Using Annual Resolution Pollen Analysis to Synchronize Varve and Tree-Ring Records
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Sites and Coring
2.2. Pollen Sampling, Preparation and Analysis
2.3. Identifying Pollen Mast Years
2.4. Tree-Ring Chronologies
2.5. Meteorological Data
2.6. Statistical Analyses
3. Results
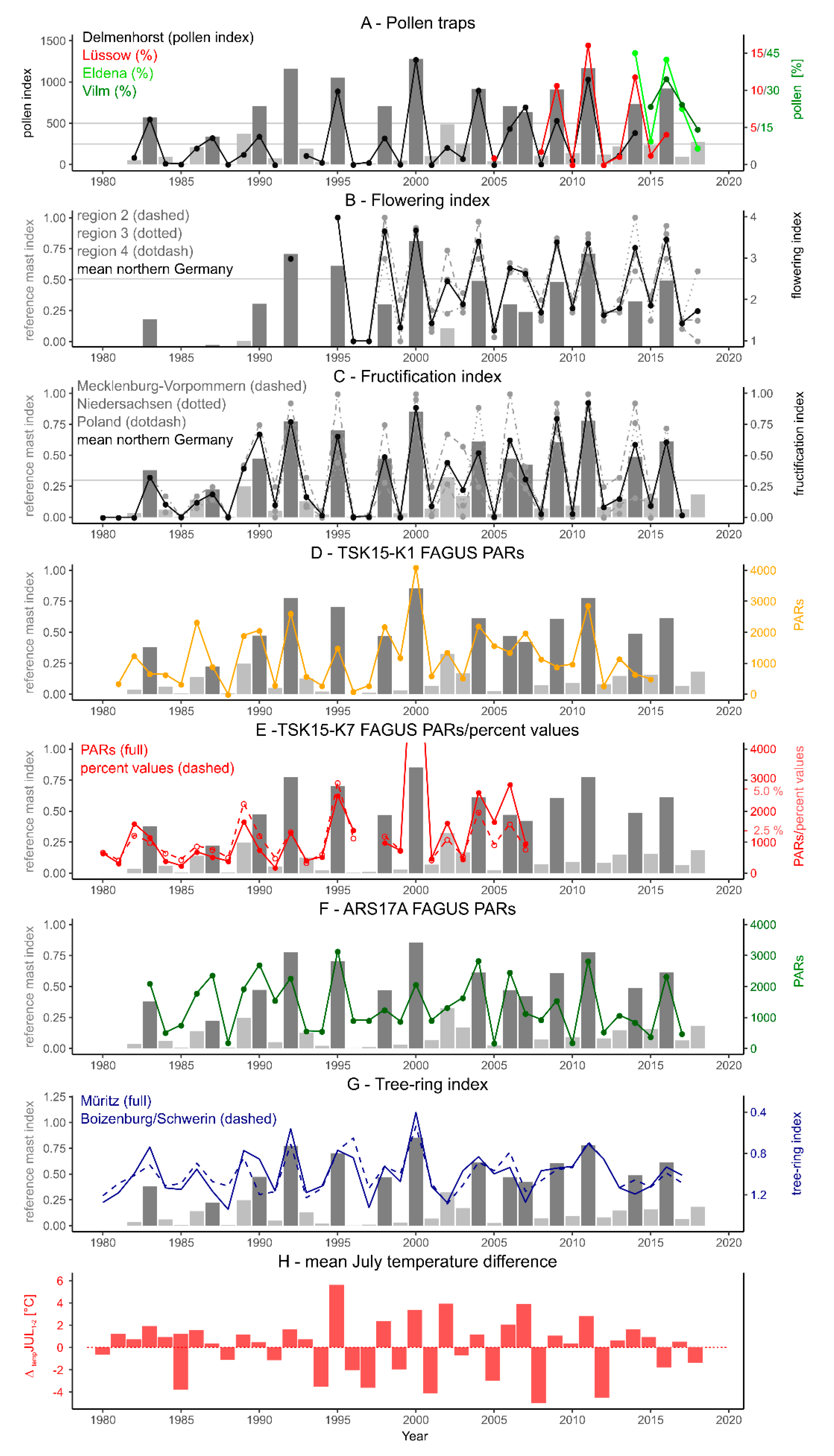
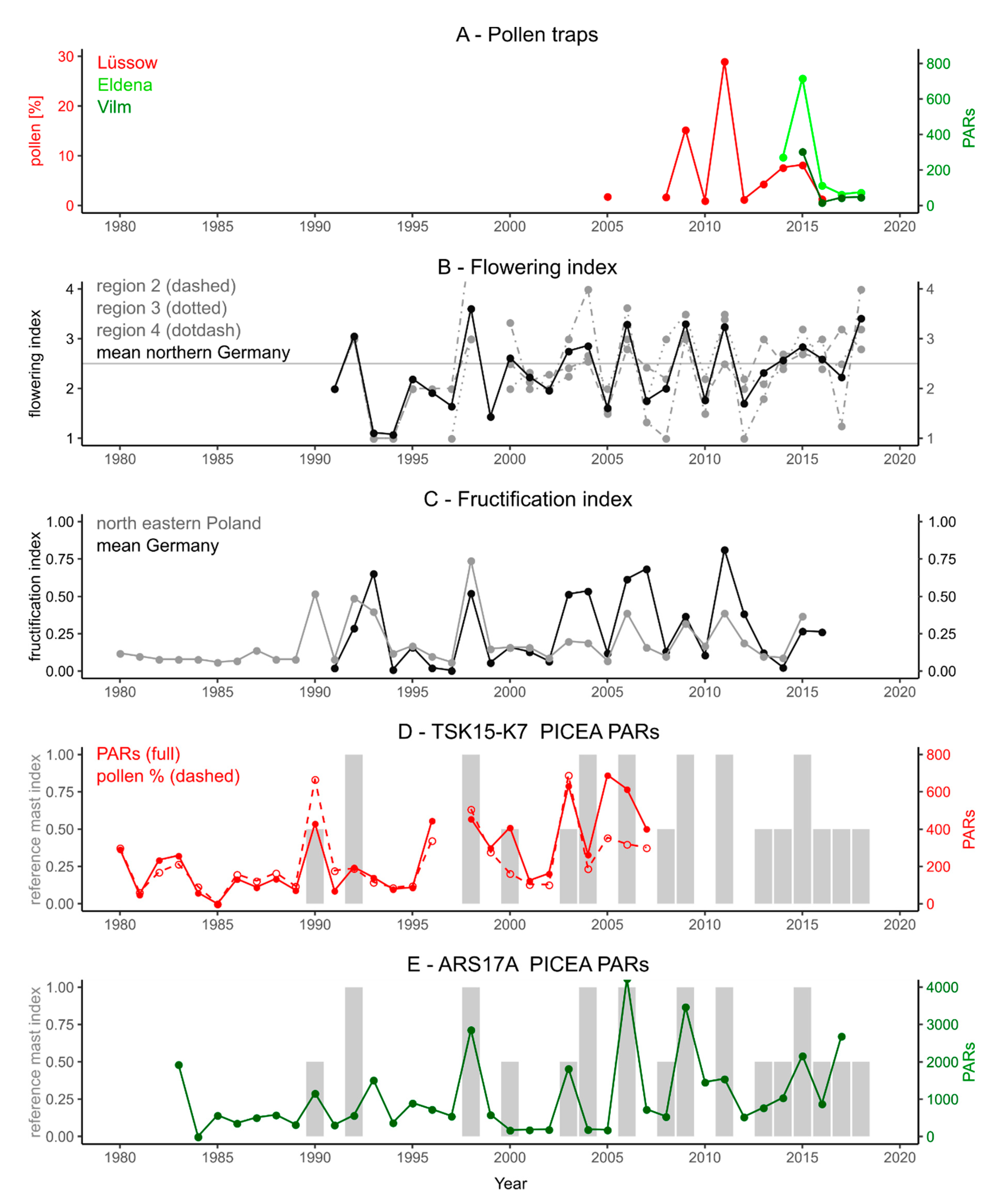
3.1. Pollen Mast Years between 1980 and 2018
3.2. Pollen Mast Years in the Lake Records
3.2.1. Fagus
3.2.2. Picea
3.3. Tree-Ring Records
4. Discussion
4.1. Annual Pollen Data
4.2. Outlook
4.2.1. Application of Annual Pollen Records
4.2.2. Practical Issues
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Wilmking, M.; Hallinger, M.; Van Bogaert, R.; Kyncl, T.; Babst, F.; Hahne, W.; Juday, G.; De Luis, M.; Novak, K.; Völlm, C. Continuously missing outer rings in woody plants at their distributional margins. Dendrochronologia 2012, 30, 213–222. [Google Scholar] [CrossRef]
- De Micco, V.; Campelo, F.; De Luis, M.; Bräuning, A.; Grabner, M.; Battipaglia, G.; Cherubini, P. Intra-annual density fluctuations in tree rings: How, when, where, and why? IAWA J. 2016, 37, 232–259. [Google Scholar] [CrossRef]
- Stokes, M.A.; Smiley, T.L. An Introduction to Tree-ring Dating; University of Arizona Press: Tucson, AZ, USA, 1996; ISBN 978-0-8165-1680-3. [Google Scholar]
- Friedrich, M.; Remmele, S.; Kromer, B.; Hofmann, J.; Spurk, M.; Kaiser, K.F.; Orcel, C.; Küppers, M. The 12,460-Year Hohenheim Oak and Pine Tree-Ring Chronology from Central Europe—A Unique Annual Record for Radiocarbon Calibration and Paleoenvironment Reconstructions. Radiocarbon 2004, 46, 1111–1122. [Google Scholar] [CrossRef]
- Grudd, H.; Briffa, K.R.; Karlén, W.; Bartholin, T.S.; Jones, P.D.; Kromer, B. A 7400-year tree-ring chronology in northern Swedish Lapland: Natural climatic variability expressed on annual to millennial timescales. Holocene 2002, 12, 657–665. [Google Scholar] [CrossRef]
- Leuschner, H.H.; Sass-Klaassen, U.; Jansma, E.; Baillie, M.G.; Spurk, M. Subfossil European bog oaks: Population dynamics and long-term growth depressions as indicators of changes in the Holocene hydro-regime and climate. Holocene 2002, 12, 695–706. [Google Scholar] [CrossRef]
- Pilcher, J.R.; Baillie, M.G.L.; Schmidt, B.; Becker, B. A 7272-year tree-ring chronology for western Europe. Nature 1984, 312, 150–152. [Google Scholar] [CrossRef]
- Kienel, U.; Dulski, P.; Ott, F.; Lorenz, S.; Brauer, A. Recently induced anoxia leading to the preservation of seasonal laminae in two NE-German lakes. J. Paleolimnol. 2013, 50, 535–544. [Google Scholar] [CrossRef]
- Żarczyński, M.; Tylmann, W.; Goslar, T. Multiple varve chronologies for the last 2000 years from the sediments of Lake Żabińskie (northeastern Poland)—Comparison of strategies for varve counting and uncertainty estimations. Quat. Geochronol. 2018, 47, 107–119. [Google Scholar] [CrossRef]
- Wulf, S.; Dräger, N.; Ott, F.; Serb, J.; Appelt, O.; Guðmundsdóttir, E.; van den Bogaard, C.; Słowiński, M.; Błaszkiewicz, M.; Brauer, A. Holocene tephrostratigraphy of varved sediment records from Lakes Tiefer See (NE Germany) and Czechowskie (N Poland). Quat. Sci. Rev. 2016, 132, 1–14. [Google Scholar] [CrossRef]
- Jones, G.; Lane, C.S.; Brauer, A.; Davies, S.M.; de Bruijn, R.; Engels, S.; Haliuc, A.; Hoek, W.Z.; Merkt, J.; Sachse, D.; et al. The Lateglacial to early Holocene tephrochronological record from Lake Hämelsee, Germany: A key site within the European tephra framework. Boreas 2018, 28, 28–40. [Google Scholar] [CrossRef]
- Wastegård, S.; Gudmundsdóttir, E.R.; Lind, E.M.; Timms, R.G.O.; Björck, S.; Hannon, G.E.; Olsen, J.; Rundgren, M. Towards a Holocene tephrochronology for the Faroe Islands, North Atlantic. Quat. Sci. Rev. 2018, 195, 195–214. [Google Scholar] [CrossRef]
- Smith, V.C.; Staff, R.A.; Blockley, S.P.; Ramsey, C.B.; Nakagawa, T.; Mark, D.F.; Takemura, K.; Danhara, T. Identification and correlation of visible tephras in the Lake Suigetsu SG06 sedimentary archive, Japan: Chronostratigraphic markers for synchronising of east Asian/west Pacific palaeoclimatic records across the last 150 ka. Quat. Sci. Rev. 2013, 67, 121–137. [Google Scholar] [CrossRef]
- Zawalna-Geer, A.; Lindsay, J.M.; Davies, S.; Augustinus, P.; Davies, S. Extracting a primary Holocene crytoptephra record from Pupuke maar sediments, Auckland, New Zealand. J. Quat. Sci. 2016, 31, 442–457. [Google Scholar] [CrossRef]
- Czymzik, M.; Muscheler, R.; Adolphi, F.; Mekhaldi, F.; Dräger, N.; Ott, F.; Słowiński, M.; Błaszkiewicz, M.; Aldahan, A.; Possnert, G.; et al. Synchronizing 10Be in two varved lake sediment records to IntCal13 14C during three grand solar minima. Clim. Past 2018, 14, 687–696. [Google Scholar] [CrossRef]
- Pidek, I.A.; Svitavská-Svobodová, H.; Van Der Knaap, W.O.; Noryśkiewicz, A.M.; Filbrandt-Czaja, A.; Noryśkiewicz, B.; Latałowa, M.; Zimny, M.; Święta-Musznicka, J.; Bozilova, E.; et al. Variation in annual pollen accumulation rates of Fagus along a N–S transect in Europe based on pollen traps. Veg. Hist. Archaeobotany 2010, 19, 259–270. [Google Scholar] [CrossRef][Green Version]
- Simoleit, A.; Wachter, R.; Gauger, U.; Werchan, M.; Werchan, B.; Zuberbier, T.; Bergmann, K.-C. Pollen season of European beech (Fagus sylvatica L.) and temperature trends at two German monitoring sites over a more than 30-year period. Aerobiologia 2016, 32, 489–497. [Google Scholar] [CrossRef]
- Nussbaumer, A.; Waldner, P.; Apuhtin, V.; Aytar, F.; Benham, S.; Bussotti, F.; Eichhorn, J.; Eickenscheidt, N.; Fabianek, P.; Falkenried, L.; et al. Impact of weather cues and resource dynamics on mast occurrence in the main forest tree species in Europe. For. Ecol. Manag. 2018, 429, 336–350. [Google Scholar] [CrossRef]
- Drobyshev, I.; Övergaard, R.; Saygin, I.; Niklasson, M.; Hickler, T.; Karlsson, M.; Sykes, M.T. Masting behaviour and dendrochronology of European beech (Fagus sylvatica L.) in southern Sweden. For. Ecol. Manag. 2010, 259, 2160–2171. [Google Scholar] [CrossRef]
- Hacket-Pain, A.J.; Friend, A.D.; Lageard, J.G.; Thomas, P.A. The influence of masting phenomenon on growth-climate relationships in trees: Explaining the influence of previous summers’ climate on ring width. Tree Physiol. 2015, 35, 319–330. [Google Scholar] [CrossRef] [PubMed]
- Vacchiano, G.; Hacket-Pain, A.; Turco, M.; Motta, R.; Maringer, J.; Conedera, M.; Drobyshev, I.; Ascoli, D.; Hacket-Pain, A. Spatial patterns and broad-scale weather cues of beech mast seeding in Europe. New Phytol. 2017, 215, 595–608. [Google Scholar] [CrossRef] [PubMed]
- Piovesan, G.; Adams, J.M.; Bernabei, M.; Selås, V. Climatic factors controlling reproduction and growth of Norway spruce in southern Norway. Can. J. For. Res. 2002, 32, 217–225. [Google Scholar]
- Hacket-Pain, A.J.; Lageard, J.G.A.; Thomas, P.A. Drought and reproductive effort interact to control growth of a temperate broadleaved tree species (Fagus sylvatica). Tree Physiol. 2017, 37, 744–754. [Google Scholar] [CrossRef] [PubMed]
- Drobyshev, I.; Niklasson, M.; Mazerolle, M.J.; Bergeron, Y. Reconstruction of a 253-year long mast record of European beech reveals its association with large scale temperature variability and no long-term trend in mast frequencies. Agric. For. Meteorol. 2014, 192, 9–17. [Google Scholar] [CrossRef]
- Kienel, U.; Kirillin, G.; Brademann, B.; Plessen, B.; Lampe, R.; Brauer, A. Effects of spring warming and mixing duration on diatom deposition in deep Tiefer See, NE Germany. J. Paleolimnol. 2017, 57, 37–49. [Google Scholar] [CrossRef]
- Dräger, N.; Theuerkauf, M.; Szeroczyńska, K.; Wulf, S.; Tjallingii, R.; Plessen, B.; Kienel, U.; Brauer, A.; Szeroczynska, K.; Wulf, S.; et al. Varve microfacies and varve preservation record of climate change and human impact for the last 6000 years at Lake Tiefer See (NE Germany). Holocene 2017, 27, 1–15. [Google Scholar] [CrossRef]
- Scharf, B.W. Eutrophication history of Lake Arendsee (Germany). Palaeogeogr. Palaeoclim. Palaeoecol. 1998, 140, 85–96. [Google Scholar] [CrossRef]
- Stüben, D.; Walpersdorf, E.; Voss, K.; Ronicke, H.; Schimmele, M.; Baborowski, M.; Luther, G.; Elsner, W. Application of lake marl at Lake Arendsee, NE Germany: First results of a geochemical monitoring during the restoration experiment. Sci. Total. Environ. 1998, 218, 33–44. [Google Scholar] [CrossRef]
- Hupfer, M.; Pöthig, R.; Brüggemann, R.; Geller, W. Mechanical resuspension of autochthonous calcite (Seekreide) failed to control internal phosphorus cycle in a eutrophic lake. Water Res. 2000, 34, 859–867. [Google Scholar] [CrossRef]
- Joosten, H.; de Klerk, P. What’s in a name? Some thoughts on pollen classification, identification, and nomenclature in Quaternary palynology. Rev. Palaeobot. Palynol. 2002, 122, 29–45. [Google Scholar] [CrossRef]
- Tauber, H. A static non-overload pollen collector. New Phytol. 1974, 73, 359–369. [Google Scholar] [CrossRef]
- Ascoli, D.; Maringer, J.; Hacket-Pain, A.; Conedera, M.; Drobyshev, I.; Motta, R.; Cirolli, M.; Kantorowicz, W.; Zang, C.; Schueler, S.; et al. Two centuries of masting data for European beech and Norway spruce across the European continent. Ecology 2017, 98, 1473. [Google Scholar] [CrossRef] [PubMed]
- Bunn, A.G. A dendrochronology program library in R (dplR). Dendrochronologia 2008, 26, 115–124. [Google Scholar] [CrossRef]
- Bunn, A.G. Statistical and visual crossdating in R using the dplR library. Dendrochronologia 2010, 28, 251–258. [Google Scholar] [CrossRef]
- Bunn, A.G.; Korpela, M.; Biondi, F.; Campelo, F.; Qeadan, F.; Mérian, P.; Zang, C. dplR: Dendrochronology Program Library in R. Available online: https://cran.r-project.org/web/packages/dplR/index.html (accessed on 8 July 2019).
- Schneck, D. Das Blühen der Waldbäume. AFZ-DerWald 2014, 69, 27–29. [Google Scholar]
- Hacket-Pain, A.J.; Ascoli, D.; Vacchiano, G.; Biondi, F.; Cavin, L.; Conedera, M.; Drobyshev, I.; Liñán, I.D.; Friend, A.D.; Grabner, M.; et al. Climatically controlled reproduction drives interannual growth variability in a temperate tree species. Ecol. Lett. 2018, 21, 1833–1844. [Google Scholar] [CrossRef] [PubMed]
- Mund, M.; Kutsch, W.L.; Wirth, C.; Kahl, T.; Knohl, A.; Skomarkova, M.V.; Schulze, E.D. The influence of climate and fructification on the inter-annual variability of stem growth and net primary productivity in an old-growth, mixed beech forest. Tree Physiol. 2010, 30, 689–704. [Google Scholar] [CrossRef] [PubMed]
- Pearse, I.S.; Koenig, W.D.; Kelly, D. Mechanisms of mast seeding: Resources, weather, cues, and selection. New Phytol. 2016, 212, 546–562. [Google Scholar] [CrossRef]
- Bogdziewicz, M.; Szymkowiak, J.; Fernández-Martínez, M.; Peñuelas, J.; Espelta, J.M. The effects of local climate on the correlation between weather and seed production differ in two species with contrasting masting habit. Agric. For. Meteorol. 2019, 268, 109–115. [Google Scholar] [CrossRef]
- Kasprzyk, I.; Ortyl, B.; Dulska-Jeż, A. Relationships among weather parameters, airborne pollen and seed crops of Fagus and Quercus in Poland. Agric. For. Meteorol. 2014, 197, 111–122. [Google Scholar] [CrossRef]
- Giesecke, T.; Fontana, S.L. Revisiting pollen accumulation rates from Swedish lake sediments. Holocene 2008, 18, 293–305. [Google Scholar] [CrossRef]
- Scharnweber, T.; Heußner, K.-U.; Smiljanic, M.; Heinrich, I.; Van Der Maaten-Theunissen, M.; Van Der Maaten, E.; Struwe, T.; Buras, A.; Wilmking, M. Removing the no-analogue bias in modern accelerated tree growth leads to stronger medieval drought. Sci. Rep. 2019, 9, 2509. [Google Scholar] [CrossRef] [PubMed]
- Skomarkova, M.V.; Vaganov, E.A.; Mund, M.; Knohl, A.; Linke, P.; Boerner, A.; Schulze, E.-D. Inter-annual and seasonal variability of radial growth, wood density and carbon isotope ratios in tree rings of beech (Fagus sylvatica) growing in Germany and Italy. Trees 2006, 20, 571–586. [Google Scholar] [CrossRef]
- Fernández-Martínez, M.; Vicca, S.; Janssens, I.A.; Espelta, J.M.; Peñuelas, J. The North Atlantic Oscillation synchronises fruit production in western European forests. Ecography 2017, 40, 864–874. [Google Scholar] [CrossRef]
- Ascoli, D.; Vacchiano, G.; Turco, M.; Conedera, M.; Drobyshev, I.; Maringer, J.; Motta, R.; Hacket-Pain, A. Inter-annual and decadal changes in teleconnections drive continental-scale synchronization of tree reproduction. Nat. Commun. 2017, 8, 2205. [Google Scholar] [CrossRef]
- Piovesan, G.; Adams, J.M. Masting behaviour in beech: Linking reproduction and climatic variation. Can. J. Bot. 2001, 79, 1039–1047. [Google Scholar]
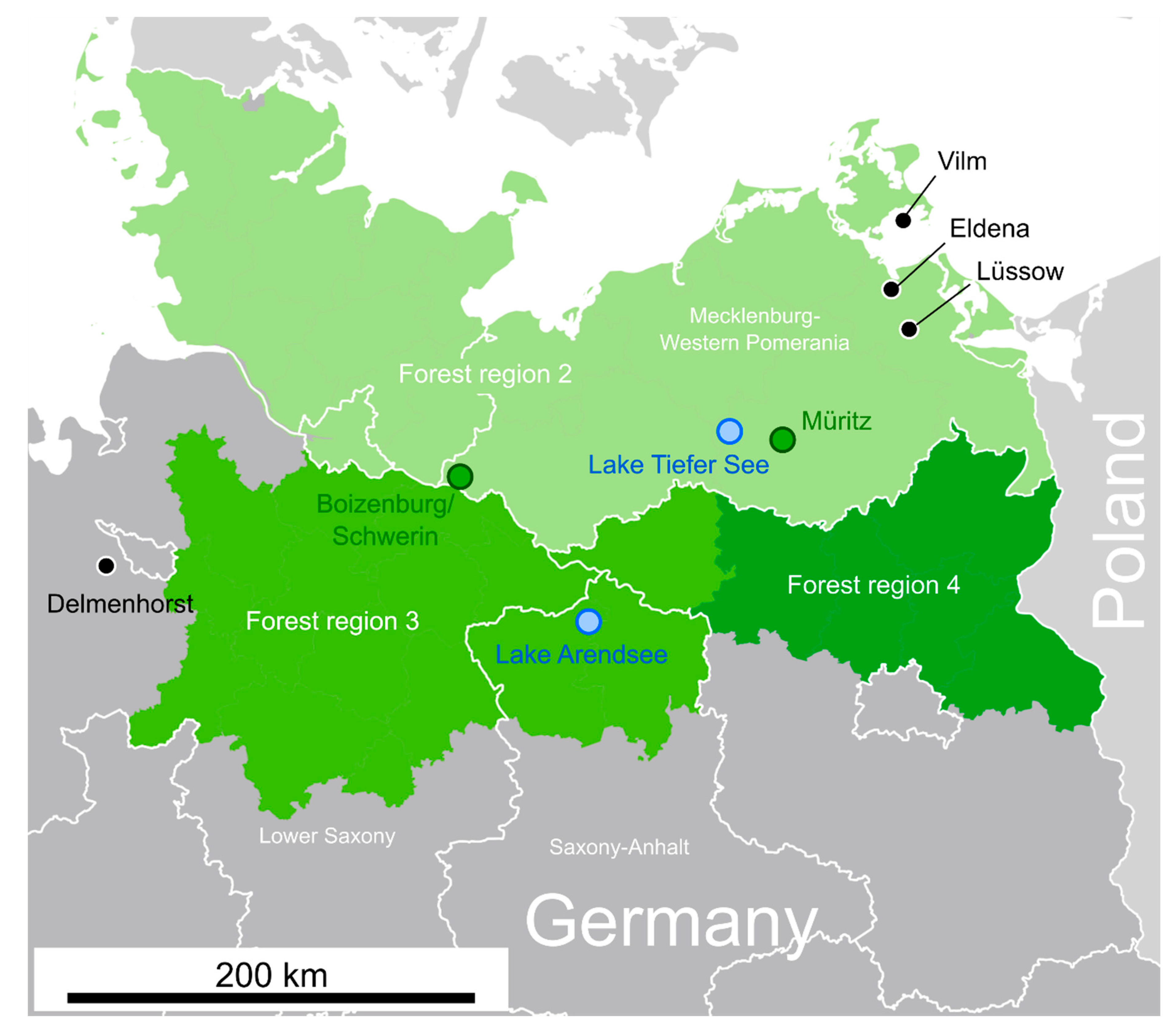
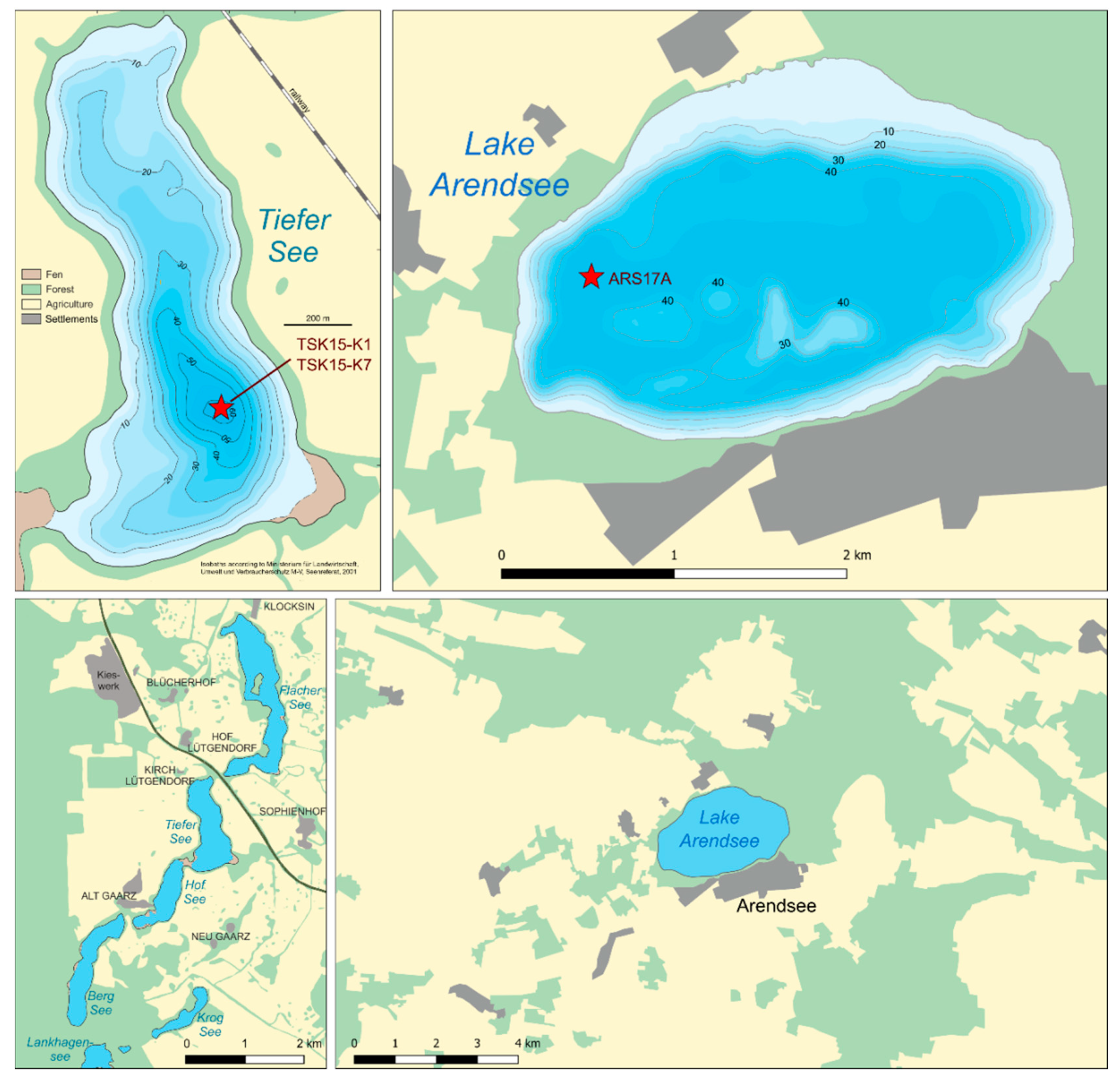
| Core | Location | Sample Period |
|---|---|---|
| TSK15-K1 | Lake Tiefer See (53.59° N, 12.53° E) | 1981–2015 |
| TSK15-K7 | Lake Tiefer See (53.59° N, 12.53° E) | 1980–2007 |
| ARS17A | Lake Arendsee (52.8891° N, 11.4598° E) | 1983–2017 |
| Trap | Location | Site Type | Sampling Period |
|---|---|---|---|
| Delmenhorst | 53.05° N 8.63° E | roof top in the city centre, 17.5 m above street level | 1982–2014 (1992 missing) |
| Lüssow | 53.8911° N 13.4781° E | Alder (Alnus glutinosa) carr (100 ha) in a large river valley mire | 2005–2016 (2006 and 2007 missing) |
| Eldena | 54.0784° N 13.4767° E | old-growth forest with Fagus sylvatica, Quercus robur, Acer spp. and Carpinus betulus | 2014–2018 |
| Vilm | 54.3273° N 13.5396° E | old-growth forest with Fagus sylvatica, Quercus robur, Acer spp. and Carpinus betulus | 2015–2018 |
| Relationship Tested | TSK15-K1 | TSK15-K7 | ARS17A |
|---|---|---|---|
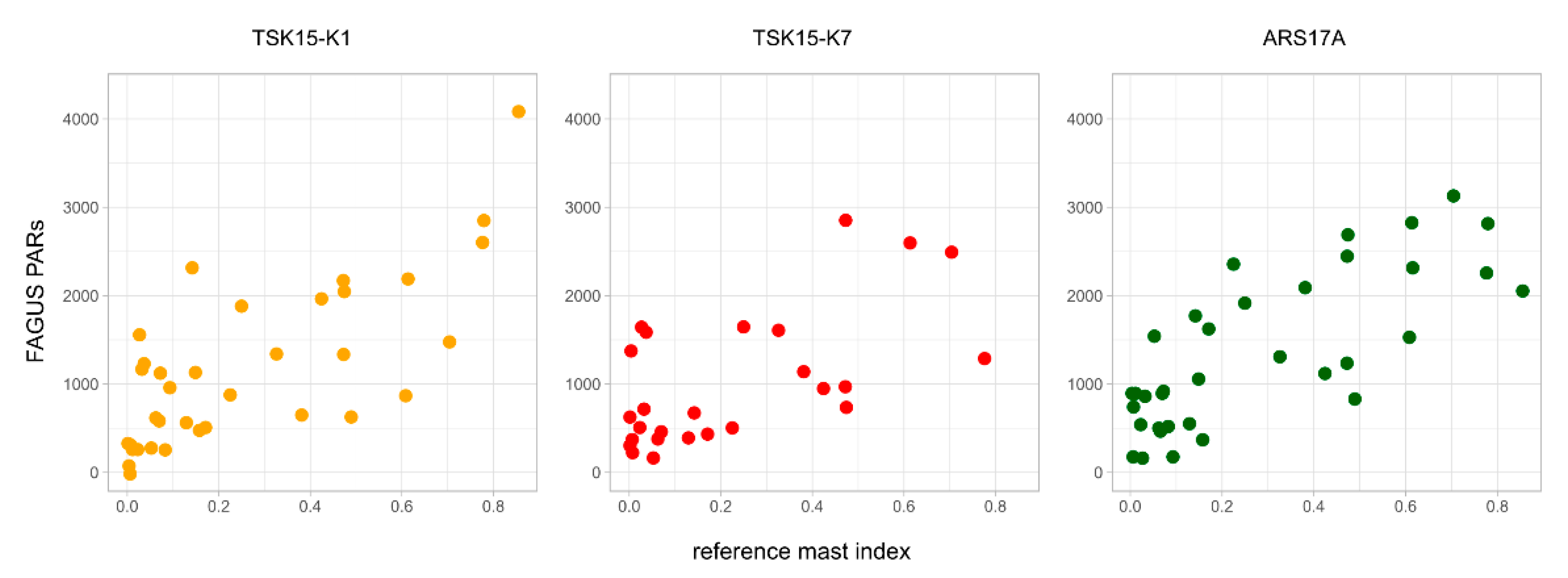
| Fagus PARs ~ reference mast index | r = 0.73 p = 1.124 × 10−6 glk = 0.79 (33 a) | r = 0.63 p = 0.00074 glk = 0.79 (25 a) | r = 0.77 p = 6.133 × 10−8 glk = 0.90 (35 a) |
| Picea PARs ~ reference mast index | - | r = 0.36 p = 0.1702 glk = 0.66 (15 a) | r = 0.56 p = 0.002192 glk = 0.73 (27 a) |
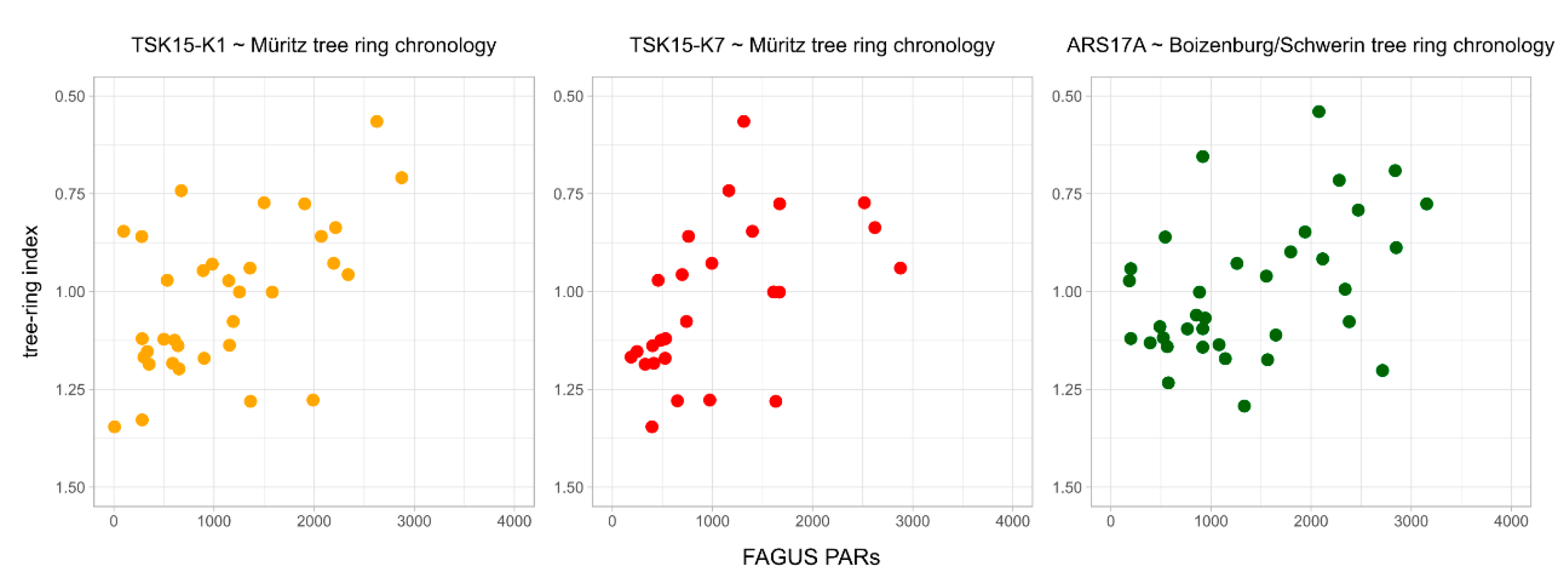
| Tree-ring index ~ Fagus PARs | r = −0.67 p = 1.129 × 10−5 1-glk = 0.65 (35 a) | r = −0.67 p = 0.00014 1-glk = 0.85 (25 a) | r = −0.44 p = 0.0082 1-glk = 0.72 (35 a) |
| Species | SD |
|---|---|
| Abies alba | 0.52 |
| Alnus glutinosa | 0.39 |
| Betula pendula | 0.29 |
| Carpinus betulus | 0.62 |
| Fagus sylvatica | 0.82 |
| Picea abies | 0.81 |
| Pinus sylvestris | 0.41 |
| Qercus petraea | 0.41 |
| Qercus robur | 0.39 |
| Tilia cordata | 0.37 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Theuerkauf, M.; Engelbrecht, E.; Dräger, N.; Hupfer, M.; Mrotzek, A.; Prager, A.; Scharnweber, T. Using Annual Resolution Pollen Analysis to Synchronize Varve and Tree-Ring Records. Quaternary 2019, 2, 23. https://doi.org/10.3390/quat2030023
Theuerkauf M, Engelbrecht E, Dräger N, Hupfer M, Mrotzek A, Prager A, Scharnweber T. Using Annual Resolution Pollen Analysis to Synchronize Varve and Tree-Ring Records. Quaternary. 2019; 2(3):23. https://doi.org/10.3390/quat2030023
Chicago/Turabian StyleTheuerkauf, Martin, Eike Engelbrecht, Nadine Dräger, Michael Hupfer, Almut Mrotzek, Anja Prager, and Tobias Scharnweber. 2019. "Using Annual Resolution Pollen Analysis to Synchronize Varve and Tree-Ring Records" Quaternary 2, no. 3: 23. https://doi.org/10.3390/quat2030023
APA StyleTheuerkauf, M., Engelbrecht, E., Dräger, N., Hupfer, M., Mrotzek, A., Prager, A., & Scharnweber, T. (2019). Using Annual Resolution Pollen Analysis to Synchronize Varve and Tree-Ring Records. Quaternary, 2(3), 23. https://doi.org/10.3390/quat2030023





