Effect of Nickel Doping on the Cure Kinetics of Epoxy/Fe3O4 Nanocomposites
Abstract
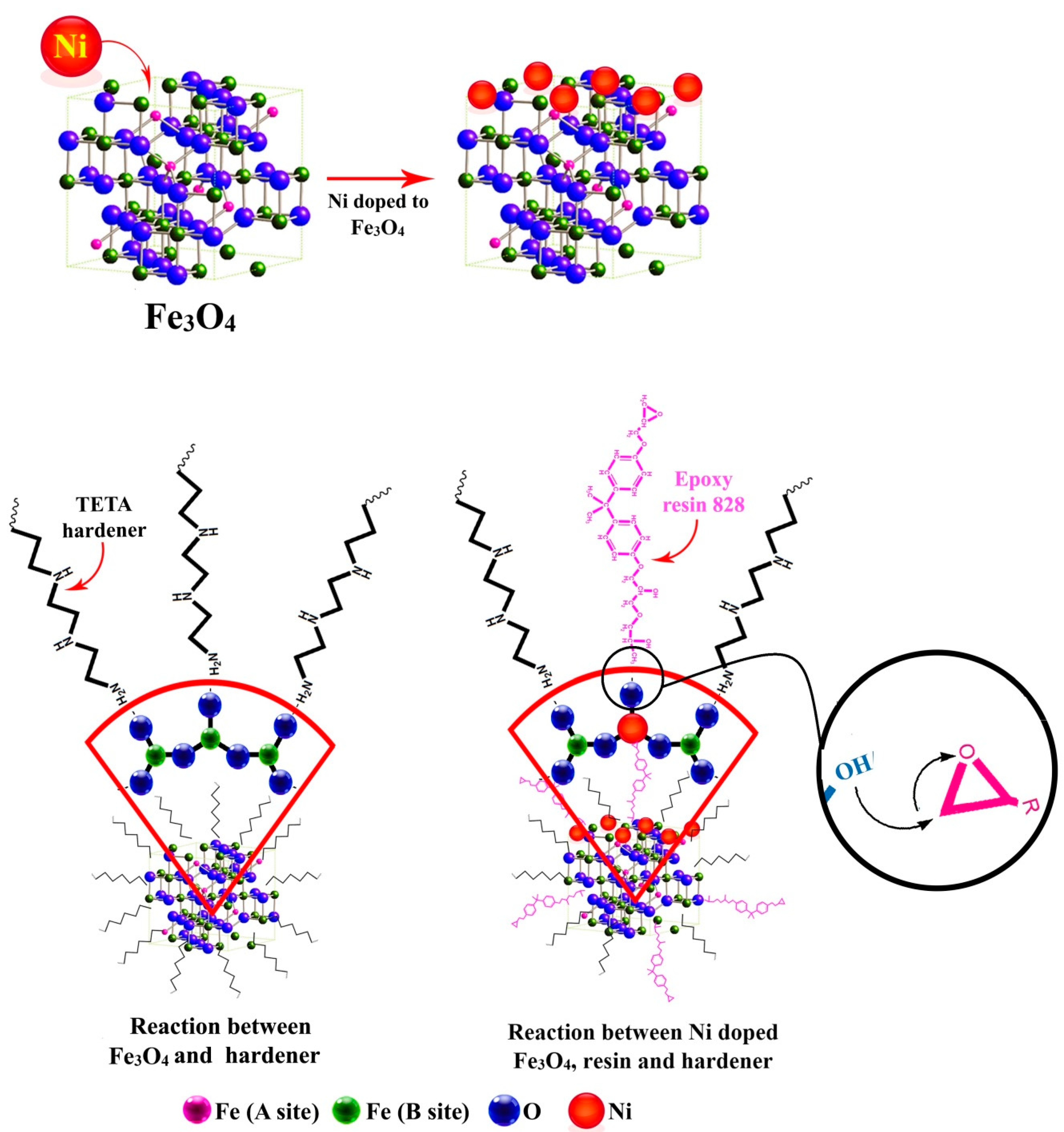
1. Problem Description
2. Development and Characterization of Nanocomposite
2.1. Chemicals
2.2. Preparation of Epoxy Nanocomposites
2.3. Characterization of Epoxy Nanocomposites
3. Quantitative Cure Analysis
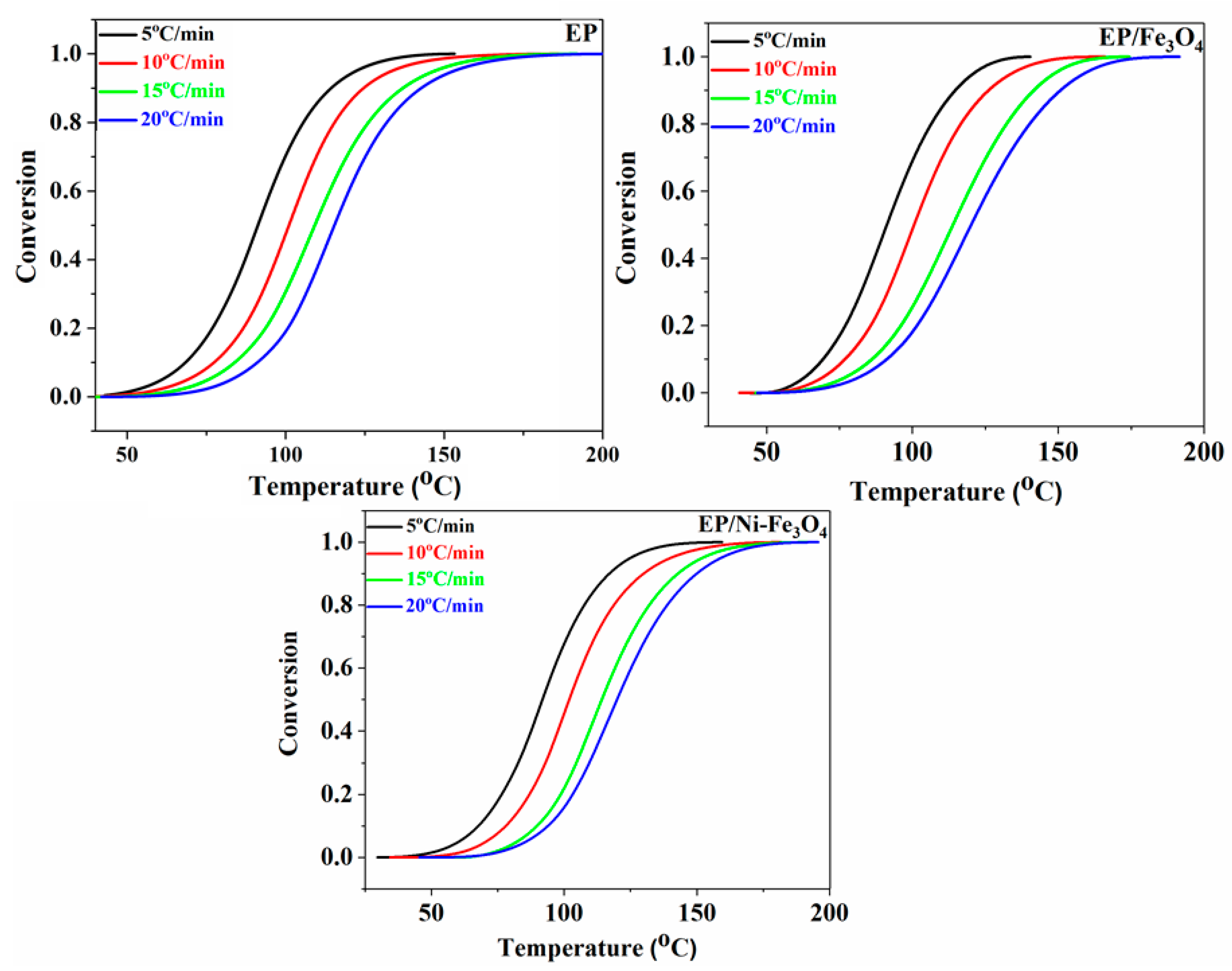
3.1. Cure Behavior Analysis
3.2. Cure Kinetics Analysis
Determining the Reaction Model and the Order of Reaction
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Jouyandeh, M.; Tikhani, F.; Hampp, N.; Akbarzadeh Yazdi, D.; Zarrintaj, P.; Reza Ganjali, M.; Reza Saeb, M. Highly curable self-healing vitrimer-like cellulose-modified halloysite nanotube/epoxy nanocomposite coatings. Chem. Eng. J. 2020, 396, 125196. [Google Scholar] [CrossRef]
- Jouyandeh, M.; Tikhani, F.; Shabanian, M.; Movahedi, F.; Moghari, S.; Akbari, V.; Gabrion, X.; Laheurte, P.; Vahabi, H.; Saeb, M.R. Synthesis, characterization, and high potential of 3D metal–organic framework (MOF) nanoparticles for curing with epoxy. J. Alloys Compd. 2020, 829, 154547. [Google Scholar] [CrossRef]
- Jouyandeh, M.; Jazani, O.M.; Navarchian, A.H.; Saeb, M.R. Epoxy Coatings Physically Cured with Hydroxyl-contained Silica Nanospheres and Halloysite nanotubes. Prog. Color Colorants Coat. 2018, 11, 199–207. [Google Scholar]
- Vryonis, O.; Virtanen, S.; Andritsch, T.; Vaughan, A.; Lewin, P. Understanding the cross-linking reactions in highly oxidized graphene/epoxy nanocomposite systems. J. Mater. Sci. 2019, 54, 3035–3051. [Google Scholar] [CrossRef]
- Throckmorton, J.A.; Watters, A.L.; Geng, X.; Palmese, G.R. Room temperature ionic liquids for epoxy nanocomposite synthesis: Direct dispersion and cure. Compos. Sci. Technol. 2013, 86, 38–44. [Google Scholar] [CrossRef]
- Mirabedini, S.; Behzadnasab, M.; Kabiri, K. Effect of various combinations of zirconia and organoclay nanoparticles on mechanical and thermal properties of an epoxy nanocomposite coating. Compos. Part A Appl. Sci. Manuf. 2012, 43, 2095–2106. [Google Scholar] [CrossRef]
- Martin-Gallego, M.; Yuste-Sanchez, V.; Sanchez-Hidalgo, R.; Verdejo, R.; Lopez-Manchado, M.A. Epoxy nanocomposites filled with carbon nanoparticles. Chem. Rec. 2018, 18, 928–939. [Google Scholar] [CrossRef]
- Samad, U.A.; Alam, M.A.; Chafidz, A.; Al-Zahrani, S.M.; Alharthi, N.H. Enhancing mechanical properties of epoxy/polyaniline coating with addition of ZnO nanoparticles: Nanoindentation characterization. Prog. Org. Coat. 2018, 119, 109–115. [Google Scholar] [CrossRef]
- Cho, J.D.; Ju, H.T.; Park, Y.S.; Hong, J.W. Kinetics of Cationic Photopolymerizations of UV-curable epoxy-based SU8-negative photoresists with and without silica nanoparticles. Macromol. Mater. Eng. 2006, 291, 1155–1163. [Google Scholar] [CrossRef]
- Ma, I.W.; Sh, A.; Ramesh, K.; Vengadaesvaran, B.; Ramesh, S.; Arof, A.K. Anticorrosion properties of epoxy-nanochitosan nanocomposite coating. Prog. Org. Coat. 2017, 113, 74–81. [Google Scholar]
- Shi, X.; Nguyen, T.A.; Suo, Z.; Liu, Y.; Avci, R. Effect of nanoparticles on the anticorrosion and mechanical properties of epoxy coating. Surf. Coat. Technol. 2009, 204, 237–245. [Google Scholar] [CrossRef]
- Madhup, M.K.; Shah, N.K.; Parekh, N.R. Investigation and improvement of abrasion resistance, water vapor barrier and anticorrosion properties of mixed clay epoxy nanocomposite coating. Prog. Org. Coat. 2017, 102, 186–193. [Google Scholar] [CrossRef]
- Jouyandeh, M.; Paran, S.M.R.; Jannesari, A.; Saeb, M.R. ‘Cure Index’ for thermoset composites. Prog. Org. Coat. 2019, 127, 429–434. [Google Scholar] [CrossRef]
- Tikhani, F.; Jouyandeh, M.; Jafari, S.H.; Chabokrow, S.; Ghahari, M.; Gharanjig, K.; Klein, F.; Hampp, N.; Ganjali, M.R.; Formela, K. Cure Index demonstrates curing of epoxy composites containing silica nanoparticles of variable morphology and porosity. Prog. Org. Coat. 2019, 135, 176–184. [Google Scholar] [CrossRef]
- Jouyandeh, M.; Ganjali, M.R.; Ali, J.A.; Aghazadeh, M.; Karimzadeh, I.; Formela, K.; Colom, X.; Cañavate, J.; Saeb, M.R. Curing epoxy with ethylenediaminetetraacetic acid (EDTA) surface-functionalized CoxFe3−xO4 magnetic nanoparticles. Prog. Org. Coat. 2019, 105248. [Google Scholar] [CrossRef]
- Jouyandeh, M.; Zarrintaj, P.; Ganjali, M.R.; Ali, J.A.; Karimzadeh, I.; Aghazadeh, M.; Ghaffari, M.; Saeb, M.R. Curing epoxy with electrochemically synthesized GdxFe3−xO4 magnetic nanoparticles. Prog. Org. Coat. 2019, 105245. [Google Scholar] [CrossRef]
- Jouyandeh, M.; Ganjali, M.R.; Ali, J.A.; Aghazadeh, M.; Paran, S.M.R.; Naderi, G.; Saeb, M.R.; Thomas, S. Curing epoxy with polyvinylpyrrolidone (PVP) surface-functionalized ZnxFe3−xO4 magnetic nanoparticles. Prog. Org. Coat. 2019, 105227. [Google Scholar] [CrossRef]
- Jouyandeh, M.; Paran, S.M.R.; Jannesari, A.; Puglia, D.; Saeb, M.R. Protocol for nonisothermal cure analysis of thermoset composites. Prog. Org. Coat. 2019, 131, 333–339. [Google Scholar] [CrossRef]
- Jouyandeh, M.; Shabanian, M.; Khaleghi, M.; Paran, S.M.R.; Ghiyasi, S.; Vahabi, H.; Formela, K.; Puglia, D.; Saeb, M.R. Acid-aided epoxy-amine curing reaction as reflected in epoxy/Fe3O4 nanocomposites: Chemistry, mechanism, and fracture behavior. Prog. Org. Coat. 2018, 125, 384–392. [Google Scholar] [CrossRef]
- Jouyandeh, M.; Paran, S.M.R.; Shabanian, M.; Ghiyasi, S.; Vahabi, H.; Badawi, M.; Formela, K.; Puglia, D.; Saeb, M.R. Curing behavior of epoxy/Fe3O4 nanocomposites: A comparison between the effects of bare Fe3O4, Fe3O4/SiO2/chitosan and Fe3O4/SiO2/chitosan/imide/phenylalanine-modified nanofillers. Prog. Org. Coat. 2018, 123, 10–19. [Google Scholar] [CrossRef]
- Jouyandeh, M.; Ganjali, M.R.; Ali, J.A.; Aghazadeh, M.; Stadler, F.J.; Saeb, M.R. Curing epoxy with electrochemically synthesized NixFe3−xO4 magnetic nanoparticles. Prog. Org. Coat. 2019, 105198. [Google Scholar] [CrossRef]
- Jouyandeh, M.; Karami, Z.; Hamad, S.M.; Ganjali, M.R.; Akbari, V.; Vahabi, H.; Kim, S.-J.; Zarrintaj, P.; Saeb, M.R. Nonisothermal cure kinetics of epoxy/ZnxFe3−xO4 nanocomposites. Prog. Org. Coat. 2019, 136, 105290. [Google Scholar] [CrossRef]
- Karami, Z.; Jouyandeh, M.; Ali, J.A.; Ganjali, M.R.; Aghazadeh, M.; Paran, S.M.R.; Naderi, G.; Puglia, D.; Saeb, M.R. Epoxy/layered double hydroxide (LDH) nanocomposites: Synthesis, characterization, and Excellent cure feature of nitrate anion intercalated Zn-Al LDH. Prog. Org. Coat. 2019, 136, 105218. [Google Scholar] [CrossRef]
- Jouyandeh, M.; Ali, J.A.; Akbari, V.; Aghazadeh, M.; Paran, S.M.R.; Naderi, G.; Saeb, M.R.; Ranjbar, Z.; Ganjali, M.R. Curing epoxy with polyvinylpyrrolidone (PVP) surface-functionalized MnxFe3−xO4 magnetic nanoparticles. Prog. Org. Coat. 2019, 105247. [Google Scholar] [CrossRef]
- Jouyandeh, M.; Ganjali, M.R.; Ali, J.A.; Aghazadeh, M.; Saeb, M.R.; Ray, S.S. Curing epoxy with polyvinylpyrrolidone (PVP) surface-functionalized NixFe3−xO4 magnetic nanoparticles. Prog. Org. Coat. 2019, 105259. [Google Scholar] [CrossRef]
- Jouyandeh, M.; Jazani, O.M.; Navarchian, A.H.; Shabanian, M.; Vahabi, H.; Saeb, M.R. Surface engineering of nanoparticles with macromolecules for epoxy curing: Development of super-reactive nitrogen-rich nanosilica through surface chemistry manipulation. Appl. Surf. Sci. 2018, 447, 152–164. [Google Scholar] [CrossRef]
- Lange, J.; Altmann, N.; Kelly, C.; Halley, P. Understanding vitrification during cure of epoxy resins using dynamic scanning calorimetry and rheological techniques. Polymer 2000, 41, 5949–5955. [Google Scholar] [CrossRef]
- Bayat, S.; Moini Jazani, O.; Molla-Abbasi, P.; Jouyandeh, M.; Saeb, M.R. Thin films of epoxy adhesives containing recycled polymers and graphene oxide nanoflakes for metal/polymer composite interface. Prog. Org. Coat. 2019, 136, 105201. [Google Scholar] [CrossRef]
- Jouyandeh, M.; Paran, S.M.R.; Khadem, S.S.M.; Ganjali, M.R.; Akbari, V.; Vahabi, H.; Saeb, M.R. Nonisothermal cure kinetics of epoxy/MnxFe3−xO4 nanocomposites. Prog. Org. Coat. 2020, 140, 105505. [Google Scholar] [CrossRef]
- Jouyandeh, M.; Jazani, O.M.; Navarchian, A.H.; Shabanian, M.; Vahabi, H.; Saeb, M.R. Bushy-surface hybrid nanoparticles for developing epoxy superadhesives. Appl. Surf. Sci. 2019, 479, 1148–1160. [Google Scholar] [CrossRef]
- Otto, S.; Engberts, J.B. Lewis-acid catalysis of a Diels-Alder reaction in water. Tetrahedron Lett. 1995, 36, 2645–2648. [Google Scholar] [CrossRef]
- Wan, J.; Li, C.; Bu, Z.-Y.; Xu, C.-J.; Li, B.-G.; Fan, H. A comparative study of epoxy resin cured with a linear diamine and a branched polyamine. Chem. Eng. J. 2012, 188, 160–172. [Google Scholar] [CrossRef]
- Vyazovkin, S.; Burnham, A.K.; Criado, J.M.; Pérez-Maqueda, L.A.; Popescu, C.; Sbirrazzuoli, N. ICTAC Kinetics Committee recommendations for performing kinetic computations on thermal analysis data. Thermochim. Acta 2011, 520, 1–19. [Google Scholar] [CrossRef]
- Miura, K. A New and Simple Method to Estimate f(E) and k0(E) in the Distributed Activation Energy Model from Three Sets of Experimental Data. Energy Fuels 1995, 9, 302–307. [Google Scholar] [CrossRef]
- Vyazovkin, S. Model-free kinetics. J. Therm. Anal. Calorim. 2006, 83, 45–51. [Google Scholar] [CrossRef]
- Ratna, D. Handbook of Thermoset Resins; ISmithers: Shawbury, UK, 2009. [Google Scholar]
- Pascault, J.-P.; Williams, R.J. Epoxy Polymers: New Materials and Innovations; John Wiley & Sons: Hoboken, NJ, USA, 2009. [Google Scholar]
- Fu, Z.; Yang, B.; Zhang, Y.; Zhang, N.; Yang, Z. Dopant segregation and CO adsorption on doped Fe3O4 (1 1 1) surfaces: A first-principle study. J. Catal. 2018, 364, 291–296. [Google Scholar] [CrossRef]
- Reddy, G.K.; Gunasekera, K.; Boolchand, P.; Dong, J.; Smirniotis, P.G. High temperature water gas shift reaction over nanocrystalline copper codoped-modified ferrites. J. Phys. Chem. C 2011, 115, 7586–7595. [Google Scholar] [CrossRef]
- Sari, M.G.; Saeb, M.R.; Shabanian, M.; Khaleghi, M.; Vahabi, H.; Vagner, C.; Zarrintaj, P.; Khalili, R.; Paran, S.M.R.; Ramezanzadeh, B.; et al. Epoxy/starch-modified nano-zinc oxide transparent nanocomposite coatings: A showcase of superior curing behavior. Prog. Org. Coat. 2018, 115, 143–150. [Google Scholar] [CrossRef]
- Rastin, H.; Saeb, M.R.; Nonahal, M.; Shabanian, M.; Vahabi, H.; Formela, K.; Gabrion, X.; Seidi, F.; Zarrintaj, P.; Sari, M.G.; et al. Transparent nanocomposite coatings based on epoxy and layered double hydroxide: Nonisothermal cure kinetics and viscoelastic behavior assessments. Prog. Org. Coat. 2017, 113, 126–135. [Google Scholar] [CrossRef]
- Zhao, K.; Wang, J.; Song, X.; Liang, C.; Xu, S. Curing kinetics of nanostructured epoxy blends toughened with epoxidized carboxyl-terminated liquid rubber. Thermochim. Acta 2015, 605, 8–15. [Google Scholar] [CrossRef]
- Huguenin, F.G.; Klein, M.T. Intrinsic and transport-limited epoxyamine cure kinetics. Ind. Eng. Chem. Prod. Res. Dev. 1985, 24, 166–171. [Google Scholar] [CrossRef]




| Sample | Tg (°C) |
|---|---|
| EP | 100.2 |
| EP/Fe3O4 | 92.6 |
| EP/Ni-Fe3O4 | 101.5 |
| Designation | Heating Rate (°C/min) | αp∞ | αm | αp |
|---|---|---|---|---|
| EP [22,29] | 5 | 0.487 | 0.144 | 0.512 |
| 10 | 0.555 | 0.073 | 0.510 | |
| 15 | 0.418 | 0.236 | 0.498 | |
| 20 | 0.383 | 0.251 | 0.483 | |
| EP/Fe3O4 [22,29] | 5 | 0.479 | 0.248 | 0.490 |
| 10 | 0.407 | 0.279 | 0.491 | |
| 15 | 0.532 | 0.193 | 0.518 | |
| 20 | 0.462 | 0.201 | 0.491 | |
| EP/Ni-Fe3O4 | 5 | 0.473 | 0.146 | 0.490 |
| 10 | 0.400 | 0.296 | 0.475 | |
| 15 | 0.361 | 0.249 | 0.463 | |
| 20 | 0.514 | 0.208 | 0.487 |
| Designation | Heating Rate (°C/min) | Friedman | KAS | ||||
|---|---|---|---|---|---|---|---|
| m | n | lnA (s−1) | m | n | lnA (s−1) | ||
| EP [22,29] | 5 | 0.41 | 1.54 | 18.80 | 0.43 | 1.52 | 18.18 |
| 10 | 0.52 | 1.59 | 19.05 | 0.51 | 1.59 | 19.13 | |
| 15 | 0.44 | 1.68 | 18.98 | 0.46 | 1.66 | 18.39 | |
| 20 | 0.55 | 1.73 | 19.12 | 0.57 | 1.71 | 18.54 | |
| EP/Fe3O4 [22,29] | 5 | 0.36 | 1.22 | 13.25 | 0.30 | 1.27 | 15.01 |
| 10 | 0.51 | 1.40 | 13.79 | 0.46 | 1.45 | 15.51 | |
| 15 | 0.32 | 1.18 | 13.24 | 0.25 | 1.23 | 14.89 | |
| 20 | 0.33 | 1.27 | 13.32 | 0.27 | 1.32 | 14.95 | |
| Epoxy/Ni-Fe3O4 | 5 | 0.43 | 1.48 | 15.78 | 0.44 | 1.47 | 15.35 |
| 10 | 0.55 | 1.68 | 16.24 | 0.57 | 1.66 | 15.82 | |
| 15 | 0.46 | 1.54 | 15.90 | 0.47 | 1.53 | 15.50 | |
| 20 | 0.41 | 1.51 | 15.83 | 0.43 | 1.49 | 15.43 | |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Jouyandeh, M.; Karami, Z.; Paran, S.M.R.; Mashhadzadeh, A.H.; Ganjali, M.R.; Bagheri, B.; Zarrintaj, P.; Habibzadeh, S.; Vijayan P., P.; Saeb, M.R. Effect of Nickel Doping on the Cure Kinetics of Epoxy/Fe3O4 Nanocomposites. J. Compos. Sci. 2020, 4, 102. https://doi.org/10.3390/jcs4030102
Jouyandeh M, Karami Z, Paran SMR, Mashhadzadeh AH, Ganjali MR, Bagheri B, Zarrintaj P, Habibzadeh S, Vijayan P. P, Saeb MR. Effect of Nickel Doping on the Cure Kinetics of Epoxy/Fe3O4 Nanocomposites. Journal of Composites Science. 2020; 4(3):102. https://doi.org/10.3390/jcs4030102
Chicago/Turabian StyleJouyandeh, Maryam, Zohre Karami, Seyed Mohammad Reza Paran, Amin Hamed Mashhadzadeh, Mohammad Reza Ganjali, Babak Bagheri, Payam Zarrintaj, Sajjad Habibzadeh, Poornima Vijayan P., and Mohammad Reza Saeb. 2020. "Effect of Nickel Doping on the Cure Kinetics of Epoxy/Fe3O4 Nanocomposites" Journal of Composites Science 4, no. 3: 102. https://doi.org/10.3390/jcs4030102
APA StyleJouyandeh, M., Karami, Z., Paran, S. M. R., Mashhadzadeh, A. H., Ganjali, M. R., Bagheri, B., Zarrintaj, P., Habibzadeh, S., Vijayan P., P., & Saeb, M. R. (2020). Effect of Nickel Doping on the Cure Kinetics of Epoxy/Fe3O4 Nanocomposites. Journal of Composites Science, 4(3), 102. https://doi.org/10.3390/jcs4030102









