Allium cepa L. Inoculation with a Consortium of Plant Growth-Promoting Bacteria: Effects on Plant Growth and Development and Soil Fertility Status and Microbial Community †
Abstract
1. Introduction
2. Material and Methods
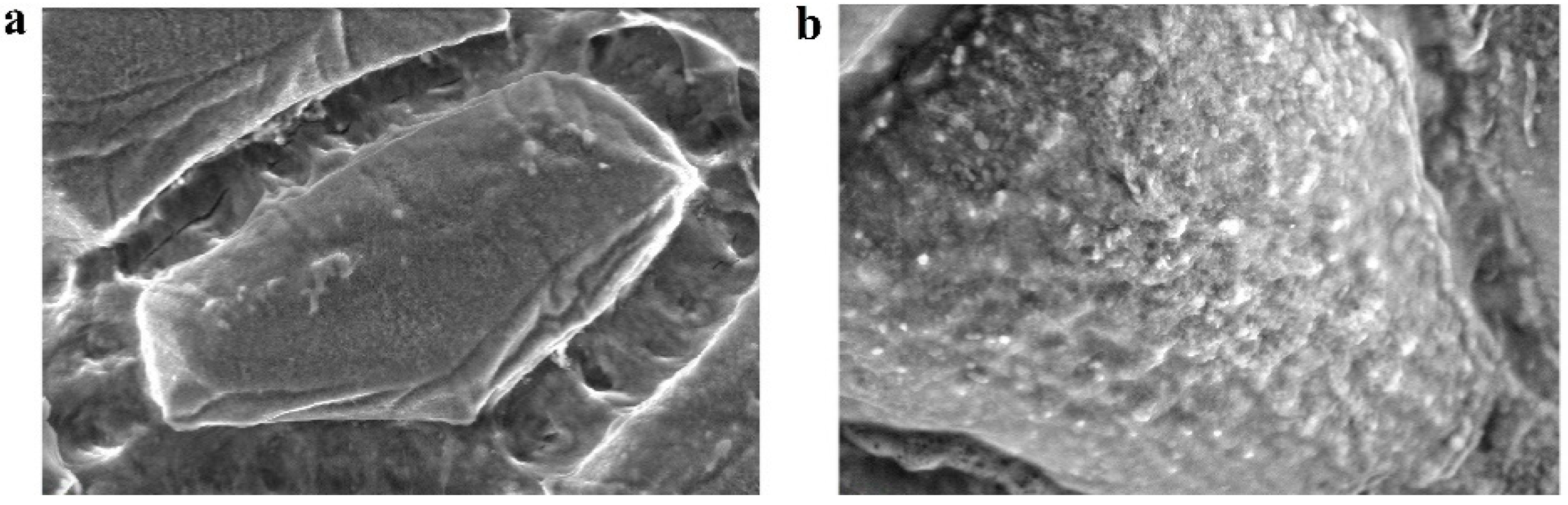
3. Results and Discussion
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Colla, G.; Rouphael, Y.; Canaguier, R.; Svecova, E.; Cardarelli, M. Biostimulant action of a plant-derived protein hydrolysate produced through enzymatic hydrolysis. Front. Plant Sci. 2014, 5, 1–6. [Google Scholar] [CrossRef] [PubMed]
- Bashan, Y.; de-Bashan, L.E.; Prabhu, S.R.; Hernandez, J.P. Advances in plant growth-promoting bacterial inoculant technology: Formulations and practical perspectives (1998–2013). Plant Soil 2014, 378, 1–33. [Google Scholar] [CrossRef]
- Botta, A.L.; Santacecilia, A.; Ercole, C.; Cacchio, P.; Del Gallo, M. In vitro and in vivo inoculation of four endophytic bacteria on Lycopersicon esculentum. N. Biotechnol. 2013, 30, 666–674. [Google Scholar] [CrossRef] [PubMed]
- Pagnani, G.; Pellegrini, M.; Galieni, A.; Egidio, S.D.; Matteucci, F.; Ricci, A.; Stagnari, F.; Sergi, M.; Lo, C.; Pisante, M.; et al. Plant growth-promoting rhizobacteria (PGPR) in Cannabis sativa ‘Finola’ cultivation: An alternative fertilization strategy to improve plant growth and quality characteristics. Ind. Crop. Prod. 2018, 123, 75–83. [Google Scholar] [CrossRef]
- Pace, L.; Pellegrini, M.; Palmieri, S.; Rocchi, R.; Lippa, L.; Del Gallo, M. Plant growth-promoting rhizobacteria for in vitro and ex vitro performance enhancement of Apennines’ Genepì (Artemisia umbelliformis subsp. eriantha), an endangered phytotherapeutic plant. Vitr. Cell. Dev. Biol. Plant 2020, 56, 134–142. [Google Scholar] [CrossRef]
- Pagnani, G.; Galieni, A.; Stagnari, F.; Pellegrini, M.; Gallo, M. Del; Pisante, M. Open field inoculation with PGPR as a strategy to manage fertilization of ancient Triticum genotypes. Biol. Fertil. Soils 2020, 56, 111–124. [Google Scholar] [CrossRef]
- Arnon, D.I. Copper enzymes in isolated chloroplasts. Polyphenoloxidase in Beta vulgaris. Plant Physiol. 1949, 24, 1–15. [Google Scholar] [CrossRef] [PubMed]
- Mirzapour, M.; Hamedi, M.; Rahimipanah, M. Sunflower Oil Stabilization by Persian Walnut Leaves Extract during Oven Storage Test. Food Sci. Technol. Res. 2010, 16, 443–446. [Google Scholar] [CrossRef]
- Pellegrini, M.; Id, A.R.; Id, A.S.; Chaves-l, C. Characterization of Essential Oils Obtained from Abruzzo Autochthonous Plants: Antioxidant and Antimicrobial Activities Assessment for Food Application. Foods 2018, 7, 19. [Google Scholar] [CrossRef] [PubMed]
- Klindworth, A.; Pruesse, E.; Schweer, T.; Peplies, J.; Quast, C.; Horn, M.; Glöckner, F.O. Evaluation of general 16S ribosomal RNA gene PCR primers for classical and next-generation sequencing-based diversity studies. Nucleic Acids Res. 2013, 41, e1. [Google Scholar] [CrossRef] [PubMed]
- Djebaili, R.; Pellegrini, M.; Smati, M.; Del Gallo, M.; Kitouni, M. Actinomycete Strains Isolated from Saline Soils: Plant-Growth-Promoting Traits and Inoculation Effects on Solanum lycopersicum. Sustainability 2020, 12, 4617. [Google Scholar] [CrossRef]
- Vejan, P.; Abdullah, R.; Khadiran, T.; Ismail, S.; Nasrulhaq Boyce, A. Role of plant growth promoting rhizobacteria in agricultural sustainability-A review. Molecules 2016, 21, 1–17. [Google Scholar] [CrossRef]
- Urana, R.; Singh, N.; Sharma, P. Effects of PGPR on growth and photosynthetic pigment of Trigonella foenum - graceum and Brassica juncea in PAH - contaminated soil. SN Appl. Sci. 2019, 1, 1–9. [Google Scholar] [CrossRef]
- Orhan, E.; Esitken, A.; Ercisli, S.; Turan, M.; Sahin, F. Effects of plant growth promoting rhizobacteria (PGPR) on yield, growth and nutrient contents in organically growing raspberry. Sci. Hortic. 2006, 111, 38–43. [Google Scholar] [CrossRef]
- Ullah, A.; Bano, A. Role of PGPR in the reclamation and revegetation of saline land. Pak. J. Bot. 2019, 51, 27–35. [Google Scholar] [CrossRef]
- Hou, J.; Liu, W.; Wang, B.; Wang, Q.; Luo, Y.; Franks, A.E. PGPR enhanced phytoremediation of petroleum contaminated soil and rhizosphere microbial community response. Chemosphere 2015, 138, 592–598. [Google Scholar] [CrossRef]
- Ren, H.; Huang, B.; Fernández-García, V.; Miesel, J.; Yan, L.; Lv, C. Biochar and rhizobacteria amendments improve several soil properties and bacterial diversity. Microorganisms 2020, 8, 502. [Google Scholar] [CrossRef]

Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Pellegrini, M.; Spera, D.M.; Ercole, C.; Gallo, M.d. Allium cepa L. Inoculation with a Consortium of Plant Growth-Promoting Bacteria: Effects on Plant Growth and Development and Soil Fertility Status and Microbial Community. Proceedings 2020, 66, 20. https://doi.org/10.3390/proceedings2020066020
Pellegrini M, Spera DM, Ercole C, Gallo Md. Allium cepa L. Inoculation with a Consortium of Plant Growth-Promoting Bacteria: Effects on Plant Growth and Development and Soil Fertility Status and Microbial Community. Proceedings. 2020; 66(1):20. https://doi.org/10.3390/proceedings2020066020
Chicago/Turabian StylePellegrini, Marika, Daniela M. Spera, Claudia Ercole, and Maddalena del Gallo. 2020. "Allium cepa L. Inoculation with a Consortium of Plant Growth-Promoting Bacteria: Effects on Plant Growth and Development and Soil Fertility Status and Microbial Community" Proceedings 66, no. 1: 20. https://doi.org/10.3390/proceedings2020066020
APA StylePellegrini, M., Spera, D. M., Ercole, C., & Gallo, M. d. (2020). Allium cepa L. Inoculation with a Consortium of Plant Growth-Promoting Bacteria: Effects on Plant Growth and Development and Soil Fertility Status and Microbial Community. Proceedings, 66(1), 20. https://doi.org/10.3390/proceedings2020066020






