Polymeric Nanocomposites of Iron–Oxide Nanoparticles (IONPs) Synthesized Using Terminalia chebula Leaf Extract for Enhanced Adsorption of Arsenic(V) from Water
Abstract
1. Introduction
2. Materials and Methods
2.1. Chemicals
2.2. Green Synthesis of Iron Nanoparticle
2.3. Stabilization of Plant-Synthesized IONPs
2.3.1. Preparation of IONP-Loaded Chitosan Nanocomposites (IONPs–C)
2.3.2. Preparation of IONP-Loaded PVA–Alginate Nanocomposites (IONPs–PA)
2.4. Material Characterization
2.5. Batch Adsorption Experiment for As(V) Removal
2.5.1. Adsorption Kinetics
2.5.2. Adsorption Isotherm
2.6. Reuse of Adsorbents and Iron Ion Release Study
3. Results and Discussion
3.1. Characterization of Iron–Oxide Nanoparticles (IONPs)
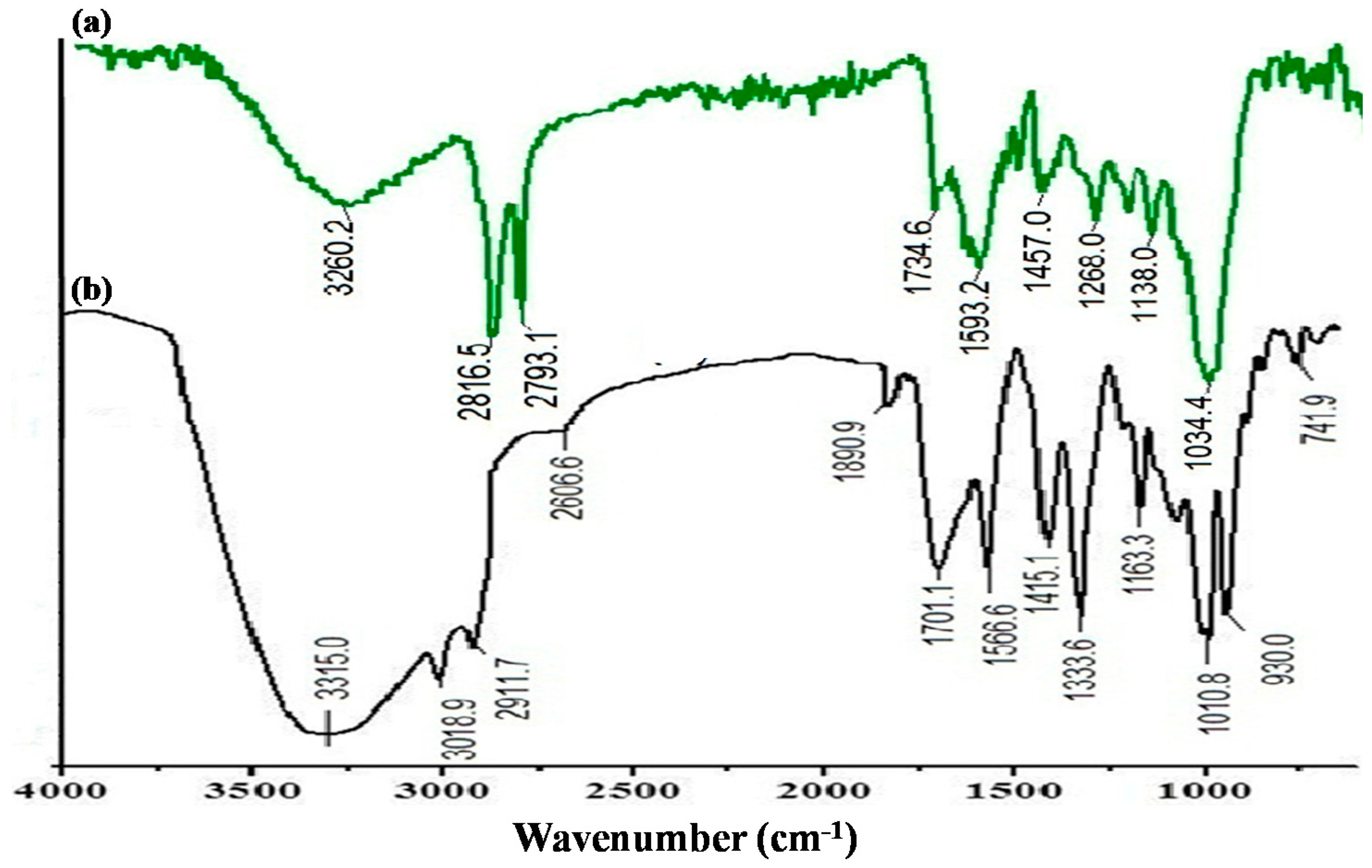
3.2. FESEM and FTIR Study of Polymer-Supported IONPs
3.3. Arsenic Adsorption Results
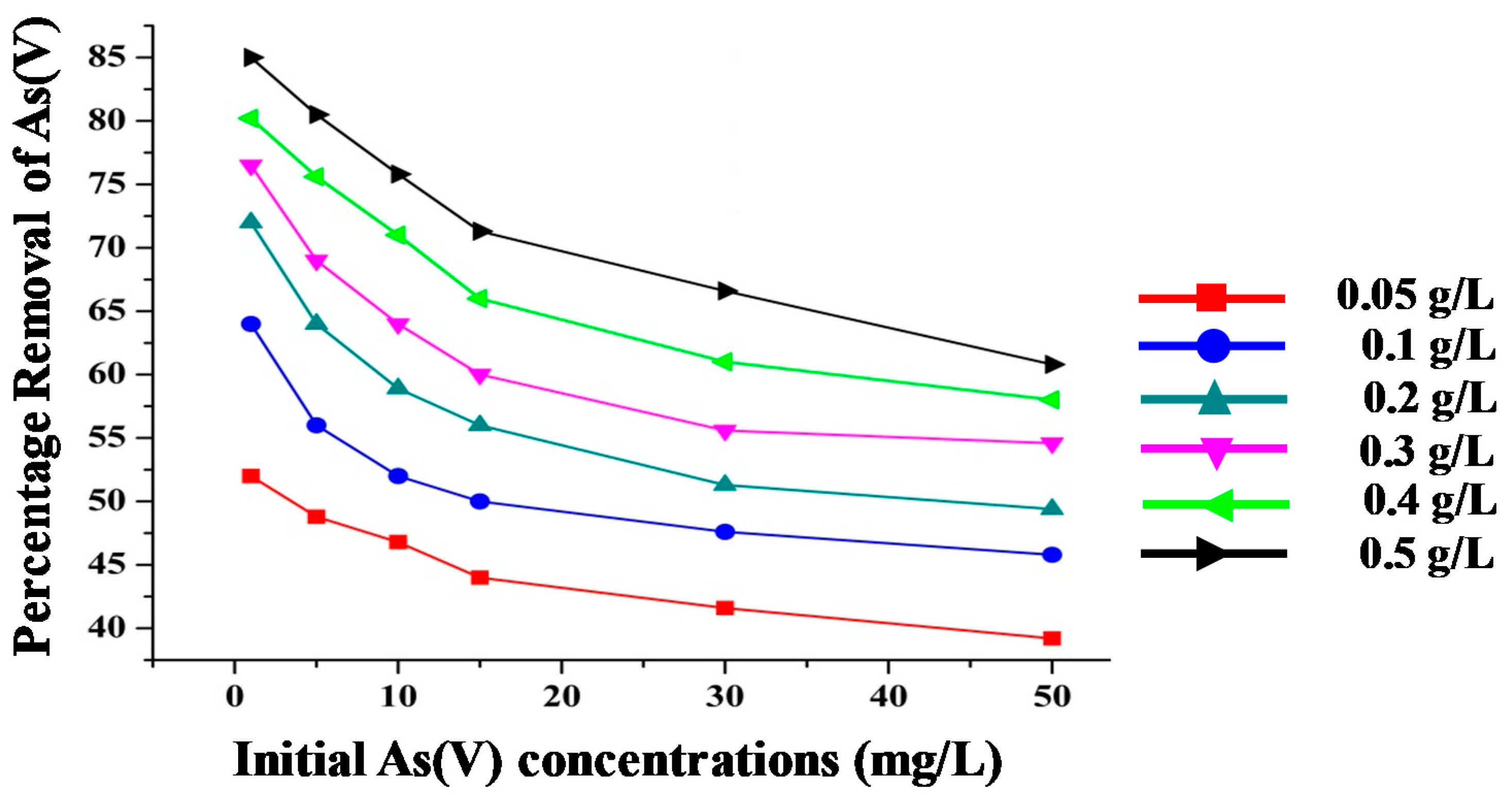
3.3.1. Effect of Nanoparticles Dosage and Initial Concentration on As(V) Removal
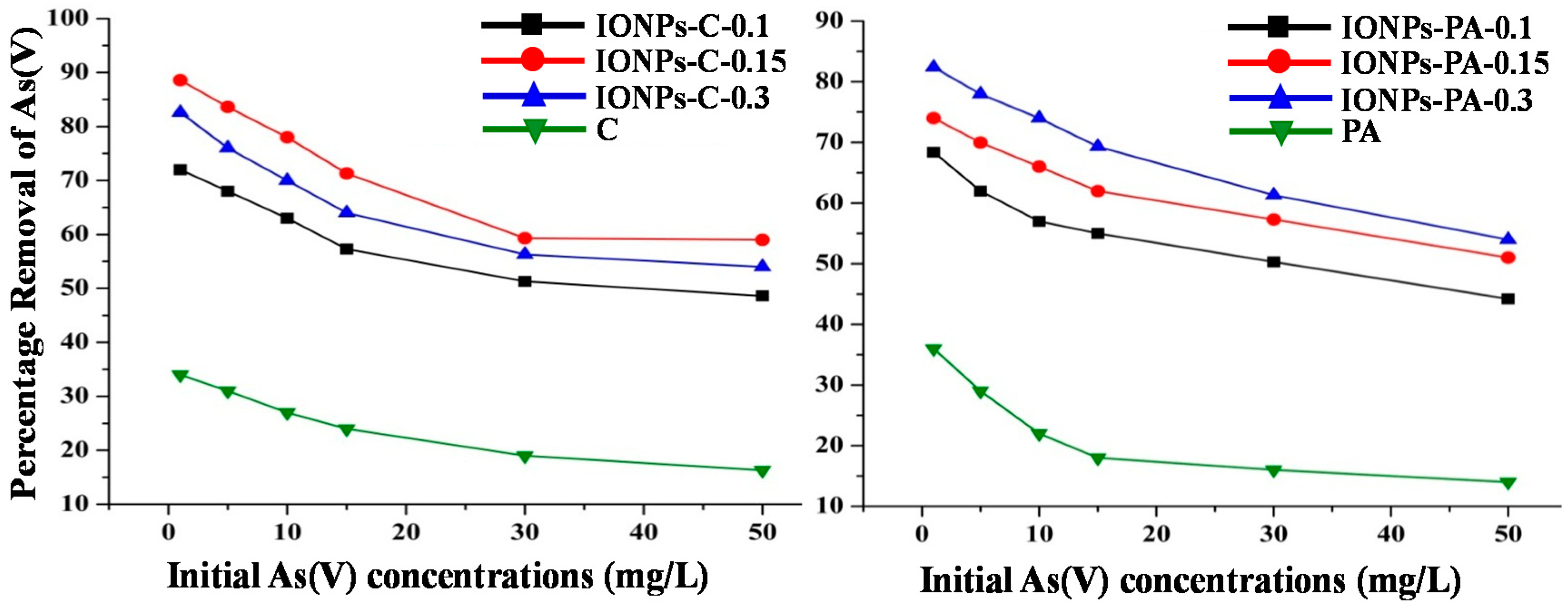
3.3.2. Effect of IONPs Loading on Chitosan (IONPs–C) and PVA–alginate (IONPs–PA) Nanocomposites for As(V) Removal
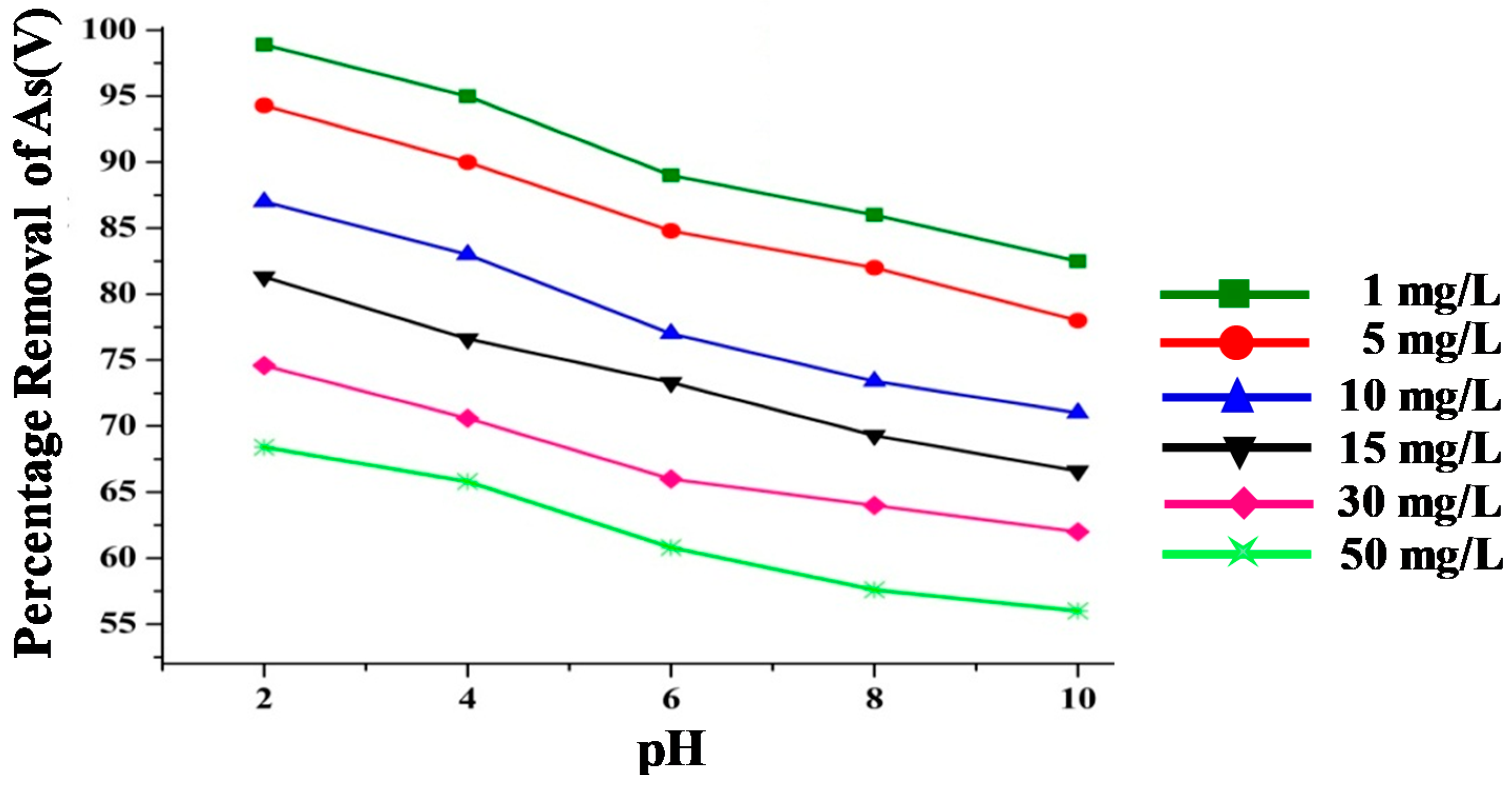
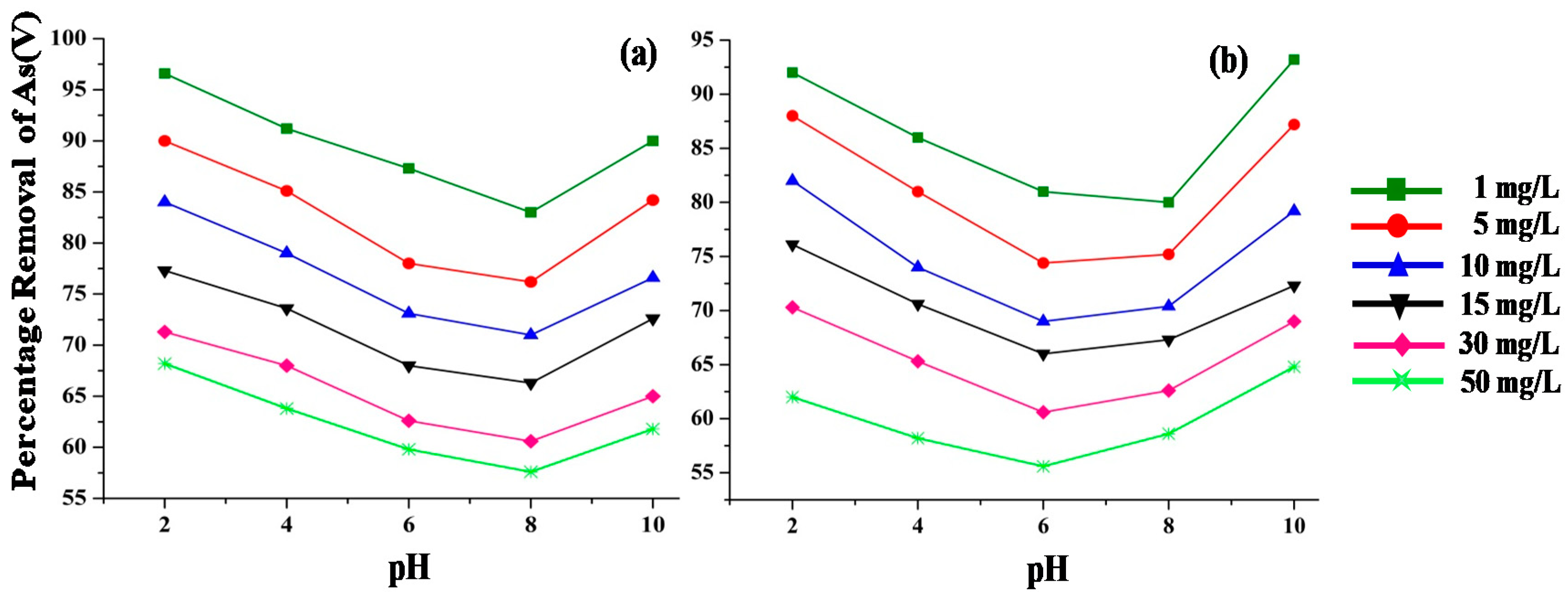
3.3.3. Effect of pH and Initial Concentration on As(V) Removal
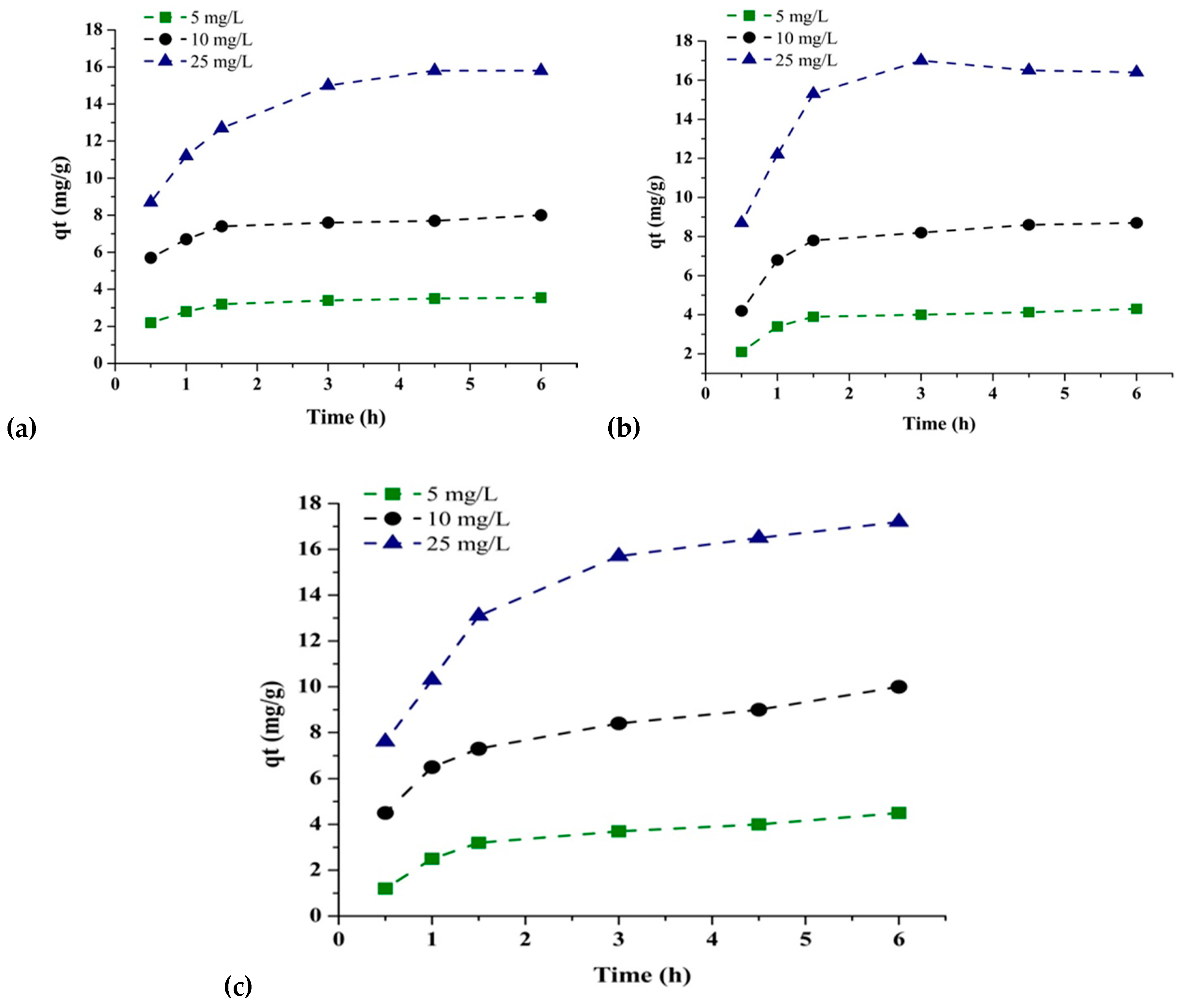
3.3.4. Adsorption Kinetics
3.3.5. Adsorption Isotherms
3.4. Reuse of Adsorbent and Iron Ion Release
4. Conclusions
- A comparative adsorption study showed that the adsorption capacity of arsenate on IONPs and polymer-supported IONPs varied in different parameters, such as pH, initial concentration of arsenic, dosages of adsorbent, and contact time, and can be improved by changing these parameters.
- Chitosan and PVA–alginate loaded with IONPs both were found to be more effective than the IONPs under different conditions of adsorption. The results also revealed that an increase of IONP loads to chitosan reduced the arsenic removal efficiency, but in the case of a PVA–alginate increase of IONPs, it increased the removal efficiency.
- In solutions with lower concentrations of arsenic, the efficiency of the IONPs and IONP–C nanocomposites was found to be up to 99% and 96.6% at a pH below 3, respectively. Adsorption at low pH as well as at high pH was observed by IONP–PA nanocomposites, and their efficiency can be enhanced by increasing the contact time.
- The adsorption kinetics was well fitted with a pseudo-second order reaction and the adsorption isotherm results were well-fitted with the Langmuir and Freundlich models.
- In the initial cycle, the sorption and desorption capacity of IONPs–C was remarkably high but decreased after a number of cycles. In the case of IONPs–PA, the reuse capacity was found to be more than that of the IONPs–C.
- Results showed that ion release was negligible for polymeric stabilized iron nanoparticles, i.e., IONPs–C and IONPs–PA, even after cycle 5 (adsorption–desorption) compared to IONPs.
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Khan, S.; Rauf, R.; Muhammad, S.; Qasim, M.; Din, I. Arsenic and heavy metals health risk assessment through drinking water consumption in the Peshawar District, Pakistan. Hum. Ecol. Risk Assess. 2016, 22, 581–596. [Google Scholar] [CrossRef]
- Ali, Z.; Malik, R.N.; Qadir, A. Heavy metals distribution and risk assessment in soils affected by tannery effluents. Chem. Ecol. 2013, 29, 676–692. [Google Scholar] [CrossRef]
- NRC. Arsenic in Drinking Water; National Academies Press: Washington, DC, USA, 2001. [Google Scholar]
- Mostafa, M.; Cherry, N. Arsenic in Drinking Water, Transition Cell Cancer and Chronic Cystitis in Rural Bangladesh. Int. J. Env. Res. Public Health 2015, 12, 13739–13749. [Google Scholar] [CrossRef] [PubMed]
- Oremland, R.S.; Stolz, J.F. The ecology of arsenic. Science 2003, 300, 939–944. [Google Scholar] [CrossRef] [PubMed]
- Lee, Y.; Um, I.-h.; Yoon, J. Arsenic (III) oxidation by iron (VI)(ferrate) and subsequent removal of arsenic (V) by iron (III) coagulation. Environ. Sci. Technol. 2003, 37, 5750–5756. [Google Scholar] [CrossRef] [PubMed]
- Hansen, H.K.; Ottosen, L.M. Removal of Arsenic from Wastewaters by Airlift Electrocoagulation: Part 3: Copper Smelter Wastewater Treatment. Sep. Sci. Technol. 2010, 45, 1326–1330. [Google Scholar] [CrossRef]
- Genç-Fuhrman, H.; Tjell, J.C.; McConchie, D. Adsorption of Arsenic from Water Using Activated Neutralized Red Mud. Environ. Sci. Technol. 2004, 38, 2428–2434. [Google Scholar] [CrossRef]
- Zhu, H.; Jia, Y.; Wu, X.; Wang, H. Removal of arsenic from water by supported nano zero-valent iron on activated carbon. J. Hazard. Mater. 2009, 172, 1591–1596. [Google Scholar] [CrossRef]
- Reisner, D.E.; Pradeep, T. Aquananotechnology: Global Prospects; CRC Press: Boca Raton, FL, USA, 2014. [Google Scholar]
- Cundy, A.B.; Hopkinson, L.; Whitby, R.L. Use of iron-based technologies in contaminated land and groundwater remediation: A review. Sci. Total Environ. 2008, 400, 42–51. [Google Scholar] [CrossRef]
- Vilardi, G.; Mpouras, T.; Dermatas, D.; Verdone, N.; Polydera, A.; Di Palma, L. Nanomaterials application for heavy metals recovery from polluted water: The combination of nano zero-valent iron and carbon nanotubes. Competitive adsorption non-linear modeling. Chemosphere 2018, 201, 716–729. [Google Scholar] [CrossRef]
- Xie, Y.; Fang, Z.; Qiu, X.; Tsang, E.P.; Liang, B. Comparisons of the reactivity, reusability and stability of four different zero-valent iron-based nanoparticles. Chemosphere 2014, 108, 433–436. [Google Scholar] [CrossRef] [PubMed]
- Xiao, Z.; Yuan, M.; Yang, B.; Liu, Z.; Huang, J.; Sun, D. Plant-mediated synthesis of highly active iron nanoparticles for Cr(VI) removal: Investigation of the leading biomolecules. Chemosphere 2016, 150, 357–364. [Google Scholar] [CrossRef] [PubMed]
- Biterna, M.; Arditsoglou, A.; Tsikouras, E.; Voutsa, D. Arsenate removal by zero valent iron: Batch and column tests. J. Hazard. Mater. 2007, 149, 548–552. [Google Scholar] [CrossRef] [PubMed]
- Tuutijärvi, T.; Lu, J.; Sillanpää, M.; Chen, G. As (V) adsorption on maghemite nanoparticles. J. Hazard. Mater. 2009, 166, 1415–1420. [Google Scholar] [CrossRef] [PubMed]
- Lin, S.; Lu, D.; Liu, Z. Removal of arsenic contaminants with magnetic γ-Fe2O3 nanoparticles. Chem. Eng. J. 2012, 211, 46–52. [Google Scholar] [CrossRef]
- Wang, F.; Yang, W.; Zheng, F.; Sun, Y. Removal of Cr (VI) from simulated and leachate wastewaters by Bentonite-supported zero-valent iron nanoparticles. Int. J. Env. Res. Public Health 2018, 15, 2162. [Google Scholar] [CrossRef] [PubMed]
- Yavuz, C.T.; Mayo, J.; William, W.Y.; Prakash, A.; Falkner, J.C.; Yean, S.; Cong, L.; Shipley, H.J.; Kan, A.; Tomson, M. Low-field magnetic separation of monodisperse Fe3O4 nanocrystals. Science 2006, 314, 964–967. [Google Scholar] [CrossRef]
- Badruddoza, A.Z.M.; Shawon, Z.B.Z.; Rahman, M.T.; Hao, K.W.; Hidajat, K.; Uddin, M.S. Ionically modified magnetic nanomaterials for arsenic and chromium removal from water. Chem. Eng. J. 2013, 225, 607–615. [Google Scholar] [CrossRef]
- Gallios, G.; Tolkou, A.; Katsoyiannis, I.; Stefusova, K.; Vaclavikova, M.; Deliyanni, E. Adsorption of arsenate by nano scaled activated carbon modified by iron and manganese oxides. Sustainability 2017, 9, 1684. [Google Scholar] [CrossRef]
- Mayo, J.; Yavuz, C.; Yean, S.; Cong, L.; Shipley, H.; Yu, W.; Falkner, J.; Kan, A.; Tomson, M.; Colvin, V. The effect of nanocrystalline magnetite size on arsenic removal. Sci. Technol. Adv. Mater. 2007, 8, 71–75. [Google Scholar] [CrossRef]
- Yean, S.; Cong, L.; Yavuz, C.T.; Mayo, J.; Yu, W.; Kan, A.; Colvin, V.; Tomson, M. Effect of magnetite particle size on adsorption and desorption of arsenite and arsenate. J. Mater. Res. 2005, 20, 3255–3264. [Google Scholar] [CrossRef]
- Wang, J.; Xu, W.; Chen, L.; Huang, X.; Liu, J. Preparation and evaluation of magnetic nanoparticles impregnated chitosan beads for arsenic removal from water. Chem. Eng. J. 2014, 251, 25–34. [Google Scholar] [CrossRef]
- Krishnamurthy, S.; Frederick, R.M. Using biopolymers to remove heavy metals from soil and water. Remediation 1994, 4, 235–244. [Google Scholar] [CrossRef]
- Gotoh, T.; Matsushima, K.; Kikuchi, K.-I. Preparation of alginate–chitosan hybrid gel beads and adsorption of divalent metal ions. Chemosphere 2004, 55, 135–140. [Google Scholar] [CrossRef] [PubMed]
- Katsoyiannis, I.A.; Zouboulis, A.I. Removal of arsenic from contaminated water sources by sorption onto iron-oxide-coated polymeric materials. Water Res. 2002, 36, 5141–5155. [Google Scholar] [CrossRef]
- Elwakeel, K.Z.; Guibal, E. Arsenic (V) sorption using chitosan/Cu (OH)2 and chitosan/CuO composite sorbents. Carbohydr. Polym. 2015, 134, 190–204. [Google Scholar] [CrossRef] [PubMed]
- Basu, H.; Singhal, R.; Pimple, M.; Reddy, A. Arsenic removal from groundwater by goethite impregnated calcium alginate beads. Water Air Soil Pollut. 2015, 226, 22. [Google Scholar] [CrossRef]
- Singh, P.; Singh, S.K.; Bajpai, J.; Bajpai, A.K.; Shrivastava, R.B. Iron crosslinked alginate as novel nanosorbents for removal of arsenic ions and bacteriological contamination from water. J. Mater Res. Technol. 2014, 3, 195–202. [Google Scholar] [CrossRef]
- Rahimi, Z.; Zinatizadeh, A.; Zinadini, S. Membrane bioreactors troubleshooting through preparation of a high antifouling PVDF ultrafiltration mixed matrix membrane blended with O-carboxymethyl chitosan-Fe3O4 nanoparticles. Environ. Technol. 2018, 1–30. [Google Scholar] [CrossRef]
- Woo, K.; Hong, J.; Choi, S.; Lee, H.-W.; Ahn, J.-P.; Kim, C.S.; Lee, S.W. Easy synthesis and magnetic properties of iron oxide nanoparticles. Chem. Mater. 2004, 16, 2814–2818. [Google Scholar] [CrossRef]
- Chin, A.B.; Yaacob, I.I. Synthesis and characterization of magnetic iron oxide nanoparticles via w/o microemulsion and Massart’s procedure. J. Mater. Process Technol. 2007, 191, 235–237. [Google Scholar] [CrossRef]
- Shahwan, T.; Abu Sirriah, S.; Nairat, M.; Boyacı, E.; Eroğlu, A.E.; Scott, T.B.; Hallam, K.R. Green synthesis of iron nanoparticles and their application as a Fenton-like catalyst for the degradation of aqueous cationic and anionic dyes. Chem. Eng. J. 2011, 172, 258–266. [Google Scholar] [CrossRef]
- Wang, Z. Iron Complex Nanoparticles Synthesized by Eucalyptus Leaves. ACS Sustain Chem. 2013, 1, 1551–1554. [Google Scholar] [CrossRef]
- Prasad, K.S.; Gandhi, P.; Selvaraj, K. Synthesis of green nano iron particles (GnIP) and their application in adsorptive removal of As(III) and As(V) from aqueous solution. Appl. Surf. Sci. 2014, 317, 1052–1059. [Google Scholar] [CrossRef]
- Pichsinee, S.; Karaked, T. Green synthesis of high dispersion and narrow size distribution of zero-valent iron nanoparticles using guava leaf (Psidium guajava L) extract. Adv. Nat. Sci. Nanosci. Nanotech. 2018, 9, 035006. [Google Scholar]
- Luo, F.; Chen, Z.; Megharaj, M.; Naidu, R. Biomolecules in grape leaf extract involved in one-step synthesis of iron-based nanoparticles. RSC Adv. 2014, 4, 53467–53474. [Google Scholar] [CrossRef]
- Smuleac, V.; Varma, R.; Sikdar, S.; Bhattacharyya, D. Green Synthesis of Fe and Fe/Pd Bimetallic Nanoparticles in Membranes for Reductive Degradation of Chlorinated Organics. J. Memb. Sci. 2011, 379, 131–137. [Google Scholar] [CrossRef]
- Madhavi, V.; Prasad, T.N.; Reddy, A.V.; Ravindra Reddy, B.; Madhavi, G. Application of phytogenic zerovalent iron nanoparticles in the adsorption of hexavalent chromium. Spectrochim. Acta. A. Mol. Biomol. Spectrosc. 2013, 116, 17–25. [Google Scholar] [CrossRef]
- Tandon, P.K.; Shukla, R.C.; Singh, S.B. Removal of arsenic(III) from water with clay-supported zerovalent iron nanoparticles synthesized with the help of tea liquor. Ind. Eng. Chem. Res. 2013, 52, 10052–10058. [Google Scholar] [CrossRef]
- Kiruba Daniel, S.C.G.; Vinothini, G.; Subramanian, N.; Nehru, K.; Sivakumar, M. Biosynthesis of Cu, ZVI, and Ag nanoparticles using Dodonaea viscosa extract for antibacterial activity against human pathogens. J. Nanopart. Res. 2012, 15. [Google Scholar] [CrossRef]
- Arokiyaraj, S.; Saravanan, M.; Udaya Prakash, N.K.; Valan Arasu, M.; Vijayakumar, B.; Vincent, S. Enhanced antibacterial activity of iron oxide magnetic nanoparticles treated with Argemone mexicana L. leaf extract: An in vitro study. Mater. Res. Bull. 2013, 48, 3323–3327. [Google Scholar] [CrossRef]
- Kuang, Y.; Wang, Q.; Chen, Z.; Megharaj, M.; Naidu, R. Heterogeneous Fenton-like oxidation of monochlorobenzene using green synthesis of iron nanoparticles. J. Colloid Interface Sci. 2013, 410, 67–73. [Google Scholar] [CrossRef] [PubMed]
- Wang, Z.; Fang, C.; Megharaj, M. Characterization of iron–polyphenol nanoparticles synthesized by three plant extracts and their Fenton oxidation of azo dye. ACS Sustain Chem. 2014, 2, 1022–1025. [Google Scholar] [CrossRef]
- Wang; Lin, J.; Chen, Z.; Megharaj, M.; Naidu, R. Green synthesized iron nanoparticles by green tea and eucalyptus leaves extracts used for removal of nitrate in aqueous solution. J. Clean. Product. 2014, 83, 413–419. [Google Scholar] [CrossRef]
- Han, Q.; Song, J.; Qiao, C.; Wong, L.; Xu, H. Preparative isolation of hydrolysable tannins chebulagic acid and chebulinic acid from Terminalia chebula by high-speed counter-current chromatography. J. Sep. Sci. 2006, 29, 1653–1657. [Google Scholar] [CrossRef] [PubMed]
- Espenti, C.S.; Rao, K.K.; Rao, K.M. Bio-synthesis and characterization of silver nanoparticles using Terminalia chebula leaf extract and evaluation of its antimicrobial potential. Mater. Lett. 2016, 174, 129–133. [Google Scholar] [CrossRef]
- Jiang, Y.-J.; Yu, X.-Y.; Luo, T.; Jia, Y.; Liu, J.-H.; Huang, X.-J. γ-Fe2O3 nanoparticles encapsulated millimeter-sized magnetic chitosan beads for removal of Cr (VI) from water: Thermodynamics, kinetics, regeneration, and uptake mechanisms. J. Chem. Eng. Data 2013, 58, 3142–3149. [Google Scholar] [CrossRef]
- Majidnia, Z.; Idris, A. Evaluation of cesium removal from radioactive waste water using maghemite PVA–alginate beads. Chem. Eng. J. 2015, 262, 372–382. [Google Scholar] [CrossRef]
- Rosli, I.; Zulhaimi, H.; Ibrahim, S.; Gopinath, S.; Kasim, K.; Akmal, H.; Nuradibah, M.; Sam, T. Phytosynthesis of iron nanoparticle from Averrhoa Bilimbi Linn. In Proceedings of the IOP Conference Series: Materials Science and Engineering, Penang, Malaysia, 6–7 December 2017; p. 012012. [Google Scholar]
- Mahdavi, M.; Namvar, F.; Ahmad, M.B.; Mohamad, R. Green biosynthesis and characterization of magnetic iron oxide (Fe3O4) nanoparticles using seaweed (Sargassum muticum) aqueous extract. Molecules 2013, 18, 5954–5964. [Google Scholar] [CrossRef]
- Mohan Kumar, K.; Mandal, B.K.; Siva Kumar, K.; Sreedhara Reddy, P.; Sreedhar, B. Biobased green method to synthesise palladium and iron nanoparticles using Terminalia chebula aqueous extract. Spectrochim. Acta. A. Mol. Biomol. Spectrosc. 2013, 102, 128–133. [Google Scholar] [CrossRef]
- Wang; Jin, X.; Chen, Z.; Megharaj, M.; Naidu, R. Green synthesis of Fe nanoparticles using eucalyptus leaf extracts for treatment of eutrophic wastewater. Sci. Total Environ. 2014, 466, 210–213. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.-T.; Yuan, Q.-B.; Duan, D.-H.; Zhang, Z.-L.; Hao, X.-G.; Wei, G.-Q.; Liu, S.-B. Electrochemical activity and stability of core–shell Fe2O3/Pt nanoparticles for methanol oxidation. J. Power Sources 2013, 243, 622–629. [Google Scholar] [CrossRef]
- Stollenwerk, K.G. Geochemical Processes Controlling Transport of Arsenic in Groundwater: A Review of Adsorption. In Arsenic in Ground Water; Welch, A.H., Stollenwerk, K.G., Eds.; Springer: New York, NY, USA, 2003; p. 475. [Google Scholar]
- Kanel, S.R.; Greneche, J.-M.; Choi, H. Arsenic (V) removal from groundwater using nano scale zero-valent iron as a colloidal reactive barrier material. Environ. Sci. Technol. 2006, 40, 2045–2050. [Google Scholar] [CrossRef] [PubMed]
- Karnitz, O., Jr.; Gurgel, L.V.A.; De Melo, J.C.P.; Botaro, V.R.; Melo, T.M.S.; de Freitas Gil, R.P.; Gil, L.F. Adsorption of heavy metal ion from aqueous single metal solution by chemically modified sugarcane bagasse. Bioresource Technol. 2007, 98, 1291–1297. [Google Scholar] [CrossRef] [PubMed]
- Gupta, A.; Yunus, M.; Sankararamakrishnan, N. Zerovalent iron encapsulated chitosan nanospheres-a novel adsorbent for the removal of total inorganic arsenic from aqueous systems. Chemosphere 2012, 86, 150–155. [Google Scholar] [CrossRef] [PubMed]
- Feng, L.; Cao, M.; Ma, X.; Zhu, Y.; Hu, C. Superparamagnetic high-surface-area Fe3O4 nanoparticles as adsorbents for arsenic removal. J. Hazard. Mater. 2012, 217, 439–446. [Google Scholar] [CrossRef] [PubMed]
- Mosaferi, M.; Nemati, S.; Khataee, A.; Nasseri, S.; Hashemi, A.A. Removal of Arsenic (III, V) from aqueous solution by nanoscale zero-valent iron stabilized with starch and carboxymethyl cellulose. J. Environ. Health. Sci. Eng. 2014, 12, 74. [Google Scholar] [CrossRef]
- Wang, C.; Luo, H.; Zhang, Z.; Wu, Y.; Zhang, J.; Chen, S. Removal of As(III) and As(V) from aqueous solutions using nanoscale zero valent iron-reduced graphite oxide modified composites. J. Hazard. Mater. 2014, 268, 124–131. [Google Scholar] [CrossRef]
- Bhowmick, S.; Chakraborty, S.; Mondal, P.; Van Renterghem, W.; Van den Berghe, S.; Roman-Ross, G.; Chatterjee, D.; Iglesias, M. Montmorillonite-supported nanoscale zero-valent iron for removal of arsenic from aqueous solution: Kinetics and mechanism. Chem. Eng. J. 2014, 243, 14–23. [Google Scholar] [CrossRef]
- Martínez-Cabanas, M.; López-García, M.; Barriada, J.L.; Herrero, R.; Sastre de Vicente, M.E. Green synthesis of iron oxide nanoparticles. Development of magnetic hybrid materials for efficient As(V) removal. Chem. Eng. J. 2016, 301, 83–91. [Google Scholar] [CrossRef]
- Chuan, L.T.; Manap, N.; Abdullah, H.Z.; Idris, M. Polyvinyl Alcohol-Alginate Adsorbent Beads for Chromium (VI) Removal. Int. Eng.Technol. 2018, 7, 95–99. [Google Scholar]
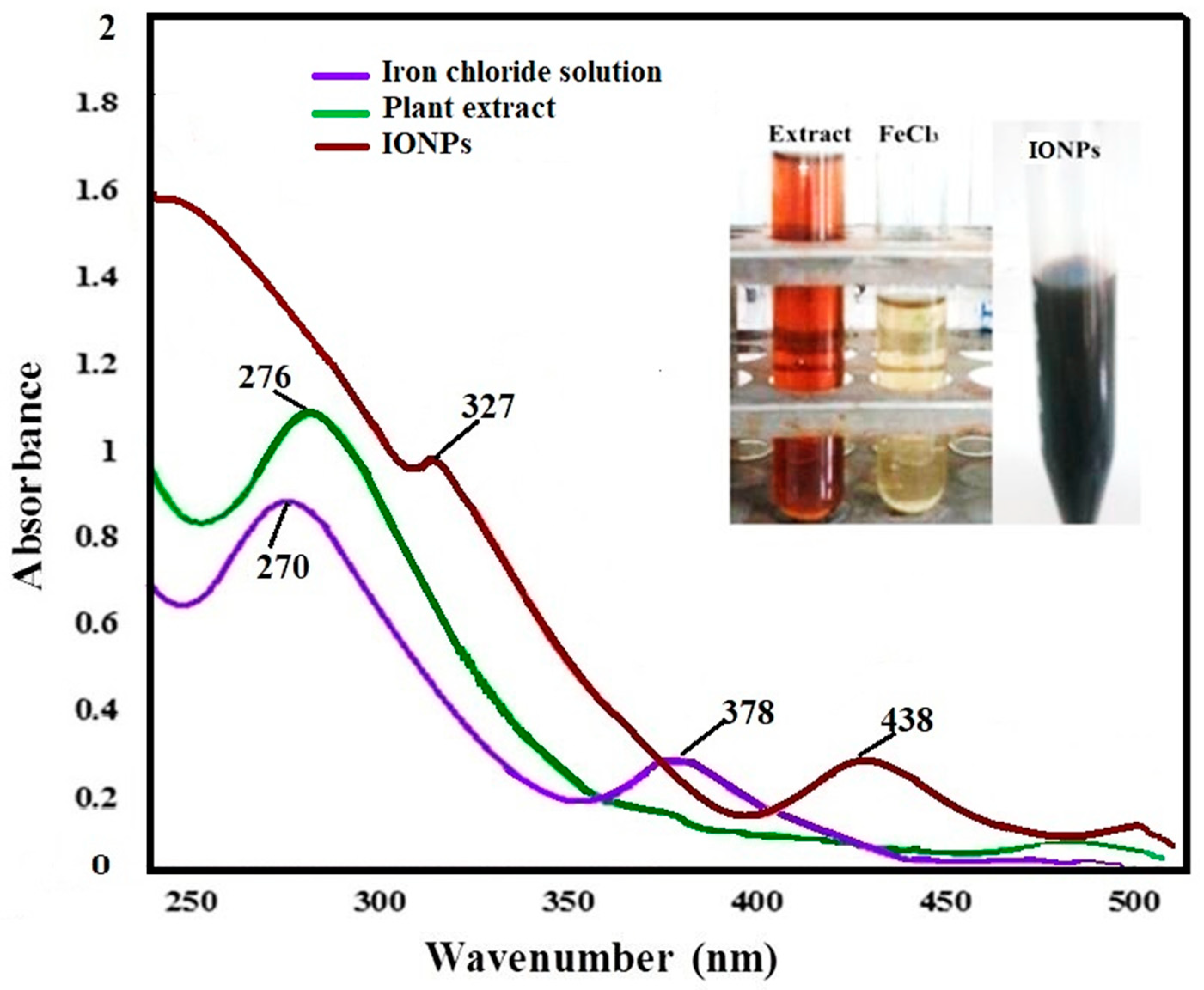
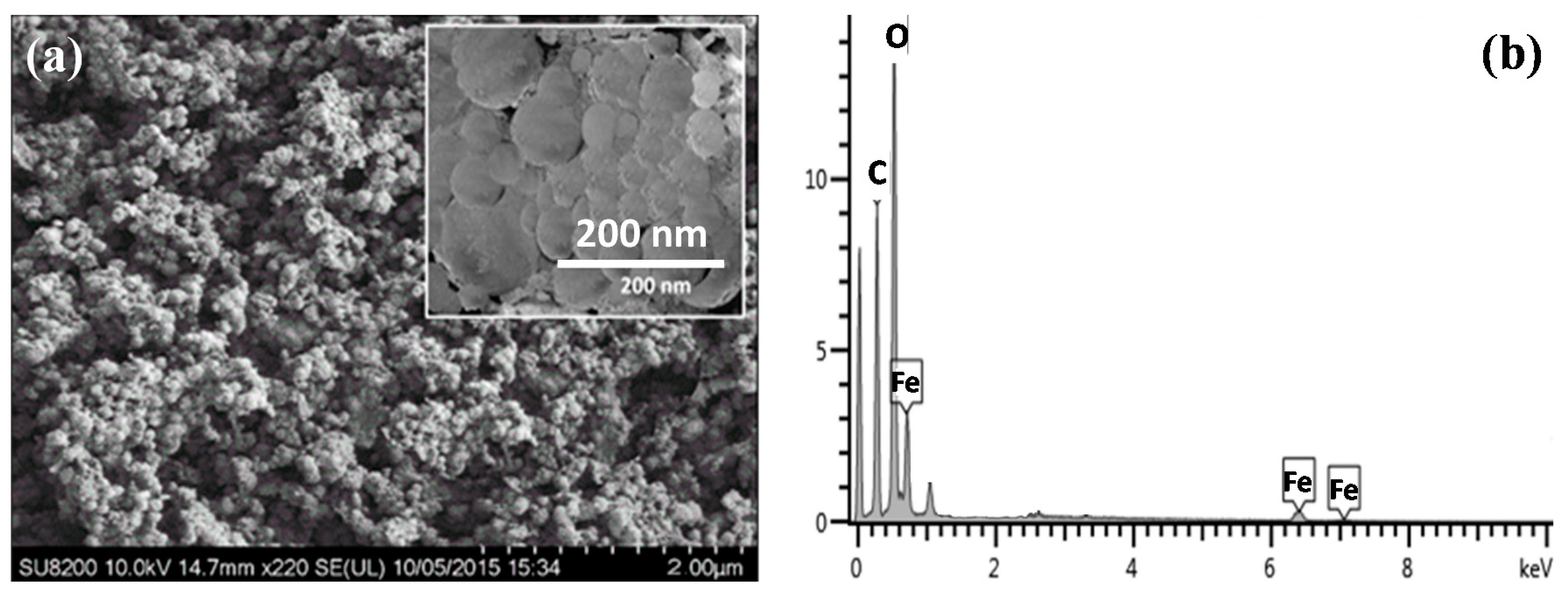
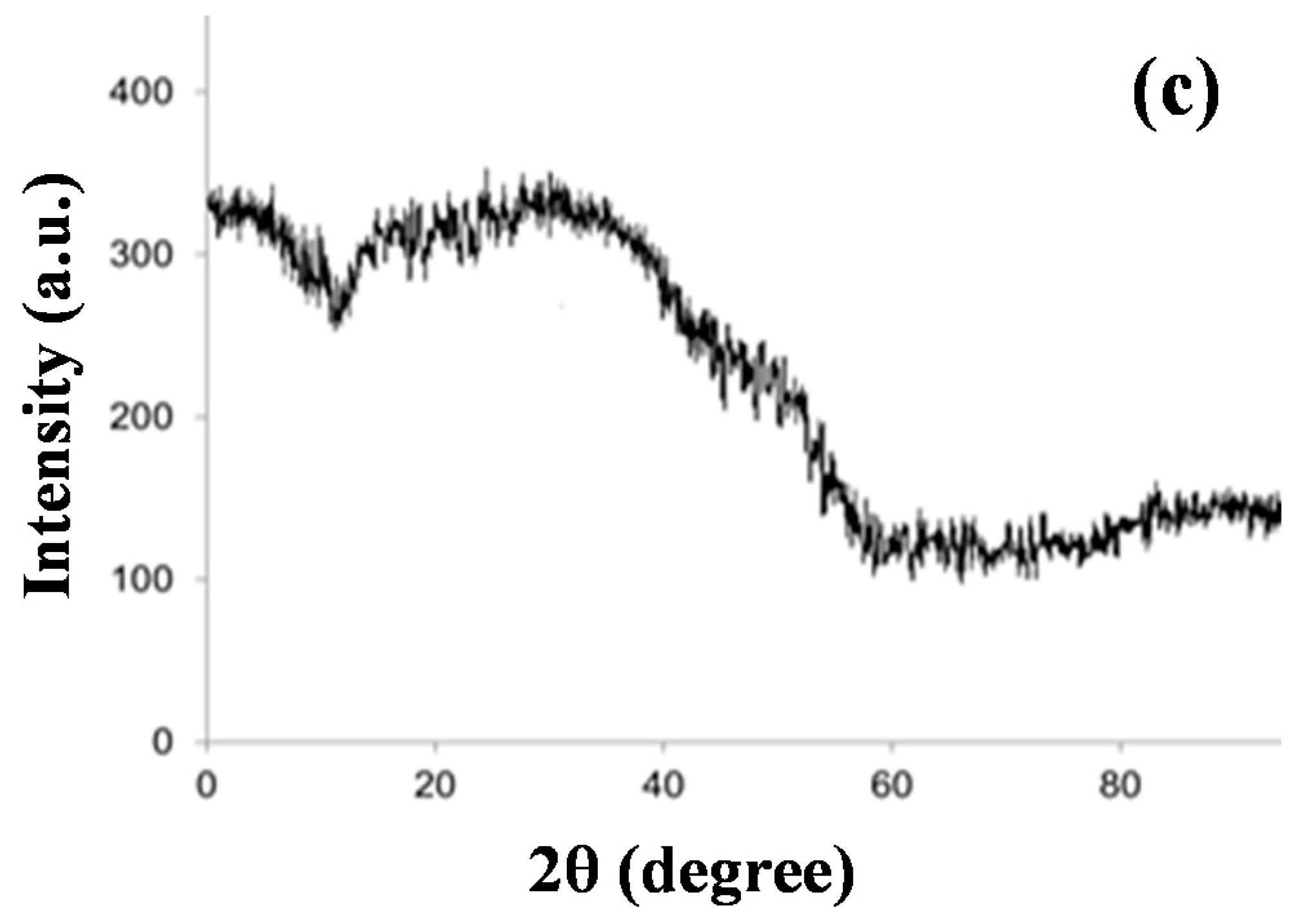
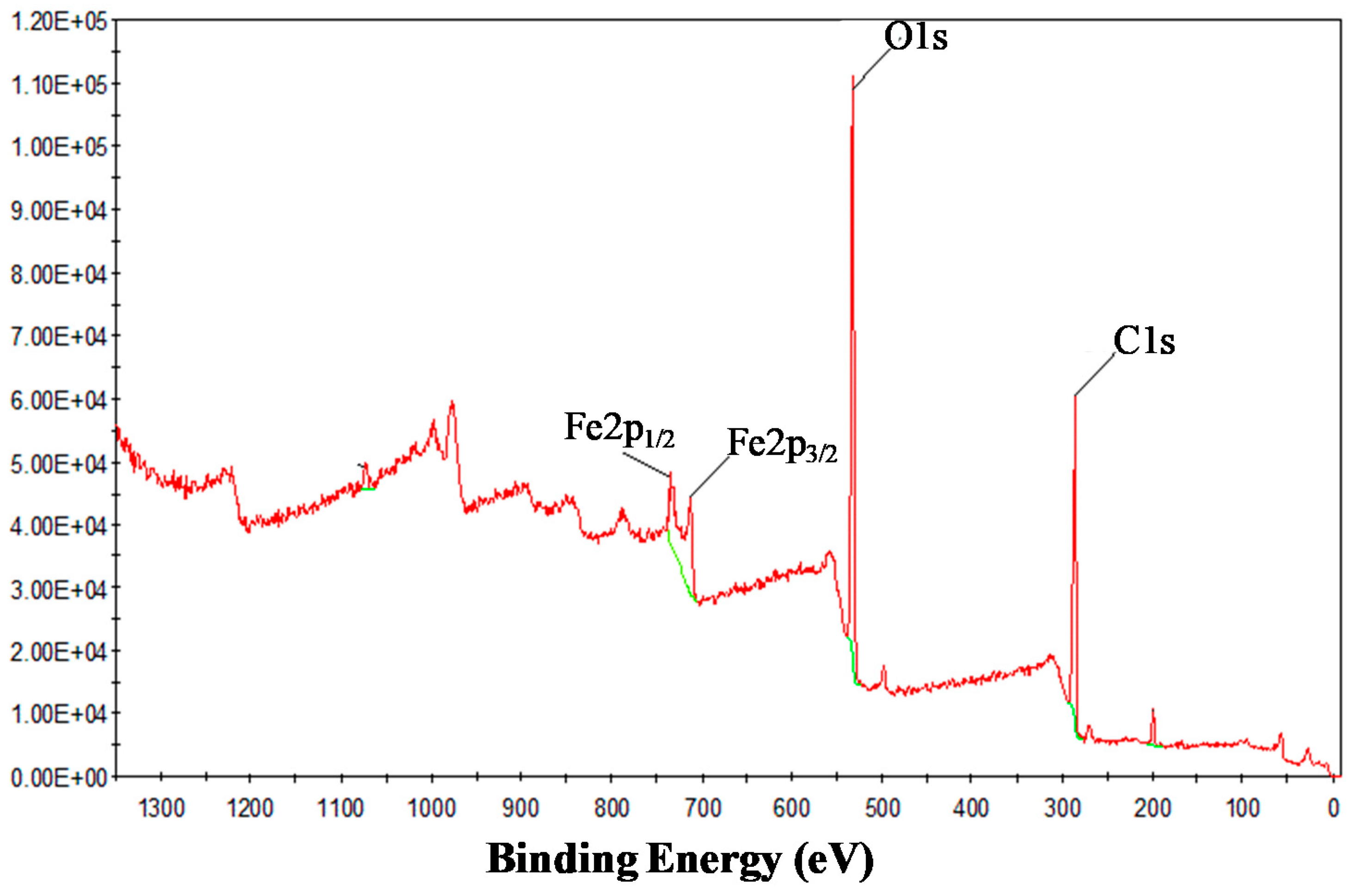

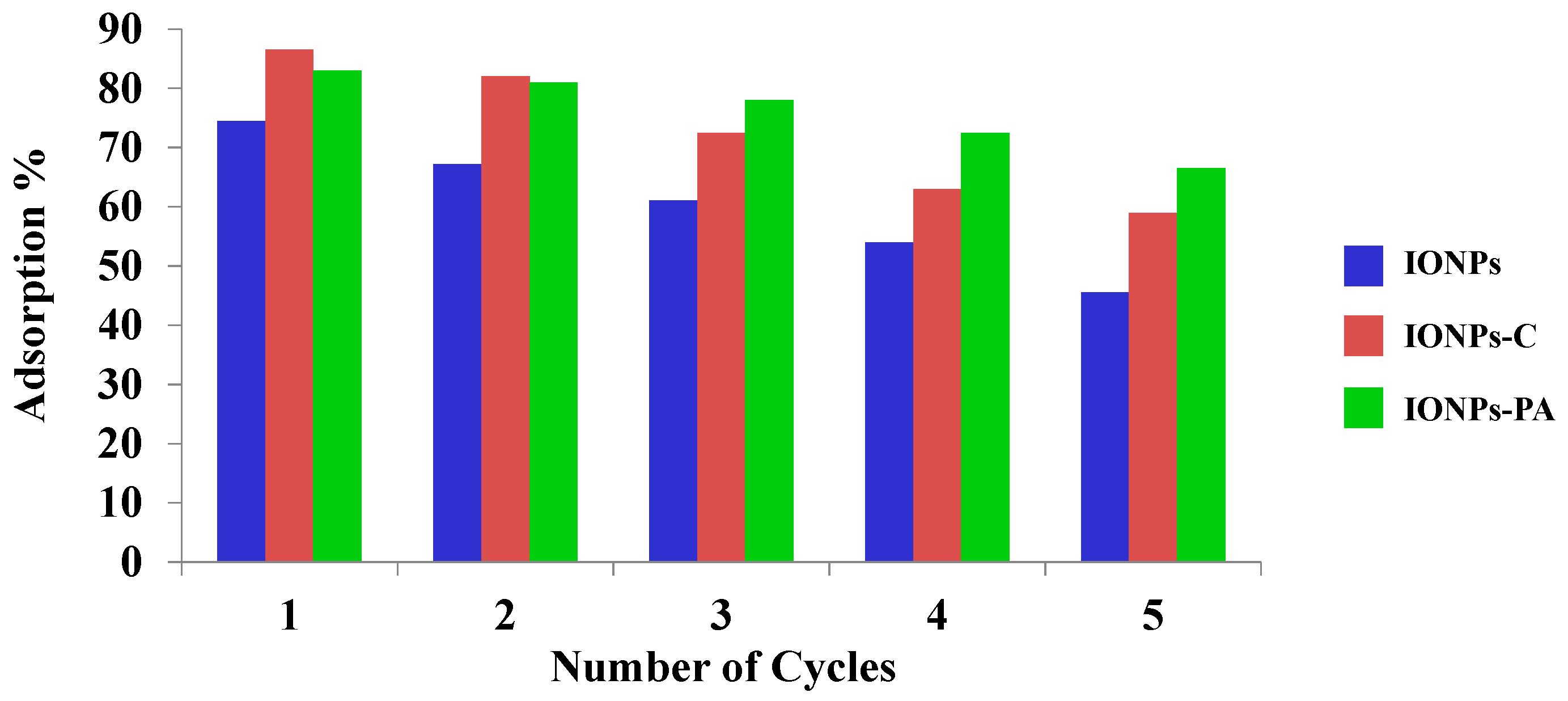
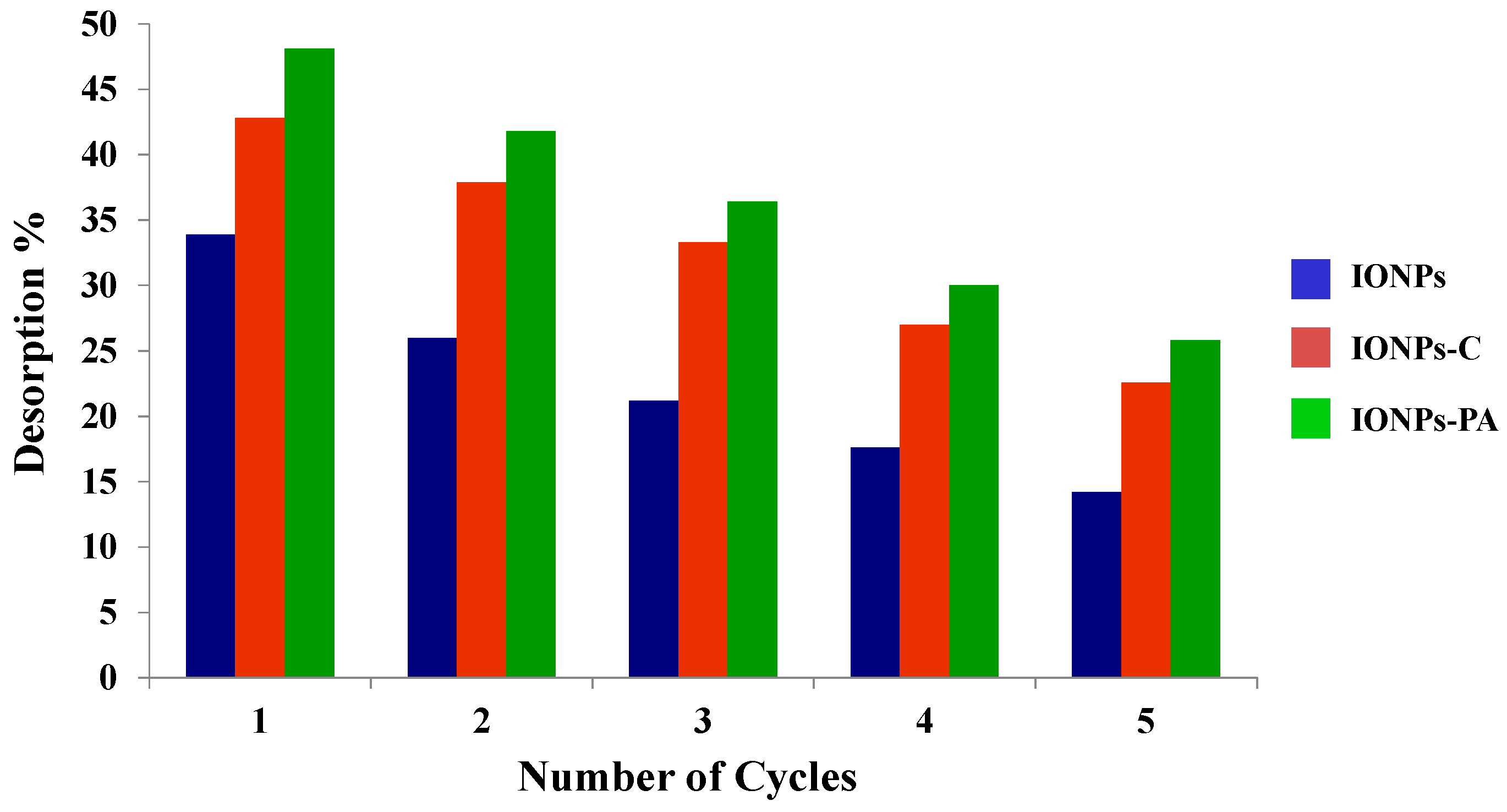
| Adsorbents | Initial As (V) (mg/L) | Pseudo-First Order Model | Pseudo-Second Order Model | ||||
|---|---|---|---|---|---|---|---|
| K1 (min−1) | qe (mg g−1) | R2 | K2 (mg g−1 min−1) | qe (mg g−1) | R2 | ||
| IONPs | 5 | 0.50 | 0.66 | 0.92 | 4.3 | 4 | 0.99 |
| 10 | 0.4 | 0.75 | 0.86 | 3.8 | 8.3 | 0.99 | |
| 25 | 0.32 | 1.7 | 0.89 | 2.7 | 14.2 | 0.99 | |
| IONPs–C | 5 | 0.581 | 1.15 | 0.98 | 2.38 | 4.62 | 0.99 |
| 10 | 0.379 | 1.52 | 0.94 | 1.42 | 11.4 | 0.99 | |
| 25 | 0.11 | 2.4 | 0.81 | 1.33 | 19.2 | 0.99 | |
| IONPs–PA | 5 | 0.62 | 0.67 | 0.99 | 0.9 | 5.10 | 0.99 |
| 10 | 0.51 | 1.12 | 0.95 | 1.5 | 9.5 | 0.999 | |
| 25 | 0.138 | 1.7 | 0.97 | 1.1 | 21.2 | 0.99 | |
| Adsorbents | Langmuir Model | Freundlich Model | ||
|---|---|---|---|---|
| qm (mg/g) | KL (L/mg) | 1/n | Kf (mg/g) | |
| IONPs | 28.57 | 0.11 | 0.68 | 2.6 |
| IONPs–C-0.1 | 27.7 | 0.088 | 0.64 | 3.17 |
| IONPs–C-0.15 | 37.0 | 0.14 | 0.571 | 4.6 |
| IONPs–C-0.3 | 34.48 | 0.05 | 0.76 | 1.84 |
| IONPs–PA-0.1 | 33.2 | 0.07 | 0.78 | 2.06 |
| IONPs–PA-0.15 | 37.03 | 0.06 | 0.76 | 2.3 |
| IONPs–PA-0.3 | 40.3 | 0.1 | 0.64 | 3.8 |
| Type of Adsorbents | Adsorption Capacity (qm) | Conditions | Ref. |
|---|---|---|---|
| Magnetic Fe2O3 nanoparticles | 88.44 mg/g at 10 °C, 95.37 mg/g at 20 °C, 105.25 mg/g at 30 °C | As = 10–200 mg/L, Adsorbent = 0.16 g, pH = 3 | [17] |
| Zerovalent iron encapsulated chitosan nanospheres | 119 ± 2.6 mg/g | As = 1–60 mg/L, Adsorbent = 0.1 g, pH = 7 | [59] |
| Ascorbic acid-coated Fe3O4 nanoparticles | 16.56 mg/g | pH = 7, 300 K, 60 mg/L adsorbent | [60] |
| Starch and Carboxymethyl cellulose (CMC) zerovalent iron | 14 mg/g | Adsorbent = 100–1000 mg/L, As = 2 mg/L, pH= 7 | [61] |
| Zero valent iron reduced graphene | 29.04 mg/g | 10 mg adsorbent, As(V) = 1–15 ppm, pH = 7.0 | [62] |
| Montmorillonite-supported nanoscale zero-valent iron | 45.5 mg/g | As = 5–250 mg/L, Adsorbent = 1 g/L, pH = 7 | [63] |
| Chitosan nanocomposites–magnetic nanoparticles | 35.7 mg/g | Adsorbent = 1 g/L, pH = 6.8 | [24] |
| Plant-synthesized IONPs chitosan hybrid | 147 ± 7 mg/g | As(V) = 300-8000 µg/L, Adsorbent = 25 g/L | [64] |
| No. of Cycles | IONPs | IONPs–C-0.15 | IONPs–PA-0.15 |
|---|---|---|---|
| Cycle-1 | 0.06 ± 0.12 | 0.007 ± 0.2 | 0.002 ± 0.06 |
| Cycle-2 | 0.14 ± 0.18 | 0.08 ± 0.21 | 0.026 ± 0.08 |
| Cycle-3 | 0.28 ± 0.11 | 0.13 ± 0.13 | 0.051 ± 0.14 |
| Cycle-4 | 0.4 ± 0.12 | 0.17 ± 0.11 | 0.078 ± 0.15 |
| Cycle-5 | 0.54 ± 0.07 | 0.2 ± 0.20 | 0.12 ± 0.04 |
| Time (h) | IONPs (0.01 g/L) | IONPs (0.5 g/L) | IONPs–C-0.15 | IONPs–PA-0.15 |
|---|---|---|---|---|
| 0.5 | 0.00 ± 0.07 | 0.003 ± 0.07 | 0.00 ± 0.02 | 0.00 ± 0.06 |
| 1 | 0.006 ± 0.11 | 0.07 ± 0.11 | 0.008 ± 0.1 | 0.003 ± 0.12 |
| 2 | 0.02 ± 0.04 | 0.13 ± 0.04 | 0.03 ± 0.16 | 0.005 ± 0.14 |
| 4 | 0.05 ± 0.03 | 0.18 ± 0.22 | 0.04 ± 0.05 | 0.008 ± 0.22 |
| 6 | 0.08 ± 0.01 | 0.32 ± 0.14 | 0.07 ± 0.24 | 0.008 ± 0.06 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Saif, S.; Tahir, A.; Asim, T.; Chen, Y.; Adil, S.F. Polymeric Nanocomposites of Iron–Oxide Nanoparticles (IONPs) Synthesized Using Terminalia chebula Leaf Extract for Enhanced Adsorption of Arsenic(V) from Water. Colloids Interfaces 2019, 3, 17. https://doi.org/10.3390/colloids3010017
Saif S, Tahir A, Asim T, Chen Y, Adil SF. Polymeric Nanocomposites of Iron–Oxide Nanoparticles (IONPs) Synthesized Using Terminalia chebula Leaf Extract for Enhanced Adsorption of Arsenic(V) from Water. Colloids and Interfaces. 2019; 3(1):17. https://doi.org/10.3390/colloids3010017
Chicago/Turabian StyleSaif, Sadia, Arifa Tahir, Tayyaba Asim, Yongsheng Chen, and Syed Farooq Adil. 2019. "Polymeric Nanocomposites of Iron–Oxide Nanoparticles (IONPs) Synthesized Using Terminalia chebula Leaf Extract for Enhanced Adsorption of Arsenic(V) from Water" Colloids and Interfaces 3, no. 1: 17. https://doi.org/10.3390/colloids3010017
APA StyleSaif, S., Tahir, A., Asim, T., Chen, Y., & Adil, S. F. (2019). Polymeric Nanocomposites of Iron–Oxide Nanoparticles (IONPs) Synthesized Using Terminalia chebula Leaf Extract for Enhanced Adsorption of Arsenic(V) from Water. Colloids and Interfaces, 3(1), 17. https://doi.org/10.3390/colloids3010017







