Factors Influencing the Rheology of Methane Foam for Gas Mobility Control in High-Temperature, Proppant-Fractured Reservoirs
Abstract
1. Introduction
2. Results and Discussion
2.1. Surfactant Characterization
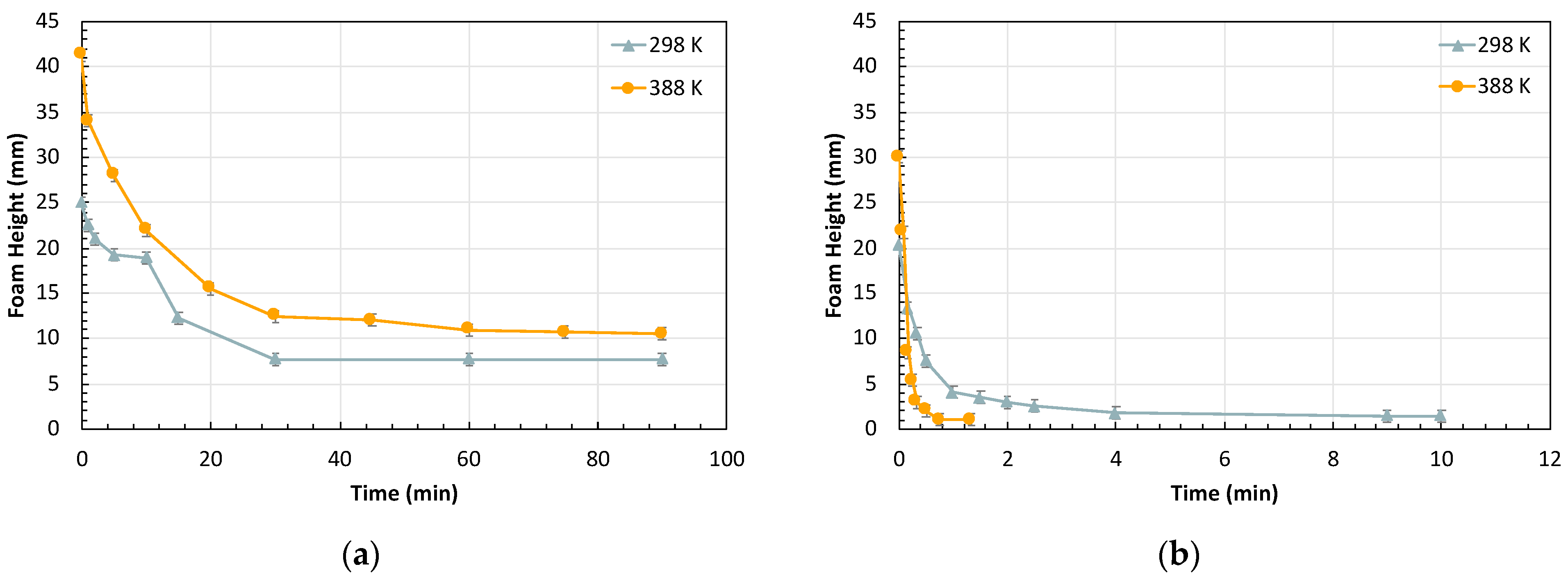
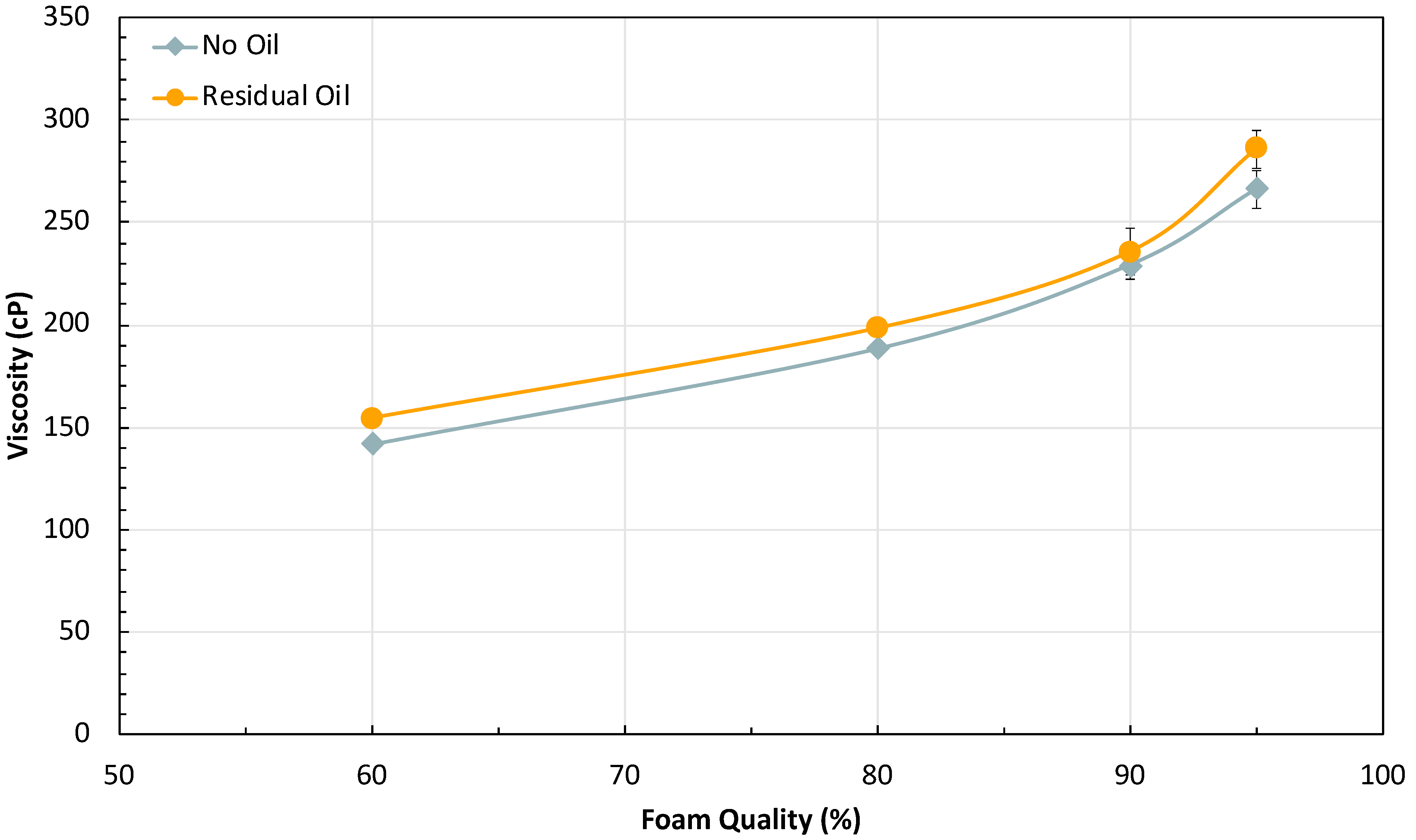
2.1.1. Effect of Oil on Foam Stability
2.1.2. Emulsion Tendency Test
2.2. Foam Transport through Proppant Pack Results
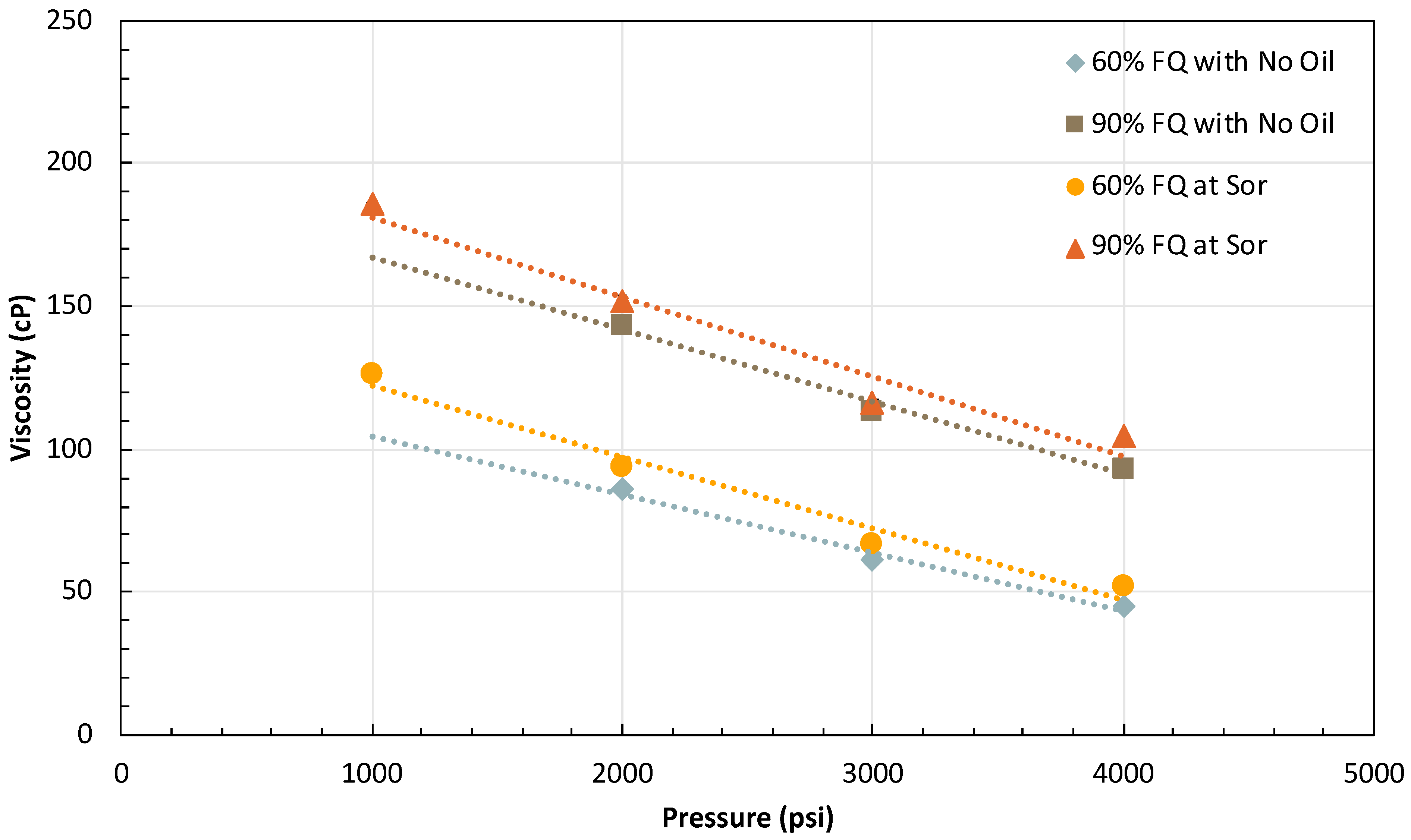
2.2.1. Foam Rheology at Residual Oil
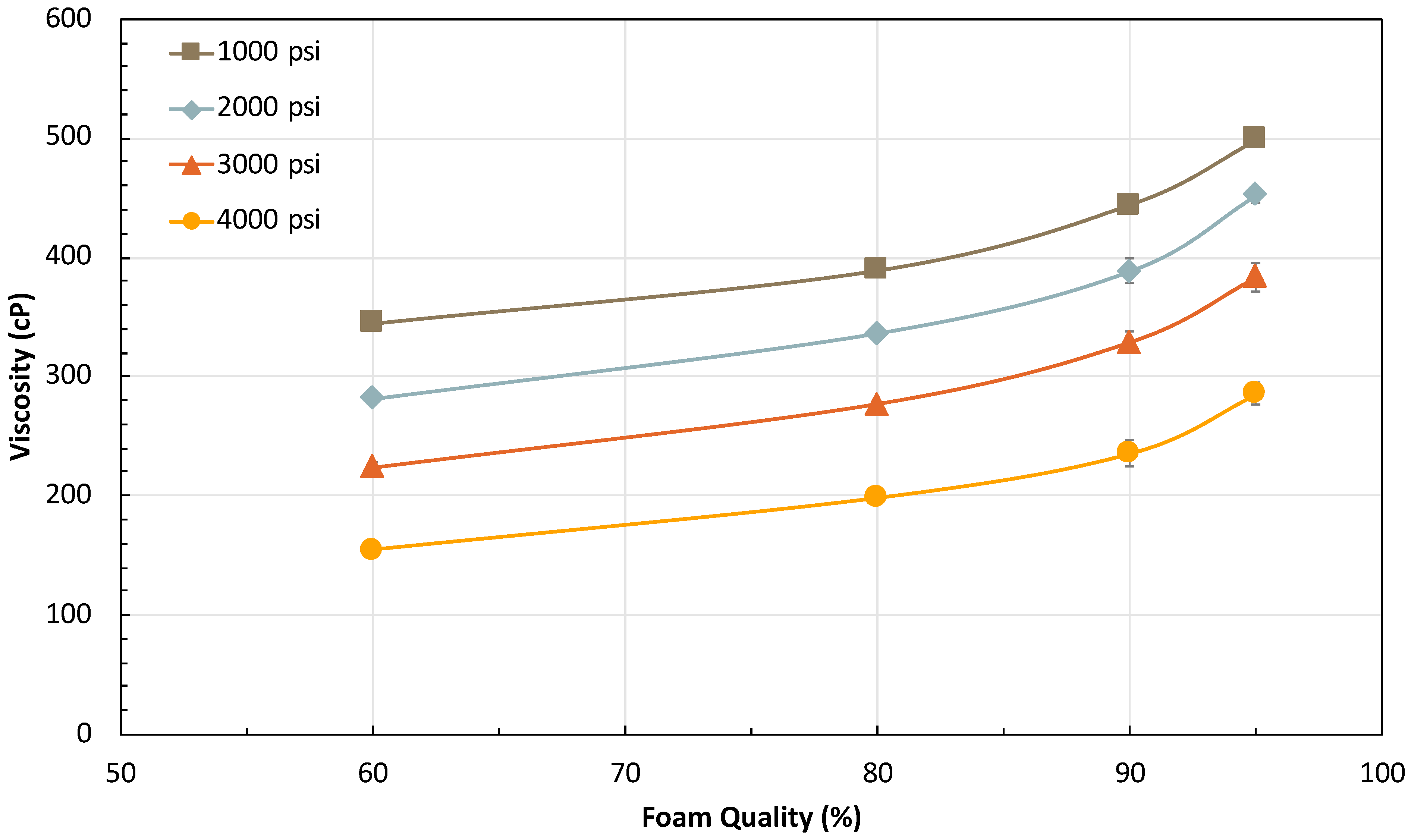
2.2.2. Effect of Foam Quality
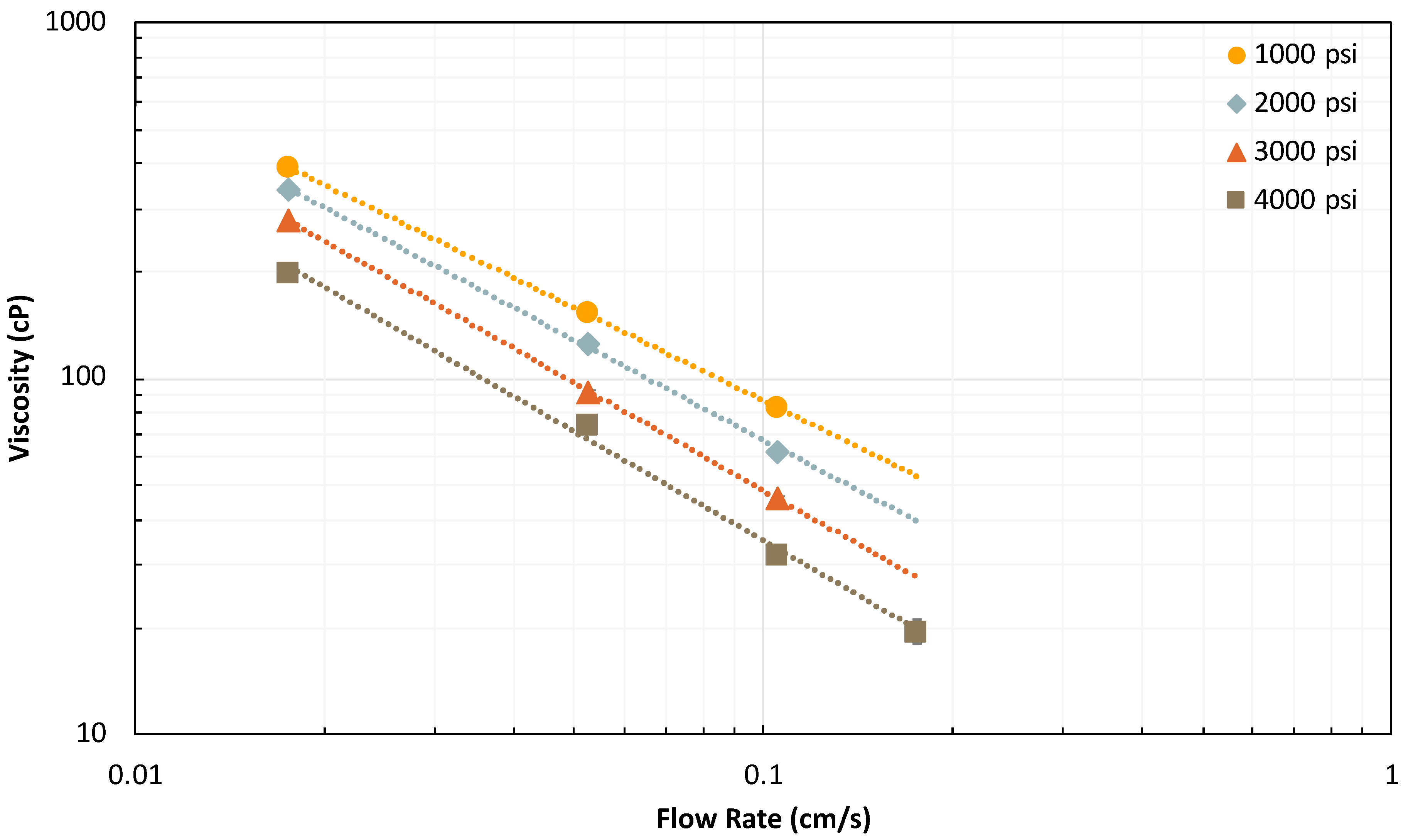
2.2.3. Effect of Total Flow Rate
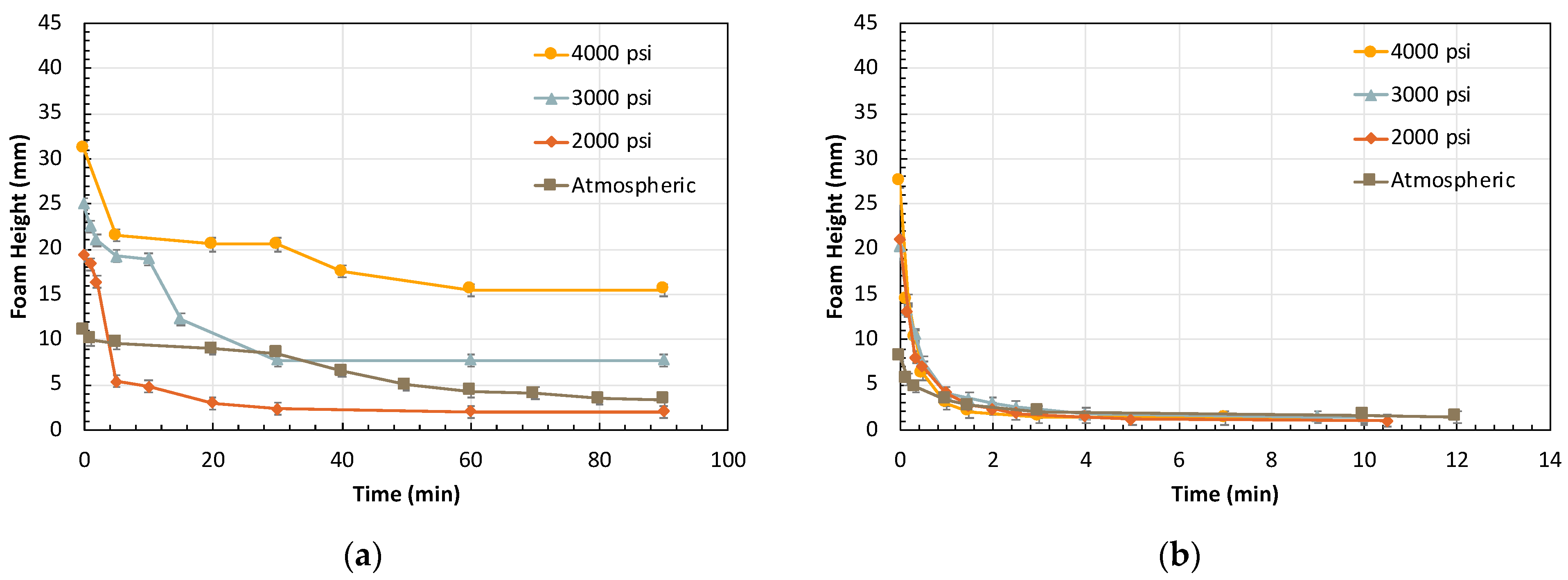
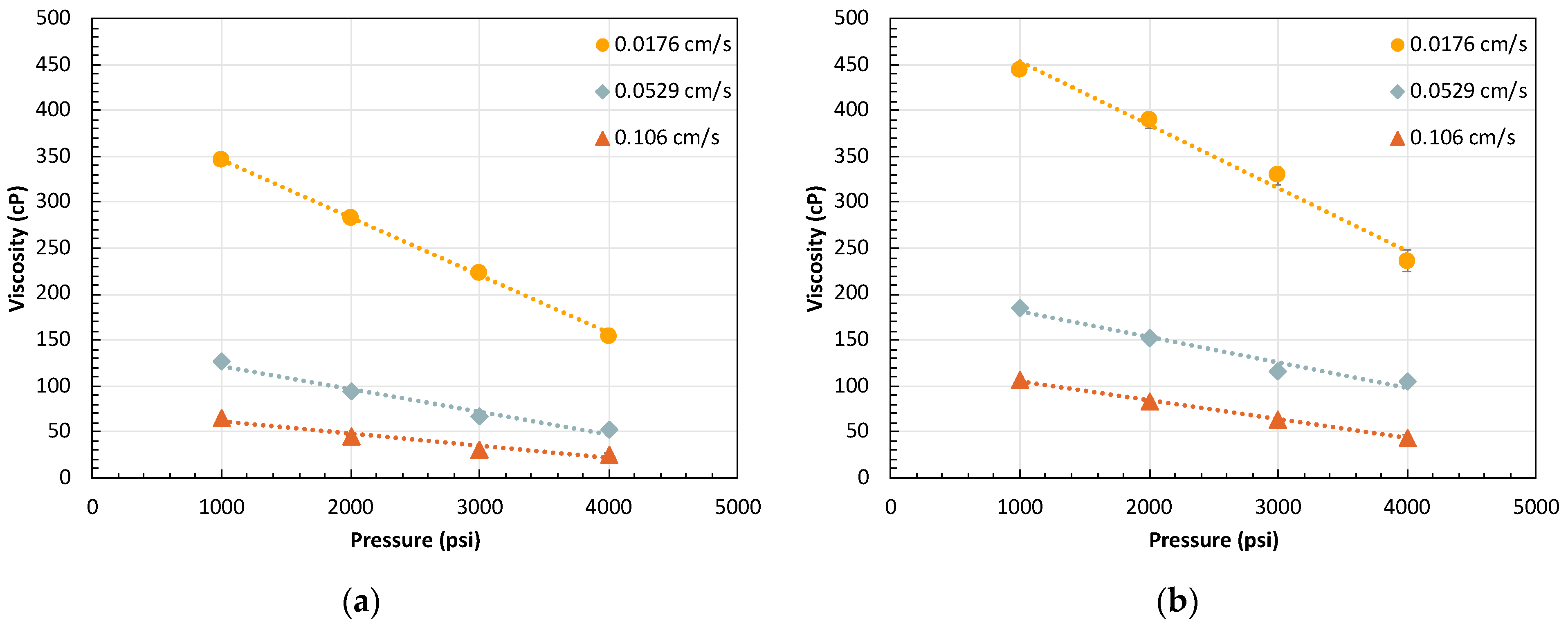
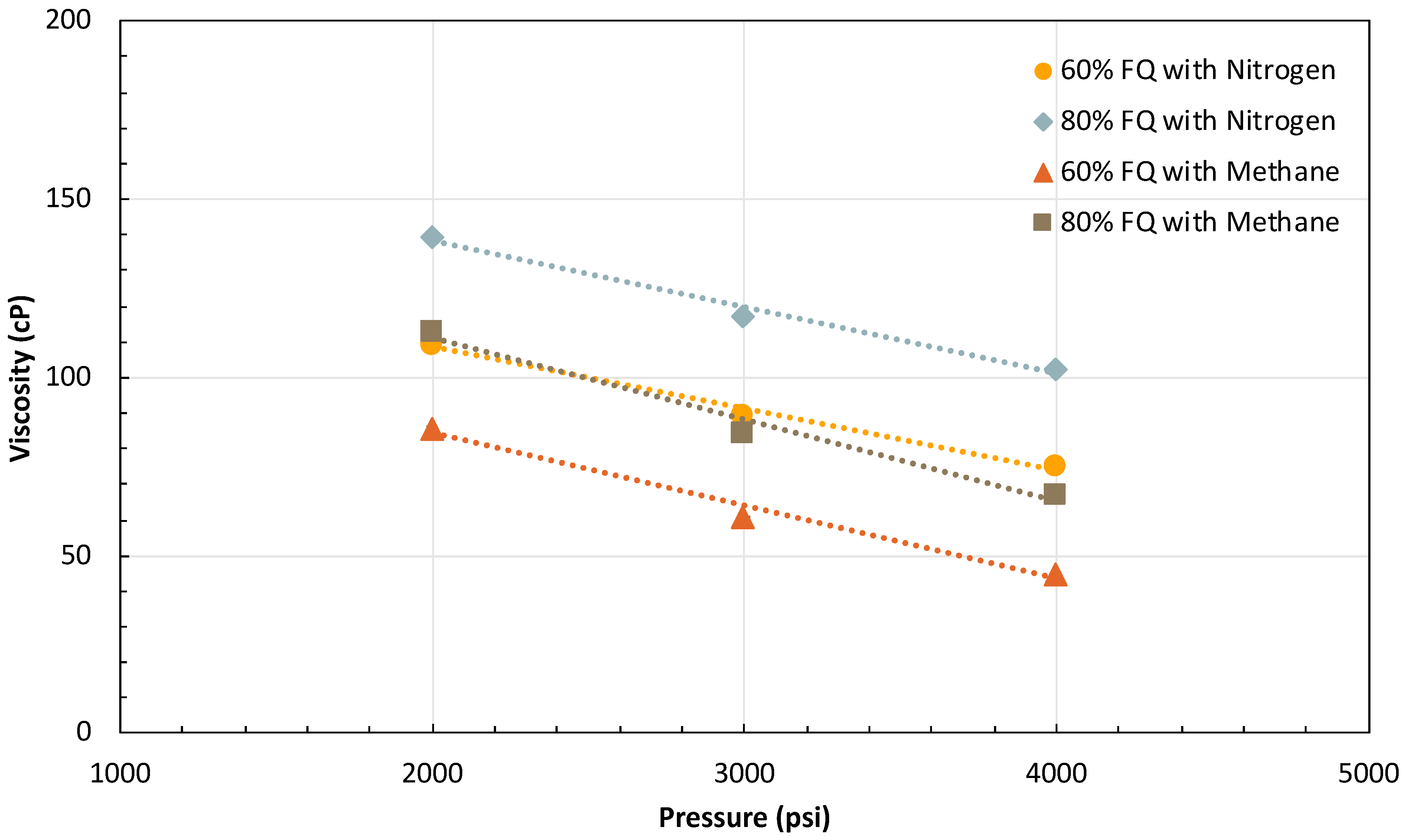
2.3. Effect of Pressure
2.3.1. Effect of Residual Oil on Pressure-Dependent Foam Rheology
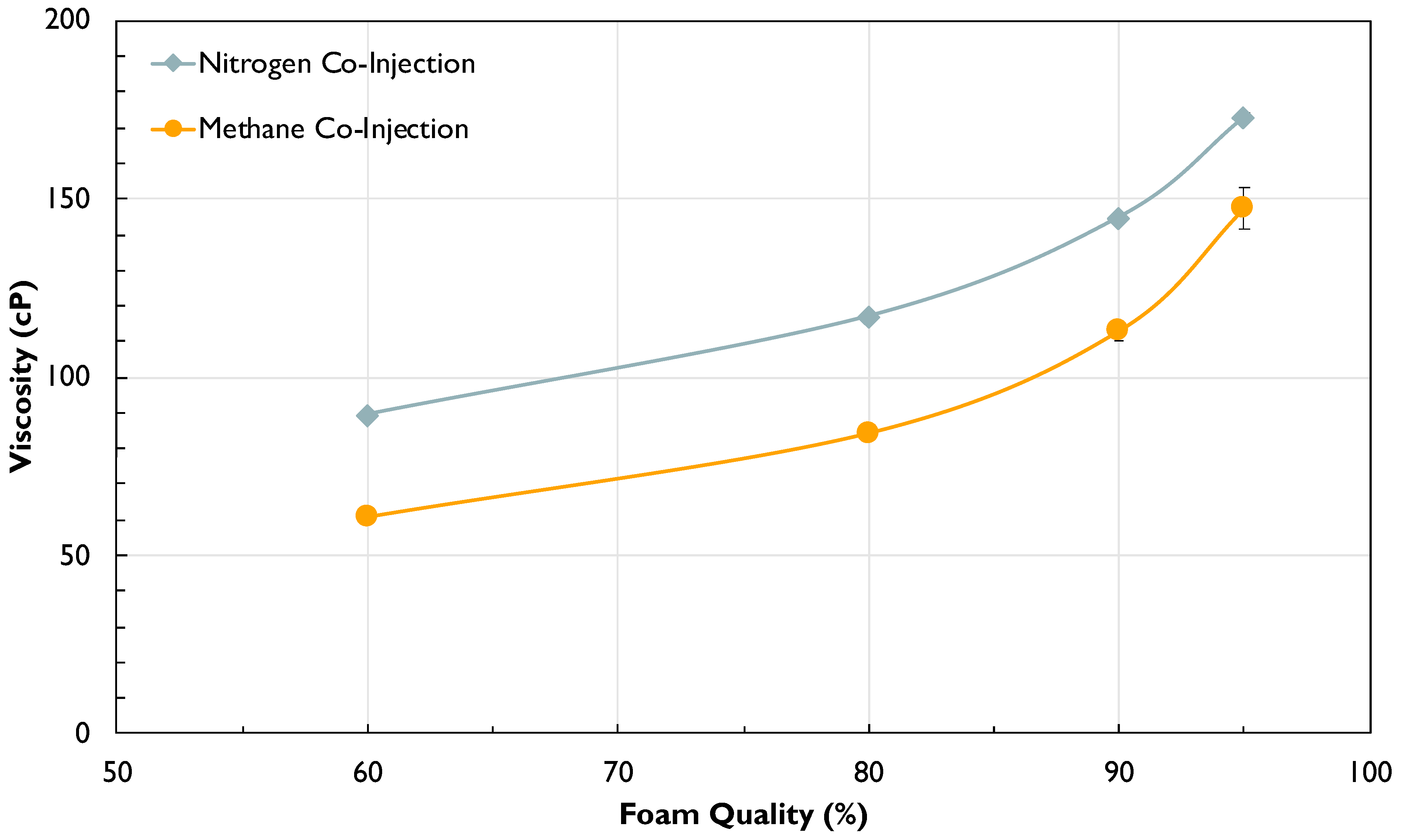
2.3.2. Effect of Injection Gas on Pressure-Dependent Foam Rheology
2.3.3. Effect of Surfactant Type on Pressure-Dependent Foam Rheology
3. Experimental Section
4. Methodology
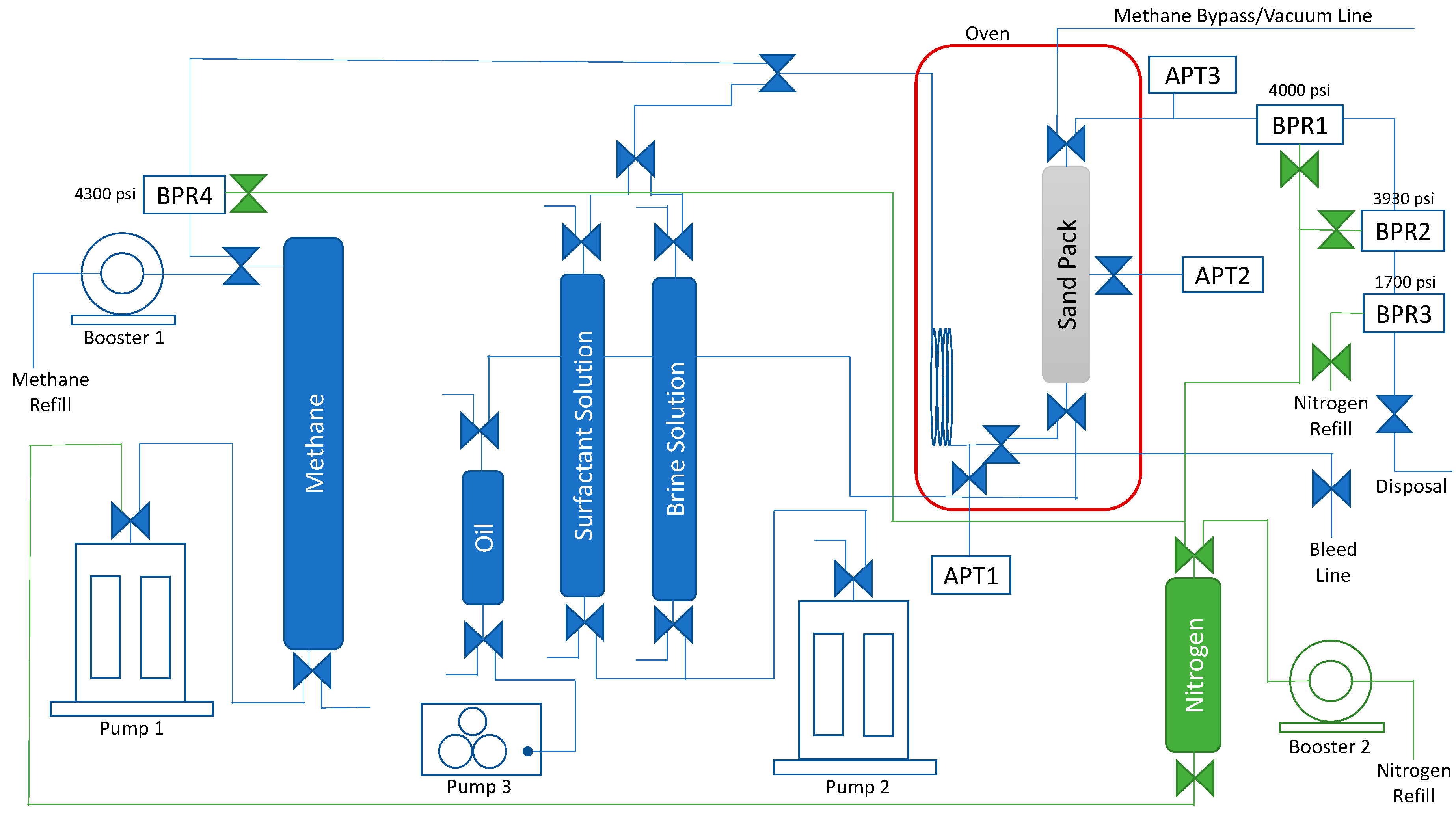
4.1. Experimental Core Flooding Setup
4.2. Surfactant Characterization
4.3. Effect of Oil on Foam Stability
4.4. Emulsion Tendency Test
4.5. Proppant Pack Preparation
4.6. Flood Procedure
5. Conclusions
- (1)
- The developed foaming formulation can generate high-viscosity (on the order of 100 cP) foam for use in the high-pressure, high-temperature, and high-permeability conditions of proppant-fractured reservoirs.
- (2)
- The foaming formulation can continue to generate foam in very dry conditions (up to 95% FQ) without sacrificing strength, which enables less water and surfactant consumption for foam generation.
- (3)
- The fluid-shear dependency confirmed strong shear-thinning rheology in the practical flow rate range for HnP processes. This shear-thinning behavior is indicative of the formulation’s ability to produce strong foam, maintain conformance control, and minimize loss of injection gas as it propagates deeper into the fracture, as well as its favorable injectivity in the near-wellbore region.
- (4)
- The inversely linear dependency of foam viscosity on system pressure is a novel finding of significant interest. Foam viscosity was observed to linearly decrease with increasing system pressure, regardless of foam quality and total flow rate. This inverse trend could be beneficial for HnP applications because it can act as a negative feedback regulator for injection gas loss and would prevent diversion of mobilized oil during the puff stage. The mechanism causing this trend was proved not to be related to oil swelling and interfacial hydrophobic attraction between surfactant and hydrocarbon gas phase at higher pressure. The observation of different pressure trends with other tested surfactants warrants the need for future work on the behaviors of surfactants and foam at high pressure, with a focus on interfacial adsorption and the properties of surfactants.
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Milad, M.; Junin, R.; Sidek, A.; Imqam, A.; Tarhuni, M. Huff-n-Puff Technology for Enhanced Oil Recovery in Shale/Tight Oil Reservoirs: Progress, Gaps, and Perspectives. Energy Fuels 2021, 35, 17279–17333. [Google Scholar] [CrossRef]
- Kathel, P.; Mohanty, K.K. EOR in Tight Oil Reservoirs through Wettability Alteration. In Proceedings of the SPE Annual Technical Conference and Exhibition, New Orleans, LA, USA, 30 September–2 October 2013; p. D021S020R008. [Google Scholar]
- Fink, J. Petroleum Engineer’s Guide to Oil Field Chemicals and Fluids, 1st ed.; Elsevier Science and Technology: Oxford, UK, 2011; ISBN 9780123838452. [Google Scholar]
- Dyni, J.R. Geology and Resources of Some World Oil-Shale Deposits; U.S. Geological Survey Scientific Investigations Report 2005–5294; U.S. Department of the Interior: Denver, CO, USA, 2006; p. 42. [Google Scholar]
- Zhang, X.-S.; Wang, H.-J.; Ma, F.; Sun, X.-C.; Zhang, Y.; Song, Z.-H. Classification and Characteristics of Tight Oil Plays. Pet. Sci. 2016, 13, 18–33. [Google Scholar] [CrossRef]
- Li, L.; Su, Y.; Sheng, J.J. Investigation of Gas Penetration Depth During Gas Huff-N-Puff EOR Process in Unconventional Oil Reservoirs. In Proceedings of the UR—Unconventional EOR/IOR & EGR, Calgary, AB, Canada, 13 March 2018; p. D012S002R001. [Google Scholar]
- Khormali, A. Effect of Water Cut on the Performance of an Asphaltene Inhibitor Package: Experimental and Modeling Analysis. Pet. Sci. Technol. 2022, 40, 2890–2906. [Google Scholar] [CrossRef]
- Bao, X.; Fragoso, A.; Aguilera, R. Geological Containment vs. Well Containment for Huff and Puff (H&P) Gas Injection in Shale Oil Reservoirs. In Proceedings of the 10th Unconventional Resources Technology Conference, Houston, TX, USA, 20 June 2022. [Google Scholar]
- Jin, L.; Wan, X.; Azzolina, N.A.; Bosshart, N.W.; Zhao, J.; Yu, Y.; Yu, X.; Smith, S.A.; Sorensen, J.A.; Gorecki, C.D. Optimizing Conformance Control for Gas Injection EOR in Unconventional Reservoirs. Fuel 2022, 324, 124523. [Google Scholar] [CrossRef]
- Elturki, M.; Imqam, A. Application of Enhanced Oil Recovery Methods in Unconventional Reservoirs: A Review and Data Analysis; American Rock Mechanics Association: Golden, CO, USA, 2020; p. ARMA-2020-1081. [Google Scholar]
- Balasubramanian, S.; Chen, P.; Bose, S.; Alzahabi, A.; Thakur, G.C. Recent Advances in Enhanced Oil Recovery Technologies for Unconventional Oil Reservoirs. In Proceedings of the HPHT Technology Verification, Validation, and Regulatory Requirements: Part One; OTC: Houston, TX, USA, 2018; p. D021S024R004. [Google Scholar]
- Mahzari, P.; Mitchell, T.M.; Jones, A.P.; Oelkers, E.H.; Striolo, A.; Iacoviello, F.; Shearing, P.R.; Ernesto Juri, J. Novel Laboratory Investigation of Huff-n-Puff Gas Injection for Shale Oils under Realistic Reservoir Conditions. Fuel 2021, 284, 118950. [Google Scholar] [CrossRef]
- Ganjdanesh, R.; Eltahan, E.; Sepehrnoori, K.; Drozd, H.; Ambrose, R. A Field Pilot of Huff-n-Puff Gas Injection for Enhanced Oil Recovery in Permian Basin. In Proceedings of the PFC Operations, Virtual, 19 October 2020; p. D021S019R002. [Google Scholar]
- Zuloaga-Molero, P.; Yu, W.; Xu, Y.; Sepehrnoori, K.; Li, B. Simulation Study of CO2-EOR in Tight Oil Reservoirs with Complex Fracture Geometries. Sci. Rep. 2016, 6, 33445. [Google Scholar] [CrossRef] [PubMed]
- Sie, C.-Y.; Nguyen, Q.P. Field Gas Huff-n-Puff for Enhancing Oil Recovery in Eagle Ford Shales—Effect of Reservoir Rock and Crude Properties. Fuel 2022, 328, 125127. [Google Scholar] [CrossRef]
- Skauge, A.; Solbakken, J.; Ormehaug, P.A.; Aarra, M.G. Foam Generation, Propagation and Stability in Porous Medium. Transp. Porous Med. 2020, 131, 5–21. [Google Scholar] [CrossRef]
- Sie, C.; Nguyen, Q.P. Field Gas Huff-n-Puff for Improving Oil Recovery from Eagle Ford Shale Reservoirs. In Proceedings of the Field Case 4 Polymer, Virtual, 30 August 2020; p. D021S023R003. [Google Scholar]
- Hoffman, B.T. Huff-N-Puff Gas Injection Pilot Projects in the Eagle Ford. In Proceedings of the UR—Unconventional EOR/IOR & EGR, Calgary, AB, Canada, 13 March 2018; p. D012S001R001. [Google Scholar]
- Hoffman, B.T.; Reichhardt, D. Quantitative Evaluation of Recovery Mechanisms for Huff-n-Puff Gas Injection in Unconventional Reservoirs. In Proceedings of the 7th Unconventional Resources Technology Conference, Denver, CO, USA, 22–24 July 2019. [Google Scholar]
- Li, L.; Su, Y.; Hao, Y.; Zhan, S.; Lv, Y.; Zhao, Q.; Wang, H. A Comparative Study of CO2 and N2 Huff-n-Puff EOR Performance in Shale Oil Production. J. Pet. Sci. Eng. 2019, 181, 106174. [Google Scholar] [CrossRef]
- Bozeman, T.; Nelle, W.; Nguyen, Q. Small Scale EOR Pilot in the Eastern Eagle Ford Boosts Production. In Proceedings of the Field Case Studies 1: Chemical, Virtual, 18 April 2022; p. D011S005R001. [Google Scholar]
- Sie, C.; Nguyen, Q.P. Laboratory Investigations on Field Gas Huff-n-Puff for Improving Oil Recovery in Eagle Ford Shale—Effect of Operating Conditions. Energy Fuels 2022, 36, 195–209. [Google Scholar] [CrossRef]
- Alfarge, D.; Alsaba, M.; Wei, M.; Bai, B. Miscible Gases Based EOR in Unconventional Liquids Rich Reservoirs: What We Can Learn. In Proceedings of the Modelling Heavy Oil Performance to Optimize the Future, Kuwait City, Kuwait, 10 December 2018; p. D022S034R002. [Google Scholar]
- Katiyar, A.; Patil, P.D.; Rohilla, N.; Crosley, M.; Miller, T.; Rozowski, P.; Evans, J.; Bozeman, T.; Nguyen, Q.P. Unconventional Reservoir Hydrocarbon-Gas Foam Enhanced-Oil-Recovery Pilot: Design, Implementation, and Performance Analysis. SPE Reserv. Eval. Eng. 2021, 24, 194–204. [Google Scholar] [CrossRef]
- Li, L.; Zhou, X.; Su, Y.; Xiao, P.; Chen, Z.; Zheng, J. Influence of Heterogeneity and Fracture Conductivity on Supercritical CO2 Miscible Flooding Enhancing Oil Recovery and Gas Channeling in Tight Oil Reservoirs. Energy Fuels 2022, 36, 8199–8209. [Google Scholar] [CrossRef]
- Fiallos Torres, M.X.; Yu, W.; Ganjdanesh, R.; Kerr, E.; Sepehrnoori, K.; Miao, J.; Ambrose, R. Modeling Interwell Fracture Interference and Huff-N-Puff Pressure Containment in Eagle Ford Using EDFM. In Proceedings of the Completions I: Fracturing Materials, Oklahoma City, OK, USA, 8 April 2019; p. D021S009R003. [Google Scholar]
- Wang, L.; Yu, W. Lean Gas Huff and Puff Process for Eagle Ford Shale with Connecting Natural Fractures: Well Interference, Methane Adsorption and Gas Trapping Effects. In Proceedings of the Learnings from Integrated Modeling Efforts, Odessa, TX, USA, 30 October 2019; p. D021S007R003. [Google Scholar]
- Wan, T.; Wang, W.; Jiang, J.; Zhang, Y. Pore-Scale Analysis of Gas Huff-n-Puff Enhanced Oil Recovery and Waterflooding Process. Fuel 2018, 215, 561–571. [Google Scholar] [CrossRef]
- Jia, H.; Sheng, J.J. Simulation Study of Huff-n-Puff Air Injection for Enhanced Oil Recovery in Shale Oil Reservoirs. Petroleum 2018, 4, 7–14. [Google Scholar] [CrossRef]
- Luo, H.; Delshad, M.; Pope, G.A.; Mohanty, K.K. Interactions Between Viscous Fingering and Channeling for Unstable Water/Polymer Floods in Heavy Oil Reservoirs. In Proceedings of the Scalable Linear and Nonlinear Solvers, Montgomery, TX, USA, 20 February 2017; p. D011S002R007. [Google Scholar]
- Sagbana, P.I.; Abushaikha, A.S. A Comprehensive Review of the Chemical-Based Conformance Control Methods in Oil Reservoirs. J. Pet. Explor. Prod. Technol. 2021, 11, 2233–2257. [Google Scholar] [CrossRef]
- Bernard, G.G.; Holm, L.W.; Harvey, C.P. Use of Surfactant to Reduce CO2 Mobility in Oil Displacement. Soc. Pet. Eng. J. 1980, 20, 281–292. [Google Scholar] [CrossRef]
- Lawson, J.B.; Reisberg, J. Alternate Slugs Of Gas And Dilute Surfactant For Mobility Control During Chemical Flooding. In Proceedings of the SPE/DOE Enhanced Oil Recovery Symposium, Tulsa, OK, USA, 20 April 1980; p. SPE-8839-MS. [Google Scholar]
- Li, R.F.; Yan, W.; Liu, S.; Hirasaki, G.J.; Miller, C.A. Foam Mobility Control for Surfactant Enhanced Oil Recovery. SPE J. 2010, 15, 928–942. [Google Scholar] [CrossRef]
- Hassanzadeh, A.; Katiyar, A.; Kalaei, H.; Pecore, D.; Schofield, E.; Nguyen, Q.P. A Novel Foam Process with CO2 Dissolved Surfactant for Improved Sweep Efficiency in EVGSAU Field. Geoenergy Sci. Eng. 2023, 231, 212310. [Google Scholar] [CrossRef]
- Seright, R.S. A Review of Gel Placement Concepts; Reservoir Sweep Improvement; New Mexico Institute of Mining and Technology: Socorro, NM, USA, 1996. [Google Scholar]
- Pope, D.S.; Leung, L.K.; Gulbis, J.; Constien, V.G. Effects of Viscous Fingering on Fracture Conductivity. SPE Prod. Facil. 1996, 11, 230–237. [Google Scholar] [CrossRef]
- Qajar, A.; Xue, Z.; Worthen, A.J.; Johnston, K.P.; Huh, C.; Bryant, S.L.; Prodanović, M. Modeling Fracture Propagation and Cleanup for Dry Nanoparticle-Stabilized-Foam Fracturing Fluids. J. Pet. Sci. Eng. 2016, 146, 210–221. [Google Scholar] [CrossRef]
- Xiong, X.; Sheng, J.J.; Wu, X.; Qin, J. Experimental Investigation of Foam-Assisted N2 Huff-n-Puff Enhanced Oil Recovery in Fractured Shale Cores. Fuel 2022, 311, 122597. [Google Scholar] [CrossRef]
- Wu, Z.; Chen, H.; Cai, X.; Gou, Q.; Jiang, L.; Chen, K.; Chen, Z.; Jiang, S. Current Status and Future Trends of In Situ Catalytic Upgrading of Extra Heavy Oil. Energies 2023, 16, 4610. [Google Scholar] [CrossRef]
- Wu, Z.; Liu, H.; Pang, Z.; Wu, Y.; Wang, X.; Liu, D.; Gao, M. A Visual Investigation of Enhanced Heavy Oil Recovery by Foam Flooding after Hot Water Injection. J. Pet. Sci. Eng. 2016, 147, 361–370. [Google Scholar] [CrossRef]
- Das, A.; Nguyen, N.; Nguyen, Q.P. Low Tension Gas Flooding for Secondary Oil Recovery in Low-Permeability, High-Salinity Reservoirs. Fuel 2020, 264, 116601. [Google Scholar] [CrossRef]
- Hao, H.; Hou, J.; Zhao, F.; Huang, H.; Liu, H. N2 -Foam-Assisted CO2 Huff-n-Puff Process for Enhanced Oil Recovery in a Heterogeneous Edge-Water Reservoir: Experiments and Pilot Tests. Rsc Adv. 2021, 11, 1134–1146. [Google Scholar] [CrossRef] [PubMed]
- AlQuaimi, B.I.; Rossen, W.R. Characterizing Foam Flow in Fractures for Enhanced Oil Recovery. In Proceedings of the 19th European Symposium on Improved Oil Recovery, Stavanger, Norway, 24 April 2017. [Google Scholar]
- Fernø, M.A.; Gauteplass, J.; Pancharoen, M.; Haugen, A.; Graue, A.; Kovscek, A.R.; Hirasaki, G. Experimental Study of Foam Generation, Sweep Efficiency, and Flow in a Fracture Network. SPE J. 2016, 21, 1140–1150. [Google Scholar] [CrossRef]
- Chen, L.; Huang, M.; Li, Z.; Liu, D.; Li, B. Experimental Study on the Characteristics of Foam Flow in Fractures. J. Pet. Sci. Eng. 2020, 185, 106663. [Google Scholar] [CrossRef]
- Osei-Bonsu, K.; Shokri, N.; Grassia, P. Fundamental Investigation of Foam Flow in a Liquid-Filled Hele-Shaw Cell. J. Colloid Interface Sci. 2016, 462, 288–296. [Google Scholar] [CrossRef]
- Steinsbø, M.; Brattekås, B.; Ersland, G.; Bø, K.; Opdal, I.E.; Tunli, R.; Graue, A.; Fernø, M.A. Foam as Mobility Control for Integrated CO2-EOR in Fractured Carbonates. In Proceedings of the 18th European Symposium on Improved Oil Recovery, Dresden, Germany, 14 April 2015. [Google Scholar]
- Haugen, A.; Fernø, M.A.; Graue, A.; Bertin, H.J. Experimental Study of Foam Flow in Fractured Oil-Wet Limestone for Enhanced Oil Recovery. SPE Reserv. Eval. Eng. 2012, 15, 218–228. [Google Scholar] [CrossRef]
- Fernø, M.A.; Eide, Ø.; Steinsbø, M.; Langlo, S.A.W.; Christophersen, A.; Skibenes, A.; Ydstebø, T.; Graue, A. Mobility Control during CO2 EOR in Fractured Carbonates Using Foam: Laboratory Evaluation and Numerical Simulations. J. Pet. Sci. Eng. 2015, 135, 442–451. [Google Scholar] [CrossRef]
- Haugen, Å.; Mani, N.; Svenningsen, S.; Brattekås, B.; Graue, A.; Ersland, G.; Fernø, M.A. Miscible and Immiscible Foam Injection for Mobility Control and EOR in Fractured Oil-Wet Carbonate Rocks. Transp. Porous Med. 2014, 104, 109–131. [Google Scholar] [CrossRef]
- Xu, Z.; Cao, A.; Chen, L.; Cui, S.; Yu, G.; Li, Z. Flow Characteristics of Foam in Fracture Networks. Ind. Eng. Chem. Res. 2020, 59, 19817–19828. [Google Scholar] [CrossRef]
- Qu, M.; Liang, T.; Hou, J. Study on Fluid Behaviors of Foam-Assisted Nitrogen Flooding on a Three-Dimensional Visualized Fracture–Vuggy Model. Appl. Sci. 2021, 11, 11082. [Google Scholar] [CrossRef]
- Fei, Y.; Johnson, R.L.; Gonzalez, M.; Haghighi, M.; Pokalai, K. Experimental and Numerical Investigation into Nano-Stabilized Foams in Low Permeability Reservoir Hydraulic Fracturing Applications. Fuel 2018, 213, 133–143. [Google Scholar] [CrossRef]
- Pancharoen, M.; Fernø, M.A.; Kovscek, A.R. Modeling Foam Displacement in Fractures. J. Pet. Sci. Eng. 2012, 100, 50–58. [Google Scholar] [CrossRef]
- Gauteplass, J.; Chaudhary, K.; Kovscek, A.R.; Fernø, M.A. Pore-Level Foam Generation and Flow for Mobility Control in Fractured Systems. Colloids Surf. A Physicochem. Eng. Asp. 2015, 468, 184–192. [Google Scholar] [CrossRef]
- Wang, F.; Du, D.; Bi, H.; Wang, H.; Chen, H.; Li, H. Quantitative Characterization of Foam Transient Structure in Porous Media and Analysis of Its Flow Behavior Based on Fractal Theory. Ind. Eng. Chem. Res. 2020, 59, 5158–5166. [Google Scholar] [CrossRef]
- Buchgraber, M.; Castanier, L.M.; Kovscek, A.R. Microvisual Investigation of Foam Flow in Ideal Fractures: Role of Fracture Aperture and Surface Roughness. In Proceedings of the SPE Annual Technical Conference and Exhibition, San Antonio, TX, USA, 8 October 2012; p. SPE-159430-MS. [Google Scholar]
- Champagne, L.M.; Zelenev, A.S.; Penny, G.S.; Travis, K. Critical Assessment of Microemulsion Technology for Enhancing Fluid Recovery from Tight Gas Formations and Propped Fractures. In Proceedings of the SPE European Formation Damage Conference, Noordwijk, The Netherlands, 7 June 2011; p. SPE-144095-MS. [Google Scholar]
- Sydansk, R.D. Polymer-Enhanced Foams Part 2: Propagation Through High-Permeability Sandpacks. SPE Adv. Technol. Ser. 1994, 2, 160–166. [Google Scholar] [CrossRef]
- Kamarul Bahrim, R.Z.; Zeng, Y.; Vincent Bonnieu, S.; Groenenboom, J.; Mohd Shafian, S.R.; Abdul Manap, A.A.; Tewari, R.D.; Biswal, S.L. A Study of Methane Foam in Reservoir Rocks for Mobility Control at High Temperature with Varied Permeabilities: Experiment and Simulation. In Proceedings of the Special Session 2: Rate Transient Analysis: A Rainbow of Real-Life Cases, Jakarta, Indonesia, 17 October 2017; p. D021S009R001. [Google Scholar]
- Sri Hanamertani, A.; Saraji, S.; Piri, M. The Effects of In-Situ Emulsion Formation and Superficial Velocity on Foam Performance in High-Permeability Porous Media. Fuel 2021, 306, 121575. [Google Scholar] [CrossRef]
- Thakore, V.; Ren, F.; Wang, H.; Wang, J.-A.J.; Polsky, Y. High Temperature, High Pressure Stability of Aqueous Foams for Potential Application in Enhanced Geothermal System (EGS). In Proceedings of the 47th Workshop on Geothermal Reservoir Engineering, Stanford, CA, USA, 1 February 2022; Volume SGP-TR-223, pp. 604–614. [Google Scholar]
- Wang, Y.; Zhang, Y.; Liu, Y.; Zhang, L.; Ren, S.; Lu, J.; Wang, X.; Fan, N. The Stability Study of CO2 Foams at High Pressure and High Temperature. J. Pet. Sci. Eng. 2017, 154, 234–243. [Google Scholar] [CrossRef]
- Nguyen, Q.P. Dynamics of Foam in Porous Media; Wageningen University: Saigon, Vietnam, 2004. [Google Scholar]
- Rashed Rohani, M.; Ghotbi, C.; Badakhshan, A. Foam Stability and Foam-Oil Interactions. Pet. Sci. Technol. 2014, 32, 1843–1850. [Google Scholar] [CrossRef]
- Osei-Bonsu, K.; Shokri, N.; Grassia, P. Foam Stability in the Presence and Absence of Hydrocarbons: From Bubble- to Bulk-Scale. Colloids Surf. A Physicochem. Eng. Asp. 2015, 481, 514–526. [Google Scholar] [CrossRef]
- Mannhardt, K.; Novosad, J.J.; Schramm, L.L. Comparative Evaluation of Foam Stability to Oil. SPE Reserv. Eval. Eng. 2000, 3, 23–34. [Google Scholar] [CrossRef]
- Le Van, S.; Youssif, M.I.; Hanamertani, A.S.; Sharma, K.V.; Elkhatib, O.; Rane, K.; Xie, Y.; Das, A.; Piri, M.; Katiyar, A.; et al. Methane Foam Performance Evaluation in Water-Wet Unconsolidated Porous Media: A Systematic Experimental Investigation at Elevated Pressure and Temperature Conditions. J. Nat. Gas. Sci. Eng. 2022, 104835. [Google Scholar] [CrossRef]
- Hirasaki, G.J.; Miller, C.A.; Raney, O.G.; Poindexter, M.K.; Nguyen, D.T.; Hera, J. Separation of Produced Emulsions from Surfactant Enhanced Oil Recovery Processes. Energy Fuels 2011, 25, 555–561. [Google Scholar] [CrossRef]
- Suniga, P.T.; Fortenberry, R.; Delshad, M. Observations of Microemulsion Viscosity for Surfactant EOR Processes. In Proceedings of the SPE Improved Oil Recovery Conference, Tulsa, OK, USA, 11 April 2016; p. SPE-179669-MS. [Google Scholar]
- Salleh, I.K.; Misra, S.; Ibrahim, J.M.B.M.; Panuganti, S.R. Micro-Emulsion-Based Dissolver for Removal of Mixed Scale Deposition. J. Pet. Explor. Prod. Technol. 2019, 9, 2635–2641. [Google Scholar] [CrossRef]
- Osterloh, W.T.; Jante, M.J. Effects of Gas and Liquid Velocity on Steady-State Foam Flow at High Temperature. In Proceedings of the SPE/DOE Enhanced Oil Recovery Symposium, Tulsa, OK, USA, 22–24 April 1992. [Google Scholar]
- Ma, K.; Lopez-Salinas, J.L.; Puerto, M.C.; Miller, C.A.; Biswal, S.L.; Hirasaki, G.J. Estimation of Parameters for the Simulation of Foam Flow through Porous Media. Part 1: The Dry-Out Effect. Energy Fuels 2013, 27, 2363–2375. [Google Scholar] [CrossRef]
- Alvarez, J.M.; Rivas, H.J.; Rossen, W.R. Unified Model for Steady-State Foam Behavior at High and Low Foam Qualities. SPE J. 2001, 6, 325–333. [Google Scholar] [CrossRef]
- Kahrobaei, S.; Vincent-Bonnieu, S.; Farajzadeh, R. Experimental Study of Hysteresis Behavior of Foam Generation in Porous Media. Sci. Rep. 2017, 7, 8986. [Google Scholar] [CrossRef]
- M’barki, O.; Ma, K.; Ren, G.; Mateen, K.; Bourdarot, G.; Morel, D.C.; Nguyen, Q.P. Repeatable Steady-State Foam Experimental Data and Investigations of Foam Hysteresis in a Sand Pack. In Proceedings of the Petroleum Education and Methods, San Antonio, TX, USA, 9 October 2017; p. D021S028R007. [Google Scholar]
- Khatib, Z.I.; Hirasaki, G.J.; Falls, A.H. Effects of Capillary Pressure on Coalescence and Phase Mobilities in Foams Flowing Through Porous Media. SPE Reserv. Eng. 1988, 3, 919–926. [Google Scholar] [CrossRef]
- Cong, Z.; Li, Y.; Pan, Y.; Liu, B.; Shi, Y.; Wei, J.; Li, W. Study on CO2 Foam Fracturing Model and Fracture Propagation Simulation. Energy 2022, 238, 121778. [Google Scholar] [CrossRef]
- Hirasaki, G.J.; Lawson, J.B. Mechanisms of Foam Flow in Porous Media: Apparent Viscosity in Smooth Capillaries. Soc. Pet. Eng. J. 1985, 25, 176–190. [Google Scholar] [CrossRef]
- Chang, S.-H.; Grigg, R.B. Effects of Foam Quality and Flow Rate on CO2-Foam Behavior at Reservoir Temperature and Pressure. SPE Reserv. Eval. Eng. 1999, 2, 248–254. [Google Scholar] [CrossRef]
- Lee, H.O.; Heller, J.P.; Hoefer, A.M.W. Change in Apparent Viscosity off CO2 Foam With Rock Permeability. SPE Reserv. Eng. 1991, 6, 421–428. [Google Scholar] [CrossRef]
- Salazar Castillo, R.O.; Ter Haar, S.F.; Ponners, C.G.; Bos, M.; Rossen, W. Fractional-Flow Theory for Non-Newtonian Surfactant-Alternating-Gas Foam Processes. Transp. Porous Med. 2020, 131, 399–426. [Google Scholar] [CrossRef]
- Holt, T.; Vassenden, F.; Svorstol, I. Effects of Pressure on Foam Stability; Implications for Foam Screening. In Proceedings of the SPE/DOE Improved Oil Recovery Symposium, Tulsa, OK, USA, 21–24 April 1996. [Google Scholar]
- Friedmann, F.; Jensen, J.A. Some Parameters Influencing the Formation and Propagation of Foams in Porous Media. In Proceedings of the SPE California Regional Meeting, Oakland, CA, USA, 2–4 April 1986. [Google Scholar]
- Farajzadeh, R.; Andrianov, A.; Bruining, H.; Zitha, P.L.J. Comparative Study of CO2 and N2 Foams in Porous Media at Low and High Pressure−Temperatures. Ind. Eng. Chem. Res. 2009, 48, 4542–4552. [Google Scholar] [CrossRef]
- Yang, C.; Gu, Y. Diffusion Coefficients and Oil Swelling Factors of Carbon Dioxide, Methane, Ethane, Propane, and Their Mixtures in Heavy Oil. Fluid Phase Equilibria 2006, 243, 64–73. [Google Scholar] [CrossRef]
- Rezk, M.G.; Foroozesh, J. Determination of Mass Transfer Parameters and Swelling Factor of CO2-Oil Systems at High Pressures. Int. J. Heat Mass Transf. 2018, 126, 380–390. [Google Scholar] [CrossRef]
- Heagle, D.J.; Ryan, D. Experimental Determination of the Interfacial Tension and Swelling Factors of Bakken and Duvernay Oils with CO2, Impure CO2, Methane, Ethane, and Propane. Energy Fuels 2022, 36, 806–817. [Google Scholar] [CrossRef]
- Wang, J.; Nguyen, A.V.; Farrokhpay, S. Effects of Surface Rheology and Surface Potential on Foam Stability. Colloids Surf. A Physicochem. Eng. Asp. 2016, 488, 70–81. [Google Scholar] [CrossRef]
- Wang, L.; Yoon, R.-H. Effects of Surface Forces and Film Elasticity on Foam Stability. Int. J. Miner. Process. 2008, 85, 101–110. [Google Scholar] [CrossRef]
- Cantat, I. Foams: Structure and Dynamics, 1st ed.; Oxford University Press: Oxford, UK; New York, NY, USA, 2013; ISBN 978-0-19-966289-0. [Google Scholar]
- Yu, Y.; García, B.F.; Saraji, S. Surfactant Viscoelasticity as a Key Parameter to Improve Supercritical CO2 Foam Stability/Foamability and Performance in Porous Media. J. Non-Newton. Fluid Mech. 2020, 282, 104311. [Google Scholar] [CrossRef]
- Stubenrauch, C.; Klitzing, R. von Disjoining Pressure in Thin Liquid Foam and Emulsion Films—New Concepts and Perspectives. J. Phys. Condens. Matter. 2003, 15, R1197–R1232. [Google Scholar] [CrossRef]
- Peng, M.; Duignan, T.T.; Nguyen, A.V. Significant Effect of Surfactant Adsorption Layer Thickness in Equilibrium Foam Films. J. Phys. Chem. B 2020, 124, 5301–5310. [Google Scholar] [CrossRef]
- Rosen, M.J.; Kunjappu, J.T. Surfactants and Interfacial Phenomena, 4th ed.; Wiley: Hoboken, NJ, USA, 2012; ISBN 978-1-118-22893-7. [Google Scholar]
- Dawson, D.R.; Offen, H.W.; Nicoli, D.F. Pressure Effects on Micellar Size. J. Colloid Interface Sci. 1981, 81, 396–401. [Google Scholar] [CrossRef]
- Espinosa Silva, Y.R.; Grigera, J.R. Micelle Stability in Water under a Range of Pressures and Temperatures; Do Both Have a Common Mechanism? RSC Adv. 2015, 5, 70005–70009. [Google Scholar] [CrossRef]
- Chu, K.-C.; Hu, S.-W.; Tsao, H.-K.; Sheng, Y.-J. Strong Competition between Adsorption and Aggregation of Surfactant in Nanoscale Systems. J. Colloid Interface Sci. 2019, 553, 674–681. [Google Scholar] [CrossRef] [PubMed]
- Gittins, P.; Iglauer, S.; Pentland, C.H.; Al-Mansoori, S.; Al-Sayari, S.; Bijeljic, B.; Blunt, M.J. Nonwetting Phase Residual Saturation in Sand Packs. J. Porous Media 2010, 13, 591–599. [Google Scholar] [CrossRef]
- Morrow, N.R.; Lim, H.T.; Ward, J.S. Effect of Crude-Oil-Induced Wettability Changes on Oil Recovery. SPE Form. Eval. 1986, 1, 89–103. [Google Scholar] [CrossRef]
- Brown, R.J.S.; Fatt, I. Measurements Of Fractional Wettability Of Oil Fields’ Rocks By The Nuclear Magnetic Relaxation Method. In Proceedings of the Fall Meeting of the Petroleum Branch of AIME, Los Angeles, CA, USA, 14 October 1956; p. SPE-743-G. [Google Scholar]
- Pentland, C.H.; Itsekiri, E.; Al Mansoori, S.K.; Iglauer, S.; Bijeljic, B.; Blunt, M.J. Measurement of Nonwetting-Phase Trapping in Sandpacks. SPE J. 2010, 15, 274–281. [Google Scholar] [CrossRef]
- Harpole, K.J.; Siemers, W.T.; Gerard, M.G. CO2 Foam Field Verification Pilot Test at EVGSAU: Phase IIIC--Reservoir Characterization and Response to Foam Injection. In Proceedings of the SPE/DOE Improved Oil Recovery Symposium, Tulsa, OK, USA, 17–20 April 1994. [Google Scholar]
- Ma, K.; Farajzadeh, R.; Lopez-Salinas, J.L.; Miller, C.A.; Biswal, S.L.; Hirasaki, G.J. Estimation of Parameters for the Simulation of Foam Flow through Porous Media: Part 3; Non-Uniqueness, Numerical Artifact and Sensitivity. In Proceedings of the SPE Enhanced Oil Recovery Conference, Kuala Lumpur, Malaysia, 2 July 2013; p. SPE-165263-MS. [Google Scholar]
- Abbaszadeh, M.; Nia Korrani, A.K.; Lopez-Salinas, J.L.; Rodriguez-de la Garza, F.; Villavicencio Pino, A.; Hirasaki, G. Experimentally-Based Empirical Foam Modeling. In Proceedings of the SPE Improved Oil Recovery Symposium, Tulsa, OK, USA, 12 April 2014; p. SPE-169888-MS. [Google Scholar]



| Salt | Concentration (ppm) |
|---|---|
| NaCl | 10,500 |
| CaCl2∙2H2O | 1000 |
| KCl | 100 |
| Temperature (K) | Pressure (psi) | Oil Volume (mL) | Surfactant Concentration (wt%) | Salinity (ppm) |
|---|---|---|---|---|
| 298 | 0, 2000, 3000, 4000 | 0, 0.4 | 0.5 | 30,000 |
| 388 | 3000 | 0, 0.4 | 0.5 | 30,000 |
| Flood Series | 1 (Baseline) | 2 (Residual Oil) | 3 (No Oil) | 4 (Nitrogen) | ||
|---|---|---|---|---|---|---|
| Temperature (K) | 388 | 388 | 388 | 388 | ||
| Pressure (psi) | 4000 | 4000 | 1000, 2000, 3000 | 4000 | 2000, 3000 | 2000, 3000, 4000 |
| Flow Rate (cm/s) | 0.0176, 0.176 | 0.0176, 0.0529, 0.106, 0.176 | 0.0176, 0.0529, 0.106 | 0.0176, 0.0529, 0.106 | 0.0529 | 0.0529 |
| Foam Quality (%) | 60, 80, 90, 95 | 60, 80, 90, 95 | 60, 80, 90, 95 | 60, 80, 90, 95 | ||
| Brine Salinity (ppm) | 30,000 | 30,000 | 30,000 | 30,000 | ||
| Surfactant Concentration (wt%) | 0 | 0.5 | 0.5 | 0.5 | ||
| Oil Saturation | Residual | Residual | None | None | ||
| Injection Gas | Methane | Methane | Methane | Nitrogen | ||
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Parekh, A.T.; Katiyar, A.; Nguyen, Q.P. Factors Influencing the Rheology of Methane Foam for Gas Mobility Control in High-Temperature, Proppant-Fractured Reservoirs. Colloids Interfaces 2024, 8, 13. https://doi.org/10.3390/colloids8010013
Parekh AT, Katiyar A, Nguyen QP. Factors Influencing the Rheology of Methane Foam for Gas Mobility Control in High-Temperature, Proppant-Fractured Reservoirs. Colloids and Interfaces. 2024; 8(1):13. https://doi.org/10.3390/colloids8010013
Chicago/Turabian StyleParekh, Aashish T., Amit Katiyar, and Quoc P. Nguyen. 2024. "Factors Influencing the Rheology of Methane Foam for Gas Mobility Control in High-Temperature, Proppant-Fractured Reservoirs" Colloids and Interfaces 8, no. 1: 13. https://doi.org/10.3390/colloids8010013
APA StyleParekh, A. T., Katiyar, A., & Nguyen, Q. P. (2024). Factors Influencing the Rheology of Methane Foam for Gas Mobility Control in High-Temperature, Proppant-Fractured Reservoirs. Colloids and Interfaces, 8(1), 13. https://doi.org/10.3390/colloids8010013







