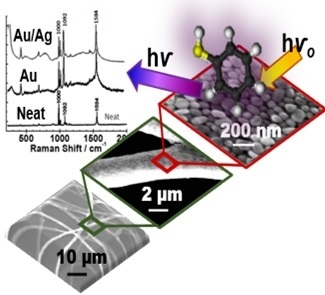
Nanostructured Assemblies of Gold and Silver Nanoparticles for Plasmon Enhanced Spectroscopy Using Living Biotemplates
Abstract
:1. Introduction
2. Materials and Methods
2.1. Synthesis
2.2. Formation of the Microtubules
2.3. Characterization
2.4. Surface-Enhanced Raman Spectroscopy (SERS)
3. Results
4. Discussion
5. Conclusions
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Maier, S.A. Plasmonics: Fundamentals and Applications; Springer: New York, NY, USA, 2007; ISBN 978-0-387-37825-1. [Google Scholar]
- Aroca, R. Surface-Enhanced Vibrational Spectroscopy; John Wiley & Sons: Chichester, UK, 2006; ISBN 978-0-471-60731-1. [Google Scholar]
- Le Ru, E.; Etchegoin, P.G. Principles of Surface Enhanced Raman Spectroscopy; Elsevier: Amsterdam, The Netherlands, 2009; ISBN 978-0-444-52779-0. [Google Scholar]
- Kawata, S. Plasmonics for nanoimaging and nanospectroscopy. Appl. Spectrosc. 2013, 67, 117–125. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Plummer, E.W.; Kempa, K. Foundations of plasmonics. Adv. Phys. 2011, 60, 799–898. [Google Scholar] [CrossRef]
- Ali, M.R.K.; Snyder, B.; El-Sayed, M.A. Synthesis and optical properties of small Au nanorods using seedless growth technique. Langmuir 2012, 28, 9807–9815. [Google Scholar] [CrossRef] [PubMed]
- Daniel, M.C.; Astruc, D. Gold nanoparticles: Assembly, supramolecular chemistry, quantum-size-related properties, and applications toward biology, catalysis, and nanotechnology. Chem. Rev. 2004, 104, 293–346. [Google Scholar] [CrossRef] [PubMed]
- Dreaden, E.C.; Alkilany, A.M.; Huang, X.; Murphy, C.J.; El-Sayed, M.A. The golden age: Gold nanoparticles for biomedicine. Chem. Soc. Rev. 2012, 41, 2740–2779. [Google Scholar] [CrossRef] [PubMed]
- Monteiro, D.R.; Gorup, L.F.; Silva, S.; Negri, M.; Camargo, E.R.; Oliveira, R.; Barbosa, D.B.; Henriques, M. Silver colloidal nanoparticles: Antifungal effect against adhered cells and biofilms of Candida albicans and Candida glabrata. Biofouling 2011, 27, 711–719. [Google Scholar] [CrossRef] [PubMed]
- Sharma, V.K.; Yngard, R.A.; Lin, Y. Silver nanoparticles: Green synthesis and their antimicrobial activities. Adv. Colloid Interface 2009, 145, 83–96. [Google Scholar] [CrossRef] [PubMed]
- Palanco, M.S.; Mogensen, K.B.; Guhlke, M.; Heiner, Z.; Kneipp, J.; Kneipp, K. Templated green synthesis of plasmonic silver nanoparticles in onion epidermal cells suitable for surface-enhanced Raman and hyper-Raman scattering. Beilstein J. Nanotechnol. 2016, 7, 834–840. [Google Scholar] [CrossRef] [PubMed]
- Jones, M.R.; Osberg, K.D.; Macfarlane, R.J.; Langille, M.R.; Mirkin, C.A. Templated techniques for the synthesis and assembly of plasmonic nanostrucutures. Chem. Rev. 2011, 111, 3736–3827. [Google Scholar] [CrossRef] [PubMed]
- Zhang, W.; Zhang, Z.P.; Zhang, X.E.; Li, F. Reaction inside a viral protein nanocage: Mineralization on a nanoparticle seed after encapsulation via self-assembly. Nano Res. 2017, 10, 3285–3294. [Google Scholar] [CrossRef]
- Maye, M.M.; Kumara, M.T.; Nykypanchuk, D.; Sherman, W.B.; Gang, O. Switching binary states of nanoparticle superlattices and dimer clusters by DNA strands. Nat. Nanotechnol. 2010, 5, 116–120. [Google Scholar] [CrossRef] [PubMed]
- Li, F.; Wang, Q. Fabrication of nanoarchitectures templated by virus-based nanoparticles: Strategies and applications. Small 2014, 10, 230–245. [Google Scholar] [CrossRef] [PubMed]
- Blum, A.S.; Soto, C.M.; Wilson, C.D.; Cole, J.D.; Kim, M.; Gnade, A.; Ochoa, W.F.; Lin, T.; Johnson, J.E.; Ratna, B.R. Cowpea mosaic virus as a scaffold for 3-D patterning of gold nanoparticles. Nano Lett. 2004, 4, 867–870. [Google Scholar] [CrossRef]
- Jain, J.K.; Huang, X.; El-Sayed, I.H.; El-Sayed, M.A. Review of some interesting surface plasmon resonance-enhanced properties of noble metal nanoparticles and their applications to biosystems. Plasmonics 2007, 2, 107–118. [Google Scholar] [CrossRef]
- Manocchi, A.K.; Seifert, S.; Lee, B.; Yi, H. On the thermal stability of surface-assembled viral-metal nanoparticle complexes. Langmuir 2010, 26, 7516–7522. [Google Scholar] [CrossRef] [PubMed]
- Moskovits, M. Surface-enhanced spectroscopy. Rev. Mod. Phys. 1985, 57, 783–826. [Google Scholar] [CrossRef]
- Kleinman, S.L.; Frontiera, R.R.; Henry, A.I.; Dieringer, J.A.; Van Duyne, R.P. Creating, characterizing, and controlling chemistry with SERS hot spots. Phys. Chem. Chem. Phys. 1985, 15, 21–36. [Google Scholar] [CrossRef] [PubMed]
- Rosi, N.L.; Thaxton, C.S.; Mirkin, C.A. Control of nanoparticle assembly using DNA-modified diatom templates. Angew. Chem. Int. Ed. 2004, 43, 5500–5503. [Google Scholar] [CrossRef] [PubMed]
- Kubo, A.M.; Gorup, L.F.; Amaral, L.S.; Filho, E.R.; Camargo, E.R. Kinetic control of microtubule morphology obtained by assembling gold nanoparticles on living fungal biotemplates. Bioconjug. Chem. 2016, 27, 2337–2345. [Google Scholar] [CrossRef] [PubMed]
- Bigall, N.C.; Reitzig, M.; Naumann, W.; Simon, P.; van Pée, K.-H.; Eychmüller, A. Fungal templates for noble-metal nanoparticles and their application in catalysis. Angew. Chem. Int. Ed. 2008, 47, 7876–7879. [Google Scholar] [CrossRef] [PubMed]
- Scheibel, T.; Parthasarathy, R.; Sawicki, G.; Lin, X.M.; Jaeger, H.; Lindquist, S. Conducting nanowires built by controlled self-assembly of amyloid fibers and selective metal deposition. Proc. Natl. Acad. Sci. USA 2010, 100, 4527–4532. [Google Scholar] [CrossRef] [PubMed]
- Zhang, T.; Wang, W.; Zhang, D.; Zhang, X.; Ma, Y.; Zhou, Y.; Qi, L. Biotemplated synthesis of gold nanoparticle–bacteria cellulose nanofiber nanocomposites and their application in biosensing. Adv. Funct. Mater. 2010, 20, 1152–1160. [Google Scholar] [CrossRef]
- Li, W.; Zhao, X.; Glushenkov, A.M.; Kong, L. Plasmonic substrates for surface enhanced Raman scattering. Anal. Chim. Acta 2017, 984, 19–41. [Google Scholar] [CrossRef] [PubMed]
- Barbillon, G.; Sandana, V.E.; Humbert, C.; Belier, B.; Rogers, D.J.; Teherani, F.H.; Bove, P.; McClintock, R.; Razeghi, M. Study of Au coated ZnO nanoarrays for surface enhanced Raman scattering chemical sensing. J. Mater. Chem. C 2017, 5, 3528–3535. [Google Scholar] [CrossRef]
- Reguera, J.; Langer, J.; Aberasturi, D.J.; Liz-Marzan, L.M. Anisotropic metal nanoparticles for surface enhanced Raman scattering. Chem. Soc. Rev. 2017, 46, 3866–3885. [Google Scholar] [CrossRef] [PubMed]
- Bryche, J.F.; Tsigara, A.; Belier, B.; Chapelle, M.L.; Canva, M.; Bartenlian, B.; Barbillon, G. Surface enhanced Raman scattering improvement of gold triangular nanoprisms by a gold reflective underlayer for chemical sensing. Sens. Actuator B Chem. 2016, 228, 31–35. [Google Scholar] [CrossRef]
- Carrasco, S.; Benito-Pena, E.; Navarro-Villoslada, F.; Langer, J.; Sanz-Ortiz, M.N.; Reguera, J.; Liz-Marzan, L.M.; Moreno-Bondi, M.C. Multibranched gold-mesoporous silica nanoparticles coated with a molecularly imprinted polymer for label-free antibiotic surface-enhanced Raman scattering analysis. Chem. Mater. 2016, 28, 7947–7954. [Google Scholar] [CrossRef]
- Stiles, P.L.; Dieringer, J.A.; Shah, N.C.; Van Duyne, R.P. Surface-enhanced Raman Spectroscopy. Annu. Rev. Anal. Chem. 2008, 1, 601–626. [Google Scholar] [CrossRef] [PubMed]
- Issaad, D.; Moustaoui, H.; Medjahed, A.; Lalaoui, L.; Spadavecchia, J.; Bouafia, M.; Chapelle, M.L.; Djaker, N. Scattering correlation spectroscopy and Raman spectroscopy of thiophenol on gold nanoparticles: Comparative study between nanospheres and nanourchins. J. Phys. Chem. C 2017, 121, 18254–18262. [Google Scholar] [CrossRef]
- Biggs, K.; Camden, J.P.; Anker, J.N.; Van Duyne, R.P. Surface-enhanced Raman spectroscopy of benzenothiol adsorbed from the gas phase onto silver film over nanosphere surfaces: Determination of the sticking probability and detection limit time. J. Phys. Chem. A 2009, 113, 4581–4586. [Google Scholar] [CrossRef] [PubMed]
- Lee, P.C.; Meisel, D. Adsorption and surface-enhanced Raman of dyes on silver and gold sols. J. Phys. Chem. 1982, 86, 3391–3395. [Google Scholar] [CrossRef]
- Zhang, S. Fabrication of novel biomaterials through molecular self-assembly. Nat. Biotech. 2003, 21, 1171–1178. [Google Scholar] [CrossRef] [PubMed]
- Sugunan, A.; Melin, P.; Schnürer, J.; Hilborn, J.G.; Dutta, J. Nutrition-driven assembly of colloidal nanoparticles: Growing fungi assemble gold nanoparticles as microwires. Adv. Mater. 2007, 19, 77–81. [Google Scholar] [CrossRef]
- Gorup, L.F.; Longo, E.; Leite, E.R.; Camargo, E.R. Moderating effect of ammonia on particle growth and stability of quasi-monodisperse silver nanoparticles synthesized by the Turkevich method. J. Colloid Interface Sci. 2011, 360, 355–358. [Google Scholar] [CrossRef] [PubMed]
- Moskovits, M. Persistent misconceptions regarding SERS. Phys. Chem. Chem. Phys. 2013, 15, 5301–5311. [Google Scholar] [CrossRef] [PubMed]
- Fan, M.; Andrade, G.F.S.; Brolo, A.G. A review on the fabrication of substrates for surface enhanced Raman spectroscopy and their applications in analytical chemistry. Anal. Chim. Acta 2011, 693, 7–25. [Google Scholar] [CrossRef] [PubMed]
- Gunnarsson, L.; Bjerneld, E.J.; Xu, H.; Petronis, S.; Kasemo, B.; Käll, M. Interparticle coupling effects in nanofabricated substrates for surface-enhanced Raman scattering. Appl. Phys. Lett. 2001, 78, 802–804. [Google Scholar] [CrossRef]
- Joo, T.H.; Kim, M.S.; Kim, K. Surface-enhanced Raman scattering of 2-butanethiol in silver sol. J. Raman Spectrosc. 1987, 160, 81–89. [Google Scholar] [CrossRef]
- Carron, K.T.; Hurley, L.G. Axial and azimuthal angle determination with surface-enhanced Raman spectroscopy: Thiophenol on copper, silver, and gold metal surfaces. J. Phys. Chem. 1991, 95, 9979–9984. [Google Scholar] [CrossRef]
- Xia, Y.; Xiong, Y.; Lim, B.; Skrabalak, S.E. Shape-controlled synthesis of metal nanocrystals: Simple chemistry meets complex physics? Angew. Chem. Int. Ed. 2009, 48, 60–103. [Google Scholar] [CrossRef] [PubMed]
- LaMer, V.K.; Dinegar, R.H. Theory, production and mechanism of formation of monodispersed hydrosols. J. Am. Chem. Soc. 1950, 72, 4847–4854. [Google Scholar] [CrossRef]
- Juárez, J.; Cambón, A.; Topete, A.; Taboada, P.; Mosquera, V. One-dimensional magnetic nanowires obtained by protein fibril biotemplating. Chem. Eur. J. 2011, 17, 7366–7373. [Google Scholar] [CrossRef] [PubMed]
- Niemeyer, C.M. Nanoparticles, proteins, and nucleic acids: Biotechnology meets materials science. Angew. Chem. Int. Ed. 2001, 40, 4128–4158. [Google Scholar] [CrossRef]
- Fakhrullin, R.F.; Zamaleeva, A.I.; Morozov, M.V.; Tazetdinova, D.I.; Alimova, F.K.; Hilmutdinov, A.K.; Zhdano, R.I.; Kahraman, M.; Culha, M. Living fungi cells encapsulated in polyelectrolyte shells doped with metal nanoparticles. Langmuir 2009, 25, 4628–4634. [Google Scholar] [CrossRef] [PubMed]
- Heynes, C.L.; Yonzon, C.R.; Zhang, X.; Van Duyne, R.P. Surface-enhanced Raman sensors: Early history and the development of sensors for quantitative biowarfare agent and glucose detector. J. Raman Spectrosc. 2005, 36, 471–484. [Google Scholar] [CrossRef]
- Dieringer, J.A.; McFarland, A.D.; Shah, N.C.; Stuart, D.A.; Whitney, A.V.; Yonzon, M.A.; Young, M.A.; Zhang, X.; Van Duyne, R.P. Surface enhanced Raman spectroscopy: New materials, concepts, characterization tools, and applications. Faraday Discuss. 2006, 132, 9–26. [Google Scholar] [CrossRef] [PubMed]
- Jana, N.R.; Gearheart, L.; Murphy, C.J. Seeding growth for size control of 5−40 nm diameter gold nanoparticles. Langmuir 2001, 17, 6782–6786. [Google Scholar] [CrossRef]
- Han, S.W.; Lee, S.J.; Kim, K. Self-assembled monolayers of aromatic thiol and selenol on silver: comparative study of adsorptivity and stability. Langmuir 2001, 17, 6981–6987. [Google Scholar] [CrossRef]
- Taylor, C.E.; Pemberton, J.E.; Goodman, G.G.; Schoenfisch, M.H. Surface enhancement factors for Ag and Au surfaces relative to Pt surfaces for monolayers of thiophenol. Appl. Spectrosc. 1999, 53, 1212–1221. [Google Scholar] [CrossRef]
- Fan, M.; Lai, F.J.; Chou, H.L.; Lu, W.T.; Hwang, B.J.; Brolo, A.G. Surface-enhanced Raman scattering (SERS) from Au:Ag bimetallic nanoparticles: The effect of the molecular probe. Chem. Sci. 2013, 4, 509–515. [Google Scholar] [CrossRef]
- Herrera, G.; Padilla, A.; Hernandez-Rivera, S. Surface enhanced Raman scattering (SERS) studies of gold and silver nanoparticles prepared by laser ablation. Nanomaterials 2013, 3, 158–172. [Google Scholar] [CrossRef] [PubMed]
- Holze, R. The adsorption of thiophenol on gold—A spectroelectrochemical study. Phys. Chem. Chem. Phys. 2015, 17, 21364–21372. [Google Scholar] [CrossRef] [PubMed]



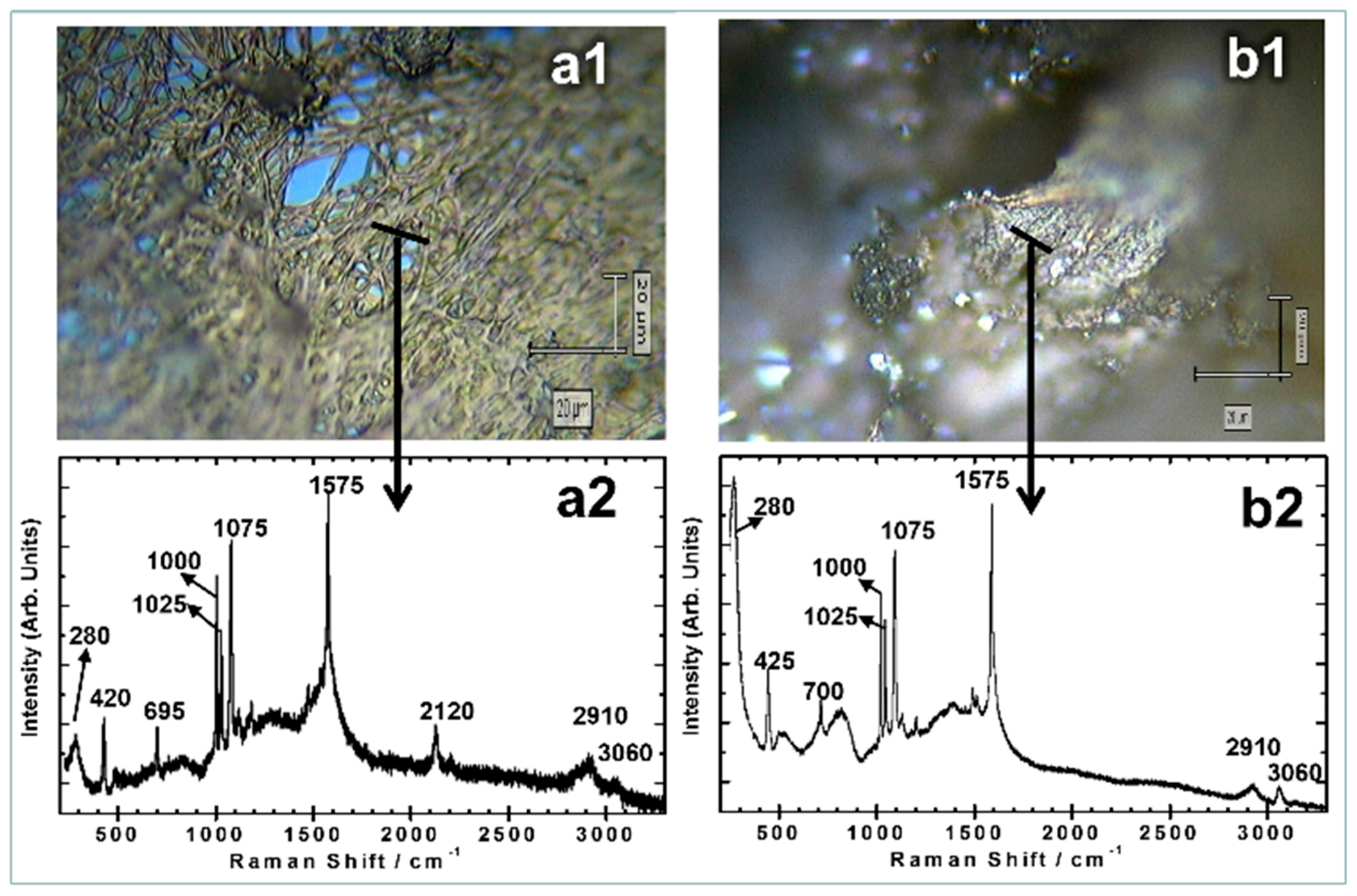
| Assignments | RS (cm−1) | SERS FG (cm−1) | SERS FGS (cm−1) |
|---|---|---|---|
| C-S stretch βCCC (a1) | 412 | 420 | 425 |
| βCCC′ υCS (a1) | 697 | 695 | 700 |
| S-H in-plane bending | 915 | ||
| βCCC (a1) | 999 | 1000 | 1000 |
| βCH (a1) | 1024 | 1025 | 1025 |
| βCCC (a1) | 1091 | 1075 | 1075 |
| C = S ring stretch (a1) | 1580 | 1575 | 1575 |
| S-H stretch | 2566 |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kubo, A.M.; Gorup, L.F.; Toffano, L.; Amaral, L.S.; Rodrigues-Filho, E.; Mohan, H.; Aroca, R.; Camargo, E.R. Nanostructured Assemblies of Gold and Silver Nanoparticles for Plasmon Enhanced Spectroscopy Using Living Biotemplates. Colloids Interfaces 2017, 1, 4. https://doi.org/10.3390/colloids1010004
Kubo AM, Gorup LF, Toffano L, Amaral LS, Rodrigues-Filho E, Mohan H, Aroca R, Camargo ER. Nanostructured Assemblies of Gold and Silver Nanoparticles for Plasmon Enhanced Spectroscopy Using Living Biotemplates. Colloids and Interfaces. 2017; 1(1):4. https://doi.org/10.3390/colloids1010004
Chicago/Turabian StyleKubo, Andressa M., Luiz F. Gorup, Leonardo Toffano, Luciana S. Amaral, Edson Rodrigues-Filho, Haider Mohan, Ricardo Aroca, and Emerson R. Camargo. 2017. "Nanostructured Assemblies of Gold and Silver Nanoparticles for Plasmon Enhanced Spectroscopy Using Living Biotemplates" Colloids and Interfaces 1, no. 1: 4. https://doi.org/10.3390/colloids1010004
APA StyleKubo, A. M., Gorup, L. F., Toffano, L., Amaral, L. S., Rodrigues-Filho, E., Mohan, H., Aroca, R., & Camargo, E. R. (2017). Nanostructured Assemblies of Gold and Silver Nanoparticles for Plasmon Enhanced Spectroscopy Using Living Biotemplates. Colloids and Interfaces, 1(1), 4. https://doi.org/10.3390/colloids1010004