Flame-Assisted Spray Pyrolysis Using an Annular Flame Nozzle with Decoupled Velocity Control
Abstract
:1. Introduction
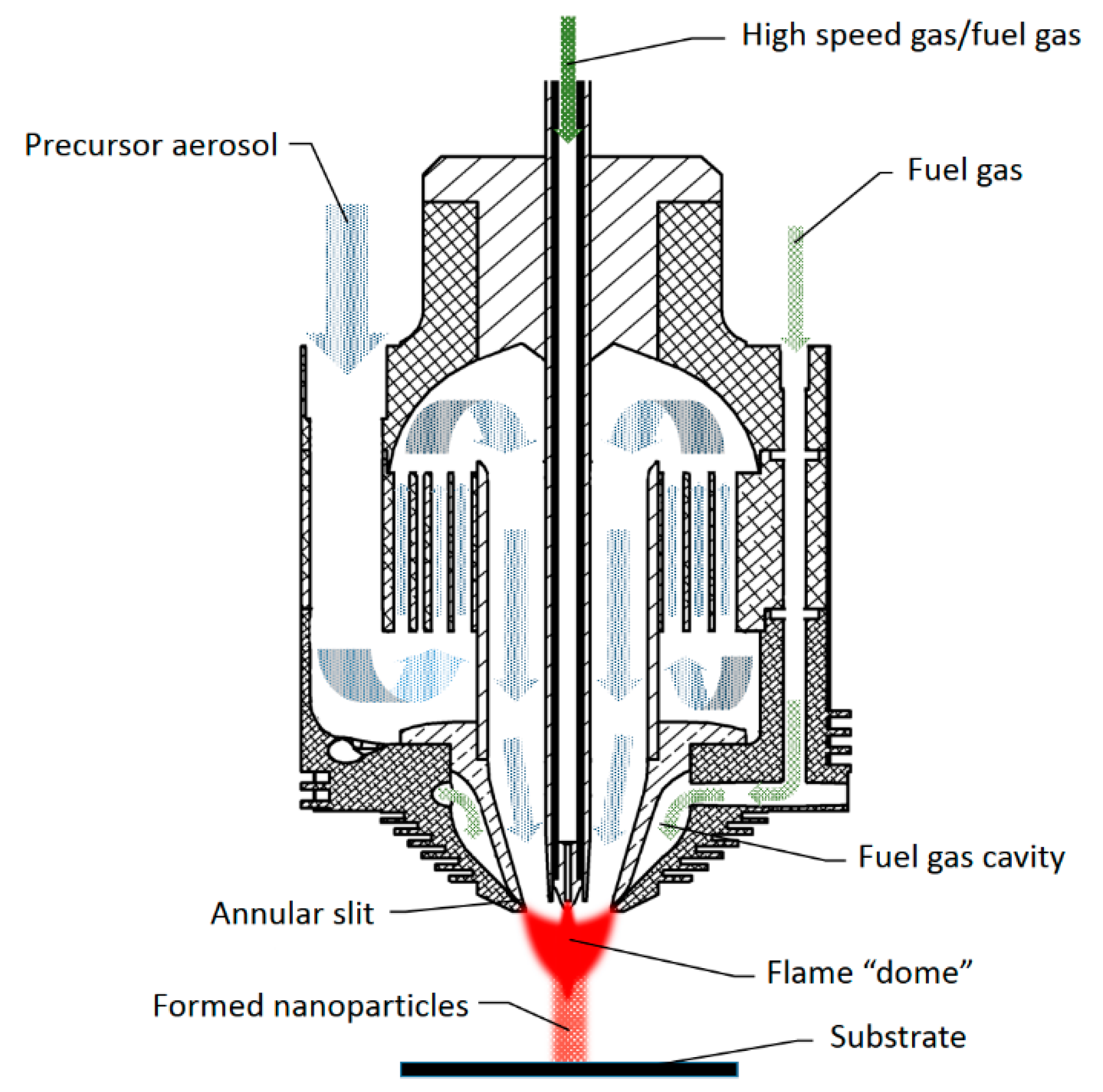
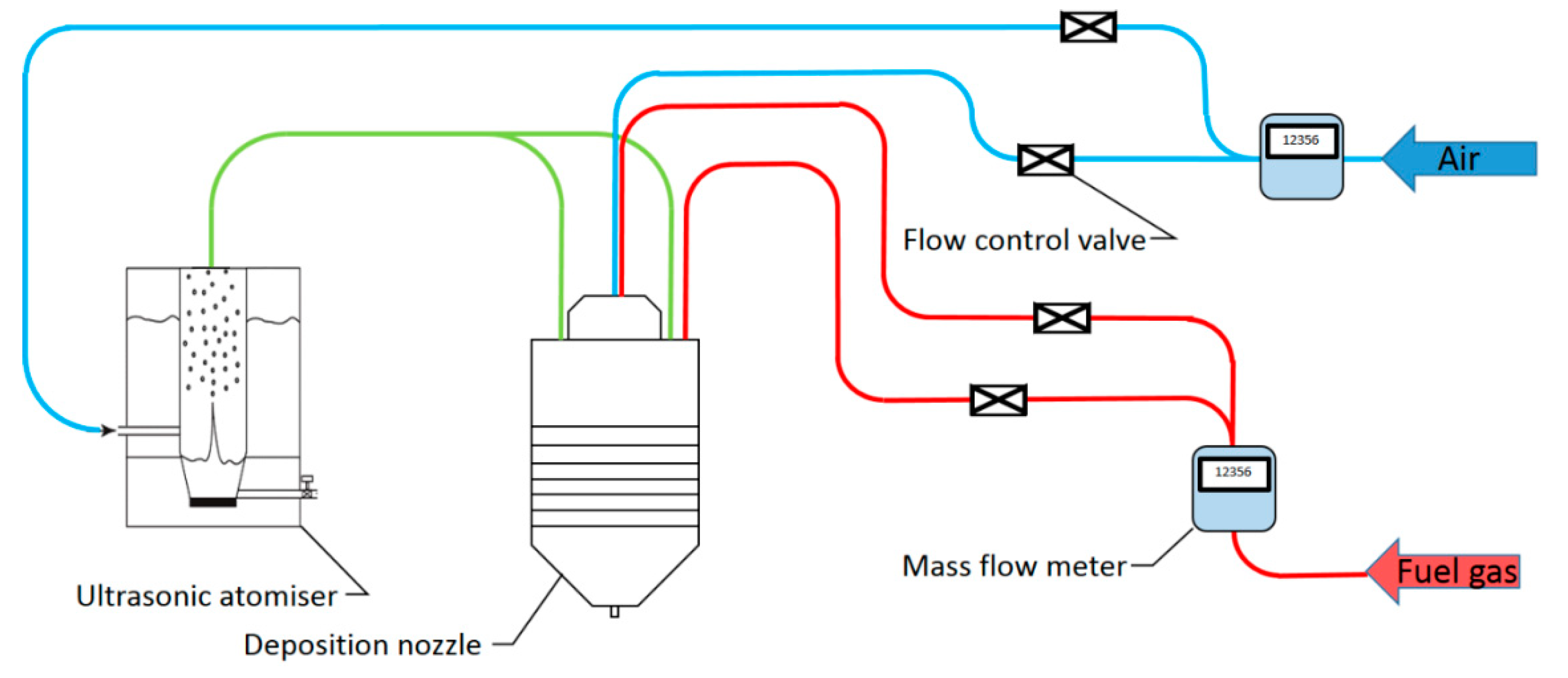
2. Nozzle Design and Testing
3. Results and Discussion
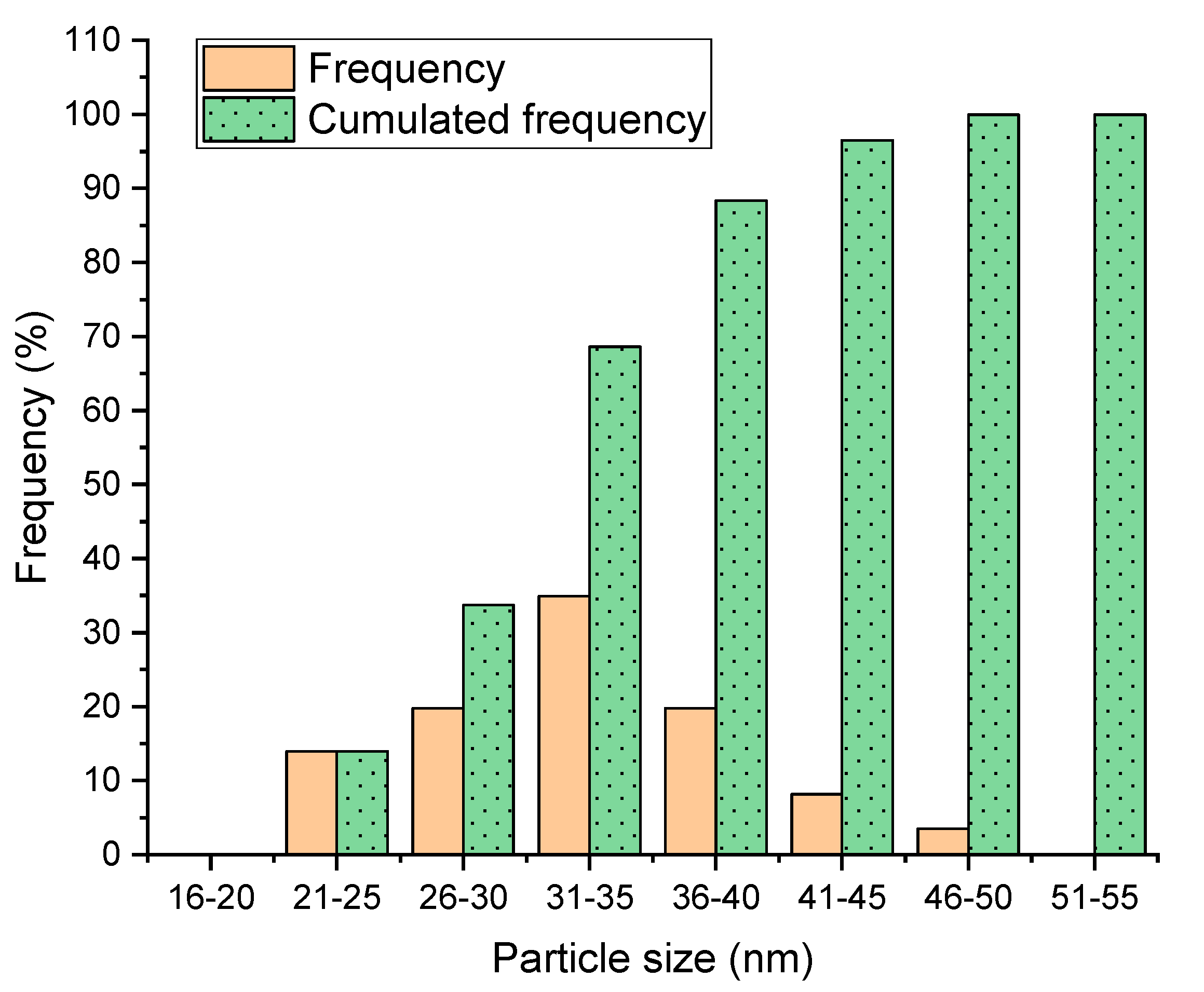
3.1. Variable Concentration and Droplet Diameter
3.2. Variable Flame Temperature
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Karthikeyan, J.; Berndt, C.; Tikkanen, J.; Wang, J.; King, A.; Herman, H. Nanomaterial powders and deposits prepared by flame spray processing of liquid precursors. Nanostruct. Mater. 1997, 8, 61–74. [Google Scholar] [CrossRef]
- Mädler, L.; Roessler, A.; Pratsinis, S.E.; Sahm, T.; Gurlo, A.; Barsan, N.; Weimar, U. Direct formation of highly porous gas-sensing films by in situ thermophoretic deposition of flame-made Pt/SnO2 nanoparticles. Sens. Actuators B Chem. 2006, 114, 283–295. [Google Scholar] [CrossRef]
- Strobel, R.; Baiker, A.; Pratsinis, S.E. Aerosol flame synthesis of catalysts. Adv. Powder Technol. 2006, 17, 457–480. [Google Scholar] [CrossRef]
- Rosner, D.E. Flame synthesis of valuable nanoparticles: Recent progress/current needs in areas of rate laws, population dynamics, and characterization. Ind. Eng. Chem. Res. 2005, 44, 6045–6055. [Google Scholar] [CrossRef]
- Teisala, H.; Tuominen, M.; Aromaa, M.; Mäkelä, J.; Stepien, M.; Saarinen, J.; Toivakka, M.; Kuusipalo, J. Development of superhydrophobic coating on paperboard surface using the liquid flame spray. Surface Coat. Technol. 2010, 205, 436–445. [Google Scholar] [CrossRef]
- Mäkelä, J.M.; Aromaa, M.; Teisala, H.; Tuominen, M.; Stepien, M.; Saarinen, J.J.; Toivakka, M.; Kuusipalo, J. Nanoparticle deposition from liquid flame spray onto moving roll-to-roll paperboard material. Aerosol Sci. Technol. 2011, 45, 827–837. [Google Scholar] [CrossRef]
- Avvaru, B.; Patil, M.N.; Gogate, P.R.; Pandit, A.B. Ultrasonic atomization: Effect of liquid phase properties. Ultrasonics 2006, 44, 146–158. [Google Scholar] [CrossRef] [PubMed]
- Hansen, J.P.; Jensen, J.R.; Livbjerg, H.; Johannessen, T. Synthesis of ZnO particles in a quench-cooled flame reactor. AIChE J. 2001, 47, 2413–2418. [Google Scholar] [CrossRef]
- Johannessen, T.; Jensen, J.R.; Mosleh, M.; Johansen, J.; Quaade, U.; Livbjerg, H. Flame synthesis of nanoparticles: Applications in catalysis and product/process engineering. Chem. Eng. Res. Des. 2004, 82, 1444–1452. [Google Scholar] [CrossRef]
- Jensen, J.R.; Johannessen, T.; Wedel, S.; Livbjerg, H. A study of Cu/ZnO/Al2O3 methanol catalysts prepared by flame combustion synthesis. J. Catal. 2003, 218, 67–77. [Google Scholar] [CrossRef]
- Perednis, D.; Gauckler, L.J. Thin film deposition using spray pyrolysis. J. Electroceram. 2005, 14, 103–111. [Google Scholar] [CrossRef]
- Pratsinis, S.E.; Zhu, W.; Vemury, S. The role of gas mixing in flame synthesis of Titania powders. Powder Technol. 1996, 86, 87–93. [Google Scholar] [CrossRef]
- Mädler, L.; Kammler, H.; Mueller, R.; Pratsinis, S.E. Controlled synthesis of nanostructured particles by flame spray pyrolysis. J. Aerosol Sci. 2002, 33, 369–389. [Google Scholar] [CrossRef]
- Mädler, L.; Stark, W.J.; Pratsinis, S.E. Flame-made ceria nanoparticles. J. Mater. Res. 2002, 17, 1356–1362. [Google Scholar] [CrossRef]
- Kammler, H.K.; Mädler, L.; Pratsinis, S.E. Flame synthesis of nanoparticles. Chem. Eng. Technol. 2001, 24, 583–596. [Google Scholar] [CrossRef]
- Pratsinis, S.E. Aerosol-based technologies in nanoscale manufacturing: From functional materials to devices through core chemical engineering. AIChE J. 2010, 56, 3028–3035. [Google Scholar] [CrossRef]
- Tricoli, A.; Elmøe, T.D. Flame spray pyrolysis synthesis and aerosol deposition of nanoparticle films. AIChE J. 2012, 58, 3578–3588. [Google Scholar] [CrossRef]
- Cho, K.; Chang, H.; Kil, D.S.; Park, J.; Jang, H.D.; Sohn, H.Y. Mechanisms of the formation of silica particles from precursors with different volatilities by flame spray pyrolysis. Aerosol Sci. Technol. 2009, 43, 911–920. [Google Scholar] [CrossRef]
- Mäkelä, J.; Hellstén, S.; Silvonen, J.; Vippola, M.; Levänen, E.; Mäntylä, T. Collection of liquid flame spray generated TiO2 nanoparticles on stainless steel surface. Mater. Lett. 2006, 60, 530–534. [Google Scholar] [CrossRef]
- Aromaa, M.; Keskinen, H.; Mäkelä, J.M. The effect of process parameters on the liquid flame spray generated titania nanoparticles. Biomol. Eng. 2007, 24, 543–548. [Google Scholar] [CrossRef] [PubMed]
- Guild, C.; Biswas, S.; Meng, Y.; Jafari, T.; Gaffney, A.M.; Suib, S.L. Perspectives of spray pyrolysis for facile synthesis of catalysts and thin films: An introduction and summary of recent directions. Catal. Today 2014, 238, 87–94. [Google Scholar] [CrossRef]
- Pimenoff, J.; Hovinen, A.; Rajala, M. Nanostructured coatings by liquid flame spraying. Thin Solid Films 2009, 517, 3057–3060. [Google Scholar] [CrossRef]
- Pratsinis, S.E. Flame aerosol synthesis of ceramic powders. Prog. Energy Combust. Sci. 1998, 24, 197–219. [Google Scholar] [CrossRef]
- You, H.; Cho, K.; Yoon, Y.; Im, J.; Shin, D. Synthesis of yttria-stabilized zirconia film by Aerosol Flame Pyrolysis Deposition. J. Anal. Appl. Pyrolysis 2008, 81, 14–19. [Google Scholar] [CrossRef]
- Mueller, R.; Mädler, L.; Pratsinis, S.E. Nanoparticle synthesis at high production rates by flame spray pyrolysis. Chem. Eng. Sci. 2003, 58, 1969–1976. [Google Scholar] [CrossRef]
- Tikkanen, J.; Gross, K.; Berndt, C.; Pitkänen, V.; Keskinen, J.; Raghu, S.; Rajala, M.; Karthikeyan, J. Characteristics of the liquid flame spray process. Surface Coat. Technol. 1997, 90, 210–216. [Google Scholar] [CrossRef]
- Teoh, W.Y.; Amal, R.; Mädler, L. Flame spray pyrolysis: An enabling technology for nanoparticles design and fabrication. Nanoscale 2010, 2, 1324–1347. [Google Scholar] [CrossRef] [PubMed]
- Purwanto, A.; Widiyandari, H.; Jumari, A. Fabrication of high-performance fluorine doped–tin oxide film using flame-assisted spray deposition. Thin Solid Films 2012, 520, 2092–2095. [Google Scholar] [CrossRef]
- Pluym, T.; Powell, Q.; Gurav, A.; Ward, T.; Kodas, T.; Wang, L.; Glicksman, H. Solid silver particle production by spray pyrolysis. J. Aerosol Sci. 1993, 24, 383–392. [Google Scholar] [CrossRef]
- Pingali, K.C.; Rockstraw, D.A.; Deng, S. Silver nanoparticles from ultrasonic spray pyrolysis of aqueous silver nitrate. Aerosol Sci. Technol. 2005, 39, 1010–1014. [Google Scholar] [CrossRef]
- Mäkelä, J.; Keskinen, H.; Forsblom, T.; Keskinen, J. Generation of metal and metal oxide nanoparticles by liquid flame spray process. J. Mater. Sci. 2004, 39, 2783–2788. [Google Scholar] [CrossRef]
- Kim, D.; Jeong, S.; Moon, J. Synthesis of silver nanoparticles using the polyol process and the influence of precursor injection. Nanotechnology 2006, 17, 4019. [Google Scholar] [CrossRef] [PubMed]
- Rukosuyev, M.V.; Barannyk, O.; Oshkai, P.; Jun, M.B. Design and application of nanoparticle coating system with decoupled spray generation and deposition control. J. Coat. Technol. Res. 2016, 13, 769–779. [Google Scholar] [CrossRef]
- Jiang, C.; Nie, J.; Ma, G. A polymer/metal core–shell nanofiber membrane by electrospinning with an electric field, and its application for catalyst support. RSC Adv. 2016, 6, 22996–23007. [Google Scholar] [CrossRef]
- Jokanović, V.; Spasić, A.M.; Uskoković, D. Designing of nanostructured hollow TiO2 spheres obtained by ultrasonic spray pyrolysis. J. Colloid Interface Sci. 2004, 278, 342–352. [Google Scholar] [CrossRef] [PubMed]




| Sample # | Precursor Concentration mg/L | Atomizer Frequency MHz |
|---|---|---|
| 1 | 50 | 3 |
| 2 | 100 | 3 |
| 3 | 200 | 3 |
| 4 | 50 | 2.4 |
| 5 | 100 | 2.4 |
| 6 | 200 | 2.4 |
| Sample # | 1 | 2 | 3 | 4 | 5 | 6 |
|---|---|---|---|---|---|---|
| Average size (nm) | 40 | 37 | 33 | 28 | 27 | 25 |
| Standard deviation (nm) | 10.6 | 9.9 | 6.5 | 10.9 | 4.8 | 5.5 |
| Sample # | Flame Temperature (°C) | Precursor Concentration (mg/L) | Size of Nanoparticle (nm) |
|---|---|---|---|
| 7 | −780 | 300 | 35~60 |
| 8 | −780 | 100 | 26~48 |
| 9 | 830 | 100 | 100~115 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Rukosuyev, M.; Baqar, S.; Nam, J.; Yun, H.; Jun, M.B.-G. Flame-Assisted Spray Pyrolysis Using an Annular Flame Nozzle with Decoupled Velocity Control. J. Manuf. Mater. Process. 2018, 2, 75. https://doi.org/10.3390/jmmp2040075
Rukosuyev M, Baqar S, Nam J, Yun H, Jun MB-G. Flame-Assisted Spray Pyrolysis Using an Annular Flame Nozzle with Decoupled Velocity Control. Journal of Manufacturing and Materials Processing. 2018; 2(4):75. https://doi.org/10.3390/jmmp2040075
Chicago/Turabian StyleRukosuyev, Maxym, Syed Baqar, Jungsoo Nam, Huitaek Yun, and Martin Byung-Guk Jun. 2018. "Flame-Assisted Spray Pyrolysis Using an Annular Flame Nozzle with Decoupled Velocity Control" Journal of Manufacturing and Materials Processing 2, no. 4: 75. https://doi.org/10.3390/jmmp2040075
APA StyleRukosuyev, M., Baqar, S., Nam, J., Yun, H., & Jun, M. B.-G. (2018). Flame-Assisted Spray Pyrolysis Using an Annular Flame Nozzle with Decoupled Velocity Control. Journal of Manufacturing and Materials Processing, 2(4), 75. https://doi.org/10.3390/jmmp2040075





