samples were oxidised by heating to produce CO2 gas. The CO2 gas samples were purified in a vacuum line and reduced to graphite by hydrogen using iron as a catalyst. The graphite samples were pressed into target holders. The graphite samples were measured against a standard of oxalic acid (HOxII) provided by the National Institute of Standards and Technology (USA), using a 14 C-AMS (accelerator mass spectrometry) system based on the tandem accelerator in the Institute of Accelerator Analysis Ltd. 14 C ages (conventional 14 C age: yrBP) were calculated with δ13 C correction [18]. 14 C ages were calibrated by OxCal v4.3 and v.4.4 [19] based on the IntCal13 [20] and IntCal20 databases [21].
3.5. Age Estimation of the Cave Lion Cubs
Individual (ontogenic) age estimation is based on Smuts’ investigation [22], which is the only reference for decidual tooth eruption detailed sequences for extant African lions (43 skulls and mandibles from South Africa). From CT scans of Sparta and Boris’ upper and lower jaws, individual ages can be estimated by comparison with the eruption stages of milk incisors and premolars for extant African lions. The nomenclature used is as follows: D/d = upper/lower decidual tooth, respectively; i = incisor, c = canine; p = premolar.
4. Results
4.1. Radiocarbon Dating
Radiocarbon dating showed that although both individuals lived during the Karginian interstadial of Late Pleistocene, Boris was estimated to be 43,448 ± 389 years BP old (IAAA-180050), while Sparta was estimated to be 27,962 ± 109 years BP old (IAAA-181499).
4.2. Sex Determination
4.2.1. Sex Determination by Using CT Scanner
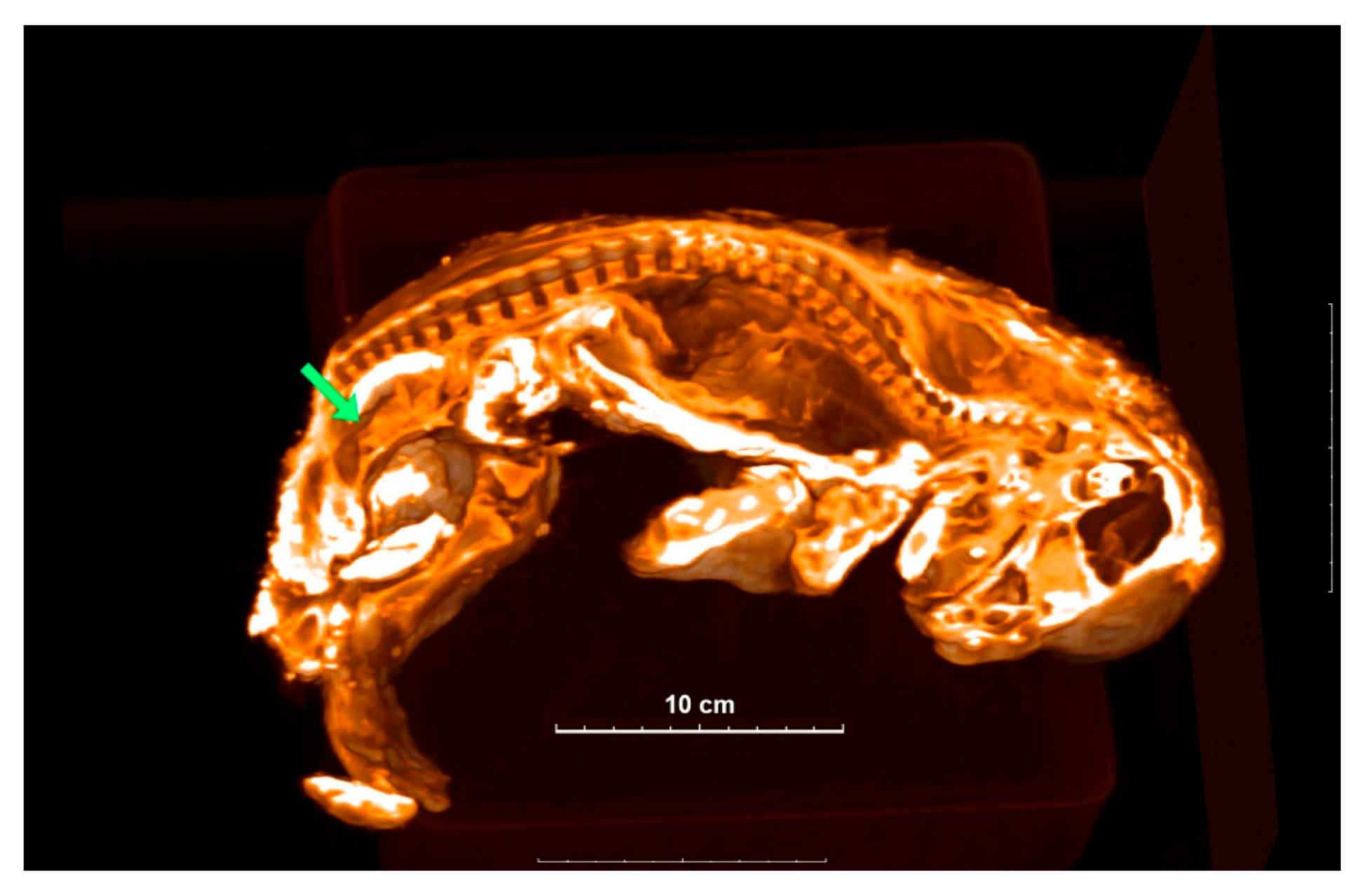
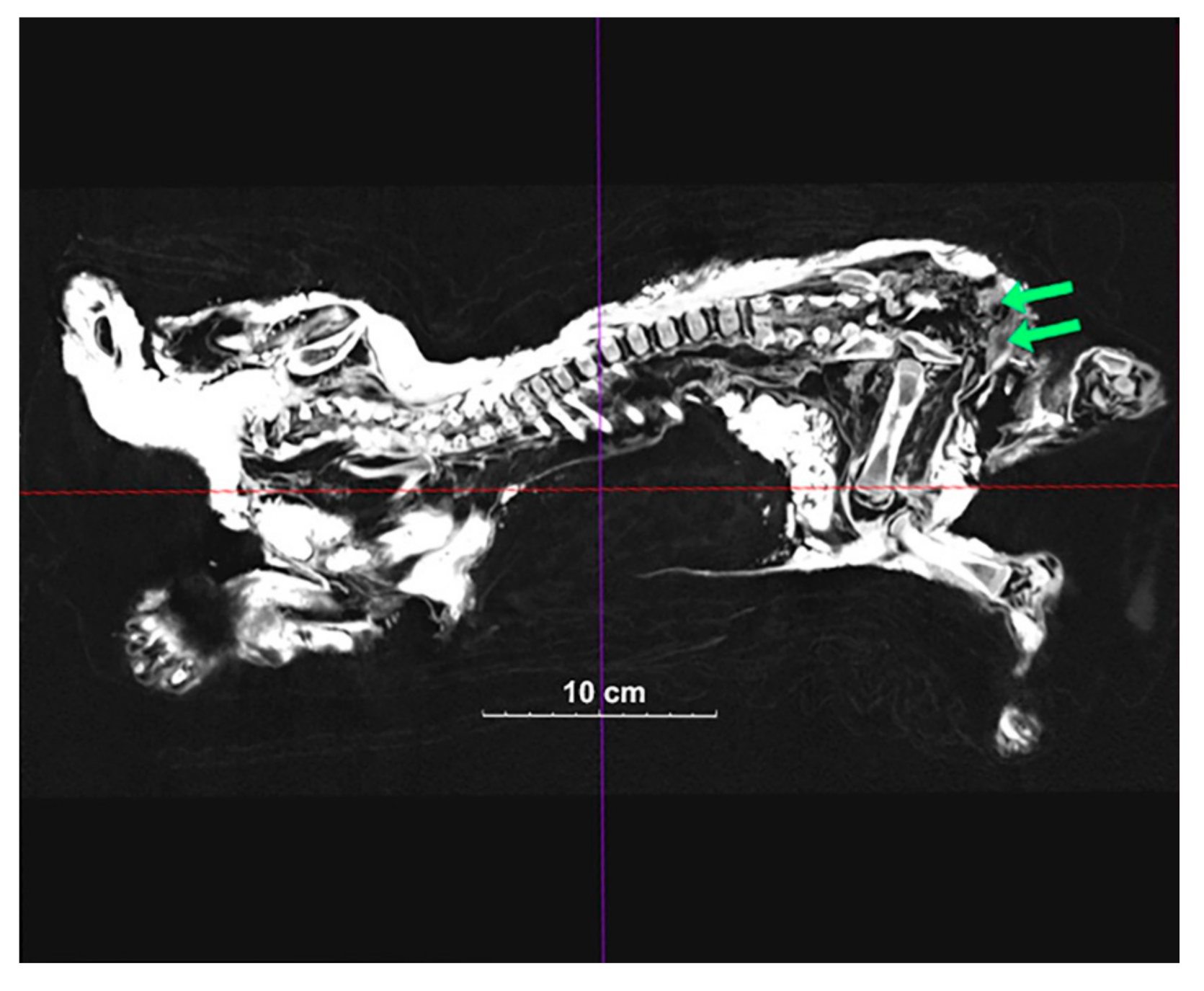
Computed tomography of Sparta and Boris identified structures that appear to be reproductive organs (Figure 2 and Figure 3). This allowed us to confidently assume that Sparta is a female, and Boris is a male. Body size does also suggest a differential sexual identification, with sexual dimorphism appearing quite early in the growing stages of extant African lions.
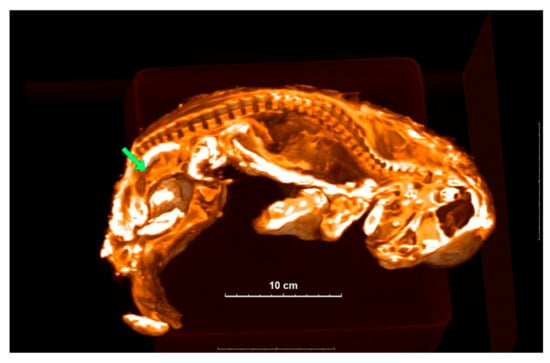
Figure 2.
A 3D image of Sparta is sectioned to show the inside structure. Green arrow shows the location of the uterus-like organ.
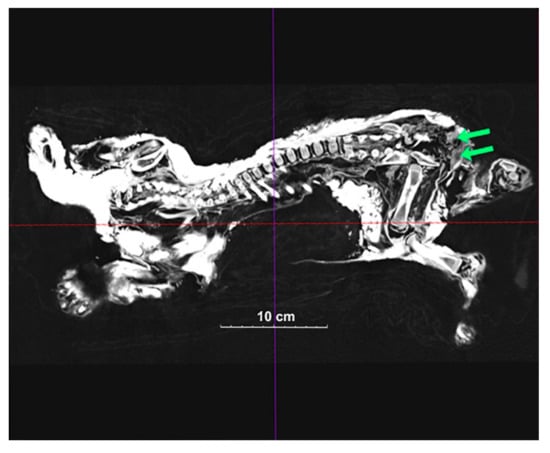
Figure 3.
A 2D image of Boris’ inside structure. Green arrows show the testicle-like organs.
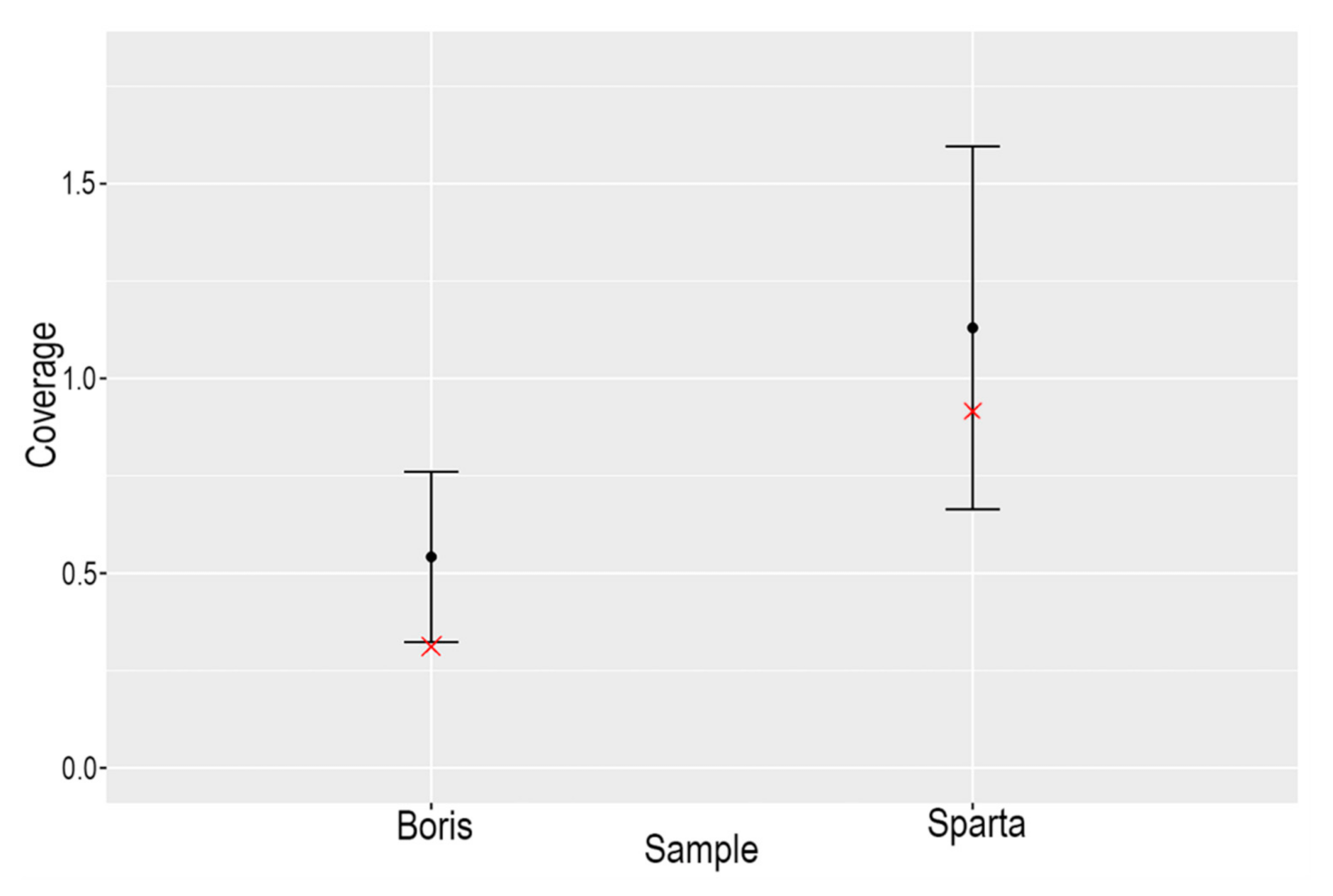
4.2.2. Molecular Sexing
Figure 4 shows the coverage of the X chromosomes versus the autosomes for ‘Boris’ and ‘Sparta’. In the case of Boris, the coverage on the X chromosome falls outside two standard deviations of the mean coverage of the autosomes, indicating that the individual is male, whereas Spartak does not, indicating that the individual is female.
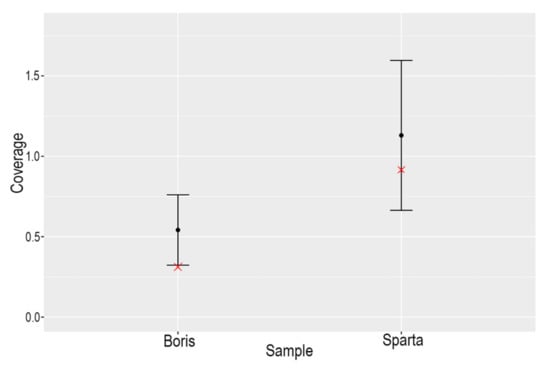
Figure 4.
Plot of the coverage of the X chromosome (red ‘✕’) for Boris and Sparta versus two standard deviations of the coverage of the autosomes for each individual.
4.3. Dentition and Age Estimation
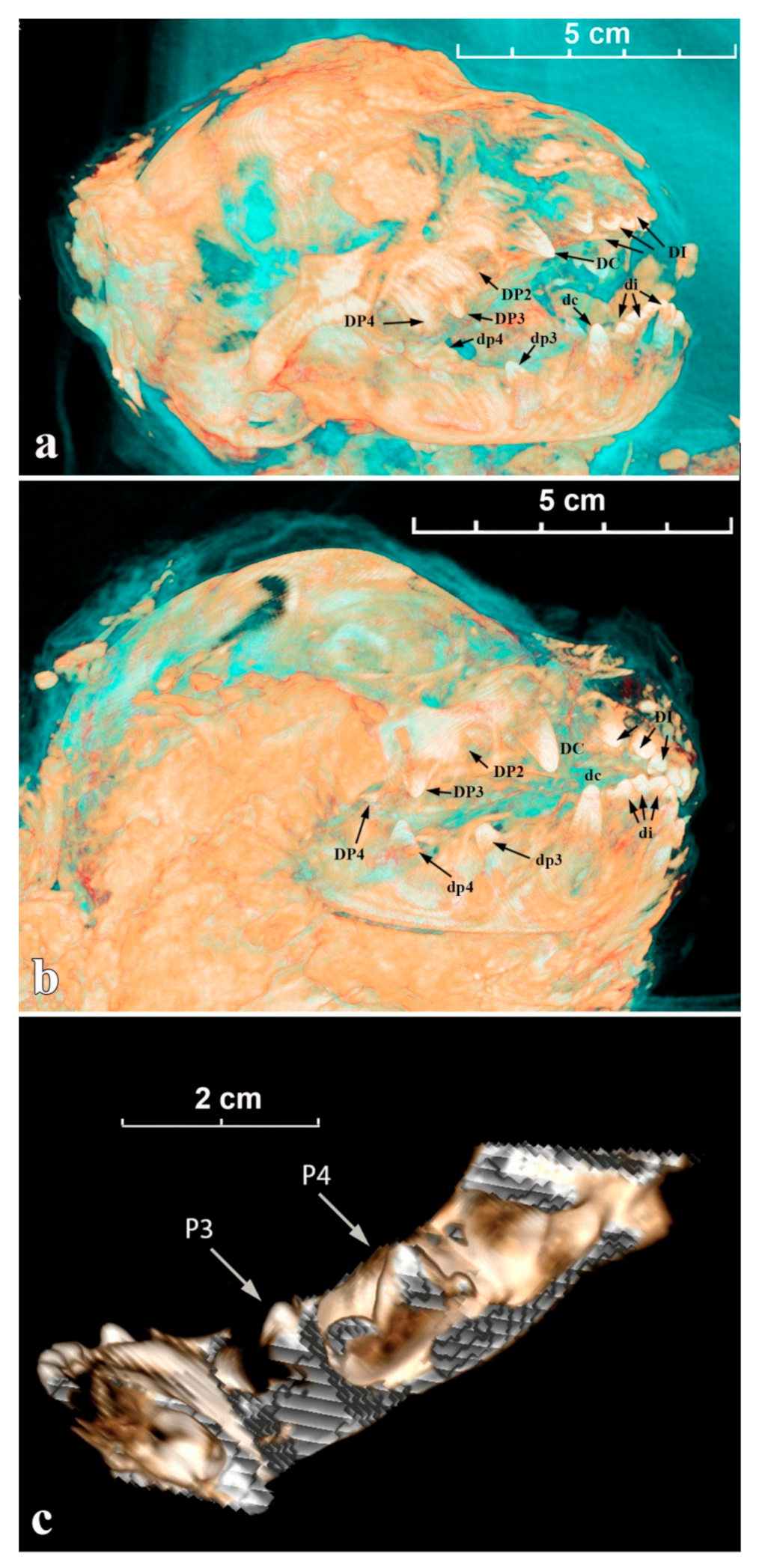
In the extant African lion, DP3 and dp4 erupt at the age of 1–2 months (±1 month), usually followed by dp3 and then DP4. DP2 is visible in the alveolus and protrudes above it at the age of 1–2 months. DP2 erupts completely and a full erupted complement of milk teeth is available at 2–3 months of age [22].
For Sparta, all incisors (DI/di) and the decidual canines (DC/dc) have started to erupt, their crowns are well visible above the gum; the top parts of DP2, DP3, and DP4 (= carnassial) are also distinguishable (Figure 5a). On the lateral view of the mandible (Figure 5c), less than ½ of the crown of dc is erupting above the gum; dp3 has just broken the gum (protoconid); dp4 is still totally in its socket and the top of the protoconid is close to the gum. For Boris, the observations are comparable to the ontogenetic data described above for Sparta (Figure 5b).
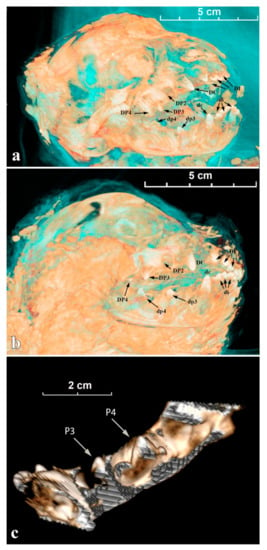
Figure 5.
The tomographic images of cave lion cubs’ skulls and mandibles with marked deciduous teeth: (a) Sparta’s skull; (b) Boris’s skull; (c) Sparta’s mandible.
For a modern P. leo, these data would show the lion cub is approximately 1–2 months old [22,23], and we, therefore, estimate that this is the individual ages of both Sparta and Boris.
The small head and body size of Sparta and Boris (see below) do not correlate with the dentition, which indicates that the postnatal development of cave lion cubs was much faster than that of a contemporary lion cub. This may be due to the short periods of warm weather harsher winters in the northern latitudes meaning cubs had to develop faster and be able to eat meat at a younger age.
4.4. Description of External Morphology and Body Position
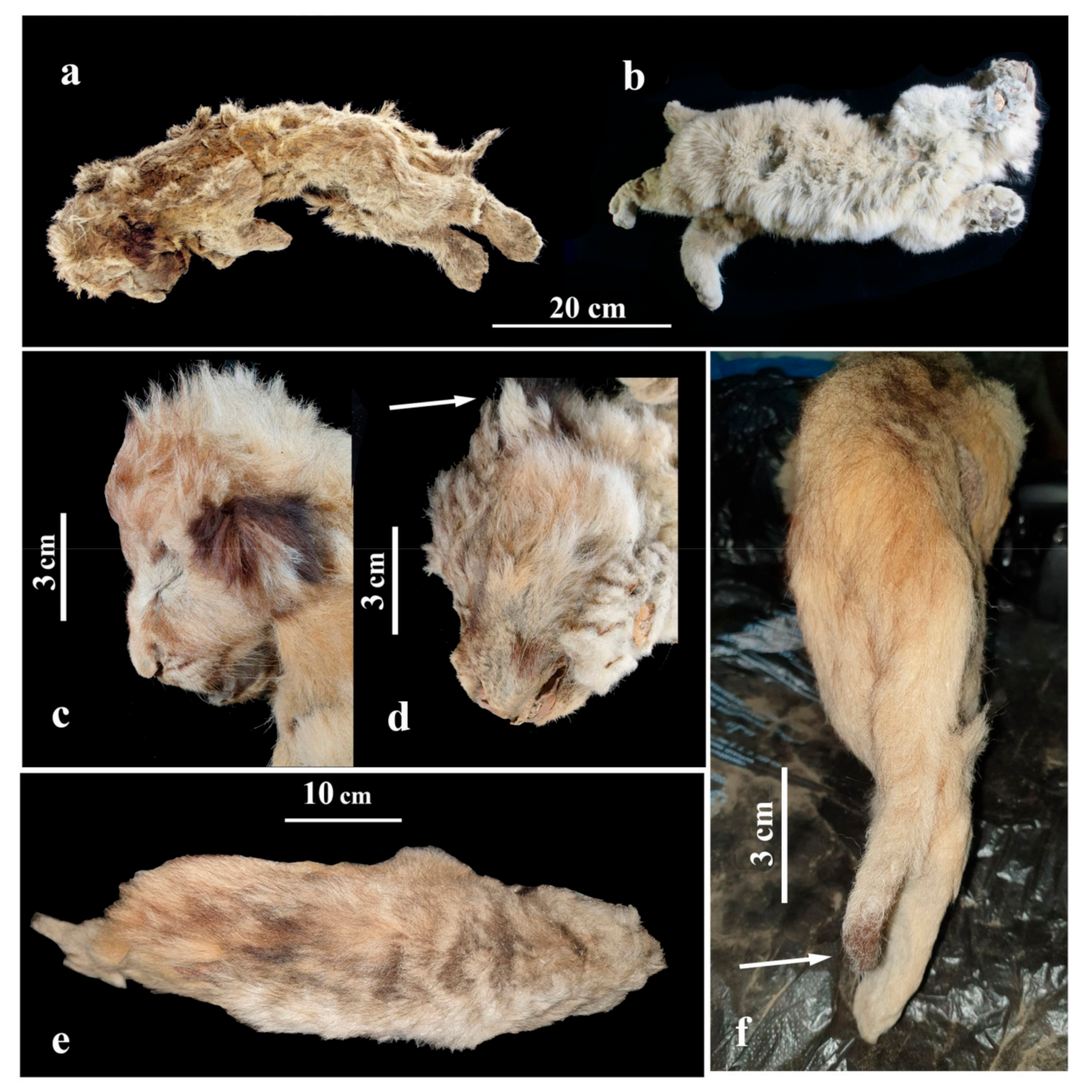
4.4.1. General Body Position and Sizes
The lion cub Sparta was lying on the right side (Figure 6a), with its body flattened in a lateral-to-medial direction. The hind limbs (at the level of a distal end of the tibia) and tail appear to have suffered the most pressure as these are the most flattened parts of the body. The skull is slightly deformed, the mouth opened, and forelimbs are bent at the joints in the elbows and wrists. Hind limbs and the tail are straightened and are parallel to each other. The stomach is sucked in, most likely from the dehydration of the internal organs and muscles. The eyes are closed.
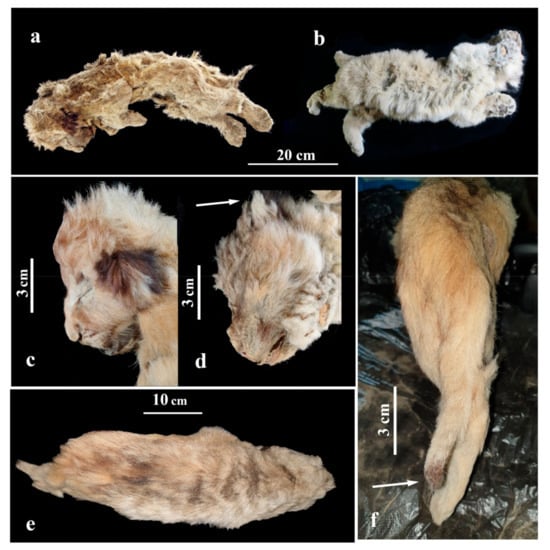
Figure 6.
Appearance of the frozen mummies of fossil cave lion cubs: (a) female Sparta; (b) male Boris. Photos of lion cubs’ heads from the side: (c) Sparta; (d) Boris; (e) Sparta mummy as seen from above; (f) dark brown ‘brush’ of Sparta’s tail.
The body of the lion cub Boris (Figure 6b) is flattened in a lateral-to-medial direction. The chest and stomach areas appear to have suffered the most pressure as they are the most flattened. The head is slightly deformed and turned to the right. Forelimbs are bent at the joints in elbows and subtly brought apart. The paws are slightly flattened in a dorsoventral direction. Hind limbs are stretched slightly and seem to be frozen in motion, which may suggest the lion cub Boris was trying to break free or find its way to the surface.
Sparta’s body length, counting its head, amounts to 515 mm, while Boris’s is 530 mm. The tail of Sparta is 123 mm in length (23.9% from the whole-body length, counting the head). The tail of the lion cub Boris is incomplete.
After defrosting the cave lion cubs and removing pieces of ice stuck to their bodies, they weigh 0.8 kg (Sparta) and 1.45 kg (Boris). A newborn cub of a contemporary African lion usually weighs between 1.15–2.1 kg (1.2 to 2.1 kg [24]; 1.15 kg to 1.785 kg [25]). In captivity, lion cubs grow much faster, and by the time they reach about 1 month in age, they weigh approximately 2.8–7.2 kg (2.8 to 5.0 kg, 4.1 kg average [25]; 3.5 to 7.2 kg [26]). However, since the bodies of these cave lion cubs have undergone frost sublimation for thousands of years, their initial weight at death is likely to be considerably different from their current weight.
4.4.2. Hair Coat
The studies on the qualities of coat hair of these mummies and a comparison to coat hair of a modern lion P. leo are already published [27]. Sparta’s coat is well preserved. The hair length on its head is 22.5 to 27.2 mm, 28 mm on the ‘sideburns’, 27.7 mm on the collar, 32 mm on the spine, 29.9 mm on the waist, 30.7 mm on the sacral region, 18.7 mm on the side, 16.8 mm on the elbows, 15.3 to 30.4 mm on the stomach. Boris’s coat is similar to Sparta’s coat. His guard hair is quite long throughout the whole body, and the undercoat is thick. However, his hair has fallen off in some areas (underarms, the right side of the body, and the muzzle).
Modern lion cub’s guard hair and fur undercoat are very similar, with the fur undercoat needed for protection and thermoregulation. The coat hair of a cave lion is similar but not identical to that of an African lion cub, with cave lions being distinguished by the presence of hair pellicle ornament. Cave lion fur also has a long thick fur undercoat consisting of strombuliform aeriferous fur hair. It covers the body of a cave lion cub evenly and most likely helped cave lion cubs adapt to the cold climate [27].
4.4.3. Hair Colour
It should be noted that frozen mummies of Pleistocene animals such as mammoths, woolly rhinoceros, or bison, originating from the frozen earth of Siberia, often have reddish-brown or yellowish coats, which is probably different from their initial hair colour (pers. observation). Both lion cubs were preserved in the frozen earth, covered by ice. It is therefore likely that their hair colour was the same when they were alive.
The primary colour of the lion cubs’ hair coat is light brown, grey, or yellowish brown (Figure 6a,b). Interestingly, although Sparta is a similar age to Boris, her fur has more reddish and brown shades, and it is darker and more saturated than Boris’s fur. The primary colour of Boris’s fur is a lighter, yellowish grey. It has to be noted that the youngest cave lion cub (found on the Uyandina River in 2015; Figure 7), had an evenly coloured fur of a yellowish-brown shade, without grey undertones, and part of its hair was dark brown on the ends [7,8]. This lion cub was assumed to be 1 to 2 weeks old, suggesting that this type of hair colour was typical for younger cubs of P. spelaea and was slowly changing to a lighter shade closer to the age of 1 to 2 months. Since Sparta is a female, this could be an indication that the juvenile colour of the fur was preserved in the cubs of female cave lions for a longer time.

Figure 7.
Frozen mummy of the cave lion cub named Uyan, Uyandina River, Yakutia.
Newborn cubs of extant African lions have dark circles on their fur, and these markings usually start to disappear after a few months [24]. We did not find any similar markings on the mummified cave lion cubs. However, a darker fur area is present around the eyes of Sparta (Figure 6c).
The guard hair on the muzzle of both cave lion pups has brown tips. There are dark spots on the forehead and above the eyes (Figure 6c). The fur is lighter around the eyes, but brown near the corners of the mouth. There are dark reddish-brown spots on the backside of Sparta’s ear conches, while Boris has black spots on his ear conches (Figure 6d), much similar to the contemporary P. leo.
Sparta’s guard hair on the back has black ends, creating a dark blurred line from the head to the tail set (Figure 6e).
Sparta’s tail is well preserved and is a bit lighter than her back. The tip of the tail is dark reddish brown, thus creating a ‘brush’ on the end (tuft) (Figure 6f). The contemporary P. leo is born without a brush, and it only starts developing when the lion cub is about 5 months old. At the age of 7 months, the brush is clearly visible [18]. Notably, the 1-to-2-week-old lion cub from the Uyandina River did not have a brush on its tail [7,8].
The fur on the stomach of Sparta and Boris is lighter than their back and has yellow undertones.
4.5. Skin
Visual observation of Boris’s body showed that it was damaged in some parts. A patch of skin is separated near the nose, and the distal part of the tail is missing. However, we were unable to check the condition of the skin of the cave lion cubs thoroughly by eye because of their thick coat fur. We, therefore, examined Sparta’s skin using computed tomography (Figure 8a). There are no visible skin injuries, but the skin is very mummified. There are a lot of unnatural wrinkles and deformations on the skin. The ears are flattened in the direction of the muzzle.
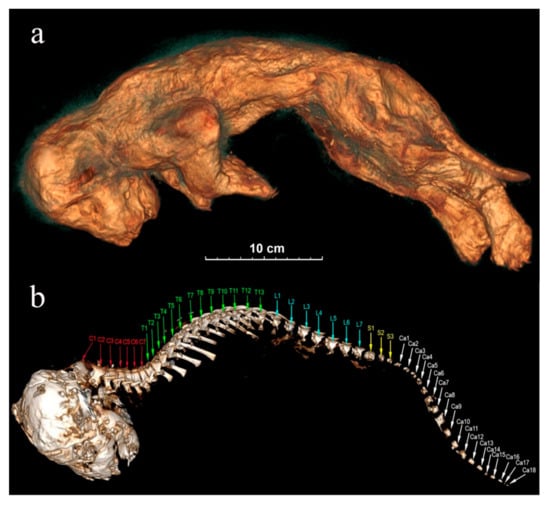
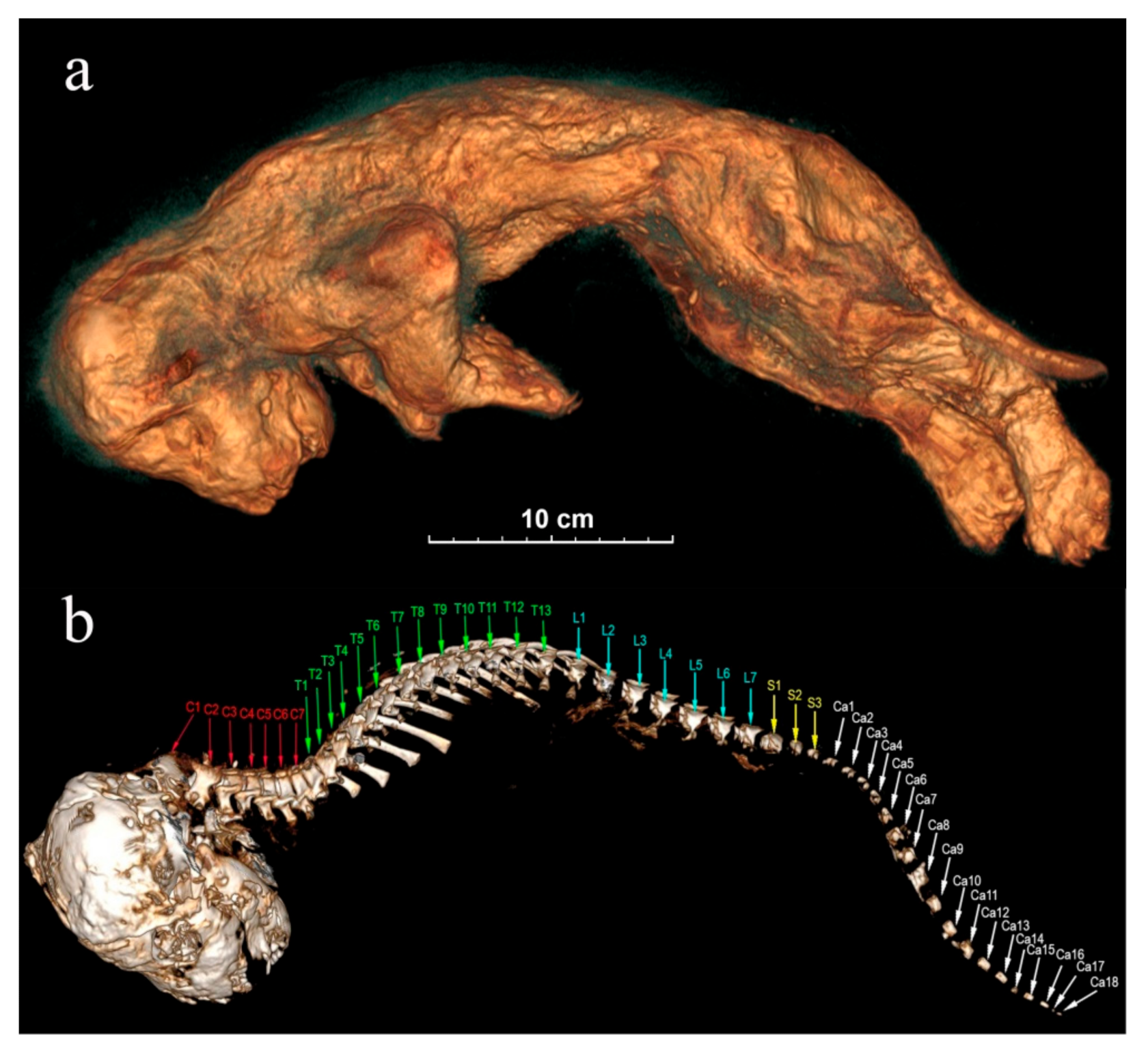
Figure 8.
The tomographic images of Sparta’s body from the left side: (a) skin; (b) skull and spine. The spinal bones: C—cervical vertebrae, T—thoracic vertebrae, L—lumbar vertebrae, S—sacral vertebrae, Ca—caudal vertebrae.
4.6. Imaging Examination of the Skeleton
Sparta’s skeleton is preserved completely, and the postcranial skeleton suffered almost no damage. Boris’s skeleton is also well preserved but very deformed. The bones in the front and hind limbs are damaged significantly. His chest is almost closed up in the medial-to-lateral direction.
Skull: Skulls of the lion cubs are fully preserved, and their sublimated brain can be seen. Cranial seams are not merged and are being held on the cartilages. The right bregma bone of Sparta is bent inside, presumably due to a physical impact. The situation of the skull towards the body is anatomically incorrect with the skull bent forward too strongly. At the same time, the atlas is visibly separated from the cranial condyles (Figure 8b). The skull of Boris has a deformation in its left part.
Spinal bones: Both lion cubs still have all 7 cervical vertebrae, 13 thoracic vertebrae, 7 lumbar vertebrae, and 3 sacral vertebrae. Sacra are not merged and consist of three segments. Sparta’s tail is fully preserved, and she still has all 18 caudal vertebrae (Figure 8b). The end part of Boris’s tail is broken off, and he has only 11 caudal vertebrae preserved.
Ribs: Sparta and Boris have all their 26 ribs, left and right. Sparta’s ribs are located incorrectly anatomically, probably because of the compression due to burial. Distal parts are not perpendicular to the spinal axis but are directed towards it in a caudal way to the extent that they are almost pressed to the vertebrae. The ribs however have no visible damage. Boris’s ribs are also not in their natural location and are separated from the vertebral glenoid cavities but not broken.
Breastbone: Sparta has all 8 segments of her breastbone preserved. The tomographic image shows 7 segments of the breastbone in Boris’s skeleton.
Pectoral girdle: The bones of the right forearm of Sparta are pressed to the humerus in an unnatural way. Due to the pressure, the right shoulder blade was separated from the cephalic end of the humerus and is located above the spinal column. The left front limb is situated in an anatomically incorrect way. The wrist area has metacarpal bones, carpal bones, phalanges, and claws preserved.
Boris’s front limbs are slightly flattened in a dorsoventral direction, but the claws are well preserved.
Pelvis: Pelvic bones are very narrow and underdeveloped in both individuals. The front and the back parts of the pelvis are connected through the cartilaginous tissue in the area of the glenoid cavity. Boris’s pelvic bones are dislocated.
Pelvic girdle: Hind limbs of the lion cubs are well preserved, and we can clearly see their astragali, bones of gaskin, and splint bones on the tomographic image, and the foot bones as well (ankle joints, instep bones, and phalanges). There is no physical damage.
On the tomographic images, we can see the internal organs of both lion cubs. Mostly they are dislocated and deformed because of dehydration and it is hard to positively identify them. However, Sparta’s womb is visible, which contributes to identifying her sex.
5. Discussion
Females of the extant African lion (P. leo) give birth to one to four cubs in a secluded place (usually in a stand of reeds, a cave, or another safe place), far away from other pride members. They often hunt by themselves in an area near the place where their cubs are being hidden [28]. African lion cubs are born blind and completely helpless, and their eyes open only about a week after birth. They start crawling after a day or two, and after the third week, they start walking [24]. A few times a month, a female carries her cubs to a new hiding place to prevent strong smells from accumulating in the den because predators can find the cubs by following their scent. Hyaenas, jackals, leopards, eagles, and snakes present a serious danger to young lion cubs. Usually, the female returns to her pride only after her cubs reach 6 to 8 weeks in age. In a pride, females with cubs usually band together to increase the chances of the cubs’ survival [28]. Adult males do not participate in raising the cubs. Moreover, if the leader of the pride is defeated by another lion, the victorious lion usually kills the defeated lion’s litter [29]. Counting all these factors and assuming that there are similarities between cave lion and African lion behaviour, it is likely that from birth, a lion cub is exposed to a large number of threats. The main threats facing cave lion cubs are likely to be food scarcity, predators, or adult male lions who are not related to them. Consequently, at least 80% of lions die before they reach the age of two years [30]. It is very difficult to identify which of these threats, if any, are more likely to be responsible for the deaths of ‘Boris’ and ‘Sparta’, or if they died as a result of falling and becoming trapped in the location that they were ultimately found. No toothmarks of carnivoran mammals have been identified on either Sparta or Boris’ bodies. Therefore, death by predation of the cave lion cubs seems unlikely. Cave lion cub mummies have been found complete (head, back, abdomen, and legs intact), suggesting a total absence of scavenger activities. Both specimens were quickly buried after death.
It is possible that cave lions had a different social system to African lions and a different set of parental care behaviours. Guthrie [31] was probably the first to hypothesise that cave lions did not live in prides, as did extant African lions, but in small family groups or even as single individuals. This assumption was based on Palaeolithic cave drawings in which adult cave lions were shown without a pronounced mane because a mane is an important visual detail in the lion pride hierarchy and territoriality. At the same time, other authors argue the importance of a mane and its connection to the social status of the extant African lion [32]. It is noted that sexual dimorphism of cave lion dentition appears greater than for modern lions [33] (this is especially the case with Beringian cave lion Panthera spelaea vereshchagini [34]), and this difference in size is much larger when compared to wild felids who live in solitude (tiger, leopard, jaguar). If we take this argument as an important factor of the social system structure, then it would suggest that cave lions and Beringian lions might have lived in bigger groups (prides) than extant African lions. In Chauvet cave, the main decorated (painted) panel depicts at least 11 cave lions (apparently females and males) in a steppe bison communal hunting scene. Hunting in groups can be more effective than solitary hunting when the prey is large [31], and cave lions would have had many such prey species available in their ecosystem, for example, mammoths and rhinoceros, when there were no other options available to them. In addition, large prides would have helped to protect their kill from the competition (e.g., wolves, brown bears, or other lions) and also to protect the cubs and young from predators (hyenas). It is also possible that pride size and relationships between its members changed seasonally, for example, to adapt to migratory changes in prey species. In this situation, cave lion prides would have large hunting territories where they followed herds of moving hoofed mammals or tried to stay near places where these hoofed mammals were gathering [34]. However, at present, there are insufficient data to reach any conclusions as to the details of the cave lion’s social structure.
If we assume that cave lions were similar to contemporary Panthera species (lion, tiger, leopard) in the way in which their cubs were brought up with male lions not participating in caring for the young, they would have been exposed to danger from other predators every time the mother went away to hunt. In the case of their mother’s death, the cubs would likely die from hunger. The harsh living conditions for large cats in high latitudes also have to be taken into account.
Siberian tiger (Panthera tigris altaica) on the south of the Russian Far East usually have newborn cubs in the warm periods, generally from May to August, but occasionally in the winter [35]. The highest mortality risk for tiger cubs is between 2.5 to 4 months old, when they are switching to a meat-based diet. They are still not able to eat large amounts of meat, but their mother’s milk does not fully sustain them. In this period, they start growing more slowly, leading to higher mortality in winter, because the food gets frozen too fast [35].
It is possible that cave lions were in a similar situation. We do not know when Sparta and Boris were born, but probably in summer (or autumn) time, and they most likely died when they were about 1 to 2 months old, in their den under the snow or the earth. This would explain why predators and scavengers did not find their bodies, and that is why we do not see any damage on the mummies that could have been inflicted by them. Maybe the cubs were buried under the ground following a landslide, and their bodies were deformed by the earth’s mass and permafrost features and froze quickly to become mummies.
The main purpose of a hair coat is protection and thermoregulation, but its colour can also be selected due to its potential influence on hunting success. The main predator of the contemporary tundra and forest tundra in the far northeast are wolves. The tundra wolf (Canis lupus albus) is usually light grey [36,37]. Animals of Eastern Europe and Kazakhstan steppe are also relatively light and grey in colour, with clear reddish-grey or reddish-brown shade on the back. This colouration seems to be the best fit for large predators in woodless areas. Judging by the mummies described in this article, cave lion cubs from the Arctic zone of Eurasia were also light grey in colour, and it is likely that the adults were as well. This would have been a part of their adaptation to life in the cold northern steppe.
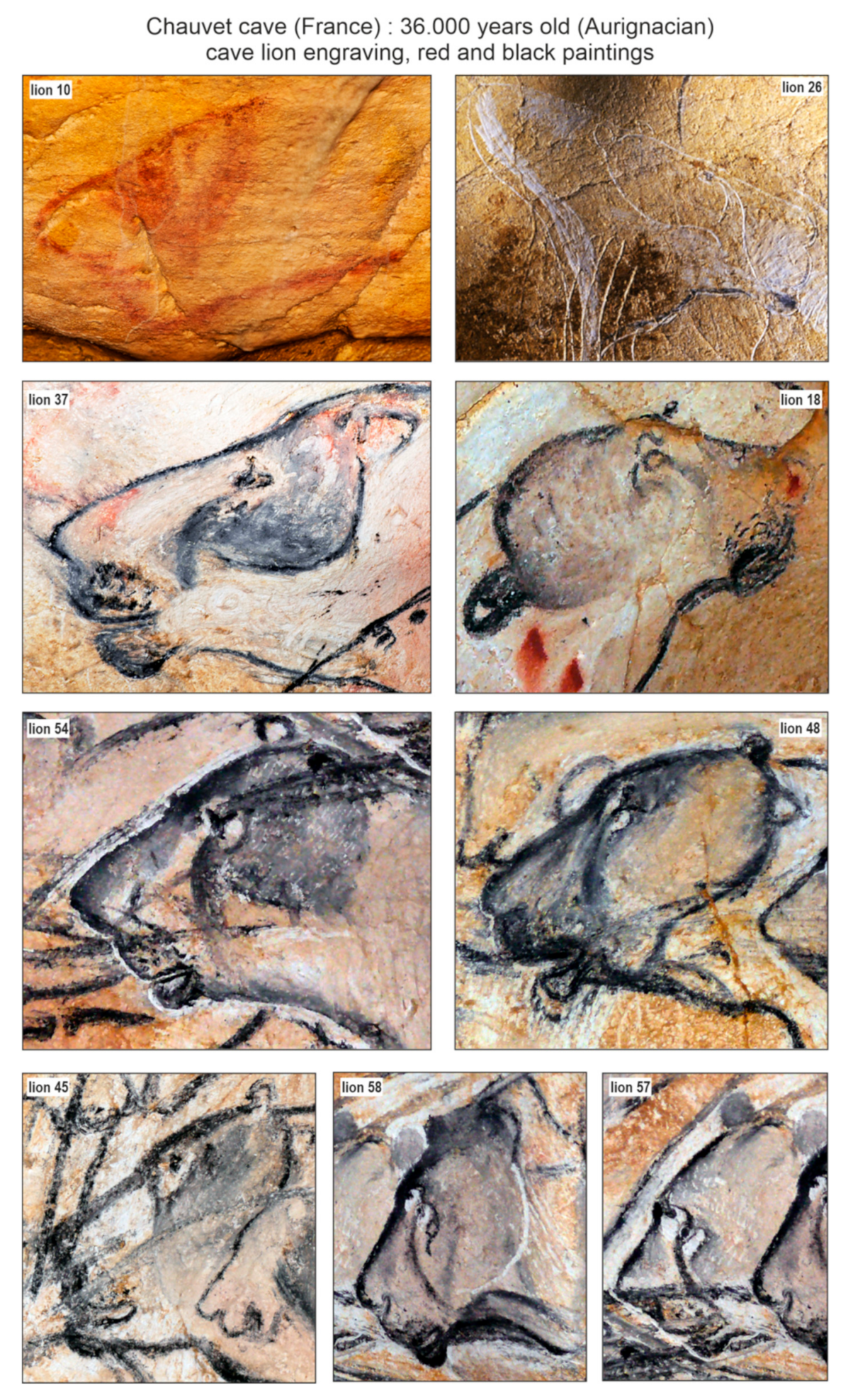
The discovery of frozen cave lion cubs provides an interesting opportunity for research on the body restitution (body mass, external aspect) of fossil lions adapted to cold climates. This adds valuable physical reconstitution data to what is currently known based on the Palaeolithic art of French caves. Analysis of cave art had demonstrated the absence or very discreet development of manes for adult males [31,38,39]. The discovery of Chauvet cave includes 75 cave lions [40], i.e., half of all cave lions Palaeolithic paintings known to date [41]. In these paintings, the heads of adult lions often have a contrasting colour around the eyes, known as a ‘drip line’ (vertical/oblique descending line, from the eye to the cheek), as well as a dark area at the level of the forehead (behind the eyes to the ears). The muzzles appear dark with 2–3 rows of abundant vibrissae. The neck also has a dark and dense, well-marked coat. Regardless of the nature of the parietal figurations (engravings, red or black paintings, see Figure 9), many heads in the Chauvet cave show this two-colour configuration, corresponding to an overall light coat punctuated by dark areas. This contrast in colouring could be related to sex or age because the fur of the cubs discovered in Yakutia is invariably light.
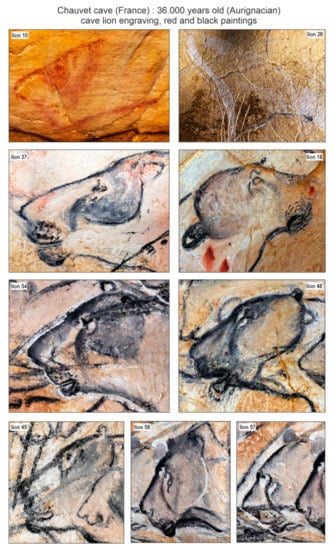
Figure 9.
Selected paintings of adult cave lions from Chauvet cave, showing interesting colouration on their head fur. Pictures: P. Fosse, numbers labelled as in Clottes and Azéma [40].
The Yakutian cave lion cubs had a light colouration, which could be considered as an adaptation to life on the northern open plains (covered the most part of a year by ice/snow cover). A darker fur area only appears around the eyes (Figure 6c). The dark colouration of adult European cave lions may be associated with habitation in closed, much more diversified landscapes: from open forests, forest steppes, mountains, and mountain forests to steppic-tundra vegetational settings, where dark colouration helps predators to be less noticeable. The differences between the fur of the Yakutian frozen cave lion cubs and Late Palaeolithic paintings could also reflect behavioural differences (in, e.g., territory size or density) that are seen in extant African lions (e.g., East versus South Africa) [42]. The main difference in hair structure between the Yakutian cave lion and the modern African lion is the strong development of the first underfur. For the Yakutian lion, the primary objective was to keep warm, while for the African lion, to avoid overheating [21].
6. Conclusions
The cave lion (P. spelaea) was widely spread throughout eastern Siberia in the Late Pleistocene period, especially during the Karginian interstadial (termochron) [34]. It seems unlikely that it is a coincidence that all four mummies of the cave lion cubs found to this day are from the Karginian interstadial and were found in a quite confined area in the river basin in the midstream of the Indigirka River (lower reaches of Uyandina River and Semyuelyakh–Tirekhtyakh River, located only c. 60 km from each other) (Figure 1). The large number of cubs found suggests that this area during the Karginian interstadial (when the climate was becoming relatively warm and tree vegetation was spreading) was a favourable breeding site for cave lions. It also seems probable that this site, during this time period, had some characteristics that made it more likely to rapidly freeze and preserve animals. The site was attractive to cave lions for making dens, but it was probably also susceptible to them collapsing.
The preliminary examination of the cubs’ mummies shows that the colour of the hair coat was changing from the juvenile yellowish-brown shade to a more ‘adult’ one, light grey to brown, at some point between 1–2 weeks and 1–2 months. Adult lions probably had light grey hair, well adapted to the Siberian Arctic, which is snow covered for two-thirds of the year.
It is known that cave lions had thick long fur undercoats consisting of strombuliform aeriferous hair. It covers the bodies of mummified lion cubs evenly and most likely helped them adapt to the cold climate [27].
We noted similarities between examined lion cubs P. spelaea and the contemporary lion cubs: their ears are light in the front but dark in the back, and they do not have a dark tuft on the end of their tail when they are born, but it is instead developed in their first few months. At the same time, cave lion cubs did not have vivid dark spots on their bodies and limbs, typical for P. leo cubs.
There is no sign of predators or scavengers damaging the fossil cave lions, but computed tomography showed skull damages, ribs dislocation, and other distortions in their skeletons that could be caused by the earth’s mass pressure due to the landslide and the following deformation due to the permafrost features.
Author Contributions
Organisation of research in Yakutsk, A.V.P.; preparing frozen mummies for research, I.S.P. and V.V.P.; external morphology studies, G.G.B., V.V.P., A.V.P., I.S.P. and A.N.T.; computed tomography studies, N.S. with the participation of V.V.P. and A.N.T.; genetic sex determination, L.D. and D.W.G.S.; discussion of images of cave lions in the caves of France, P.F.; writing—original draft preparation, G.G.B., V.V.P., G.F.B., P.F., D.W.G.S. and A.N.T.; discussion and writing—review and editing, all authors. All authors have read and agreed to the published version of the manuscript.
Funding
This research was conducted within the frameworks of the next scientific programs: the project on Funding: government assignment of the Diamond and Precious Metals Geology Institute, SB RAS No. 0381-2019-0002 (G.G.B.); the Federal theme of the Laboratory of Theriology ZIN RAS No. AAAA-A19-119032590102-7 ‘Phylogeny, morphology and systematics of placental mammals’ (G.F.B., A.N.T.). The study of G.G.B. was also supported by a grant from the Russian Science Foundation No. 21-17-00054, https://rscf.ru/project/21-17-00054/ (accessed on 3 November 2020). The study of A.V.P. was supported by a grant from the Russian Foundation for Basic Research (project no. 18-45-140007 р_а). D.W.G.S. received funding for this project from the European Union’s Horizon 2020 research and innovation programme under the Marie Skłodowska-Curie grant agreement (grant nr. 796877). L.D. acknowledges funding from FOR-MAS (grant nr. 2018–01640).
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
Not applicable.
Acknowledgments
The authors would like to express their gratitude to Anna A. Tikhonova, Saint-Petersburg, Russia, for the translation of the manuscript from Russian into English and to David W.G. Stanton for improving English language of the manuscript. Acknowledgements are addressed to the French Ministry of Culture and to the scientific team of Chauvet cave (Carole Fritz, Gilles Tosello).
Conflicts of Interest
The authors declare no conflict of interest.
References
- Vereshchagin, N.K.; Tikhonov, A.N. Exterior of the mammoth. Cranium 1999, 1, 4–44. [Google Scholar]
- Lazarev, P.A. Large Mammals of the Antropogene of Yakutia; Nauka Publ.: Novosibirsk, Russia, 2008; pp. 1–160. (In Russian) [Google Scholar]
- Boeskorov, G.G. Le mammoth Laineux. Biologie et Adaptation aux Conditions Glaciaires. In Memoire de Mammoth; Cretin, C., Madelaine, S., Eds.; Musee National de Prehistoire Print: Les Eyzies-de-Tayac, France, 2018; pp. 19–30. [Google Scholar]
- Boeskorov, G.G.; Lazarev, P.A.; Sher, A.V.; Davydov, S.P.; Bakulina, N.T.; Shchelchkova, M.V.; Binladen, J.; Willerslev, E.; Buigues, B.; Tikhonov, A. Woolly rhino discovery in the lower Kolyma River. Quat. Sci. Rev. 2011, 30, 2262–2272. [Google Scholar] [CrossRef]
- Boeskorov, G.G.; Potapova, O.R.; Protopopov, A.V.; Plotnikov, V.V.; Maschenko, E.N.; Shchelchkova, M.V.; Petrova, E.A.; Kowalczyk, R.; van der Plicht, J.; Tikhonov, A.N. A study of a frozen mummy of a wild horse from the Holocene of Yakutia, East Siberia, Russia. Mammal. Res. 2018, 63, 307–314. [Google Scholar] [CrossRef]
- Boeskorov, G.G.; Potapova, O.R.; Protopopov, A.V.; Plotnikov, V.V.; Kirikov, K.S.; Agenbroadd, L.D.; Pavlov, I.S.; Shchelchkova, M.V.; Belolyubskii, I.N.; Tomshin, V.D.; et al. The Yukagir Bison: Analysis of a complete frozen mummy of the bison, Bison priscus from the early Holocene of Northern Eurasia. Quat. Int. 2016, 406, 94–110. [Google Scholar] [CrossRef]
- Mashchenko, E.; Boeskorov, G.; Potapova, O.; Protopopov, A. Lions—From the Pleistocene to the present. What did the cubs of a lion from Yakutia tell us about? Nauka I Zhizn Sci. Life 2016, 6, 24–32. (In Russian) [Google Scholar]
- Protopopov, A.V.; Potapova, O.; Kharlamova, A.; Boeskorov, G.G.; Maschenko, E.N.; Shapiro, B.; Soares, A.; Pavlov, I.; Plotnikov, V.; Klimovskii, A. The Frozen Cave Lion ((Panthera spelaea (Goldfuss, 1810)) Newborn Cubs from Eastern Siberia, Russia: The first data on early ontogeny of the extinct species. PA J. Vertebr. Paleontol. 2016, 209. Available online: https://www.researchgate.net/publication/309910192 (accessed on 1 July 2021).
- Dussex, N.; Stanton, D.W.G.; Sigeman, H.; Ericson, P.G.; Gill, J.; Fisher, D.C.; Protopopov, A.V.; Herridge, V.L.; Plotnikov, V.; Hansson, B.; et al. Biomolecular analyses reveal the age, sex and species identity of a near-intact Pleistocene bird carcass. Commun. Biol. 2020, 3, 84. [Google Scholar] [CrossRef] [Green Version]
- Gromov, I.M.; Gureev, A.A.; Novikov, G.A.; Sokolov, I.I.; Strelkov, P.P.; Chapskii, K.K. Fauna SSSR. Mlekopitayushie. Opredelitel mlekopitaiushikh. Chast 2. (Kitoobraznye, Khishnye, Lastonogie, Neparnopalye, Parnopalye); Academy of Sciences of the USSR Publ. House: Moscow-Leningrad, Soviet Union, 1963; pp. 641–2000. (In Russian) [Google Scholar]
- Stanton, D.W.; Alberti, F.; Plotnikov, V.; Androsov, S.; Grigoriev, S.; Fedorov, S.; Kosintsev, P.; Nagel, D.; Vartanyan, S.; Barnes, I.; et al. Early Pleistocene origin and extensive intra-species diversity of the extinct cave lion. Sci. Rep. 2020, 10, 1–7. [Google Scholar] [CrossRef]
- Gilbert, M.T.P.; Tomsho, L.P.; Rendulic, S.; Packard, M.; Drautz, D.I.; Sher, A.; Tikhonov, A.; Dalén, L.; Kuznetsova, T.; Kosintsev, P.; et al. Whole-Genome Shotgun Sequencing of Mitochondria from Ancient Hair Shafts. Science 2007, 317, 1927–1930. [Google Scholar] [CrossRef]
- Yang, D.Y.; Eng, B.; Waye, J.S.; Dudar, J.C.; Saunders, S.R. Improved DNA Extraction from Ancient Bones Using Silica-Based Spin Columns. Am. J. Phys. Anthropol. 1998, 105, 539–543. [Google Scholar] [CrossRef]
- Meyer, M.; Kircher, M. Illumina Sequencing Library Preparation for Highly Multiplexed Target Capture and Sequencing. Cold Spring Harb. Protoc. 2010, 5. [Google Scholar] [CrossRef]
- Palkopoulou, E.; Mallick, S.; Skoglund, P.; Enk, J.; Rohland, N.; Li, H.; Omrak, A.; Vartanyan, S.; Poinar, H.; Götherström, A.; et al. Complete Genomes Reveal Signatures of Demographic and Genetic Declines in the Woolly Mammoth. Curr. Biol. 2015, 25, 1395–1400. [Google Scholar] [CrossRef] [Green Version]
- Flynn, J.M.; Hubley, R.; Goubert, C.; Rosen, J.; Clark, A.G.; Feschotte, C.; Smit, A.F. RepeatModeler2 for Automated Genomic Discovery of Transposable Element Families. Proc. Natl. Acad. Sci. USA 2020, 117, 9451–9457. [Google Scholar] [CrossRef] [PubMed]
- Li, H.; Durbin, R. Fast and Accurate Short Read Alignment with Burrows-Wheeler Transform. Bioinformatics 2009, 25, 1754–1760. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Stuiver, M.; Polach, H.A. Discussion: Reporting of 14C data. Radiocarbon 1977, 19, 355–363. [Google Scholar] [CrossRef] [Green Version]
- Bronk Ramsey, C. Bayesian analysis of radiocarbon dates. Radiocarbon 2009, 51, 337–360. [Google Scholar] [CrossRef] [Green Version]
- Reimer, P.J.; Bard, E.; Bayliss, A.; Beck, J.W.; Blackwell, P.G.; Bronk Ramsey, C.; Buck, C.E.; Cheng, H.; Edwards, R.L.; Friedrich, M.; et al. IntCal13 and Marine13 Radiocarbon age calibration curves 0–50,000 years cal BP. Radiocarbon 2013, 55, 1869–1887. [Google Scholar] [CrossRef] [Green Version]
- Reimer, P.J.; Austin, W.E.N.; Bard, E.; Bayliss, A.; Blackwell, P.G.; Ramsey, C.B.; Butzin, M.; Cheng, H.; Edwards, R.L.; Friedrich, M.; et al. The IntCal20 Northern Hemisphere radiocarbon age calibration curve (0–55 cal kBP). Radiocarbon 2020, 62, 725–757. [Google Scholar] [CrossRef]
- Smuts, G.L.; Anderson, J.L.; Austin, J.C. Age determination of the African lion (Panthera leo). J. Zool. 1978, 185, 115–146. [Google Scholar] [CrossRef]
- Smuts, G.L.; Robinson, G.A.; Whyte, I.J. Comparative growth of wild male and female lions (Panthera leo). J. Zool. 1980, 190, 365–373. [Google Scholar] [CrossRef]
- Schaller, G. The Serengeti Lion: A Study of Predator-Prey Relations; University of Chicago Press: Chicago, IL, USA, 1972; pp. 1–504. [Google Scholar]
- Colahan, H.; Asa, C.; Azzarello-Dole, C.; Boutelle, S.; Briggs, M.; Cox, K.; Kellerman, E.; Murray, S.; New, E.; Pukazhenthi, B.; et al. AZA Lion Species Survival Plan. Lion Care Manual; Association of Zoos and Aquariums: Silver Spring, MD, USA, 2012; pp. 1–143. [Google Scholar]
- De Carvalho, C.T. Comparative growth rates of hand-reared big cats. Int. Zoo Yearb. 1968, 8, 56–59. [Google Scholar] [CrossRef]
- Chernova, O.F.; Protopopov, A.V.; Boeskorov, G.G.; Pavlov, I.S.; Plotnikov, V.V.; Suzuki, N. First Description of the Fur of Two Cubs of Fossil Cave Lion Panthera spelaea (Goldfuss, 1810) Found in Yakutia in 2017 and 2018. Dokl. Biol. Sci. 2020, 492, 93–98. [Google Scholar] [CrossRef]
- Scott, J.; Scott, A. Big Cat Diary: Lion; Harper Collins Press: New York, NY, USA, 2002; pp. 1–45. [Google Scholar]
- Packer, C.; Pusey, A.E. Adaptations of female lions to infanticide by incoming males. Am. Nat. 1983, 121, 716–728. [Google Scholar] [CrossRef]
- Macdonald, D. The Encyclopedia of Mammals; Facts on File: New York, NY, USA, 1984; p. 31. [Google Scholar]
- Guthrie, R.D. Frozen Fauna of the Mammoth Steppe. The Story of Blue Babe; University of Chicago Press: Chicago, IL, USA, 1990; pp. 1–323. [Google Scholar]
- Yamaguchi, N.; Cooper, A.; Werdelin, L.; Macdonald, D.W. Evolution of the mane and group-living in the lion (Panthera leo): A review. J. Zool. 2004, 263, 329–342. [Google Scholar] [CrossRef]
- Turner, A. Dental sex dimorphism in European lions (Panthera leo L.) of the Upper Pleistocene: Palaeoecological and palaeoethological implications. Ann. Zool. Fenn. 1984, 21, 1–8. [Google Scholar]
- Boeskorov, G.G.; Baryshnikov, G.F. Late Quaternary Carnivorous Mammals of Yakutia; Nauka Publ. House: St. Petersburg, Russia, 2013; pp. 1–199. (In Russian) [Google Scholar]
- Yudin, I.G.; Yudina, E.V. Tiger of the Russian Far East; Dalnauka Publ.: Vladivostok, Russia, 2009; pp. 1–485. (In Russian) [Google Scholar]
- Heptner, V.G.; Naumov, N.P.; Yurgenson, P.B.; Sludsky, A.A.; Chirkova, A.F.; Bannikov, A.G. Mammals of the Soviet Union. Sea Cows and Carnivores; Vysshaya Shkola Publ.: Moscow, USSR, 1967; Volume II, pp. 136–140. (In Russian) [Google Scholar]
- Sokolov, V.E.; Rossolimo, O.L. Systematics and Variability. In The Wolf. History, Systematics, Morphology, Ecology; Bibikov, D.I., Ed.; Nauka Publ.: Moscow, USSR, 1985; pp. 22–50. (In Russian) [Google Scholar]
- Nougier, L.R.; Robert, R. Les félins dans l’art quaternaire. Préhistoire Spéléologie Ariégeoises 1965, 20, 17–62. [Google Scholar]
- Guthrie, R.D. The Nature of Paleolithic Art; University of Chicago Press: Chicago, IL, USA, 2006; pp. 1–520. [Google Scholar]
- Clottes, J.; Azéma, M. Les félins de la grotte Chauvet; Seuil Publ.: Paris, France, 2005; pp. 1–125. [Google Scholar]
- Fritz, C.; Fosse, P.; Tosello, G.; Sauvet, G.; Azéma, M. Ours et lion; réfléxion sur la place des carnivores dans l’art paléolithique. In Prédateurs dans tous leurs états: Évolution, Biodiversité, Interactions, Mythes, Symboles. XXXI Rencontres Internationales D’archéologie et D’histoire d’Antibes; Brugal, J.P., Gardeisen, A., Zucker, A., Eds.; Éditions APDCA: Antibes, France, 2011; pp. 299–323. [Google Scholar]
- Haas, S.K.; Hayssen, V.; Krausman, P.R. Panthera Leo. Mamm. Species 2005, 762, 1–11. [Google Scholar] [CrossRef]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).