Thermodynamic Behaviour of Mixed Films of an Unsaturated and a Saturated Polar Lipid. (Oleic Acid-Stearic Acid and POPC-DPPC)
Abstract
1. Introduction
2. Materials and Methods
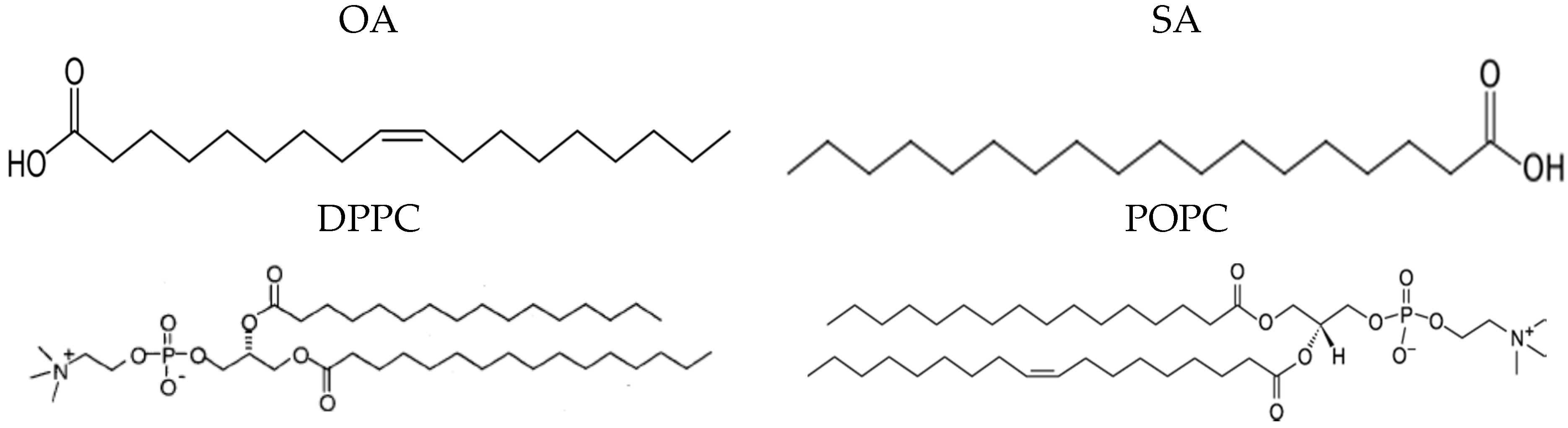
2.1. Materials
2.2. Techniques
3. Results and Discussion
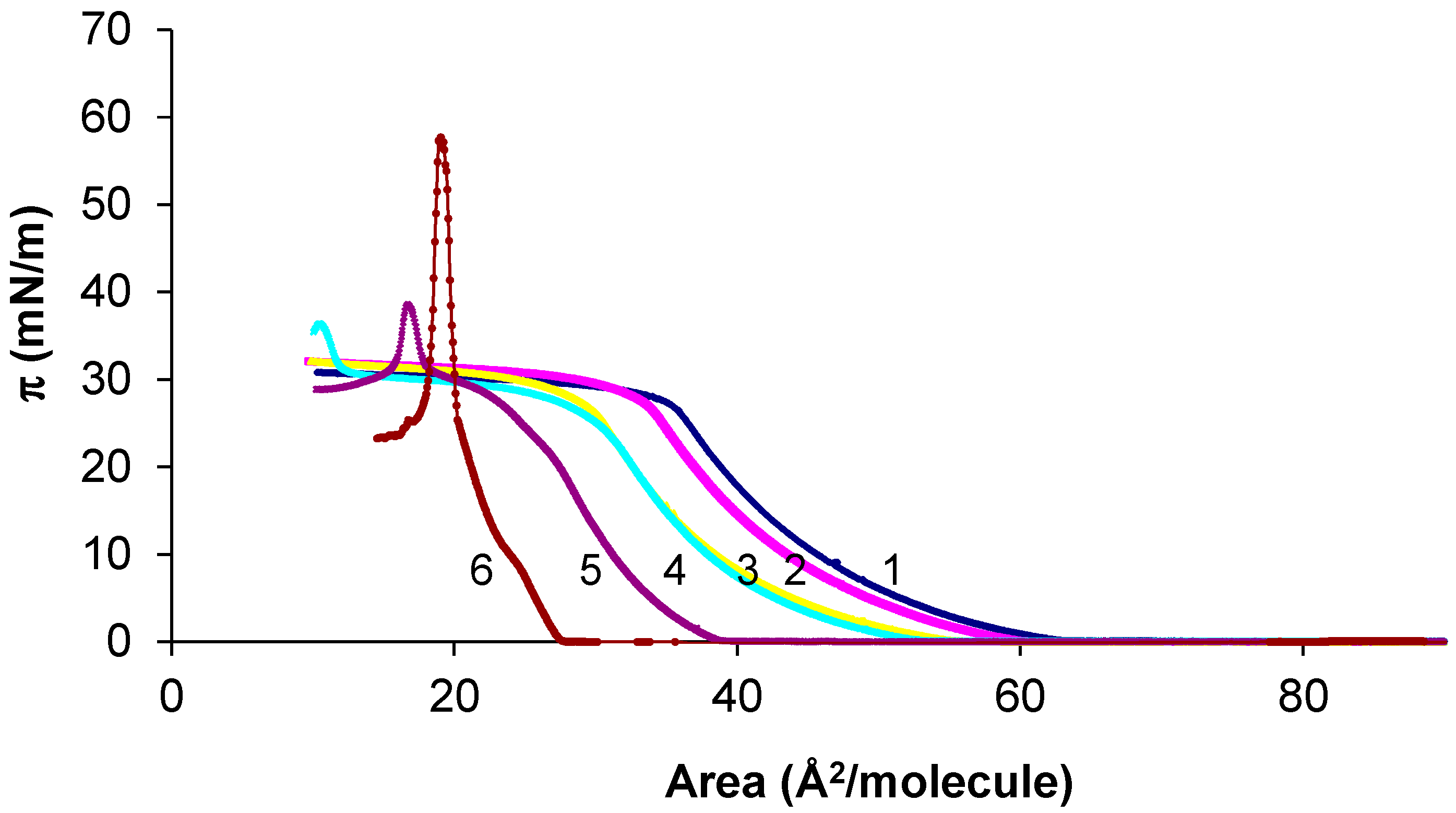
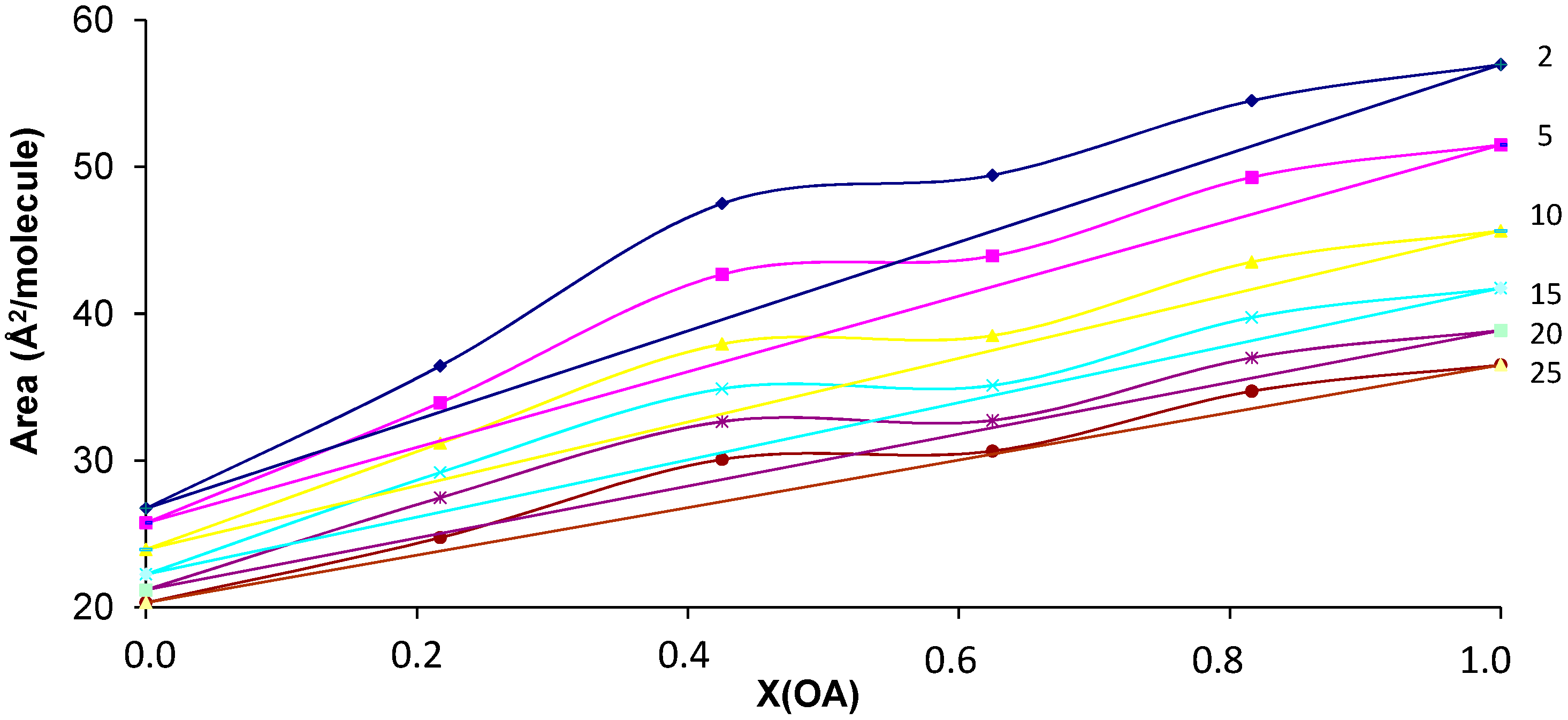
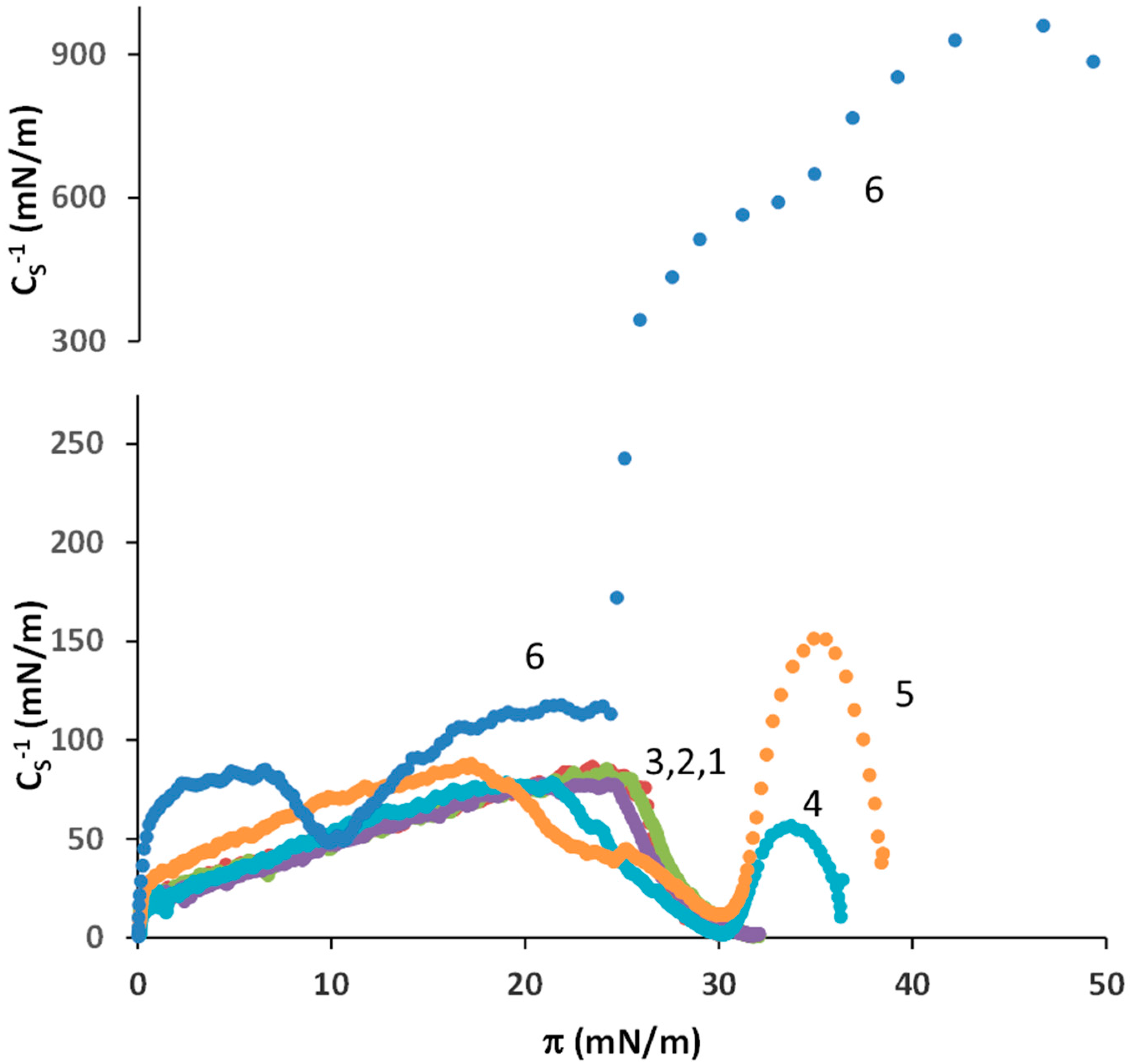
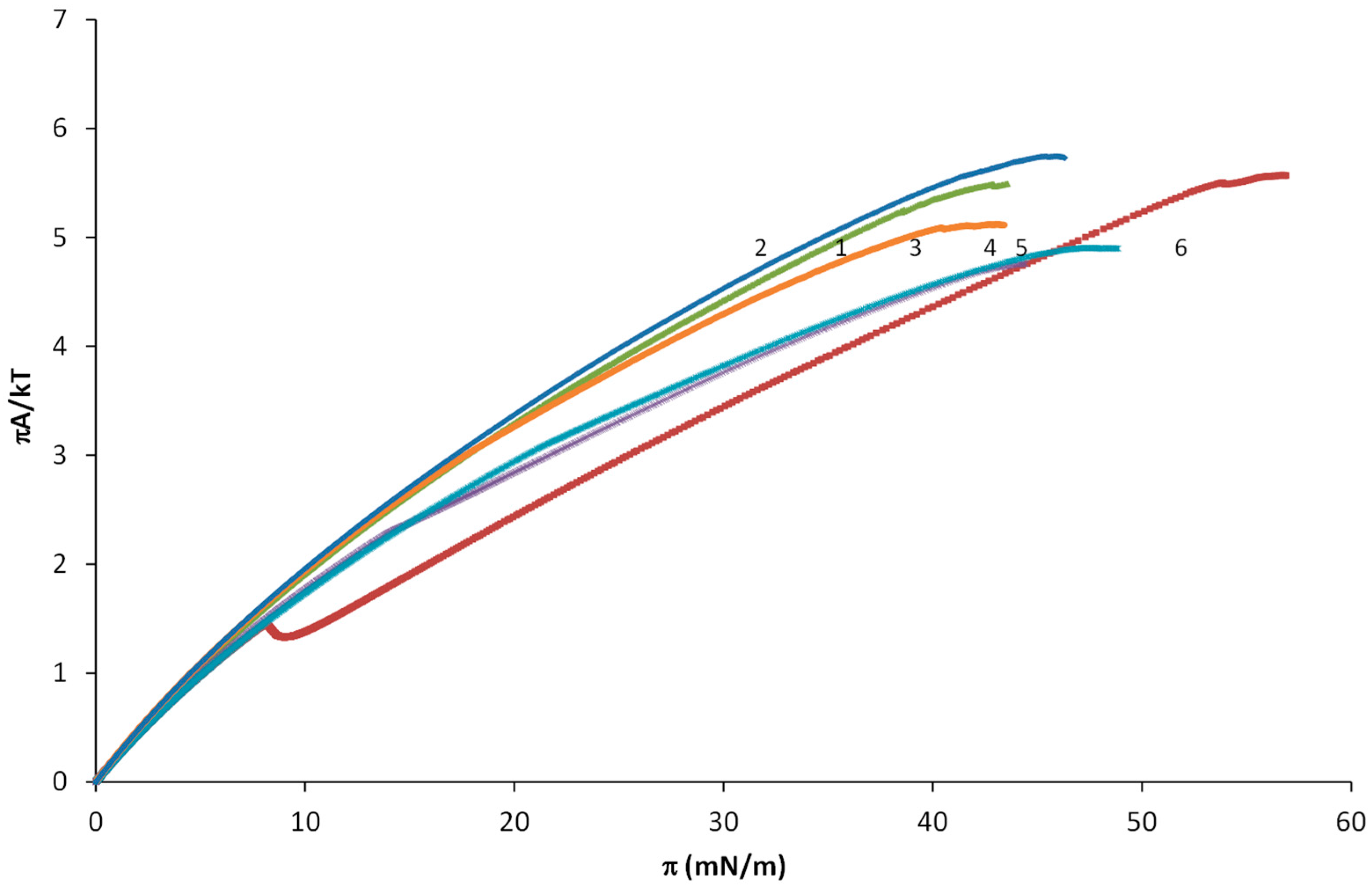
3.1. SA-OA Mixture
3.2. DPPC-POPC Mixture
3.3. Discussion
4. Conclusions
Funding
Conflicts of Interest
References
- Ulman, A. An Introduction to Ultrathin Organic Films; Academic Press: Boston, MA, USA, 1990. [Google Scholar]
- Petty, M.C. Langmuir-Blodgett Films, An Introduction; Cambridge University Press: Cambridge, MA, USA, 1996. [Google Scholar]
- Richardson, T.H. (Ed.) Functional Organic and Polymeric Materials: Molecular Functionality—Macroscopic Reality; Wiley: Chichester, UK, 2000. [Google Scholar]
- Baba, T.; Takai, K.; Takagi, T.; Kanamori, T. Effect of perfluoroalkyl chain length on monolayer behavior of partially fluorinated oleic acid molecules at the air–water interface. Chem. Phys. Lipids 2013, 172, 31–39. [Google Scholar] [CrossRef] [PubMed]
- Barzyk, W.; Vuorinen, J. Application of the vibrating plate technique to measuring electric surface potential, ΔV, of solutions; the flow cell for simultaneous measurement of the ΔV and the surface pressure. Colloids Surf. A 2011, 385, 1–10. [Google Scholar] [CrossRef]
- Dhathathreyan, A. Dissociation constants of long-chain hydroxy fatty acids in Langmuir-Blodgett films. Colloids Surf. A 2008, 318, 307–314. [Google Scholar] [CrossRef]
- Dupres, V.; Cantin, S.; Benhabib, F.; Perrot, F.; Fontaine, P.; Goldmann, M.; Daillant, J.; Konovalov, O. Superlattice formation in fatty acid monolayers on a divalent ion subphase: Role of chain length, temperature, and subphase concentration. Langmuir 2003, 19, 10808–10815. [Google Scholar] [CrossRef]
- Kundu, S.; Raychaudhuri, A.K. Effect of water and air-water interface on the structural modification of Ni-arachidate Langmuir-Blodgett films. J. Colloid Interf. Sci. 2011, 353, 316–321. [Google Scholar] [CrossRef] [PubMed]
- Mildner, J.; Dynarowicz-Latka, P. β-Carotene does not form a true Langmuir monolayer at the air-water interface. Colloids Surf. B 2012, 90, 244–247. [Google Scholar] [CrossRef] [PubMed]
- Maheshwari, R.; Dhathathreyan, A. Influence of ammonium nitrate in phase transitions of Langmuir and Langmuir–Blodgett films at air/solution and solid/solution interfaces. J. Colloid Interface Sci. 2004, 275, 270–276. [Google Scholar] [CrossRef] [PubMed]
- Snow, A.W.; Jernigan, G.G.; Ancona, M.G. Equilibrium spreading pressure and Langmuir–Blodgett film formation of omega-substituted palmitic acids. Thin Solid Films 2014, 556, 475–484. [Google Scholar] [CrossRef]
- Yang, G.; Jiang, X.; Dai, S.; Cheng, G.; Zhang, X.; Du, Z. Morphology, Defect evolutions and nano-mechanical anisotropy of behenic acid monolayer. Thin Solid Films 2010, 518, 7086–7092. [Google Scholar] [CrossRef]
- Ortiz-Collazos, S.; Gonçalves, Y.M.H.; Horta, B.A.C.; Picciani, P.H.S.; Louro, S.R.W.; Oliveira, O.N., Jr.; Pimentel, A.S. Langmuir films and mechanical properties of polyethyleneglycol fatty acids esters at the air-water interface. Colloids Surf. A 2016, 498, 50–57. [Google Scholar] [CrossRef]
- Pantoja-Romero, W.S.; Estrada-López, E.D.; Picciani, P.H.S.; Oliveira, O.N., Jr.; Lachter, E.R.; Pimentel, A.S. Efficient molecular packing of glycerol monostearate in Langmuir monolayers at the air-water interface. Colloids Surf. A 2016, 508, 85–92. [Google Scholar] [CrossRef]
- Rodríguez, J.F.F.; Dynarowicz-Latka, P.; Miñones-Conde, J. Structure of unsaturated fatty acids in 2D system. Colloids Surf. B 2017, 158, 634–642. [Google Scholar] [CrossRef] [PubMed]
- Lunkenheimer, K.; Geggel, K.; Prescher, D. Role of counterion in the adsorption behavior of 1:1 ionic surfactants at fluid interfaces-Adsorption properties of alkali perfluoro-n-octanoates at the air/water interface. Langmuir 2017, 33, 10216–10224. [Google Scholar] [CrossRef] [PubMed]
- Baba, T.; Takai, K.; Takagi, T.; Kanamori, T. Effect of the fluorination degree of hydrophobic chains on the monolayer behavior of unsaturated diacylphosphatidylcholines bearing partially fluorinated 9-octadecynoyl (stearoloyl) groups at the air–water interface. Colloids Surf. B 2014, 123, 246–253. [Google Scholar] [CrossRef] [PubMed]
- Flasinski, M.; Wydro, P.; Broniatowski, M. Lyso-phosphatidylcholines in Langmuir monolayers–Influence of chain length on physicochemical characteristics of single-chained lipids. J. Colloid Interface Sci. 2014, 418, 20–30. [Google Scholar] [CrossRef] [PubMed]
- Kaviratna, A.S.; Banerjee, R. The effect of acids on dipalmitoyl phosphatidylcholine (DPPC) monolayers and liposomes. Colloids Surf. A 2009, 345, 155–162. [Google Scholar] [CrossRef]
- Rodríguez Niño, M.R.; Lucero, A.; Rodríguez Patino, J.M. Relaxation phenomena in phospholipid monolayers at the air–water interface. Colloids Surf. A 2008, 320, 260–270. [Google Scholar] [CrossRef]
- Weis, M.; Ou-Yang, W.; Aida, T.; Yamamoto, T.; Manaka, T.; Iwamoto, M. Study of electrostatic energy contribution on monolayer domains size. Thin Solid Films 2008, 517, 1317–1320. [Google Scholar] [CrossRef]
- Adams, E.M.; Casper, C.B.; Allen, H.C. Effect of cation enrichment on DPPC monolayers at the air-water interface. J. Colloid Interface Sci. 2016, 478, 353–364. [Google Scholar] [CrossRef] [PubMed]
- Broniatowski, M.; Dynarowicz-Latka, P. Semifluorinated chains at the air/water interface: Studies of the interaction of a semifluorinated alkane with fluorinated alcohols in mixed Langmuir monolayers. Langmuir 2006, 22, 2691–2696. [Google Scholar] [CrossRef] [PubMed]
- Brzozowska, A.M.; Mugele, F.; Duits, M.H.G. Stability and interactions in mixed monolayers of fatty acid derivatives on Artificial Sea Water. Colloids Surf. A 2013, 433, 200–211. [Google Scholar] [CrossRef]
- Eftaiha, A.F.; Paige, M.F. Phase-separation of mixed surfactant monolayers: A comparison of film morphology at the solid–air and liquid–air interfaces. J. Colloid Interface Sci. 2012, 380, 105–112. [Google Scholar] [CrossRef] [PubMed]
- Imae, T.; Takeshita, T.; Kato, M. Phase separation in hybrid Langmuir-Blodgett films of perfluorinated and hydrogenated amphiphiles. Examination by AFM. Langmuir 2000, 16, 612–621. [Google Scholar] [CrossRef]
- Matsumoto, M.; Tanaka, K.; Azumi, R.; Kondo, Y.; Yoshino, N. Structure of phase-separated LB films of hydrogenated and perfluorinated carboxylic acids investigated by IR spectroscopy, AFM and FFM. Langmuir 2003, 19, 2802–2807. [Google Scholar] [CrossRef]
- Matsumoto, M.; Tanaka, K.; Azumi, R.; Kondo, Y.; Yoshino, N. Template-directed patterning using phase-separated LB films. Langmuir 2004, 20, 8728–8734. [Google Scholar] [CrossRef] [PubMed]
- Qaqish, S.E.; Paige, M.F. Characterization of domain growth kinetics in a mixed perfluorocarbon-hydrocarbon Langmuir-Blodgett monolayer. J. Colloid Interface Sci. 2008, 325, 290–293. [Google Scholar] [CrossRef] [PubMed]
- Watanabe, S.; Okuda, R.; Azumi, R.; Sakai, H.; Abe, M. Effect of subphase temperature on the phase-separated structures of mixed Langmuir and Langmuir-Blodgett films of fatty acids and hybrid carboxylic acids. J. Colloid Interface Sci. 2011, 363, 379–385. [Google Scholar] [CrossRef] [PubMed]
- Rehman, J.; Sowah-Kuma, D.; Stevens, A.L.; Bu, W.; Paige, M.F. Mixing behaviour in binary anionic gemini surfactant-perfluorinated fatty acid Langmuir monolayers. Langmuir 2017, 33, 10205–10215. [Google Scholar] [CrossRef] [PubMed]
- Paige, M.F.; Eftaiha, A.F. Phase-separated surfactant monolayers: Exploiting immiscibility of fluorocarbons and hydrocarbons to pattern interfaces. Adv. Colloid Interface Sci. 2017, 248, 129–146. [Google Scholar] [CrossRef] [PubMed]
- Ocko, B.M.; Kelley, M.S.; Nikova, A.T.; Schwartz, D.K. Structure and Phase Behavior of Mixed Monolayers of Saturated and Unsaturated Fatty Acids. Langmuir 2002, 18, 9810–9815. [Google Scholar] [CrossRef]
- Seoane, R.; Dynarowicz-Latka, P.; Miñones, J., Jr.; Rey, I. Mixed Langmuir monolayers of cholesterol and essential fatty acids. Colloid Polym. Sci. 2001, 279, 562–570. [Google Scholar] [CrossRef]
- Domenech, O.; Torrent-Burgués, J.; Merino, S.; Sanz, F.; Montero, M.T.; Hernández-Borrell, J. Surface thermodynamics study of monolayers formed with heteroacid phospholipids of biological interest. Colloids Surf. B 2005, 41, 233–238. [Google Scholar] [CrossRef] [PubMed]
- Domènech, O.; Sanz, F.; Montero, M.T.; Hernández-Borrell, J. Thermodynamic and structural study of the main phospholipid components comprising the mitochondrial inner membrane. Biochim. Biophys. Acta 2006, 1758, 213–221. [Google Scholar] [CrossRef] [PubMed]
- Dynarowicz-Łatka, P.; Wnetrzak, A.; Broniatowski, M.; Flasinski, M. Miscibility and phase separation in mixed erucylphosphocholine–DPPC Monolayers. Colloids Surf. B 2013, 107, 43–52. [Google Scholar] [CrossRef] [PubMed]
- Garcia-Manyes, S.; Domènech, O.; Sanz, F.; Montero, M.T.; Hernandez-Borrell, J. Atomic force microscopy and force spectroscopy study of Langmuir–Blodgett films formed by heteroacid phospholipids of biological interest. Biochim. Biophys. Acta 2007, 1768, 1190–1198. [Google Scholar] [CrossRef] [PubMed]
- Ohki, S.; Müller, M.; Arnold, K.; Ohshima, H. Surface potential of phosphoinositide membranes: Comparison between theory and experiment. Colloids Surf. B 2010, 79, 210–218. [Google Scholar] [CrossRef] [PubMed]
- Park, J.-W. Individual leaflet phase effect on nanometer-scale surface properties of phospholipid bilayers. Colloids Surf. B 2009, 71, 128–132. [Google Scholar] [CrossRef] [PubMed]
- Stefaniu, C.; Brezesinski, G. Grazing incidence X-ray diffraction studies of condensed double-chain phospholipid monolayers formed at the soft air/water interface. Adv. Colloid Interface Sci. 2014, 207, 265–279. [Google Scholar] [CrossRef] [PubMed]
- Wydro, P. The influence of cardiolipin on phosphatidylglycerol/ phosphatidylethanolamine monolayers—Studies on ternary films imitating bacterial membranes. Colloids Surf. B 2013, 106, 217–223. [Google Scholar] [CrossRef] [PubMed]
- Wydro, P. The influence of cholesterol on multicomponent Langmuir monolayers imitating outer and inner leaflet of human erythrocyte membrane. Colloids Surf. B 2013, 103, 67–74. [Google Scholar] [CrossRef] [PubMed]
- Wydro, P.; Witkowska, K. The interactions between phosphatidylglycerol and phosphatidylethanolamines in model bacterial membranes. The effect of the acyl chain length and saturation. Colloids Surf. B 2009, 72, 32–39. [Google Scholar] [CrossRef] [PubMed]
- Olżyńska, A.; Zubek, M.; Roeselova, M.; Korchowiec, J.; Cwiklik, L. Mixed DPPC/POPC monolayers: All-atom molecular dynamics simulations and Langmuir monolayer experiments. Biochim. Biophys. Acta 2016, 1858, 3120–3130. [Google Scholar] [CrossRef] [PubMed]
- Hoyo, J.; Torrent-Burgués, J.; Guaus, E. Biomimetic monolayer films of monogalactosyldiacylglycerol incorporating ubiquinone. J. Colloid Interface Sci. 2012, 384, 189–197. [Google Scholar] [CrossRef] [PubMed]
- Vitovič, P.; Nikolelis, D.P.; Hianik, T. Study of calix [4] resorcinarene–dopamine com-plexation in mixed phospholipid monolayers formed at the air|water interface. Biochim. Biophys. Acta 2006, 1758, 1852–1861. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Hoyo, J.; Guaus, E.; Torrent-Burgués, J. Monogalactosyldiacylglycerol and digalactosyldiacylglycerol role, physical states, applications and biomimetic monolayer films. Eur. Phys. J. E 2016, 39, 39. [Google Scholar] [CrossRef] [PubMed]
- Hoyo, J.; Guaus, E.; Torrent-Burgués, J. Tuning ubiquinone position in biomimètic monolayer membranes. Eur. Phys. J. E 2017, 40, 62. [Google Scholar] [CrossRef] [PubMed]
- Birdi, K.S.; Gevod, V.S. Melittin and ionic surfactant interactions in monomolecular films. Colloid Polym. Sci. 1987, 265, 257–261. [Google Scholar] [CrossRef]
- Davies, J.T.; Rideal, E.K. Interfacial Phenomena; Academic Press: Cambridge, MA, USA, 1993. [Google Scholar]
- Avantilipids. Available online: https://avantilipids.com/tech-support/physical-properties/ (accessed on 10 April 2018).
- Nakata, S.; Seki, Y.; Nomura, M.; Fukuhara, K.; Denda, M. Characteristic Isotherms for a Mixed Molecular Layer Composed of Phospholipid and Fatty Acid. Bull. Chem. Soc. Jpn. 2017, 90, 801–806. [Google Scholar] [CrossRef]
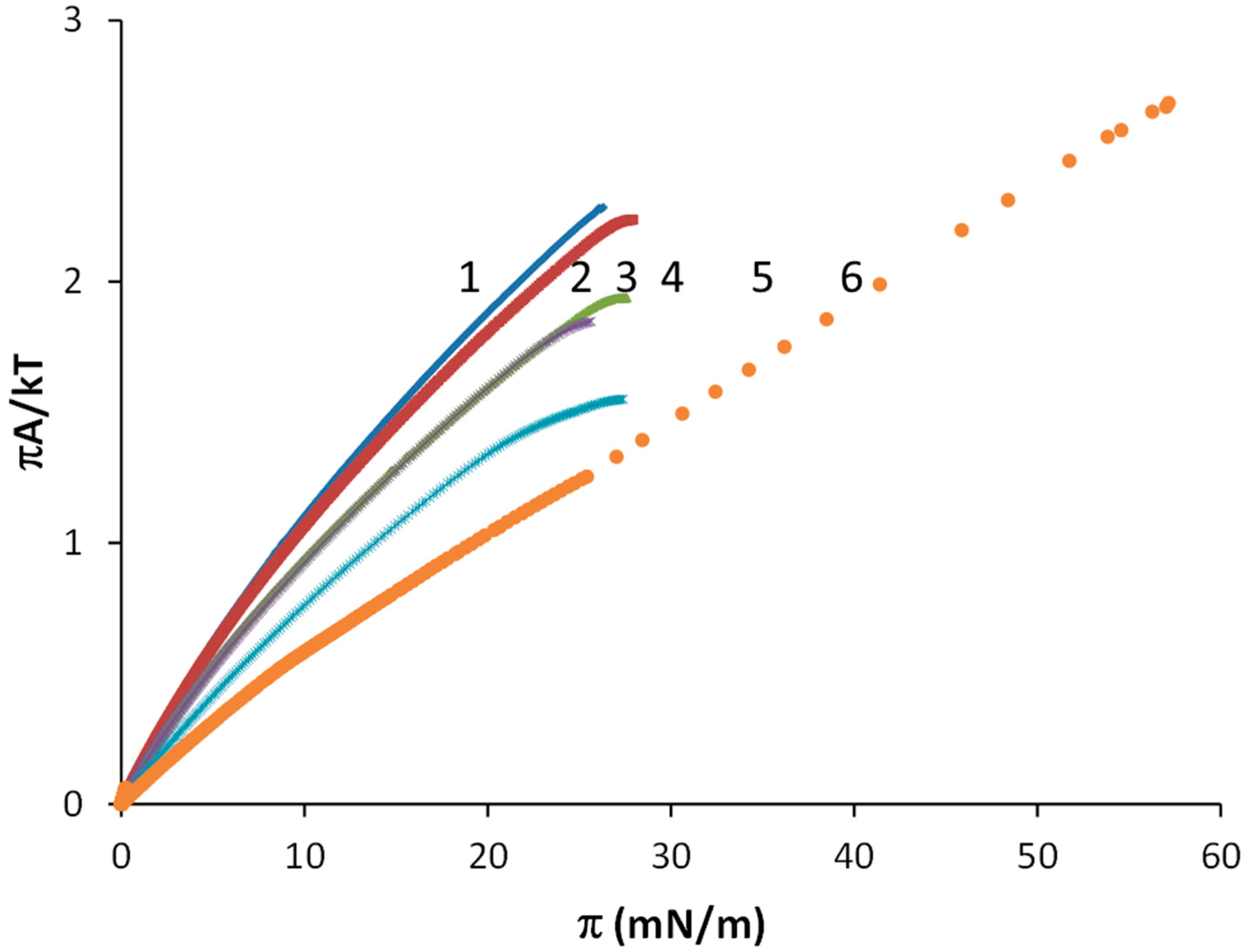
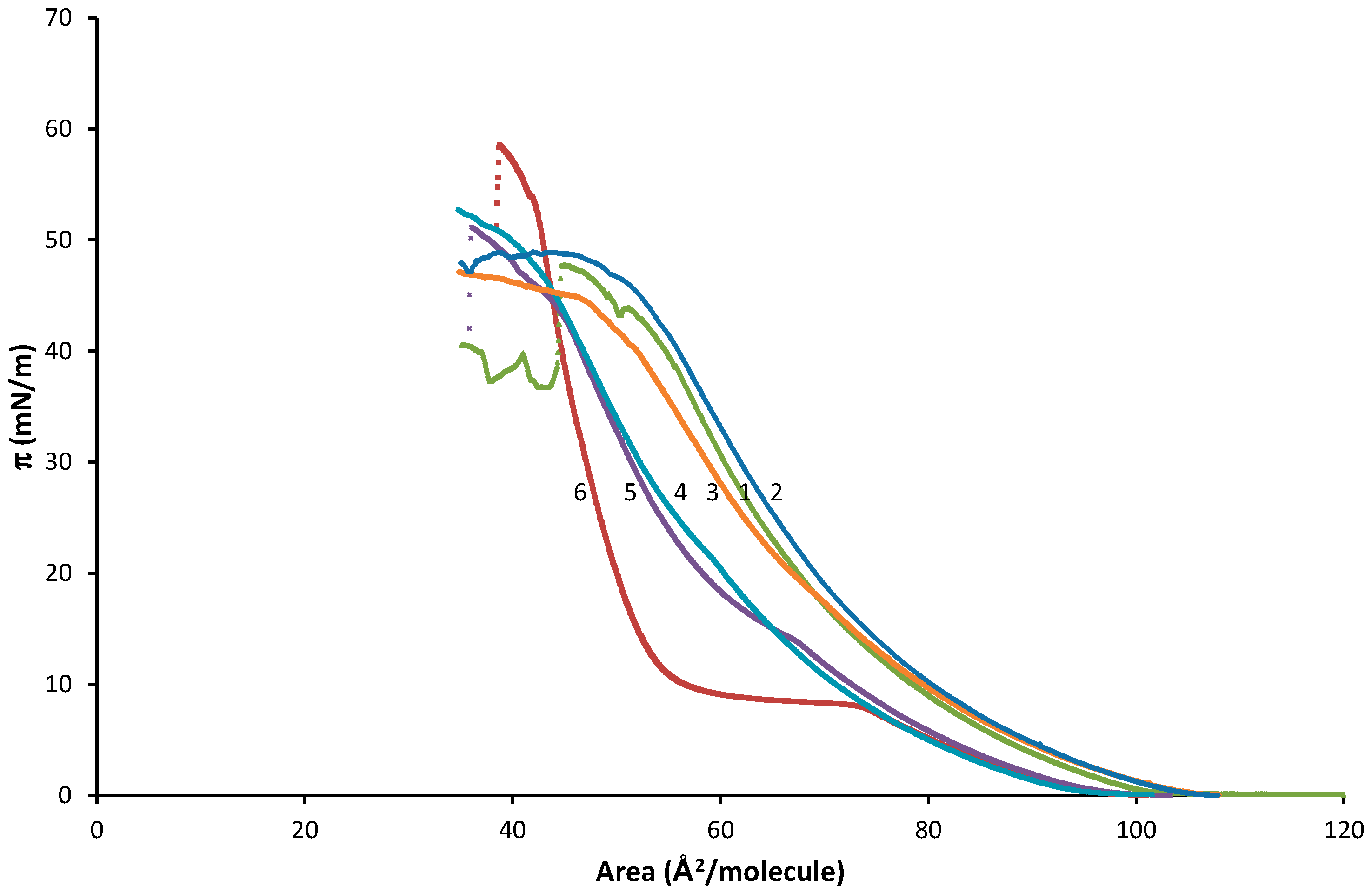
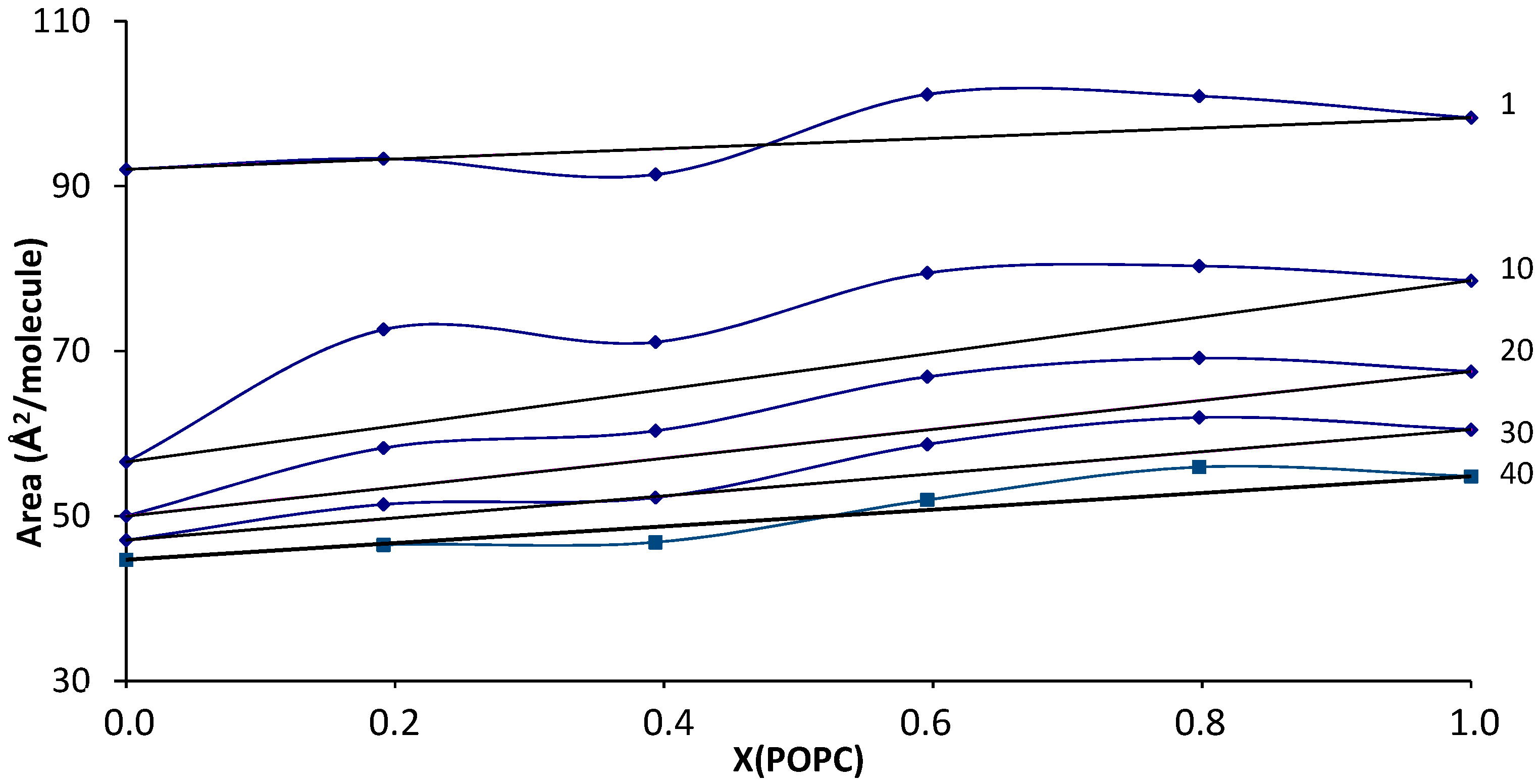
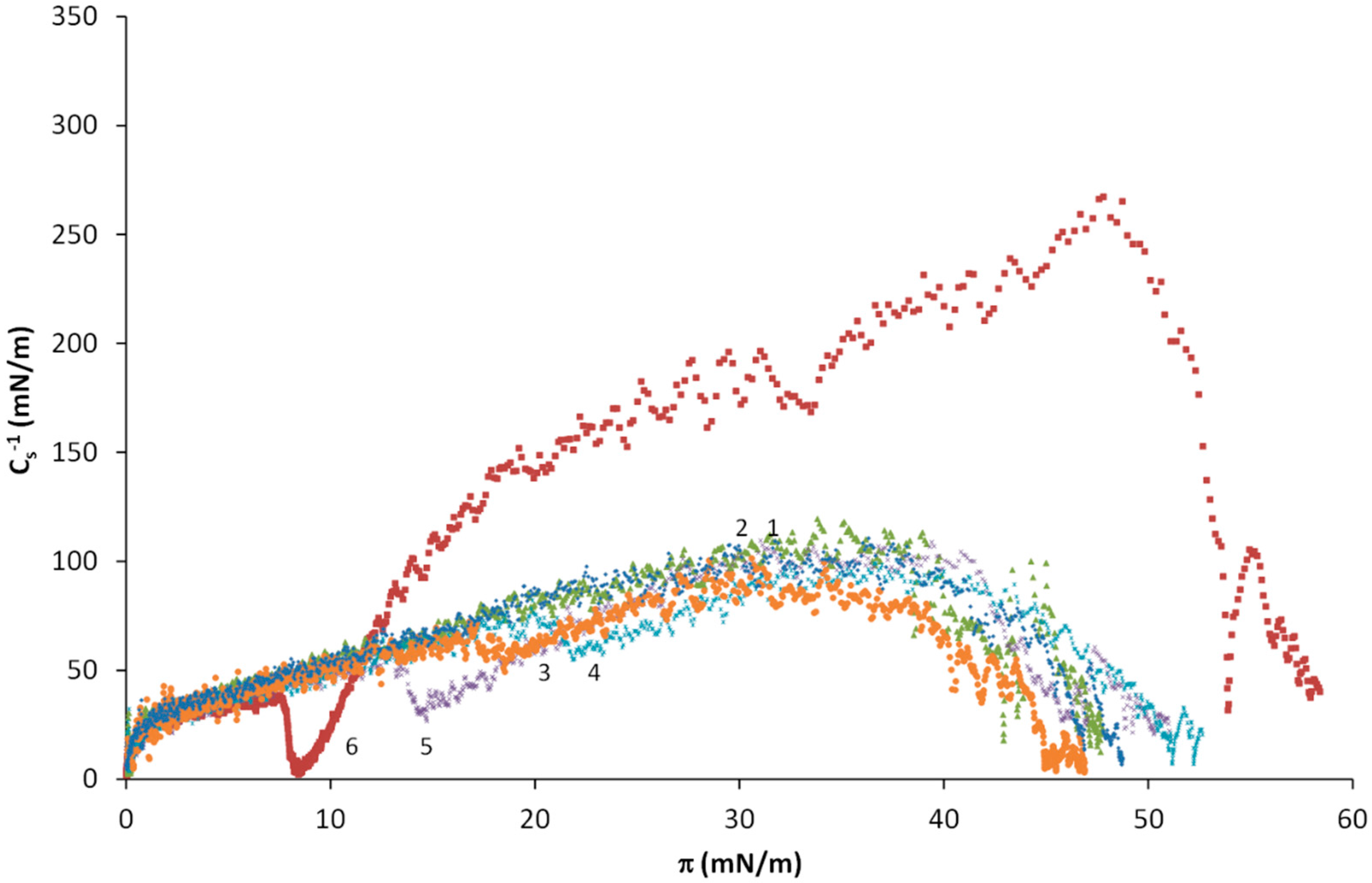
| Virial Coefficient | OA | OA-SA XOA = 0.816 | OA-SA XOA = 0.625 | OA-SA XOA = 0.425 | OA-SA XOA = 0.217 | SA |
|---|---|---|---|---|---|---|
| b0 | 0.0055 | 0.0050 | 0.0046 | 0.0048 | 0.0007 | −0.0021 |
| b1 | 0.1255 | 0.1208 | 0.1080 | 0.1064 | 0.0883 | 0.0566 |
| b2 | −0.0015 | −0.0015 | −0.0014 | −0.0013 | −0.0011 | −0.00018 |
| R2 | 0.9994 | 0.9994 | 0.9994 | 0.9997 | 0.9999 | 0.9974 |
| XOA | b1)m | b1)12 | b1E |
|---|---|---|---|
| 0 | 0.0566 | 0 | |
| 0.217 | 0.0883 | 0.1403 | 0.0167 |
| 0.425 | 0.1064 | 0.1330 | 0.0205 |
| 0.625 | 0.1080 | 0.1088 | 0.0083 |
| 0.816 | 0.1208 | 0.1176 | 0.0080 |
| 1 | 0.1255 | 0 |
| π (mN/m) | XOA | 0.816 | 0.625 | 0.425 | 0.217 |
|---|---|---|---|---|---|
| 5 | GE (J/mol) | 75 | 78 | 199 | 150 |
| ΔGmix (J/mol) | −1096 | −1545 | −1473 | −1133 | |
| 15 | GE (J/mol) | 213 | 191 | 575 | 409 |
| ΔGmix (J/mol) | −958 | −1432 | −1097 | −874 | |
| 25 | GE (J/mol) | 291 | 204 | 809 | 510 |
| ΔGmix (J/mol) | −880 | −1419 | −863 | −773 |
| Virial Coefficient | POPC | POPC:DPPC XPOPC = 0.798 | POPC:DPPC XPOPC = 0.596 | POPC:DPPC XPOPC = 0.394 | POPC:DPPC XPOPC = 0.191 | DPPC |
|---|---|---|---|---|---|---|
| b0 | 0.0319 | 0.0856 | 0.0923 | 0.0832 | 0.0978 | 0.1930 |
| b1 | 0.2002 | 0.1991 | 0.1984 | 0.1744 | 0.1736 | 0.1333 |
| b2 | −0.0017 | −0.0016 | −0.0019 | −0.0016 | −0.0016 | −0.00068 |
| R2 | 0.9994 | 0.9995 | 0.9992 | 0.9989 | 0.9970 | 0.9916 |
| XPOPC | b1)m | b1)12 | b1E |
|---|---|---|---|
| 0 | 0.1333 | 0 | |
| 0.191 | 0.1736 | 0.256 | 0.0275 |
| 0.394 | 0.1744 | 0.198 | 0.0147 |
| 0.596 | 0.1984 | 0.219 | 0.0252 |
| 0.798 | 0.1991 | 0.205 | 0.0124 |
| 1 | 0.2002 | 0 |
| π (mN/m) | XPOPC | 0.798 | 0.596 | 0.394 | 0.191 |
|---|---|---|---|---|---|
| 5 | GE (J/mol) | 202 | 211 | −53 | −6 |
| ΔGmix (J/mol) | −1032 | −1444 | −1698 | −1202 | |
| 15 | GE (J/mol) | 538 | 667 | 56 | 318 |
| ΔGmix (J/mol) | −696 | −988 | −1588 | −878 | |
| 25 | GE (J/mol) | 817 | 980 | 106 | 556 |
| ΔGmix (J/mol) | −417 | −675 | −1539 | −640 | |
| 35 | GE (J/mol) | 1061 | 1144 | 48 | 645 |
| ΔGmix (J/mol) | −174 | −510 | −1597 | −551 |
© 2018 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Torrent-Burgués, J. Thermodynamic Behaviour of Mixed Films of an Unsaturated and a Saturated Polar Lipid. (Oleic Acid-Stearic Acid and POPC-DPPC). Colloids Interfaces 2018, 2, 17. https://doi.org/10.3390/colloids2020017
Torrent-Burgués J. Thermodynamic Behaviour of Mixed Films of an Unsaturated and a Saturated Polar Lipid. (Oleic Acid-Stearic Acid and POPC-DPPC). Colloids and Interfaces. 2018; 2(2):17. https://doi.org/10.3390/colloids2020017
Chicago/Turabian StyleTorrent-Burgués, Juan. 2018. "Thermodynamic Behaviour of Mixed Films of an Unsaturated and a Saturated Polar Lipid. (Oleic Acid-Stearic Acid and POPC-DPPC)" Colloids and Interfaces 2, no. 2: 17. https://doi.org/10.3390/colloids2020017
APA StyleTorrent-Burgués, J. (2018). Thermodynamic Behaviour of Mixed Films of an Unsaturated and a Saturated Polar Lipid. (Oleic Acid-Stearic Acid and POPC-DPPC). Colloids and Interfaces, 2(2), 17. https://doi.org/10.3390/colloids2020017






