1. Introduction
Airborne particulate matter (PM), particularly ultrafine particles such as PM2.5, is a major environmental contributor to skin aging and dysfunction. Upon dermal exposure, PM can penetrate the skin barrier via intercellular routes or hair follicles, initiating oxidative stress, lipid peroxidation, DNA damage, and inflammatory cascades through the activation of signaling pathways such as nuclear factor kappa B (NF-κB), mitogen-activated protein kinase (MAPK), and the aryl hydrocarbon receptor (AhR) [
1,
2,
3,
4,
5,
6]. These processes degrade essential structural proteins, including filaggrin and loricrin, leading to increased transepidermal water loss (TEWL), impaired hydration, and visible signs of skin aging such as wrinkles, hyperpigmentation, and sensitivity [
7,
8,
9,
10]. Epidemiological studies have further linked PM exposure to inflammatory dermatoses such as atopic dermatitis, psoriasis, and acne [
11]. While topical formulations containing antioxidants or barrier-supporting agents are frequently used to mitigate pollution-induced skin damage, these treatments often provide limited penetration and superficial protection [
12,
13]. As a result, there is growing interest in nutricosmetic approaches—systemic or oral supplementation of bioactive compounds that enhance skin defense mechanisms from within [
14].
Cannabidiol (CBD), a non-psychoactive compound derived from
Cannabis sativa, has received increasing attention for its anti-inflammatory, antioxidant, sebostatic, and regenerative properties [
15,
16,
17,
18]. These effects are mediated through multiple molecular targets, including transient receptor potential vanilloid (TRPV) channels, peroxisome proliferator-activated receptors (PPARs), and cannabinoid receptors CB1 and CB2 [
17,
18,
19,
20]. Studies using in vitro and ex vivo models have shown that CBD can reduce reactive oxygen species (ROS), downregulate pro-inflammatory cytokines, inhibit MMP-1, and promote collagen synthesis [
18,
21,
22,
23,
24,
25]. However, its clinical utility is limited by unfavorable physicochemical properties, including high lipophilicity (log P ~6.3), low aqueous solubility, and instability under heat, light, and oxygen exposure, resulting in low and variable bioavailability in both oral and systemic formats [
21,
22,
23,
24].
To overcome these challenges, nanoemulsions (NEs) have been extensively studied as delivery platforms for lipophilic and poorly soluble compounds. Owing to their small droplet size (typically 20–200 nm), oil-in-water (O/W) NEs offer several advantages over conventional emulsions, including enhanced solubilization, protection from environmental degradation, increased surface area for absorption, and kinetic stability [
25,
26,
27]. In this study, we employed polyoxyethylene (10) oleyl ether (C18:1E10), commercially known as Brij
® O10, as the surfactant. Brij
® O10 is a nonionic emulsifier with a high hydrophilic-lipophilic balance and has demonstrated efficient emulsification behavior and physicochemical stability in NE systems [
28,
29]. It has also been investigated for its functional role in enhancing intestinal permeability of certain drug substrates [
30]. Olive oil was selected as the lipid phase due to its established compatibility with Brij
® O10 in forming stable, transparent NEs [
28,
29,
31,
32,
33], as well as its own antioxidant and anti-inflammatory activity through constituents like oleic acid, squalene, and polyphenols [
34,
35].
To fabricate the NEs, we utilized a low-energy phase inversion temperature (PIT) method, a thermodynamically driven emulsification process that eliminates the need for high-shear equipment or organic solvents [
36,
37]. The PIT method exploits the temperature-dependent shift in surfactant curvature: heating the surfactant–oil–water system induces a transition at which the surfactant changes from hydrophilic to lipophilic, allowing for spontaneous formation of fine, stable droplets upon cooling [
36,
37]. This approach is especially suitable for thermolabile compounds such as CBD, as it avoids harsh mechanical or chemical conditions [
37].
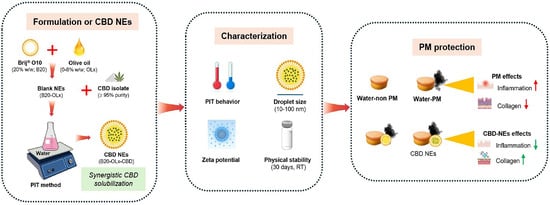
Using this approach, we prepared five CBD-loaded NE systems and systematically characterized their PIT behavior, droplet size, zeta potential, morphology, visual appearance, and CBD solubilization capacity. Based on these metrics, two optimized formulations—designated B20-OL4-CBD and B20-OL8-CBD, corresponding to specific Brij
® O10 and olive oil ratios—were selected for biological evaluation. To assess their efficacy, we employed a full-thickness human ex vivo skin model, which preserves the epidermal and dermal architecture, extracellular matrix components, and barrier function [
38]. The NEs were added to the culture medium to simulate systemic exposure, while the skin surface was challenged with PM to mimic environmental stress [
8]. Biomarkers related to inflammation (IL-6), matrix degradation (MMP-1), and skin integrity (PIP) were assessed to evaluate the anti-pollution potential of each formulation. This study bridges formulation engineering with biological assessment, integrating low-energy NE fabrication with ex vivo human skin testing. Our findings offer insights into how colloidal encapsulation strategies can enhance the delivery and functional activity of CBD in the context of pollution-related skin damage.
2. Materials and Methods
2.1. Preparation of Cannabidiol (CBD)-Loaded Nanoemulsions
Oil-in-water NEs were prepared using the phase inversion temperature (PIT) method. Olive oil (Thai-China Flavours and Fragrances Industry Co., Ltd., Ayutthaya, Thailand), Brij® O10 (polyoxyethylene-10-oleyl ether, #P6136, Sigma-Aldrich, St. Louis, MO, USA), and distilled water were weighed sequentially in the following order: oil, surfactant, and water. The mixture was magnetically stirred while being heated to 70–80 °C for 10 min, followed by gradual cooling to room temperature under continuous vigorous stirring. Micellar solutions were prepared using the same procedure, omitting the oil phase.
To prepare CBD-loaded micelles and NEs, an excess amount of CBD (Leapdelab Co., Ltd., Samut Prakan, Thailand; ≥95% purity by HPLC) was added to 1 mL aliquots (n = 3) of each formulation in Eppendorf tubes. Samples were sealed, wrapped in aluminum foil to protect them from light, and agitated on a rotating wheel at room temperature for 7 days to reach solubilization equilibrium. Importantly, this 7-day period did not reflect NE production time, as PIT-based emulsification was completed before CBD loading. Equilibration was carried out under controlled ambient conditions (25 ± 1 °C) without additional heating or cooling. After equilibrium was reached, excess undissolved CBD was removed by centrifugation at 6500 rpm for 20 min. Blank formulations (NEs without CBD) were prepared and processed under the same conditions.
2.2. Stability Assessment and Nanoemulsion Phase Diagram Construction
All formulations were stored in tightly sealed glass bottles at room temperature. Physical stability was assessed by visual inspection immediately after preparation, and again on days 1, 7, and 30. Samples that remained isotropic (non-birefringent), monophasic, and fluid throughout the 30-day storage period were classified as stable NEs.
Stable formulations were prepared in triplicate and used to define the NE existence region with ±1% w/w component accuracy. A partial ternary phase diagram was constructed with apexes representing 100% oil, surfactant, and water, respectively, with each point corresponding to a unique formulation composition.
2.3. Cloud Point and Phase Inversion Temperature (PIT) Determination
The PIT of the NEs and the cloud point (CP) of corresponding micellar solutions were determined using a water bath method. Each sample was transferred into a sealed glass bottle equipped with an internal thermometer and immersed in a temperature-controlled water bath placed on a hot plate. Samples were heated at approximately 1 °C/min under continuous vigorous stirring until turbidity was visually observed, and then cooled at the same rate until clarity was restored.
The PIT was defined as the temperature at which turbidity first appeared during heating, and CP as the temperature at which clarity returned during cooling. Each sample underwent three heating–cooling cycles (six total measurements), and the average temperature values were reported.
2.4. Particle Size and Zeta Potential Measurement by Photon Correlation Spectroscopy
Particle size and zeta potential of micelles and oil-in-water (O/W) NEs were measured by photon correlation spectroscopy (PCS) and electrophoretic light scattering, respectively, using a (Malvern Panalytical Ltd., Worcestershire, UK) at 25 ± 0.1 °C. The water used to dilute the samples was filtered through a 0.22 µm nylon membrane (National Scientific Company, Rockwood, TN, USA). Instrument parameters for water were set to a refractive index of 1.33 and a viscosity of 0.89 cP. The instrument was equipped with a 4 mW He–Ne laser operating at 633 nm, and size measurements were acquired in backscattering mode at a detection angle of 173°.
All samples were stored as concentrates and freshly diluted with filtered deionized water to a final surfactant concentration of 2% w/w immediately before analysis, in order to minimize multiple scattering at high surfactant levels. For particle size determination, three independent batches of each formulation were measured, and each batch was analyzed in triplicate. The apparent hydrodynamic diameter was calculated assuming spherical particles, with each reported value obtained as the average of at least 20 sub-runs per measurement.
Zeta potential was determined on the same diluted samples using folded capillary cells (Malvern Panalytical Ltd., Worcestershire, UK). Zeta potential (
) was calculated from the measured electrophoretic mobility (
) using the Henry equation [
39]:
where
is the dispersant viscosity and
is the dielectric permittivity of the dispersant
The Hückel approximation was applied
= 1.0) as selected in the Zetasizer software v3.30 for low-ionic-strength dispersions. For each formulation, three independent batches were analyzed in triplicate, and zeta potential values are reported as mean ± standard deviation.
2.5. Transmission Electron Microscopy (TEM)
The size and morphology of CBD-free and CBD-loaded NEs were examined by transmission electron microscopy (TEM, Hitachi HT7800, Tokyo, Japan). Fresh O/W NEs were diluted in filtered deionized water by gentle vortex mixing for 30 s at dilution ratios of 1:10 and 1:50 (v/v). A 5 µL aliquot of the diluted sample was deposited onto a carbon-coated copper grid (200 mesh) and allowed to adsorb. Excess liquid was removed with filter paper, and the grids were placed in a dehumidification cabinet at room temperature until completely dry (up to 3 days). The dried samples were examined at an accelerating voltage of 80 kV. For most formulations, images were acquired at 50,000× magnification with a nominal scale bar of 200 nm, whereas the CBD-loaded NEs with 8% olive oil (B20-OL8-CBD) were imaged at 10,000× magnification with a scale bar of 1.0 µm to provide a wider field of view. TEM was used for qualitative assessment of morphology.
2.6. Visual Assessment of CBD-Loaded Nanoemulsions
CBD-loaded formulations containing the maximum solubilized amount of CBD were compared with their corresponding blank NEs by visual inspection to assess changes in physical appearance and dispersion stability. Samples were examined for signs of instability, including increased turbidity, phase separation, flocculation, or precipitation, during storage under the same conditions used for stability assessment. Any visible differences between CBD-loaded and blank NEs were recorded and interpreted together with the physicochemical data.
2.7. Evaluation of Micellar and Nanoemulsion Capacity for CBD Solubilization
CBD solubility in the micellar and NE system was evaluated by quantifying drug content in the supernatant after centrifugation. Supernatants were diluted with isopropanol (Fisher Scientific, Breda, Netherlands) and analyzed using a LAMBDA 650 UV/Vis Spectrophotometer (Perkin-Elmer, Shelton, CT, USA) at 276 nm. Absorbance values from isopropanol-diluted blank formulations were subtracted, and CBD concentrations were calculated using a standard calibration curve. The calibration curve was prepared by dissolving 50 mg of CBD in 10 mL of isopropanol to create a stock solution, followed by serial dilutions (25–275 µL in 5 mL of isopropanol). Each dilution was analyzed in triplicate.
For comparison, CBD solubility was also determined in individual bulk components, including various oils and distilled water. Excess, undissolved CBD was removed by centrifugation at 6000 rpm for 20 min. Visual inspection confirmed that centrifugation did not affect the physical integrity of the formulations.
2.8. Calculation of Theoretical CBD Solubility in NE Systems
To estimate the theoretical solubilization capacity of CBD in NE formulations, a weighted average model was applied based on the experimentally determined solubility of CBD in each individual component: Brij
® O10 and olive oil [
40]. The theoretical solubility (
Scalc) was calculated using the following equation:
where
foil and
fsurf represent the weight fractions of olive oil and Brij
® O10 in the formulation, and
Soil and
Ssurf are the experimentally determined CBD solubilities in olive oil (27.29% ± 0.01%
w/
v) and Brij
® O10 (0.38 ± 0.09%
w/
v), respectively.
To evaluate whether the mixed oil–surfactant system provides enhanced solubilization beyond ideal additive behavior, we further calculated the synergistic solubilization percentage (ΔS%), adapted from established approaches used to assess solubilization synergy in mixed surfactant and binary solvent systems [
41,
42].
where
is the experimentally measured CBD solubility in each NE formulation. A positive ΔS% indicates non-ideal, synergistic solubilization in the Brij
® O10–olive oil environment.
2.9. Human Ex Vivo Skin Model Preparation
Full-thickness human abdominal skin was obtained from a 35-year-old female donor undergoing elective abdominoplasty, with written informed consent and ethical approval from the Institutional Ethics Committee of Yanhee International Hospital, Thailand (Protocol No. FM-YGH-024; approved 8 June 2023). All procedures complied with the ethical principles outlined in the Declaration of Helsinki.
Subcutaneous fat was carefully removed, and the skin was sequentially rinsed in sterile saline (Thai Nakorn Patana Co., Ltd., Nonthaburi, Thailand) containing Amphotericin B (#A9528, Sigma-Aldrich), 70% ethanol (PURE SERV Co., Ltd., Bangkok, Thailand), and PBS containing Amphotericin B. Full-thickness 6 mm punch biopsies were placed onto 24-well transwell inserts coated with a collagen-Matrigel
® matrix (Corning, Tewksbury, MA, USA). Skin explants were incubated overnight in Dulbecco’s Modified Eagle Medium (DMEM; Gibco, Waltham, MA, USA) supplemented with 10% fetal bovine serum (FBS; Gibco, MA, USA) GlutaMAX™ (Gibco, MA, USA), insulin (Millipore Sigma, Burlington, MA, USA), and Primocin (InvivoGen, San Diego, CA, USA) to allow tissue recovery. This protocol was adapted from previously established methods, with minor modifications [
8].
2.10. Anti-Pollution Efficacy Testing Using a Human Ex Vivo Skin Model
Pollution-induced skin damage was modeled by simultaneous topical application of particulate matter (PM) and systemic delivery of NEs with or without CBD. Urban Dust Standard Reference Material 1649b (Sigma-Aldrich, MO, USA) was applied topically at 0.54 mg/cm2 (10 µL), while CBD-loaded NE were added to the culture medium at 0.05% and 0.1% final concentrations. Blank NEs (CBD-free) served as vehicle controls.
L-ascorbic acid (250 µM, Sigma-Aldrich, MO, USA) was used as a positive control, and distilled water served as the vehicle control. Additional control groups were included to assess PM-specific effects: (i) a water–non-PM, consisting of explants treated with distilled water without PM exposure, and (ii) a water–PM, exposed to PM and treated with water only [
8].
Skin explants were incubated for 48 h, with daily renewal of the culture medium and test formulations. Following treatment, the medium was replaced with serum-free fibroblast medium (ATCC, Manassas, VA, USA), and the explants were incubated for an additional 7 h. Conditioned media were collected for subsequent biomarker analysis.
2.11. Lactate Dehydrogenase (LDH) Assay
Cytotoxicity was assessed by quantifying LDH release using the LDH Cytotoxicity Detection Kit (#CK12-05, Dojindo, Kumamoto, Japan). Absorbance was measured using a SpectraMax iD5 microplate reader (Molecular Devices, San Jose, CA, USA). LDH levels were normalized to total protein content, determined using the Pierce™ BCA Protein Assay Kit (#23225, Thermo Fisher Scientific, Waltham, MA, USA), and results are expressed as a percentage relative to the water–non-PM group. Each condition was tested in duplicate using three independent skin explants.
2.12. Quantification of IL-6, MMP-1, and PIP
Conditioned media were analyzed for IL-6 and MMP-1 using AlphaLISA® kits (#AL242C and #AL353C, Revvity, Waltham, MA, USA), with luminescence read using an EnVision-Alpha Reader (Revvity, Waltham, MA, USA). PIP was quantified using an ELISA kit (#ab272198, Abcam, Cambridge, MA, USA), with absorbance measured using the SpectraMax iD5 reader. All values were normalized to total protein content and expressed as a percentage relative to the water–non-PM control. Each assay was performed in duplicate using conditioned media from three independent skin explants per treatment group.
2.13. Statistical Analysis
Statistical analyses were performed using GraphPad Prism version 7 (GraphPad Software, San Diego, CA, USA). All results are reported as mean ± standard deviation (SD).
For LDH assays, one-way analysis of variance (ANOVA) followed by Dunnett’s post hoc test was used to compare all treatment groups against the water–non-PM control. To evaluate the direct impact of PM exposure, an unpaired t-test was performed between the water–non-PM and water–PM groups. Within the PM-exposed group, selected pairwise comparisons among CBD-loaded NEs, blank NEs, water, and ascorbic acid were analyzed using one-way ANOVA followed by Bonferroni post hoc tests. A p-value < 0.05 was considered statistically significant after correction.
3. Results
3.1. Nanoemulsion Formation and Stability
Olive oil (OL) was selected as the oil phase due to its high molar volume and previously reported compatibility with non-ionic surfactants such as Brij
® O10 for forming broad NE regions [
28,
29,
31,
33,
43]. A representative O/W NE formulated with Brij
® O10 and OL remained clear, flowable, and physically stable after 30 days of storage at room temperature, exhibiting no visible sign of turbidity, phase separation, or creaming (
Figure 1a).
To define the compositional range supporting stable NE formation, a partial ternary phase diagram was constructed by varying the weight ratios of OL, Brij
® O10, and water (
Figure 1b). Only formulations that remained clear, monophasic, and non-birefringent after 30 days were classified as stable. The stable NE region was identified within compositions containing 5–30%
w/
w Brij
® O10, with optimal stability and clarity observed near 25%
w/
w surfactant. At Brij
® O10 concentration exceeding 30%
w/
w, the formulation developed increased viscosity and gel-like consistency, impairing flowability and ease of handling. Based on these observations, the formulation containing 20%
w/
w Brij
® O10 (designated B20) was selected for further evaluations, as it resided within the stable NE region while maintaining desirable physical characteristics.
3.2. CBD Solubility in Water, Olive Oil (OL), and 20% w/w Brij® O10 (B20)
CBD, a highly lipophilic compound with limited aqueous solubility, was selected as a model active to evaluate the solubilization performance of the NE system. To assess the individual contribution of each component, CBD solubility was first quantified in water, olive oil (OL), and 20% Brij® O10 micelles (B20).
As shown in
Table 1, CBD exhibited negligible solubility in water (0.001%
w/
v), as reported previously [
44], consistent with its high hydrophobicity and poor aqueous compatibility. In contrast, OL demonstrated the highest solubilization capacity (27.29 ± 0.01%
w/
v), highlighting CBD’s strong affinity for lipid-rich environments. The micellar solution (20%
w/
w Brij
® O10) enabled moderate solubilization (0.38 ± 0.09%
w/
v), likely due to incorporation of CBD into the hydrophobic cores of micelles. These findings informed the NE formulation strategy, suggesting that combining oil and surfactant phases may synergistically enhance CBD solubilization and facilitate the development of stable NE carriers.
3.3. Phase Inversion Temperature Behavior of CBD-Free and CBD-Loaded Nanoemulsions
The phase inversion temperature (PIT) of NEs formulated with 20%
w/
w Brij
® O10 (B20) and varying concentrations of olive oil (OL) (denoted as B20-OLx) was evaluated to investigate the thermal behavior of blank (CBD-free) systems. As shown in
Figure 2, the PIT initially decreased with the addition of low OL concentrations (<2%
w/
w), followed by a gradual increase as OL content was further elevated. This U-shaped trend likely reflects changes in the hydrophilic–lipophilic balance (HLB) of the system: at low oil level, micellar organization is disrupted, whereas higher oil concentrations stabilize the interfacial layer and facilitate emulsion formation.
To assess the effect of CBD incorporation on PIT behavior, equivalent CBD-loadedformulations (B20-OLx-CBD) were tested. However, all CBD-containing NEs with added oil exhibited turbidity or phase separation during heating, precluding accurate PIT determination. The only measurable PIT value was obtained from the CBD-loaded micellar formulation (B20-CBD, 0% OL), which showed a significantly reduced PIT (~35 °C) compared to its blank counterpart (~67 °C) (
Figure 2). This marked reduction suggests that CBD perturbs micellar architecture, possibly by inserting into both hydrophobic and hydrophilic domains of the surfactant assembly. These findings indicate that CBD disrupts the thermal stability of micelles and NEs—particularly in the presence of oil—posing formulation challenges for the system requiring thermal robustness during processing or storage.
3.4. Particle Size and Morphology of CBD-Free and CBD-Loaded Nanoemulsions
The apparent hydrodynamic diameter of NE droplets was measured by photon correlation spectroscopy (PCS) following appropriate dilution. As shown in
Figure 3a, the CBD-free micellar formulations (B20) exhibited an average particle size of approximately 16 nm. Upon initial addition of olive oil (OL), droplet size decreased, reaching a minimum of ~10 nm at 1%
w/
w OL (B20-OL1). As the OL concentration increased beyond 1%, droplet size gradually increased, reaching ~16.2 nm at 8%
w/
w OL (B20-OL8). All CBD-free formulations maintained particle sizes below 25 nm, consistent with their clear and flowable appearance.
In contrast, CBD-loaded formulations (B20-OLx-CBD) displayed a nonlinear particle size trend with varying OL concentrations. At 1% OL, particle size markedly increased (~105 nm), likely due to micelle swelling or partial destabilization as CBD partitioned between the surfactant core and limited oil phase. Droplet size then sharply decreased at 4% OL (~17 nm), suggesting efficient droplet formation and optimal encapsulation of CBD. However, further increasing the OL content resulted in progressively larger droplets (up to ~90 nm at 8% OL), possibly reflecting insufficient surfactant coverage, leading to coalescence or formation of loosely packed aggregates (
Figure 3a). These results highlight the critical role of oil-to-surfactant ratio in maintaining colloidal stability and controlling droplet size in CBD-loaded NEs.
The size and morphology of the NEs were further examined by TEM. Representative images for selected formulations (0, 1, 4, and 8%
w/
w OL, with and without CBD) are shown in
Figure 3b, and the complete set is provided in
Figure S1. In these images, CBD-free NEs appear as relatively homogeneous dispersions of small, roughly spherical droplets at all OL contents, consistent with the nearly constant hydrodynamic diameters below 25 nm measured by PCS. In the CBD-loaded series, pronounced clustering was most evident at 1% OL, consistent with the highest PCS hydrodynamic diameter. At 8% OL, TEM showed larger, more discrete nanoscale features, in line with the increased hydrodynamic diameter measured by PCS. At 4% OL, TEM still revealed some clustered features, but these were generally less prominent than at 1% OL, consistent with the minimum hydrodynamic diameter observed by PCS. Because TEM specimens were prepared by deposition and drying on grids, the extent of clustering can be accentuated relative to the hydrated dispersion [
46]; therefore, TEM was used qualitatively to assess morphology and support the PCS size trends rather than as a primary quantitative sizing method.
3.5. Zeta Potential of CBD-Free and CBD-Loaded Nanoemulsions
The zeta potential of B20-OLx NEs with and without CBD was measured at all investigated olive oil (OL) concentrations (0–8%
w/
w). The mean values ranged between approximately −10 to +3 mV (
Figure 4). For each OL concentration, CBD-loaded formulations (B20-OLx-CBD) showed zeta potentials that were comparable to their corresponding CBD-free NEs (B20-OLx), and no systematic change in zeta potential was observed upon CBD incorporation.
3.6. Visual Appearance of CBD-Free and CBD-Loaded Nanoemulsions
The physical appearance of NE formulations was assessed to evaluate the impact of CBD incorporation on optical clarity and dispersion stability (
Table 2). Micellar formulations composed of 20%
w/
w Brij
® O10 without olive oil (B20) remained optically clear after CBD addition, indicating that CBD was effectively solubilized without disrupting micellar integrity or inducing turbidity.
In contrast, formulations containing both Brij
® O10 and increasing concentrations of olive oil (OL) exhibited progressive changes in appearance upon CBD loading. While all CBD-free NEs (B20-OLx) remained uniformly clear across the tested oil concentrations (1–8%
w/
w), the corresponding CBD-loaded NEs (B20-OLx-CBD) showed increased turbidity with higher OL content. Specifically, B20-OL1-CBD and B20-OL2-CBD appeared translucent, B20-OL4-CBD showed a bluish tint, and B20-OL6-CBD and B20-OL8-CBD became noticeably cloudy (
Table 2).
Despite these visual changes, no phase separation, flocculation, or precipitation was observed in any CBD-loaded formulations throughout the study period, suggesting maintained colloidal stability. The increased turbidity likely resulted from light scattering by larger droplets or structural rearrangement induced by CBD partitioning into the oil phase—consistent with the particle size trends shown in
Figure 3.
3.7. CBD Solubilization in Micelles and Nanoemulsions
The solubilization capacity of micelles (B20) and NEs composed of 20%
w/
w Brij
® O10 with varying concentrations of olive oil (OL) was quantitatively evaluated (
Table 3). In the micellar system (B20), CBD solubility was limited to 0.38 ± 0.09%
w/
v, reflecting the modest capacity of non-ionic surfactant micelles to accommodate highly lipophilic compounds. Incorporation of OL substantially enhanced CBD solubilization across all formulations, with solubility increasing from 1.25 ± 0.53%
w/
v at 1% OL (B20-OL1) to 4.05 ± 1.02%
w/
v at 8% OL (B20-OL8) (
Table 3). The progressive enhancement confirms a synergistic effect between oil and surfactant phase in solubilizing CBD.
To determine whether this enhancement was merely additive or truly synergistic, theoretical solubility values were calculated based on the individual solubilities of CBD in Brij
® O10 micelles (0.38 ± 0.09%
w/
v) and olive oil (27.29 ± 0.01%
w/
v), weighted by their respective proportions in each formulation. As shown in
Table 3, the experimentally measured solubilities in all NE formulations significantly exceeded their theoretical counterparts, indicating that the structure of the NE environment facilitates solubilization beyond simple physical mixing.
To further quantify this non-ideal enhancement, the synergistic solubilization percentage (ΔS%) was calculated based on the deviation of experimental solubility () from its theoretical additive value (). All NEs showed positive ΔS% values (+79% to +257%), confirming that the Brij® O10–olive oil system creates a cooperative solubilization environment that enhances CBD partitioning into both the droplet core and interfacial regions. Although B20-OL1 displayed the highest ΔS%, this arises from its very low theoretical solubility at 1% oil—meaning even modest experimental solubility produces a large relative synergy. Thus, the high ΔS% reflects strong non-ideal behavior rather than superior absolute CBD solubilization.
3.8. Tissue Viability of CBD-Loaded Nanoemulsions in a PM- Exposed Ex Vivo Skin Model
CBD-loaded NEs, B20-OL4-CBD and B20-OL8-CBD, were selected for tissue viability evaluation based on their overall physicochemical performance, including nanoscale droplet size, colloidal stability, and high absolute CBD solubilization. Although B20-OL1-CBD exhibited the highest ΔS% value, this reflected a low theoretical solubility at 1% oil rather than superior solubilization. In practice, B20-OL1-CBD showed larger and more variable droplet sizes and reduced structural robustness upon CBD incorporation, making it unsuitable for biological testing. By contrast, B20-OL4-CBD combined optimal droplet size with good optical clarity, while B20-OL8-CBD provided the highest absolute CBD solubilization despite its turbidity. These two formulations were therefore selected as representative NE systems for subsequent ex vivo evaluation.
Cytocompatibility was assessed using LDH release as an indicator of membrane integrity in a full-thickness ex vivo human skin model exposed to PM. As shown in
Figure 5, treatment with blank NEs at 0.1% (B20-OL4 and B20-OL8) led to significantly elevated LDH release compared to the non-PM control, suggesting mild cytotoxicity effects potentially due to surfactant-related membrane disruption. In contrast, CBD-loaded counterparts at the same concentration (B20-OL4-CBD and B20-OL8-CBD) did not increase LDH release, suggesting a protective effect of CBD against PM-induced tissue damage.
At the lower concentration of 0.05%, all tested formulations—both blank and CBD-loaded—exhibited LDH levels comparable to the non-PM water control, indicating good tissue compatibility at this dose. Based on these results, 0.05% B20-OL4-CBD and B20-OL8-CBD were selected for subsequent anti-pollution efficacy studies, focusing on biomarkers of inflammation and skin integrity.
3.9. Modulation of Inflammatory and Extracellular Matrix Markers by CBD-Loaded Nanoemulsions in PM-Exposed Ex Vivo Skin Model
To investigate the anti-inflammatory and extracellular matrix (ECM)-preserving effects of CBD-loaded NEs, levels of IL-6, MMP-1, and PIP were quantified in the culture medium of PM-exposed human skin explants. As shown in
Figure 6a, PM exposure induced a strong inflammatory response, as evidenced by a ~3-fold increase in IL-6 secretion relative to the water–non-PM control. Ascorbic acid treatment reduced IL-6 levels by 24% compared to the water–PM group. All NE formulations—both CBD-free and CBD-loaded—significantly decreased IL-6 secretion by 45–70% relative to the water–PM control. Notably, CBD-loaded NEs (B20-OL4-CBD and B20-OL8-CBD) further reduced IL-6 expression by 30% and 46%, respectively, compared to their corresponding blank NEs, indicating an additive anti-inflammatory effect from CBD incorporation.
MMP-1 secretion, associated with ECM degradation, was elevated ~2-fold upon PM exposure (
Figure 6b). All NE formulations, regardless of CBD content, substantially suppressed MMP-1 levels by over 90% compared to the water–PM control. Among them, B20-OL8-CBD showed the greatest reduction—55% lower than its blank—suggesting superior protective capacity. Although B20-OL4-CBD showed a downward trend, the reduction did not reach statistical significance.
Pollution exposure resulted in a 15% decrease in PIP, a marker of collagen synthesis (
Figure 6c). Treatment with CBD-loaded NEs effectively reversed this decline. B20-OL4-CBD increased PIP expression by ~70%, and B20-OL8-CBD by ~41%, relative to their respective blanks. Both treatments restored PIP levels above those observed in the PM-exposed water control, indicating that CBD supports collagen maintenance under environmental stress.
Together, these results support the hypothesis that CBD-loaded NEs—especially B20-OL4-CBD and B20-OL8-CBD—can mitigate pollution-induced inflammation and ECM degradation while promoting collagen synthesis, offering promise as functional colloidal systems for dermal protection.
4. Discussion
This study aimed to develop a cannabidiol (CBD)-loaded nanoemulsion (CBD-NE) system with optimal physicochemical properties for dermal delivery and to assess its anti-pollution efficacy using an ex vivo human skin model exposed to PM. The formulated oil-in-water (O/W) NEs, composed of Brij® O10 and olive oil and prepared by the PIT method, formed nanosized droplets with markedly enhanced and synergistic CBD solubilization, a reduced PIT, and nonlinear droplet size behavior, moderately low zeta potentials, and TEM-confirmed colloidal structures that remained physically stable over 30 days. In the PM-challenged skin model, these CBD-NEs significantly mitigated inflammatory responses, reduced matrix degradation, and restored extracellular matrix (ECM) integrity following pollutant exposure.
The choice of olive oil—a long-chain triglyceride with high molecular volume—played a key role in shaping the internal NE structure and formation behavior [
47,
48]. Due to its low compatibility with the hydrophobic regions of surfactant micelles, olive oil is expected to localize primarily in the oil core of NE droplets [
47]. This core is stabilized by the oleyl chains of Brij
® O10, while the poly(ethylene oxide) segments form a hydrated shell, yielding a core–shell structure. This arrangement is consistent with previous reports on O/W NEs prepared from high molecular weight oils and nonionic surfactants with a C18:1E10 architecture [
28,
29,
32,
33,
43]. Ternary phase mapping (
Figure 1b) identified a stable NE region within 5–30%
w/
w Brij
® O10. A 20%
w/
w surfactant concentration (B20) was selected as optimal, balancing droplet stability with formulation processability, and avoiding the high viscosity and gelation seen at elevated surfactant levels.
Figure 2 and
Figure 3 and
Table 2 illustrate the effects of olive oil and CBD incorporation on the phase inversion temperature (PIT), particle size, morphology, and visual clarity of the NE formulations. In CBD-free systems, a U-shaped PIT trend was observed with increasing olive oil content: initial decreases in PIT at low oil levels likely resulted from oil incorporation into the surfactant monolayer, while subsequent increases at higher concentrations reflected a structural shift toward spherical, oil-core droplets (
Figure 2). These PIT changes were mirrored by corresponding alterations in hydrodynamic diameter, supporting a transition from elongated or asymmetric micelles to more thermodynamically stable droplets (
Figure 3a). This behavior is consistent with micelle-to-droplet transformations previously reported for nonionic and ionic surfactants [
32,
49].
CBD incorporation markedly disrupted the structural behavior of the NEs, as evidenced by a sharp reduction in PIT—from ~67 °C in blank formulations to ~35 °C—and a significant increase in particle size (
Figure 2 and
Figure 3). These changes were accompanied by turbidity and thermal instability, preventing reliable PIT determination in CBD-loaded systems. The nonlinear particle size profile of CBD-loaded NEs—an increase at 1% OL, a minimum at 4% OL, and a subsequent increase at higher oil levels (
Figure 3a)—is consistent with colloidal restructuring when amphiphilic actives partition between the micellar palisade layer and the oil core. Similar behavior has been reported for α-tocopherol and curcumin in Brij surfactants, where incorporation of these molecules induces micelle swelling, interfacial packing disruption, and transitions toward swollen droplets or microemulsion-like structures [
50]. In our system, the markedly enlarged droplets at low oil levels (e.g., ~105 nm in B20-OL1-CBD) can be attributed to CBD-induced micelle swelling under conditions of limited oil content, whereas the reduced size at 4% OL (~17 nm in B20-OL4-CBD) suggests an optimal oil–surfactant balance for CBD accommodation. At higher oil levels (B20-OL6-CBD and B20-OL8-CBD), droplet sizes increase again, consistent with insufficient surfactant coverage and the formation of more loosely packed structures. TEM images (
Figure 3b and
Figure S1) qualitatively support these trends. Pronounced clustering is most evident at 1% OL, whereas at 8% OL, larger, more discrete features are observed. At 4% OL, clustered features are still present but are generally less prominent than at 1% OL. Because TEM specimens were prepared by deposition and drying on grids, TEM was used qualitatively to assess morphology and complement the PCS measurements rather than as a primary quantitative sizing method [
46].
In addition to PIT and droplet size, interfacial and macroscopic properties further supported these structural transitions. All formulations showed moderately low zeta potential values in the range of approximately −10 to +3 mV, and CBD loading did not markedly alter these values (
Figure 4). The limited magnitude of the zeta potential is consistent with the nonionic nature of Bri
j® O10, for which steric stabilization by hydrated poly(ethylene oxide) chains is expected to dominate over electrostatic repulsion [
33]. The near-neutral to slightly negative values are also in line with previous observations for PEG-based formulations containing Brij L9, which reported zeta potentials of approximately −10 mV and −5 mV immediately and 4 h after preparation, respectively, with values around −10 mV commonly seen for PEG-based systems [
51]. Visual inspection over 30 days (
Table 2) was consistent with these colloidal characteristics. CBD-free NEs remained optically clear, whereas CBD-loaded formulations showed increasing turbidity and bluish hues at higher oil contents, consistent with light scattering from larger, perturbed droplets. Nevertheless, all formulations remained physically stable without phase separation throughout the 30-day study, confirming their short-term storage suitability (
Table 2). Together,
Figure 2 and
Figure 4 and
Table 2 therefore provide a coherent picture of CBD-induced restructuring of the NEs without loss of macroscopic stability.
Solubility profiling of CBD across different media revealed key insights into its formulation behavior (
Table 1 and
Table 3). CBD exhibited negligible solubility in water but showed high solubility in olive oil and moderate solubility in Brij
® O10 micelles, consistent with its lipophilic character. When incorporated into Brij
® O10–olive oil NEs, the experimentally measured CBD solubility consistently exceeded values predicted from simple additive models based on individual component solubilities (
Table 3), indicating non-ideal interactions within the mixed system. To quantify this deviation, the synergistic solubilization percentage (ΔS%) was calculated. All NEs displayed positive ΔS% values—approximately +79% to +257%—demonstrating cooperative, non-ideal solubilization behavior between the oil and surfactant phases (
Table 3). The highest ΔS% occurred in B20-OL1, consistent with reports for mixed micelle and mixed solvent systems where small additions of a hydrophobic co-component can induce disproportionately large structural reorganization and amplified solubilization efficiency. Importantly, this high ΔS% reflects strong relative synergy rather than superior absolute CBD loading, because B20-OL1 has a very low theoretical solubility at 1% oil. For this reason, despite its strong synergistic signature, B20-OL1 was not selected for biological evaluation. Across all formulations, the positive ΔS% values indicate that the NE environment provides a more efficient solubilization domain than either component alone. The improved performance is likely driven by structural features of the NEs—such as enlarged interfacial area, dynamic surfactant layers, and enhanced droplet core accessibility—that collectively promote more efficient CBD partitioning [
52,
53]. These findings support the design of O/W NEs as effective platforms for solubilizing poorly water-soluble actives and highlight the importance of optimizing both oil content and surfactant architecture to maximize solubilization capacity.
Based on their favorable physicochemical profiles—including high CBD solubility, nanoscale droplet size, and good physical stability—B20-OL4-CBD and B20-OL8-CBD were selected for biological evaluation. Cytotoxicity was first assessed using an LDH release assay in ex vivo human skin. At 0.05% CBD concentration, both formulations exhibited excellent tissue compatibility, with LDH levels comparable to untreated controls, supporting the use of 0.05% as the optimal dose for further testing (
Figure 5). In the PM2.5-exposed skin model, treatment with 0.05% B20-OL4-CBD and B20-OL8-CBD significantly attenuated PM-induced upregulation of IL-6 and MMP-1 while restoring expression of PIP, a collagen synthesis marker, compared to water–PM control (
Figure 6a–c). These effects were more pronounced than those observed with blank NEs, which conferred partial protection likely due to antioxidant constituents of olive oil [
34,
35]. Compared to its corresponding blank, B20-OL8-CBD reduced IL-6 and MMP-1 by 46% and 55%, respectively, and increased PIP by 41%, underscoring the synergistic anti-inflammatory and ECM-restorative activity of CBD in combination with the NE delivery system.
Overall, this study emphasizes the importance of oil–surfactant ratios, PIT-driven formulation strategies, and molecular interactions in designing functional NE delivery systems. Although direct encapsulation efficiency was not quantified, the observed physicochemical and biological outcomes strongly suggest successful CBD incorporation. Moreover, administering CBD via the culture medium offers a preliminary model for systemic or nutricosmetic delivery, simulating circulation-based skin exposure [
8,
54,
55].