The Use of Plant Macrofossils for Paleoenvironmental Reconstructions in Southern European Peatlands
Abstract
1. Introduction
2. Material and Methods
3. Results and Discussion
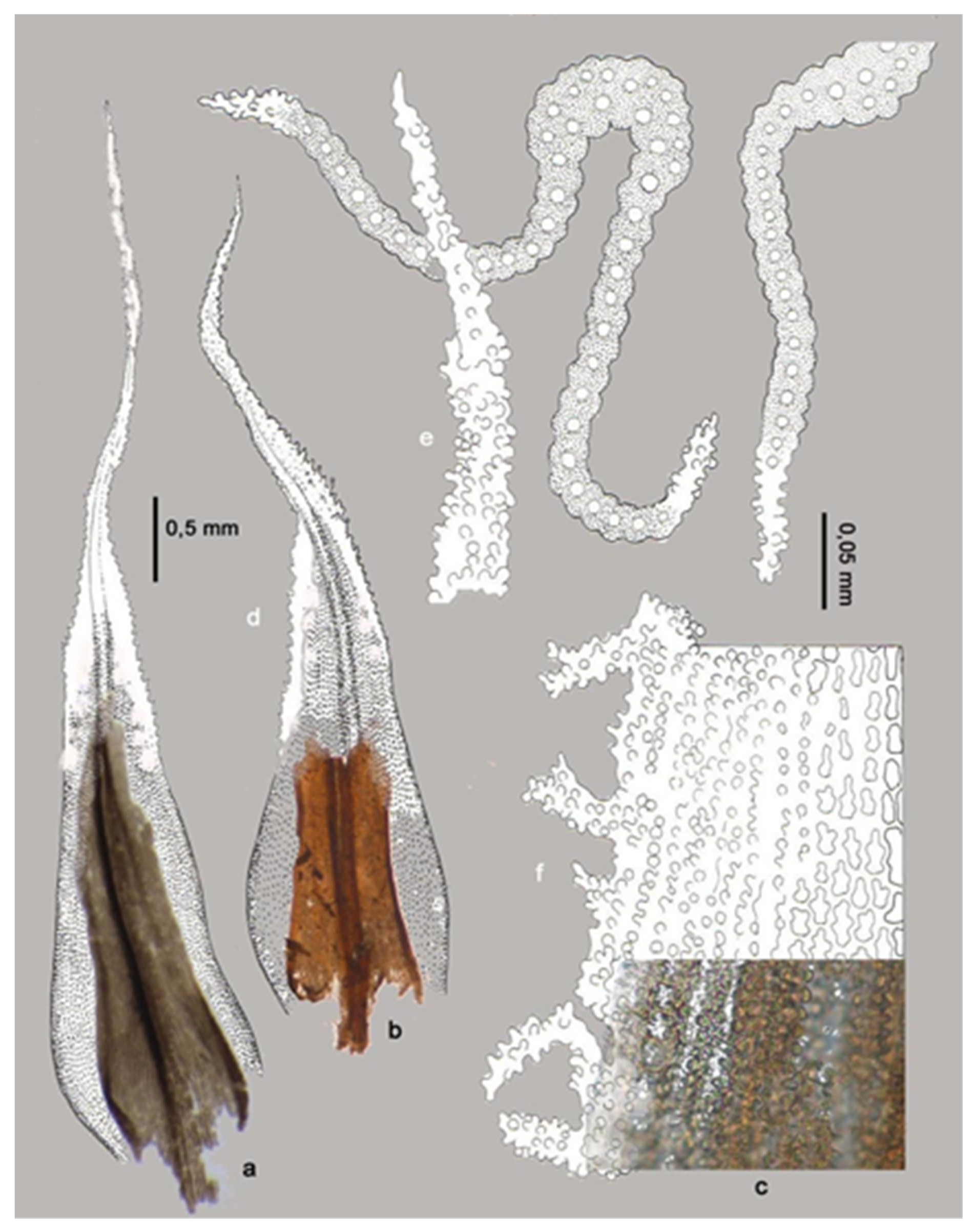
3.1. Problems in the Identification of Plant Macrofossils
3.2. Macrofossils: West Zone (Iberian Peninsula)
3.3. Macrofossils: Central Zone (Alps)
3.4. Macrofossils East Zone
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Clymo, R.S. Peat. In Ecosystems of the World. Mires: Swamp, Bog, Fen and Moor; Gore, A.J.P., Ed.; Elsevier: Amsterdam, The Netherlands, 1983; Volume 4A, pp. 159–224. [Google Scholar]
- Joosten, H. Mires in Europe: A preliminary status report. Int. Mire Conserv. Group Memb. Newslett. 1997, 3, 10–13. [Google Scholar]
- Joosten, H. Peat the final frontier: Mires and peatlands outside the tropics. In Peatlands under Pressure. Arctic to Tropical Peatlands; Maltby, E., Maclean, L., Eds.; Royal Holloway Institute for Environmental Research: Egham, UK, 1999; pp. 9–17. [Google Scholar]
- European Commission. European Commission Interpretation Manual of European Union Habitats, Vers. EUR28; European Commission, DG Environment: Brussel, Belgium, 2013. [Google Scholar]
- Barber, K.E.; Chambers, F.M.; Maddy, D.; Stoneman, R.E.; Brew, J.S. A sensitive highresolution record of late Holocene climate change from a raised bog in northern England. Holocene 1994, 4, 198–205. [Google Scholar] [CrossRef]
- Blackford, J. Palaeoclimatic records from peat bogs. Trends Ecol. Evol. 2000, 15, 193–198. [Google Scholar] [CrossRef]
- Birks, H.H.; Birks, H.J.B. Future uses of pollen analysis must include plant macrofossils. J. Biogeogr. 2000, 27, 31–35. [Google Scholar] [CrossRef]
- Birks, H.H.; Birks, H.J.B. Reconstructing Holocene climates from pollen and plant macrofossils. In Global Change in the Holocene; Mackay, A., Battarbee, R.W., Birks, H.J.B., Oldfield, F., Eds.; Arnold: London, UK, 2003; pp. 342–357. [Google Scholar]
- Kołaczek, P.; Gałka, M.; Apolinarska, K.; Gębica, P.; Superson, S.; Michno, A.; Harmata, K.; Szczepanek, K.; Płóciennik, M.; Gąsiorowski, M.; et al. Lost in dating—Problems with the absolute chronologies and sedimentation rates of late glacial and early Holocene oxbow lake deposits in Central Europe. Quat. Geochronol. 2017, 41, 187–201. [Google Scholar] [CrossRef]
- Kołaczek, P.; Gałka, M.; Lamentowiczac, M.; Marcisz, K.; Kajukało-Drygalska, K.; Karpińska-Kołaczeka, M. Increased radiocarbon dating resolution of ombrotrophic peat profiles reveals periods of disturbance which were previously undetected. Quat. Geochronol. 2019, 52, 21–28. [Google Scholar] [CrossRef]
- Feurdean, A.; Gałka, M.; Tanţău, I.; Geantă, A.; Hutchinson, S.M.; Hickler, T. Tree and timberline shifts in the northern Romanian Carpathians during the Holocene and the responses to environmental changes. Quat. Sci. Rev. 2016, 134, 100–113. [Google Scholar] [CrossRef]
- Gałka, M.; Tobolski, K.; Górska, A.; Lamentowicz, M. Resilience of plant and testate amoeba communities after climatic and anthropogenic disturbances in a Baltic bog in Northern Poland: Implications for ecological restoration. Holocene 2017, 27, 130–141. [Google Scholar] [CrossRef]
- Gałka, M.; Tobolski, K.; Lamentowicz, L.; Ersek, V.; Jaseey, V.E.; van der Knaap, W.O.; Lamentowicz, M. Unveiling exceptional Baltic bog ecohydrology, autogenic succession and climate change during the last 2000 years in CE Europe using replicate cores, multi-proxy data and functional traits of testate amoebae. Quat. Sci. Rev. 2017, 156, 90–106. [Google Scholar] [CrossRef]
- Coulson, J.C.; Butterfield, J. An investigation of the biotic factors determining the rates of plant decomposition on blanket bog. J. Ecol. 1978, 66, 631–650. [Google Scholar] [CrossRef]
- Yeloff, D.; Mauquoy, D. The influence of vegetation composition on peat humification: Implications for palaeoclimatic studies. Boreas 2006, 35, 662–673. [Google Scholar] [CrossRef]
- Buurman, P.; Nierop, K.G.J.; Pontevedra-Pombal, X.; Martínez-Cortizas, A. Molecular chemistry by pyrolysis-GC/MS of selected samples of the Penido Vello peat deposit, Galicia, NW Spain. In Peatlands: Basin Evolution and Repositories of Records of Environmental and Climate Changes; Martini, I.P., Martínez, A., Chesworth, W., Eds.; Elsevier: London, UK, 2006; pp. 219–242. [Google Scholar]
- Disnar, J.R.; Jacob, J.; Morched-Issa, M.; Lottier, N.; Arnaud, F. Assessment of peat quality by molecular and bulk geochemical analysis: Application to the Holocene record of the Chautagne marsh (Haute Savoie, France). Chem. Geol. 2008, 254, 101–112. [Google Scholar] [CrossRef]
- Ortiz, J.E.; Gallego, J.L.R.; Torres, T.; Díaz-Bautista, A.; Sierra, C. Palaeoenvironmental reconstruction of Northern Spain during the last 8000 cal yr BP based on the biomarker content of the Roñanzas peat bog (Asturias). Org. Geochem. 2010, 41, 454–466. [Google Scholar] [CrossRef]
- Schellekens, J.; Buurman, P.; Fraga, I.; Martínez-Cortizas, A. Holocene vegetation and hydrologic changes inferred from molecular vegetation markers in peat, Penido Vello (Galicia, Spain). Palaeogeogr. Palaeoclimatol. Palaeoecol. 2011, 299, 56–69. [Google Scholar] [CrossRef]
- Schellekens, J.; Buurman, P.; Kuyper, T.W.; Abbott, G.D.; Pontevedra-Pombal, X.; Martínez-Cortizas, A. Influence of source vegetation and redox conditions on lignin-based decomposition proxies in graminoid-dominated ombrotrophic peat (Penido Vello, NWSpain). Geoderma 2015, 237, 270–282. [Google Scholar] [CrossRef]
- Schellekens, J.; Bradley, J.A.; Kuyper, T.W.; Fraga, I.; Pontevedra-Pombal, X.; Vidal-Torrado, P.; Abbott, G.D.; Peter Buurman, P. The use of plant-specific pyrolysis products as biomarkers in peat deposits. Quat. Sci. Rev. 2015, 123, 254–264. [Google Scholar] [CrossRef]
- Moen, A.; Joosten, H.; Tanneberger, F. Mire diversity in Europe: Mire regionality. In Mires and Peatlands of Europe: Status, Distribution, and Nature Conservation; Joosten, H., Tanneberger, F., Moen, A., Eds.; Schweizerbart Science Publishers: Stuttgart, Germany, 2017; pp. 97–149. [Google Scholar]
- Katz, N.J.; Katz, S.V.; Kipiani, M.G. Atlas and Keys of Fruits and Seeds Occurring in the Quaternary Deposits of the USSR; Publishing House Nauka: Moscow, Russia, 1965; pp. 1–367. [Google Scholar]
- Katz, N.J.; Katz, S.V.; Skobeyeva, E.I. Atlas of Plant Remains in Peat Soil; Publishing House Nauka: Moscow, Russia, 1977; p. 736. [Google Scholar]
- Velichkevich, F.U.; Zastawniak, E. Atlas of the Pleistocene Vascular Plant Macrofossils of Central and Eastern Europe. Part 1: Pteridophytes and Monocotylendons; Polish Academy of Sciences, W Szafer Institute of Botany: Krakow, Poland, 2006. [Google Scholar]
- Velichkevich, F.U.; Zastawniak, E. Atlas of the Pleistocene Vascular Plant Macrofossils of Central and Eastern Europe. Part 2: Pteridophytes and Monocotylendons; Polish Academy of Sciences, W Szafer Institute of Botany: Krakow, Poland, 2008. [Google Scholar]
- Godwin, H. The History of the British Flora: A Factual Basis for Phytogeography; Cambridge University Press: Cambridge, UK, 1975. [Google Scholar]
- Grosse-Brauckmann, G. Über pflanzliche Makrofossilien mitteleuropäischer Torfe. I. Gewebereste krautiger Pflanzen und ihre Merkmale. Telma 1972, 2, 19–55. [Google Scholar]
- Grosse-Brauckmann, G. Über pflanzliche Makrofossilien mitteleuropäischer Torfe. II. Weitere Reste (Früchte und Samen, Moose u.a.) und ihre Bestimmungsmöglichkeiten. Telma 1974, 4, 51–117. [Google Scholar]
- Grosse-Brauckmann, G.; Streitz, B. Pflanzliche Makrofossilien mitteleuropäischer Torfe. III. Früchte, Samen undeinige Gewebe (Fotos von fossilen Pflanzenresten). Telma 1992, 22, 53–102. [Google Scholar]
- Mauquoy, D.; Van Geel, B. Plant macrofossil methods and studies: Mire and peat macros. In Encyclopedia of Quaternary Science; Elias, S.A., Ed.; Elsevier Science: Amsterdam, The Netherlands, 2007; pp. 2315–2336. [Google Scholar]
- Berggren, G. Atlas of Seeds and Small Fruits of Northwest-European Plant Species, Part 2. Cyperaceae; Swedish Natural Science Research Council: Stockholm, Sweden, 1969; pp. 1–107. [Google Scholar]
- Körber-Grohne, U. Bestimmungsschlüssel für subfossile Juncus-samen und Gramineen-Früchtchen (Identification keys for subfossil Juncus seeds and grasses fruits). In Probleme der Küstenforschung im Südlichen Nordseegebiet (Problems of Coastal Research in the Southern North Sea Area); Haarnagel, W., Ed.; Lax: Hildesheim, Germany, 1964; Volume 1, pp. 1–47. [Google Scholar]
- Tallantire, P.A. Provisional key for the identification of sub-fossil seeds of Vaccinium spp. Folia Quat. 1976, 47, 39–40. [Google Scholar]
- Truchanowiczówna, J. Fossil seeds of the genus Menyanthes in Eurasia. Acta Paleobot. 1964, 5, 25–69. [Google Scholar]
- Bialobrzeska, M.; Truchanowiczówna, J. Zmiennose ksztaltu owoców i lusek europejskich brzóz (Betula L.) oraz oznaczanie ich w stanie kopalnym. (The variability of shape of fruits and scales of the European birches (Betula L.) and their determination in fossil materials.). Mon. Bot. 1964, 9, 1–93. [Google Scholar]
- Souto, M.; Castro, D.; Pontevedra-Pombal, X.; Garcia-Rodeja, E.; Fraga, M.I. Characterisation of Holocene plant macrofossils from North Spanish ombrotrofic mires: Vascular plants. Mires Peat 2016, 18, 1–21. [Google Scholar]
- Souto, M. Reconstrucción Paleoambiental de Turberas del Norte de la Península Ibérica Mediante Análisis de Macrofósiles Vegetales y Grado de Humificación de la Turba. Ph.D. Thesis, Universidade de Santiago de Compostela, Santiago, Spain, 2018; pp. 1–207. [Google Scholar]
- Birks, H.J.B. Quaternary bryophyte palaeoecology. In Bryophyte Ecology; Smith, A.J.E., Ed.; Chapman & Hall: London, UK, 1982; pp. 473–490. [Google Scholar]
- Barry, T.A.; Synnott, D.M. Further studies into bryophyte occurrence and succession in the Hochmoor peat types of Ireland. Glasra 1987, 10, 1–21. [Google Scholar]
- Tallis, J.H. Climate and erosion signals in British blanket peats: The significance of Racomitrium lanuginosum remains. J. Ecol. 1995, 83, 1021–1030. [Google Scholar] [CrossRef]
- Jakab, G.; Sümegi, P. The role of bryophyte paleoecology in quaternary climate reconstructions. In Bryophyte Ecology and Climate Change; Tuba, Z., Slack, N.G., Stark, Y.R., Eds.; Cambridge University Press: Cambridge, UK, 2010; pp. 335–358. [Google Scholar]
- Souto, M.; Castro, D.; Pontevedra-Pombal, X.; Garcia-Rodeja, E.; Fraga, M.I. Characterisation of Holocene plant macrofossils from North Spanish ombrotrofic mires: Bryophytes. Mires Peat 2017, 19, 1–12. [Google Scholar]
- Maldonado, F.J.; Roch, V.; Rubiales, J.M.; Morla, C.; García-Amorena, I. Caracterización y Significación de los Yacimientos Paleobotánicos (Paleoxilológicos) de la Sierra de Gredos. In Proceedings of the IV Congreso Forestal Nacional, Zaragoza, Spain, 26–30 September 2005. [Google Scholar]
- García-Antón, M.; Franco Múgica, F.; Maldonado Ruiz, J.; Morla Juaristi, C.; Sainz Ollero, H. Una secuencia polínica en Quintana Redonda (Soria). Evolución holocena del tapiz vegetal en el Sistema Ibérico Septentrional. An. Jard. Bot. 1995, 52, 187–195. [Google Scholar]
- Muñoz Sobrino, C.; Ramil Rego, P.; Delibes de Castro, G.; Rojo Guerra, M. Datos paleobotánicos sobre la turbera de la Piedra (Páramo de Tozo, Burgos). In Biogeografía Pleistocena-Holocena de la Península Ibérica; Ramil Rego, P., Fernández Rodríguez, C., Rodríguez Guitián, M., Eds.; Xunta de Galicia: Santiago, Spain, 1996; pp. 149–162. [Google Scholar]
- Roig, S.; Gómez Manzaneque, F.; Masedo, F.; Morla, C.; Sanchez Hernando, L.J. Estudio paleobotánico de estróbilos y maderas subfósiles holocenas en el yacimiento de Cevico Navero (Palencia, España). An. Jard. Bot. 1997, 55, 111–123. [Google Scholar]
- Alcalde, C.; García-Amorena, I.; Gómez Manzaneque, F.; Maldonado, J.; Morla, C.; Postigo Mijarra, J.M. Estudio de los macrorrestos vegetales del yacimiento de Lomilla (Aguilar de Campo, Palencia, España). An. Jard. Bot. 2000, 59, 101–112. [Google Scholar]
- García-Amorena, I.; Morla, C.; Rubiales, J.M.; Gómez Manzaneque, F. Taxonomic composition of the Holocene forests of the northern coast of Spain, as determined from their macroremains. Holocene 2008, 18, 819–829. [Google Scholar] [CrossRef]
- Oldfield, F. Late-quaternary deposits at le Moura, Biarritz, south-west France. New Phytol. 1964, 63, 374–409. [Google Scholar] [CrossRef]
- Castro, D.; Souto, M.; Garcia-Rodeja, E.; Pontevedra-Pombal, X.; Fraga, M.I. Climate change records between the mid and late Holocene in a peat bog from Serra do Xistral (SW Europe) using plant macrofossils and peat humification analyses. Palaeogeogr. Palaeoclimatol. Palaeoecol. 2015, 420, 82–95. [Google Scholar] [CrossRef]
- Castro, D. Reconstrucción Paleoambiental das Turbeiras da Serra do Xistral Mediante Análises Paleobotánicas. Ph.D. Thesis, Universidade de Santiago de Compostela, Santiago, Spain, 2017; p. 207. [Google Scholar]
- Stefanini, B.S.; Oksanen, P.O.; Corcoran, J.P.; Mitchell, F.G. Appraising the cohesion of palaeoenvironmental reconstructions in north-west Spain since the mid-Holocene from a high temporal resolution multi-proxy peat record. Holocene 2018, 28, 681–694. [Google Scholar] [CrossRef]
- Pontevedra-Pombal, X.; Nóvoa-Muñoz, J.C.; García-Rodeja, E.; Martínez-Cortizas, A. Mountain mires from Galicia (NW Spain). In Peatlands: Basin Evolution and Repositories of Records of Environmental and Climate Changes; Martini, I.P., Martínez, A., Chesworth, W., Eds.; Elsevier: London, UK, 2006; pp. 83–108. [Google Scholar]
- Pontevedra-Pombal, X.; Castro, D.; Carballeira, R.; Souto, M.; López-Sáez, J.A.; Pérez-Díaz, S.; Fraga, M.I.; Valcárcel, M.; García-Rodeja, E. Iberian acid peatlands: Types, origin and general trends of development. Mires Peat 2017, 19, 1–19. [Google Scholar]
- Pontevedra-Pombal, X. Turberas de Montaña de Galicia. Génesis, Propiedades y su Aplicación Como Registros Ambientales Geoquímicos. Ph.D. Thesis, Universidade de Santiago de Compostela, Santiago, Spain, 2002; pp. 1–483. [Google Scholar]
- Heras, P.; Infante, M.; Pontevedra-Pombal, X.; Nóvoa-Muñoz, J.C. Mires and peatlands of Spain. In Mires and Peatlands of Europe: Status, Distribution, and Nature Conservation; Joosten, H., Tanneberger, F., Moen, A., Eds.; Schweizerbart Science Publishers: Stuttgart, Germany, 2017; pp. 639–656. [Google Scholar]
- Ponel, P.; de Beaulieu, J.L.; Tobolsk, K. Holocene palaeoenvironments at the timberline in the taillefer massif, French Alps: A study of pollen, plant macrofossils and fossil insects. Holocene 1992, 2, 117–130. [Google Scholar] [CrossRef]
- Ponel, P.; Court-Picon, M.; Badura, M.; Guiter, F.; de Beaulieu, J.L.; Andrieu-Ponel, V.; Djamali, M.; Leydet, M.; Gandouin, E.; Buttler, A. Holocene history of Lac des Lauzons (2180 m a.s.l.), reconstructed from multiproxy analyses of Coleoptera, plant macroremains and pollen (Hautes-Alpes, France). Holocene 2011, 21, 565–582. [Google Scholar] [CrossRef]
- Field, M.H.; de Beaulieu, J.L.; Guiot, J.; Ponel, P. Middle Pleistocene deposits at La Cote, Val-de-Lans, Isère department, France: Plant macrofossil, palynological and fossil insect investigations. Palaeogeogr. Palaeoclimatol. Palaeoecol. 2000, 159, 53–83. [Google Scholar] [CrossRef]
- Tobolski, K.; Ammann, B. Macrofossils as records of plant responses to rapid Late Glacial climatic changes at three sites in the Swiss Alps. Palaeogeogr. Palaeoclimatol. Palaeoecol. 2000, 159, 251–259. [Google Scholar] [CrossRef]
- Finsinger, W.; Tinner, W. Pollen and plant macrofossils at Lac de Fully (2135 m a.s.l.): Holocene forest dynamics on a highland plateau in the Valais, Switzerland. Holocene 2007, 17, 1119–1127. [Google Scholar] [CrossRef]
- Stähli, M.; Finsinger, W.; Tinner, W.; Allgöwer, B. Wildfire history and fire ecology of the Swiss National Park (Central Alps): New evidence from charcoal, polen and plant macrofossils. Holocene 2006, 16, 805–817. [Google Scholar] [CrossRef]
- Kalis, A.J.; van der Knaap, W.O.; Schweizer, A.; Urz, R. Three thousand year succession of plant communities on a valley bottom in the Vosges Mountains, NE France, reconstructed from fossil pollen, plant macrofossils, and modern phytosociological communities. Veget. Hist. Archaeobot. 2006, 15, 377–390. [Google Scholar] [CrossRef]
- Fauvart, N.; Ali, A.; Terral, J.F.; Roiron, P.; Blarquez, O.; Carcaillet, C. Holocene upper tree-limits of Pinus section sylvestris in the Western Alps as evidenced from travertine archives. Rev. Palaeobot. Palynol. 2012, 169, 96–102. [Google Scholar] [CrossRef]
- Ammann, B.; van der Knaap, W.O.; Lang, G.; Gaillard, M.J.; Kaltenrieder, P.; Rösch, M.; Finsinger, W.; Wright, H.E.; Tinner, W. The potential of stomata analysis in conifers to estimate presence of conifer trees: Examples from the Alps. Veget. Hist. Archaeobot. 2014, 23, 249–264. [Google Scholar] [CrossRef]
- Avigliano, R.; Anastasio, G.D.; Improta, S.; Peresani, M.; Ravazzi, C. A new late glacial to early Holocene palaeobotanical and archaeological record in the Eastern Pre-Alps: The Palughetto basin (Cansiglio Plateau, Italy). J. Quat. Sci. 2000, 15, 789–803. [Google Scholar] [CrossRef]
- Heiss, A.G.; Kofler, W.; Oeggl, K. The ulten valley in South Tyrol, Italy: Vegetation and settlement history of the area, and macrofossil record from the Iron Age cult site of St. Walburg. Palyno Bull. 2005, 1, 63–73. [Google Scholar]
- Krisai, R.; van Leeuwen, J.F.; van der Knaap, W.O. Present-day vegetation and the Holocene and recent development of Egelsee-Moor, Salzburg province, Austria. Veget. Hist. Archaeobot. 2016, 25, 555–568. [Google Scholar] [CrossRef]
- Sjögren, P.; Lamentowicz, M. Human and climatic impact on mires: A case study of Les Amburnex mire, Swiss Jura Mountains. Veget. Hist. Archaeobot. 2008, 17, 185–197. [Google Scholar] [CrossRef]
- Krisai, R. Zum rezenten und subfossilen Vorkommen subarktischer Moose im salzburgisch/oberösterreichischen Alpenvorland Vorh. Zool. Bot. Gos. Osterreich. 1985, 123, 143–150. [Google Scholar]
- Hölzer, A.; Hölzer, A. Ein torfprofil vom westabfall der hornisgride im nordschwarzwald mit Messia triquetra Angstr. Carolinea 2000, 58, 139–148. [Google Scholar]
- Mitchell, E.A.D.; Payne, R.J.; van der Knaap, W.O.; Lamentowicz, L.; Gąbka, M.; Lamentowicz, M. The performance of single- and multi-proxy transfer functions (testate amoebae, bryophytes, vascular plants) for reconstructing mire surface wetness and pH. Quat. Res. 2013, 79, 6–13. [Google Scholar] [CrossRef]
- Miola, A.; Bondesan, A.; Corain, L.; Favaretto, S.; Mozzi, P.; Piovan, S.; Sostizzo, I. Wetlands in the Venetian Po Plain (northeastern Italy) during the Last Glacial Maximum: Interplay between vegetation, hydrology and sedimentary environment. Rev. Palaeobot. Palynol. 2006, 141, 53–81. [Google Scholar] [CrossRef]
- Miola, A.; Favaretto, S.; Sostizzo, I.; Valentini, G.; Asioli, A. Holocene salt marsh plant communities in the North Adriatic coastal plain (Italy) as reflected by pollen, non-pollen palynomorphs and plant macrofossil analyses. Veget. Hist. Archaeobot. 2010, 19, 513–529. [Google Scholar] [CrossRef]
- Zaccone, C.; Lobianco, D.; Shotyk, W.; Ciavatta, C.; Appleby, P.; Brugiapaglia, E.; Casella, L.; Miano, T.M.; D’Orazio, V. Highly anomalous accumulation rates of C and N recorded by a relic, free- floating peatland in Central Italy. Sci. Rep. 2017, 7, 43040. [Google Scholar] [CrossRef] [PubMed]
- Hájek, M.; Horsák, M.; Tichy, L.; Hájkova, P.; Díte, D.; Jamrichová, E. Testing a relict distributional pattern of fen plant and terrestrial snail species at the Holocene scale: A null model approach. J. Biogeogr. 2011, 38, 742–755. [Google Scholar] [CrossRef]
- Hájková, P.; Stechová, T.; Soltés, R.; Smerdová, E.; Plesková, Z.; Díte, D.; Bradácová, J.; Mútnanová, M.; Singh, P.; Hájek, M. Using a new database of plant macrofossils of the Czech and Slovak Republics to compare past and present distributions of hypothetically relict fen mosses. Preslia 2018, 90, 367–386. [Google Scholar] [CrossRef]
- Hájková, P.; Grootjans, A.B.; Lamentowicz, M.; Rybnickova, E.; Madaras, M.; Opravilova, V.; Hajkova, T. How a Sphagnum fuscum-dominated bog changed into a calcareous fen: The unique Holocene history of a Slovak spring-fed mire. J. Quat. Sci. 2012, 27, 233–243. [Google Scholar] [CrossRef]
- Peterka, T.; Hájek, M.; Dítě, D.; Hájková, P.; Palpurina, S.; Goia, I.; Grulich, V.; Kalníková, V.; Plesková, Z.; Šímová, A.; et al. Relict occurrences of boreal brown-moss quaking rich fens in the Carpathians and adjacent territories. Folia Geobot. 2018, 53, 265–276. [Google Scholar] [CrossRef]
- Kołaczek, P.; Gałka, M.; Apolinarska, K.; Płóciennik, M.; Gąsiorowski, M.; Brooks, S.J.; Hutchinson, S.M.; Karpińska-Kołaczek, M. A multi-proxy view of exceptionally early postglacial development of riparian woodlands with Ulmus in the Dniester River valley, western Ukraine. Rev. Palaeobot. Palynol. 2018, 250, 27–43. [Google Scholar] [CrossRef]
- Šímová, A.; Pánek, T.; Gałka, M.; Zernitskaya, V.; Hájková, P.; Brodská, H.; Jamrichova, E.; Hájek, M. Landslides increased Holocene habitat diversity on a flysch bedrock in the Western Carpathians. Quat. Sci. Rev. 2019, 219, 68–83. [Google Scholar] [CrossRef]
- Stachowicz-Rybka, R.; Gałka, M.; Alexandrowicz, W.P.; Alexandrowicz, S.W. Plant macrofossils and malacocoenoses of Quaternary mineral-organic sediments at Starunia palaeontological site and vicinity (Carpathian region, Ukraine). Ann. Soc. Geol. Pol. 2009, 79, 297–313. [Google Scholar]
- Gałka, M.; Tantau, I.; Ersek, V.; Feurdean, A. A 9000 year record of cyclic vegetation changes identified in a montane peatland deposit located in the Eastern Carpathians (Central-Eastern Europe): Autogenic succession or regional climatic influences? Palaeogeogr. Palaeoclimatol. Palaeoecol. 2016, 449, 52–61. [Google Scholar] [CrossRef]
- Magyari, E.; Sümegi, P.; Braun, M.; Jakab, G.; Molnar, M. Retarded wetland succession: Anthropogenic and climatic signals in a Holocene peat bog profile from north-east Hungary. J. Ecol. 2001, 89, 1019–1032. [Google Scholar] [CrossRef]
- Magyari, E.; Jakab, G.; Sümegi, P.; Szoör, G.Y. Holocene vegetation dynamics im the Bereg Plain, NE Hungary—The Báb-tava pollen and plant macrofossil record. Acta GGM Debrecina 2008, 3, 33–50. [Google Scholar]
- Sümegi, P.; Jakab, G.; Majkut, P.; Törocsik, T.; Zatykó, C. Middle age paleoecological and paleoclimatological reconstruction in the Carparthian Basin. Idöjárás 2009, 113, 265–298. [Google Scholar]
- Gałka, M.; Feurdean, A.; Hutchinson, S.; Milecka, K.; Tantau, I.; Apolinarska, K. Response of a spring-fed ecosystem in Central Eastern Europe (NW Romania) to climate changes during the last 4000 years: A high resolution multi-proxy reconstruction. Palaeogeogr. Palaeoclimatol. Palaeoecol. 2018, 504, 170–185. [Google Scholar] [CrossRef]
- Bozilova, E.; Tonkov, S. Pollen from Lake Sedmo Rilsko reveals southeast European postglacial vegetation in the highest mountain area of the Balkans. New Phytol. 2000, 148, 315–325. [Google Scholar] [CrossRef]
- Marinova, E.; Tonkov, S. Holocene vegetation history of the northwestern Pirin Mountain (Bulgaria). Plant fossil record from peat-bog Mozgovitsa. C. R. Acad. Bulg. Sci. 2012, 65, 1087–1094. [Google Scholar]
- Tonkov, S.; Possnert, G.; Bazilova, E.; Marinova, E.; Pavlova, D. On the Holocene vegetation history of the Central Rila Mountains, Bulgaria: The palaeoecological record of peat bog Vodniza (2113 m). Rev. Palaeobot. Palynol. 2018, 250, 16–26. [Google Scholar] [CrossRef]
- Tonkov, S.; Marinova, E. Pollen and plant macrofossil analyses of mid-Holocene ra-diocarbon dated profiles from two subalpine lakes in Rila Mountains, Bulgaria. Holocene 2005, 15, 663–671. [Google Scholar] [CrossRef]
- Stefanova, I. New data on the late Holocene vegetative succession in the Northern Pirin Mts: Pollen and macrofossil analysis of depositions from peat bogs Goce Delchev and Visokata Ela. Phytol. Balc. 1997, 3, 53–63. [Google Scholar]
- Stefanova, I.; Atanassova, J.; Delcheva, M.; Wright, H.E. Chronological framework for the Lateglacial pollen and macrofossil sequence in the Pirin Mountains, Bulgaria: Lake Besbog and Lake Kremensko-5. Holocene 2006, 16, 877–892. [Google Scholar] [CrossRef]
- Lazarova, M.; Marinova, E.; Tonkov, S.; Snowball, I. A 5000-year pollen and plant macrofossil record from the Osogovo Mountain, Southwestern Bulgaria: Vegetation history and human impact. Rev. Palaeobot. Palynol. 2015, 223, 1–9. [Google Scholar] [CrossRef]
- Stefanova, I.; Ammann, B. Lateglacial and Holocene vegetation belts in the Pirin Mountains (southwestern Bulgaria). Holocene 2003, 13, 97–107. [Google Scholar] [CrossRef]
- Finsinger, W.; Morales-Molino, C.; Gałka, M.; Valsecchi, V.; Bojovic, S.; Tinner, W. Holocene vegetation and fire dynanics at Crveni Potok, a small mire in the Dinaric Alps (Tara National Park, Serbia). Quat. Sci. Rev. 2017, 167, 63–77. [Google Scholar] [CrossRef]
- Markgraf, V. Pollen dispersal in a mountain area. Grana 1989, 19, 127–146. [Google Scholar] [CrossRef]
- Chytrý, M.; Hennekens, S.M.; Jiménez-Alfaro, B.; Knollová, I.; Dengler, J.; Schaminée, J.H.J.; Aćić, S.; Agrillo, E.; Ambarlı, D.; Angelini, P.; et al. European vegetation archive (EVA): An integrated database of European vegetation plots. Appl. Veg. Sci. 2016, 19, 173–180. Available online: https://onlinelibrary.wiley.com/doi/full/10.1111/avsc.12191 (accessed on 8 October 2019). [CrossRef]
- Mucina, L.; B€ultmann, H.; Dierßen, K.; Theurillat, J.-P.; Raus, T.; Carni, A.; Sumberova, K.; Willner, W.; Dengler, J.; Garcia, R.G.; et al. Vegetation of Europe: Hierarchical floristic classification system of vascular plant, bryophyte, lichen, and algal communities. Appl. Veg. Sci. 2016, 19, 3–264. [Google Scholar] [CrossRef]
- Pokorná, A.; Dreslerová, D.; Křivánková, D. Archaeobotanical Database of the Czech Republic, an Interim Report. IANSA 2011, 1, 49–53. [Google Scholar] [CrossRef]
- Rochefort, L.; Lode, E. Restoration of degraded boreal peatlands. In Boreal Peatland Ecosystems; Ecological Studies (Analysis and Synthesis); Wieder, R.K., Vitt, D.H., Eds.; Springer: Berlin/Heidelberg, Germany, 2006; Volume 188. [Google Scholar]
- Schouten, M.G.C. (Ed.) Conservation and Restoration of Raised Bogs; Geological, Hydrological and Ecological Studies; Department of the Environment and Local Government Ireland, Geological Survey of Ireland: Staatsbosbeheer, The Netherlands, 2002.
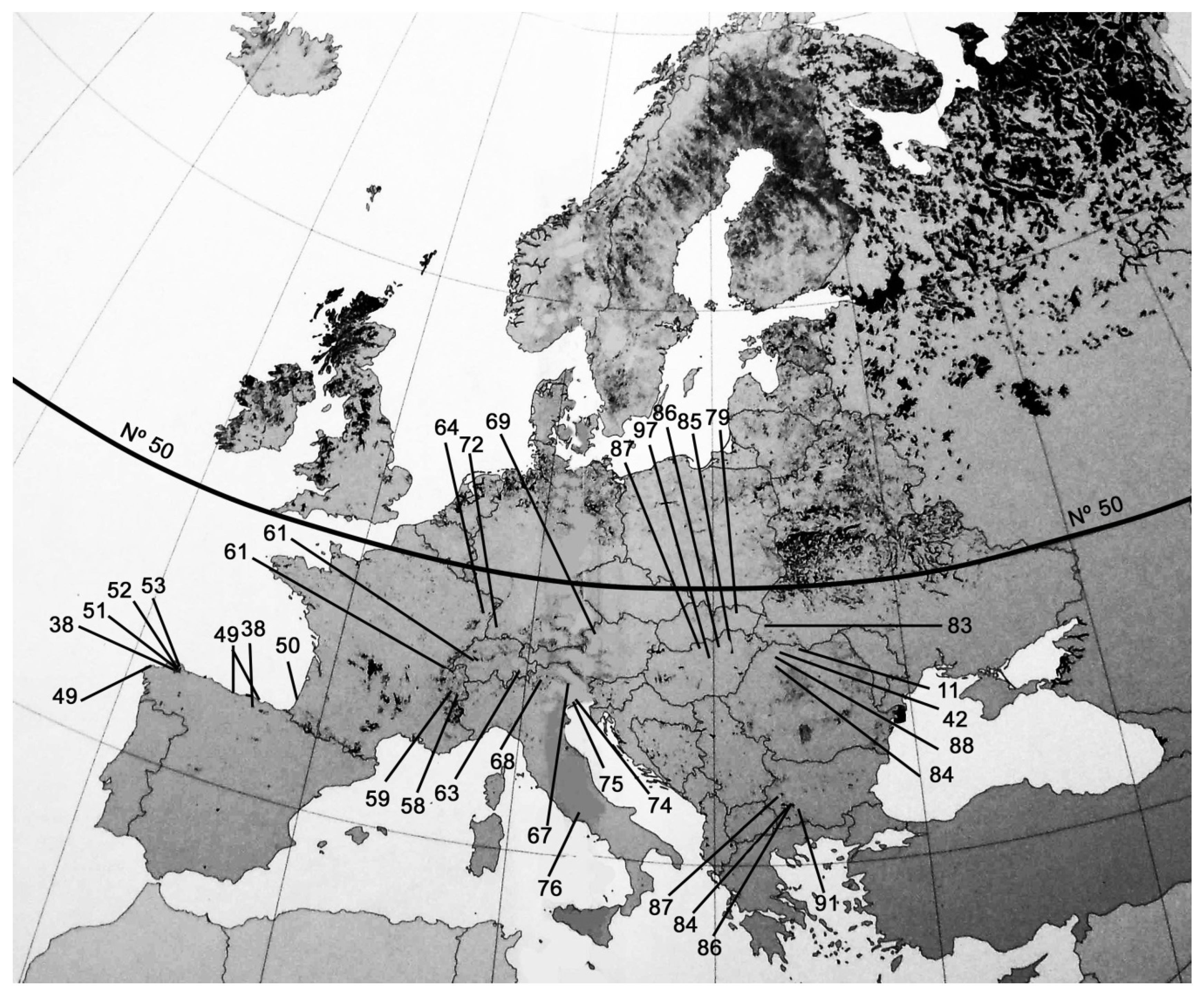

| SITE LOCATION | DEPTH (cm) | Yrs. Cal. BP | MATERIAL | ALT (m a.s.l.) | REF | AUTOR |
|---|---|---|---|---|---|---|
| WEST ZONE | ||||||
| Zalama, De Ordunte Mts. (ES) | 226 | 8000 | Peat | 1330 | 38 | Souto, 2018 |
| Borralleiras, Cabaleiros Mts. (ES) | 230 | 5500 | Peat | 600 | 38 | Souto, 2018 |
| Pena da Cadela, Serra do Xistral (ES) | 183 | 5500 | Peat | 970 | 51 | Castro et al., 2015 |
| Chan de Veiga Mol,Serra do Xistral (ES) | 845 | 8000 | Peat | 695 | 52 | Castro, 2017 |
| Pedrido, Serra do Xistral (ES) | 250 | 4750 | Peat | 770 | 53 | Stefanini et al., 2018 |
| Noja, Cantabric Coast (ES) | 100 | 3200–4600 | Coast peat deposit | 50 | 49 | García et al., 2008 |
| Merón, Cantabric Coast (ES) | 20 | 6000–7000 | Coast peat deposit | 50 | 49 | García et al., 2008 |
| Baldaio, Cantabric Coast (ES) | 30 | 870 | Coast peat deposit | 25 | 49 | García et al., 2008 |
| Le Moura, Cantabric Coast (FR) | 600 | 10,000 | Peat | 50 | Oldfield, 1964 | |
| CENTRAL ZONE | ||||||
| Grande Basse, Vosges Mts. (FR) | 200 | 3000 | Peat | 945 | 64 | Kalis et al., 2006 |
| Canard Ib, Taillefer Massif (FR) | 125 | 10,000 | Peat | 2200 | 58 | Ponel et al., 1992 |
| Lac Lauzons, Hautes-Alpes (FR) | 170 | 10,000 | Peat | 2180 | 59 | Ponel et al., 2011 |
| Egelsee-Moor Mire, Salzburg (AT) | 600 | 10,000 | Peat | 700 | 69 | Krisai et al., 2016 |
| Fuorn Mire, Fuorn Valley (CH) | 255 | 8500 | Peat-Gyttja | 1805 | 63 | Stahii et al., 2006 |
| Lac de Fully, Rhône Valley (CH) | 270 | 11,000 | Gyttja | 2135 | 62 | Finsinger and Tinner, 2007 |
| Gerzensee (CH) | 190–200 | 11,500 | Gyttja | 630 | 61 | Tobolski and Ammann, 2000 |
| Leysin (CH) | 320–350 | 10,850–12,050 | Gyttja | 1230 | 61 | Tobolski and Ammann, 2000 |
| Northern Black Forest (GR) | 80 | 2000 | Peat | 430 | 72 | Hölzer and Hólzer, 2000 |
| Palughetto, Lapisina Valley (IT) | 50 | 12,200 | Paleo peat | 1040 | 67 | Avigliano et al., 2000 |
| Totenmoos, South Tyrol (IT) | 800 | 15,000 | Peat | 1718 | 68 | Heiss et al., 2005 |
| Paludetto S2, Venetian plain (IT) | 500–1050 | 8700 | Peat | 50 | 75 | Miola et al., 2010 |
| Fiorentina, Venetian plain (IT) | 1100–1800 | 3000–17,000 | Peat-clay-silt | 50 | 74 | Miola et al., 2006 |
| La Rota, Posta Fibreno (IT) | 400 | 600 | Peat | 76 | Zaccone et al., 2017 | |
| EAST ZONE | ||||||
| Begbunar, Osogovo Mts (BG) | 105 | 5000 | Peat | 1800 | 95 | Lazarova et al., 2015 |
| Lake Ostrezko, Rila Mts (BG) | 200 | 6000 | Peat | 2340 | 92 | Tonkov and Marinova, 2005 |
| Lake Besbog,Pirin Mts (BG) | 380 | 15,000 | Peat-Gyttja | 2200 | 94 | Stefanova et al., 2006 |
| Vodniza, Rila Mts (BG) | 500 | 9500 | Peat-Gyttja | 2113 | 91 | Tonkov et al., 2018 |
| Tara, Mts. (RS) | 270 | 9000 | Peat | 1600 | 97 | Fisinger et al., 2017 |
| Nagymohos (HU) | 140–280 | 4000–7500 | Peat | 300 | 85 | Magyari et al., 2001 |
| Báb-tava, Bereg Plain, (HU) | 142–176 | 1000–2300 | Peat | 100 | 86 | Magyari et al., 2008 |
| Sirok Nyírjes-to, Mátra Mts, (HU) | 400 | 9000 | Peat | 250 m | 42 | Jakab & Sümegi, 2010 |
| Gärgäläu fen, Rodna Mts. (RO) | 150 | 9500 | Peat | 1810 | 11 | Feurdean et al., 2016 |
| Valea Morii, North western (RO) | 100 | 1100 | Peat | 640 | 88 | Galka et al., 2018 |
| Tăul Muced, Rodna Mts. (RO) | 500 | 9000 | Peat | 1360 | 84 | Galka et al., 2016 |
| Belanské Lú́ky, Tatra Mts. (SK) | 230 | 10,000 | Peat | 700 | 79 | Hájková et al., 2012 |
| Nádas Lake, Cserhát Mts. (HU) | 340 | 8000 | Peat-Clay | 360 | 87 | Sümegi et al., 2009 |
| Starunia, Carpathos (UA) | 300–500 | 12,000 | Peat-Clay | 400 | 83 | Stachowicz-Rybka et al., 2009 |
| REF. BIBLIOGRAPHY | [53] | [52] | [38] | [51] | [38] |
|---|---|---|---|---|---|
| MACROFOSSIL TAXA | |||||
| Betula pubescens | S | ||||
| Ericaceae | LF, F, W | LF, F, W | |||
| Erica tetralix | LF | LF, S | |||
| Erica mackaiana | LF, S | LF, S | LF, S | LF, S | |
| Calluna vulgaris | S | LF, S | LF, S | LF, S | LF, S |
| Andromeda polifolia | LF, S | ||||
| Daboecia cantabrica | S | S | |||
| Vaccinium myrtillus | LF | LF, W | |||
| Poaceae | LF | ||||
| Agrostis curtisii | LF,R | LF,R | |||
| Molinia caerulea | LF,R,F | LF,R,F | LF,R,F | LF,R,F | |
| Potentilla erecta | LF, F | F | LF, F | ||
| Drosera intermedia | S | ||||
| Drosera rotundifolia | S | S | S | S | |
| Narthecium ossifragum | S | S | S | ||
| Caltha palustris | S | S | |||
| Cyperaceae | LF | LF, R | LF, R | LF, R | |
| Eriophorum sp. | S, LF | LF, R, S | LF, R, S | LF, R, S | S, LF, R |
| Carex echinata | F | ||||
| Carex demissa | F | ||||
| Carex binervis | F | ||||
| Carex durieui | F | F | |||
| Rhynchospora alba | F | ||||
| Trichophorum sp | LF | ||||
| Luzula multiflora | S | ||||
| Juncus bulbosus | S | S | S | ||
| Juncus squarrosus | S | S | S | ||
| BRYOPHYTA | |||||
| Aulacomnium palustre | VP | VP | |||
| Calliergonella cuspidata | VP | ||||
| Campylopus sp | VP | VP | |||
| Dicranum scoparium | VP | ||||
| Hypnum cupressiforme | VP | VP | VP | VP | VP |
| Leucobryum juniperoideum | VP | VP | VP | ||
| Polytrichum sp. | VP | ||||
| Racomitrium lanuginosum | VP | VP | VP | ||
| Spagnum capillifolium | VP | ||||
| Spagnum tenellum | VP | VP | VP | ||
| Sphagnum acutifolia | VP | ||||
| Sphagnum compactum | VP | VP | |||
| Sphagnum cuspidatum | VP | ||||
| Sphagnum molle | VP | ||||
| Sphagnum papillosum | VP | VP | |||
| Sphagnum sec. acutifolia | VP | VP | VP | VP | |
| Sphagnum sec. cuspidata | VP | VP | |||
| Thuidium tamariscinum | VP | ||||
| Calypogeia sphagnicola | VP | ||||
| Odontochisma sphagni | VP | ||||
| FUNGI | |||||
| Cenoccocum geophilum | SCL | SCL | SCL |
| REF. BIBLIOGRAPHY | [50] | [64] | [58] | [69] | [63] | [62] | [61] | [61] |
|---|---|---|---|---|---|---|---|---|
| MACROFOSSIL TAXA | ||||||||
| Pinus sp. | ST | S | ||||||
| Pinus cembra | BS, NE | NE | NE, S, W | |||||
| Pinus mugo | W, NE | |||||||
| Pinus uncinata | NE, S | |||||||
| Pinus sylvestris | NE, BS,S | NE,BS,S | ||||||
| Picea abies | NE, S | NE, W, S | NE, ST | S | ||||
| Abies alba | NE | NE | ||||||
| Juniperus sp. | W | NE | ||||||
| Juniperus communis | NE, S | NE, W | ||||||
| Larix sp. | Ne | BS,BK | ||||||
| Larix decidua | NE, ST | NE, S, W | ||||||
| Salix sp. | BS | BS | ||||||
| Populus tremula | BS,F | BS,F | ||||||
| Alnus glutinosa | S, BS | S, W | ||||||
| Betula sp. | F, W | BS | S, W | |||||
| Betula alba | F | F, BS | F, BS | F | ||||
| Betula carpatica | F | |||||||
| Betula pendula | S, BS | F | ||||||
| Betula pubescens | F, BS | F | ||||||
| Betula nana | F | |||||||
| Sambucus racemosa | S | |||||||
| Fagus sylvatica | BS, W, F |
| COD. SITE LOCATION | [79] | [85] | [97] | [87] | [86] | [84] | [88] |
|---|---|---|---|---|---|---|---|
| MACROFOSSIL TAXA | |||||||
| Amblystegium kochii | VP | ||||||
| Amblystegium serpens | VP | ||||||
| Anomodon sp. | VP | ||||||
| Aulacomnium palustre | VP | VP | |||||
| Brachythecium mildeanum | VP | ||||||
| Bryum pseudotriquetum | VP | VP | |||||
| Calliergon giganteum | VP | ||||||
| Calliergonella cuspidate | VP | ||||||
| Campylium stellatum | VP | VP | |||||
| Campylopus sp. | VP | ||||||
| Depranocladus | VP | ||||||
| Depranocladus aduncus | VP | VP | |||||
| Herzogiella seligeri | VP | ||||||
| Leucodon sciuroides | VP | ||||||
| Meesia cf hexasticha | VP | ||||||
| Meesia longiseta | VP | VP | VP | VP | |||
| Meesia trinquetra | VP | ||||||
| Philonotis calcarean | VP | ||||||
| Polytrichum strictum | VP | VP | |||||
| Scorpidium cossonii | VP | ||||||
| Scorpidium revolvens | VP | ||||||
| Sphagnum sp. | VP | VP | VP | ||||
| Sphagnum acutifolia | VP | VP | |||||
| Sphagnum angustifolium | VP | ||||||
| Sphagnum cuspidatum | VP | ||||||
| Sphagnum fuscum | VP | ||||||
| Sphagnum magellanicum | VP | VP | |||||
| Sphagnum obtusum | VP | ||||||
| Sphagnum palustre | VP | VP | VP | VP | VP | ||
| Sphagnum sec. cuspidate | VP | VP | VP | VP | |||
| Sphagnum squarrosum | VP | VP | |||||
| Sphagnum subsecundum | VP | ||||||
| Thuidium recognitum | VP | ||||||
| Warnstorfia exannulata | VP | VP | VP | ||||
| Warnstorfia fluitans | VP |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Souto, M.; Castro, D.; García-Rodeja, E.; Pontevedra-Pombal, X. The Use of Plant Macrofossils for Paleoenvironmental Reconstructions in Southern European Peatlands. Quaternary 2019, 2, 34. https://doi.org/10.3390/quat2040034
Souto M, Castro D, García-Rodeja E, Pontevedra-Pombal X. The Use of Plant Macrofossils for Paleoenvironmental Reconstructions in Southern European Peatlands. Quaternary. 2019; 2(4):34. https://doi.org/10.3390/quat2040034
Chicago/Turabian StyleSouto, Martin, Daniel Castro, Eduardo García-Rodeja, and Xabier Pontevedra-Pombal. 2019. "The Use of Plant Macrofossils for Paleoenvironmental Reconstructions in Southern European Peatlands" Quaternary 2, no. 4: 34. https://doi.org/10.3390/quat2040034
APA StyleSouto, M., Castro, D., García-Rodeja, E., & Pontevedra-Pombal, X. (2019). The Use of Plant Macrofossils for Paleoenvironmental Reconstructions in Southern European Peatlands. Quaternary, 2(4), 34. https://doi.org/10.3390/quat2040034





