Abstract
Adenine complexes were prepared with some of the first series transition metals in a stoichiometric ratio of 1: 2 (Mn+: L), where Mn+ = Mn2+, Fe3+, Co2+, Ni2+, Cu2+, Zn2+, and Cd2+ ions. The Complexes were characterized by the physicochemical and spectroscopic techniques as electric conductivity, metal contents, IR, UV–Visible, and molar conductance techniques. The stoichiometric ratios of the synthesized complexes were confirmed by using molar ratio method. The dissociation constant of adenine ligand was determined spectrophotometrically. Solvent effect on the electronic spectra of the adenine ligand was examined using solvents with different polarities. The biological activity of adenine ligand and its metal complexes were tested in vitro against some selected species of fungi and bacteria. The results showed a satisfactory spectrum against the tested organisms.
1. Introduction
Heterocyclic compounds play a significant role in many biological systems, especially N-donor ligand systems being a component of several vitamins and drugs such as nicotinamide. Nicotinamide is known as a component of the vitamin B complex as well as a component of the coenzyme, nicotinamide adenine dinucleotide (NAD) [1]. The interaction of nucleic acids and their constituents with metal ions has been a matter of extensive studies in the recent years, because of their chemical and biological interest, covering structural, thermodynamic and kinetic works. In this broad context, certain recent studies have focused on metal complexes that can specifically recognize nucleobases [2,3,4].
1.1. Adenine Metal Complexes and Its Binding Sites
The complexation of metal ions with adenine has been well studied and its binding sites has also been clarified [5,6]. Various coordination sites have been observed for adenine in copper complexes as indicated by X-ray studies. Among the four nitrogen’s N(1), N(3), N(7) and N(9) of adenine, the N(9) is the most basic and hence bears a proton rendering it is the most preferred metal binding site. The understanding of the structure and function of RNA and DNA systems depends partly on the binding sites of metal ions to nucleobases [7,8].
Adenine (Ad) is a nucleo-base (a purine derivative) with a variety of roles in biochemistry including cellular respiration, in the form of both the energy-rich adenosine triphosphate (ATP) and the cofactors nicotinamide adenine dinucleotide (NAD) and flavin adenine dinucleotide (FAD), and protein synthesis, as a chemical component of DNA and RNA.
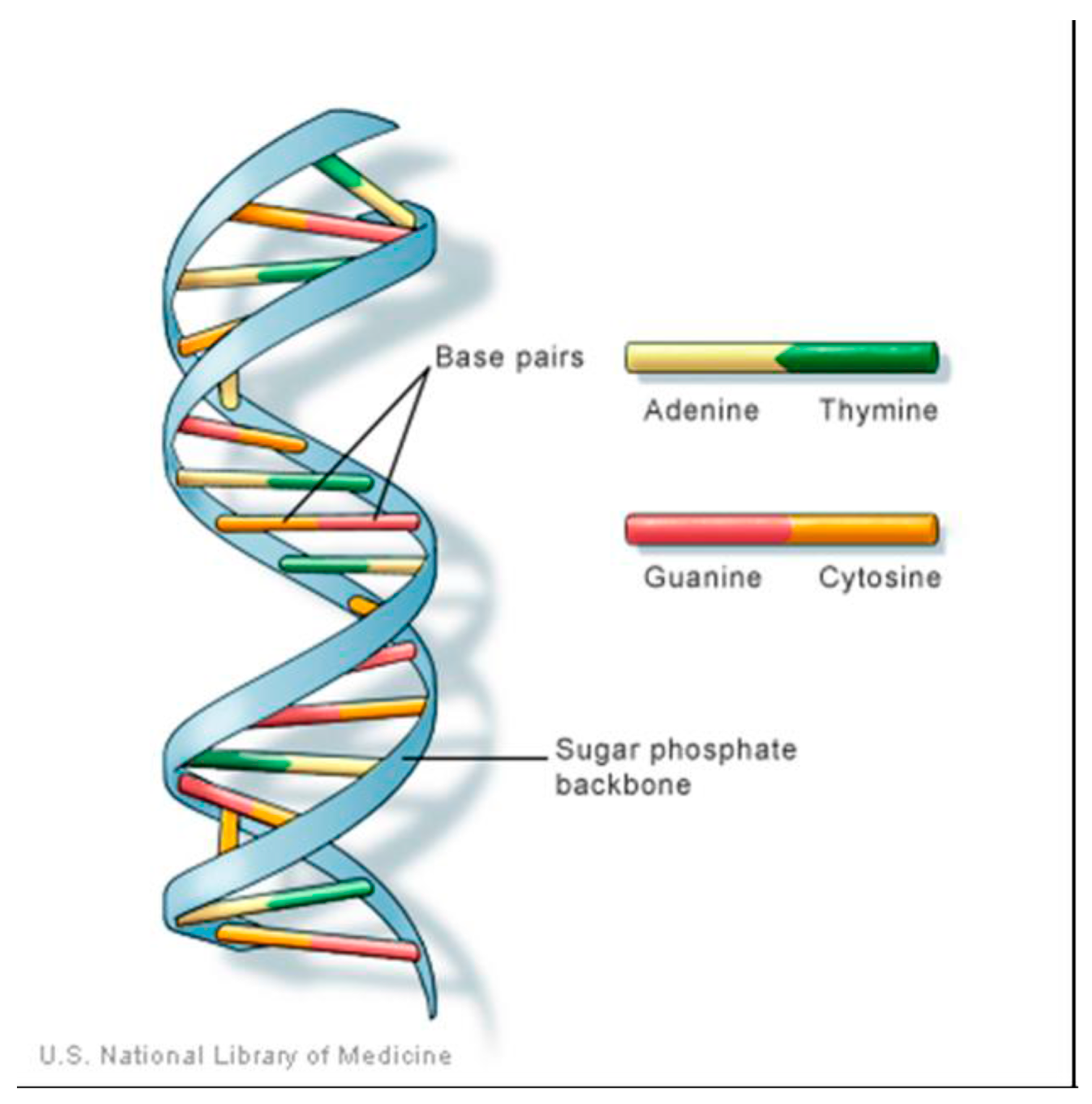
Adenine (Ad) is one of four chemical bases in DNA as in Figure 1, with the other three being cytosine (C), guanine (G), and thymine (T). Within the DNA molecule, adenine bases located on one strand form chemical bonds with thymine bases on the opposite strand [9].
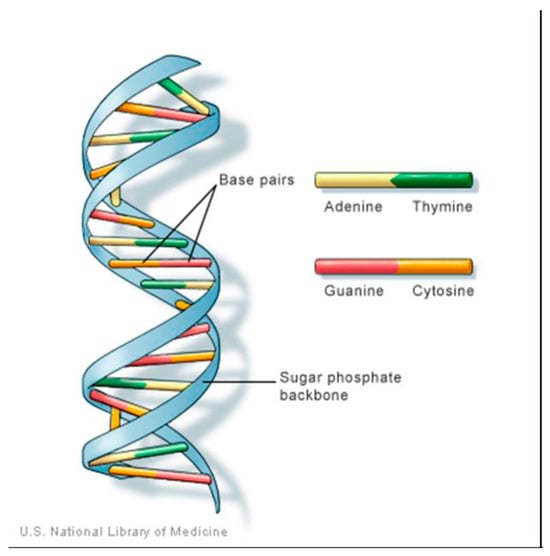
Figure 1.
structure of DNA showing the Adenine position.
1.2. Adenine Metabolism
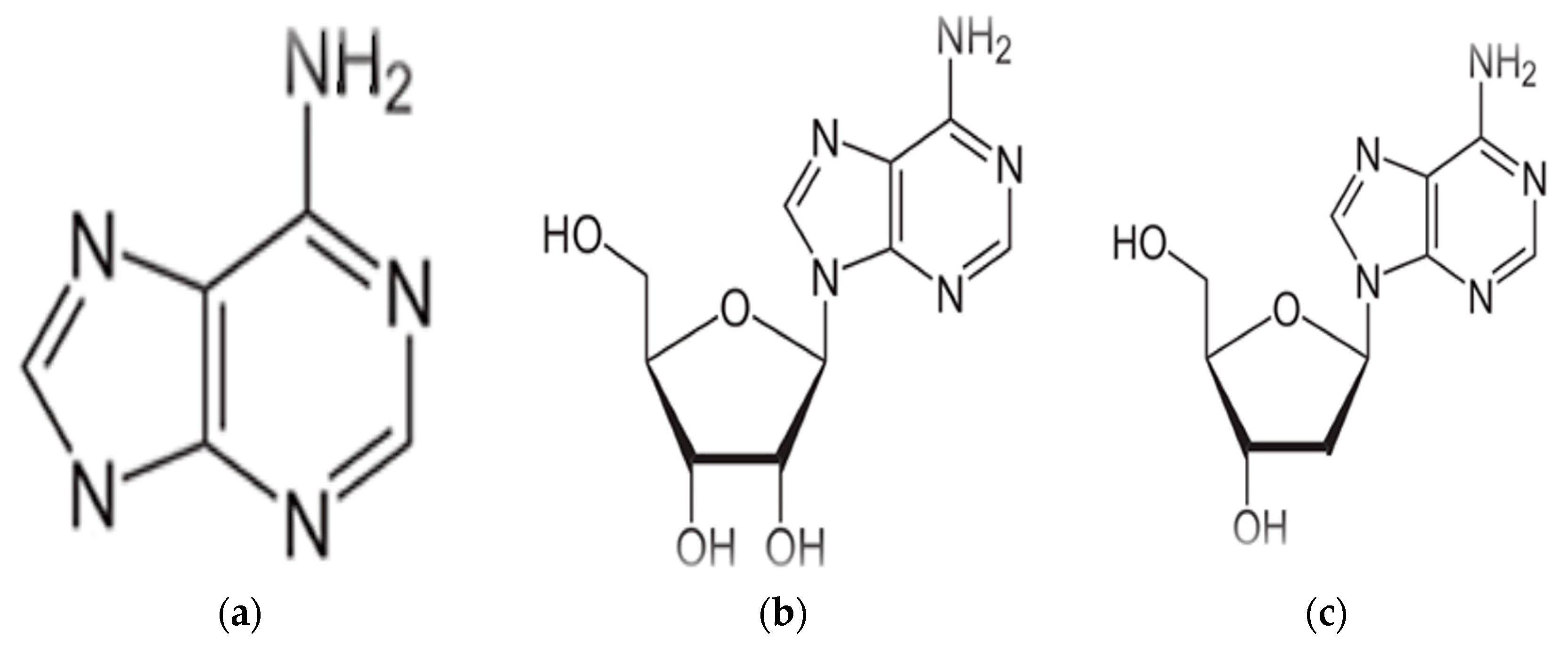
A form of adenine called adenosine tri phosphate (ATP) serves as an energy storage molecule and is used to power many chemical reactions within the cell. Adenosine tri phosphate is used in cellular metabolism as one of the basic methods of transferring chemical energy between reactions. In DNA, adenine bonds only to thymine. It does so with two strong hydrogen bonds, so the bond is difficult to break and the code is difficult to damage. Adenine, has the chemical structure as shown in (Figure 2a). Which is known as organic compound belong to the purine family, characterized by its nucleic acid, in this case called nucleotides, where the phosphate esters of adenosine (Figure 2b) and deoxy adenosine (Figure 2c) are clear in the structure, The importance of Adenine to RNA is similar to that of DNA. Adenosine tri phosphate is the nitrogenous base adenine bonded to a five carbon sugar. Adenosine tri phosphate is used in cellular metabolism as one of the basic methods of transferring chemical energy between, chemical reactions [10].
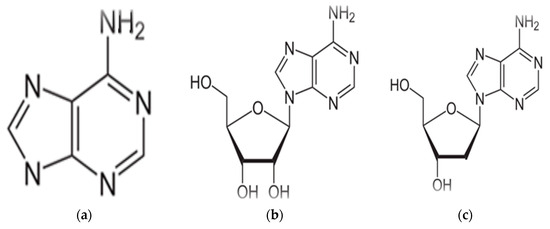
Figure 2.
(a) Structure of Adenine, (b) structure of Adenosine, (c) structure of Deoxyadenosine.
Adenine is one of the two purine nucleo bases used in forming nucleotides of the nucleic acids DNA and RNA. The derivatives of purine are called adenine(A) and guanine(G). The other three bases—thymine(T), cytosine(C), and uracil(U)—are derivatives of pyrimidine, These four "code letters" allow cells to store their blueprint about how that life form is built. All cells of all living organisms, no matter how simple or complex, share this design [11].
Spectroscopic study of the Cu(II) complexes with adenosine and adenine nucleotides, as adenosine mono phosphate (AMP), adenosine diphosphate (ADP), adenosine tri phosphate (ATP) and with inorganic polyphosphate solutions has been studied as a function of pH in the range 3 to 12, resulted in Cu(II)-adenosine phosphate complexes exist with a variety of forms in equilibrium, according to the state of ionization of the intrinsic adenine, phosphate and ribose groups. Mixed-ligand palladium(II) complexes with amino acids, cytosine, and adenine pH titration have been synthesized and characterized as 1:1:1 mixed-ligand complexes showing, palladium(II) (C5H5N5) (C5H8NO4)Cl complex. The coordination mode of amino acids, cytosine, and adenine to the palladium(II) ion is determined [12,13].
In the human body, adenine is synthesized in the liver. Biological systems tend to conserve energy, The vitamin folic acid is essential for adenine synthesis, Adenine forms adenosine, a nucleoside, when attached to ribose, and deoxy adenosine it forms adenosine triphosphate (ATP), a nucleotide, when three phosphate groups are added to adenosine Vitamin B4. Adenine is one of the two purine nucleo-bases (the other being guanine) used in forming nucleotides of the nucleic acids. In DNA, adenine binds to thymine via two hydrogen bonds to assist in stabilizing the nucleic acid structures. Adenine is one of the most interesting biological ligands which used for preparing and synthesizing different types of complexes [12].
Here in, we report the synthesis, characterizations and thermal stabilities of Adenine complexes with some of the first series transition metals in a stoichiometric ratio of 1:2 (Mn+:L), where Mn+ = Mn2+, Fe3+, Co2+, Ni2+, Cu2+, Zn2+, and Cd2+ ions. The Complexes were characterized by the physicochemical techniques as, IR, UV–Visible, and molar conductance techniques. The stoichiometric ratios of the synthesized complexes were confirmed by using molar ratio method.
2. Experimental
All chemicals were obtained from commercial sources and were used without further purifications (MnCl2·4H2O, FeCl3·H2O, CoNO3·6H2O, NiSO4·7H2O, CuSO4·5H2O, ZnSO4·7H2O, Cd(NO3)2·4H2O). Adenine were obtained from BDH, methanol, ethanol, dimethylsulfoxide (DMSO), DMF and distilled water were used as solvents. Molar conductance of the complexes was measured using conductivity bridge at room temperature in DMSO. Conductivity measurements (cm2 mol−1) were carried out in DMF using HANA conduct meter. Perkin-Elmer type FT-IR spectrophotometers were used to record the IR spectra using KBr pellets, covering the range from 600 to 4000 cm−1. UV–visible spectra were recorded on DU-800 spectrometer covering range of 190 to 800 nm. The absorbance values were plotted against [L]/[M] ratios. The result curves are composed of straight lines with inflection points, [L]/[M] ratio corresponding to the infection point indicates to the actual stoichiometry of the complex. Metal content was determined by absorption spectrometry. Thermal analyses (TGA) were performed on a Shimadzu DTA/TGA-50 thermal analyzer in a synthetic air atmosphere (79%N2/21%O2) with a heating rate of 5 °C min−1. The melting points were determined using a Stuart-Melting Point apparatus.
2.1. Synthesis of the Metal Complexes
Standard procedure were used to prepare Adenine metal complexes [14], where 0.2 mole of Adenine in 50 mL ammonia was added to 0.2 mole of metal chloride, Nitrate or sulphate. nH2O, to a conical flask, the mixture was refluxed for one hour. The complex was formed (with different color), then leaving in darkened place even of second day, then filtrated and derided on P2O5.
2.2. Antimicrobial Studies
The antibacterial tests were prepared and characterized according to the standard method [15]. The strains of bacteria used were Gram-positive bacteria (Bacillus) Gram negative bacteria (Escherichia coli), and two types of fungi (Pencilium & Rhizopns) All strains were isolated from laboratory of microbiology. The identity of all the strains was confirmed. A bacterial suspension was prepared and added to the Nutrient Agar, the fungi added to surrounded Agar, All this before medium solidification and under aseptic condition. Then different concentration of complexes were placed on the surface of the culture, The bacteria incubated at 37 °C for 24 h, The fungi incubated at 28 °C for 72 h.
2.3. Bacterial & FUNGI Cultures
Plate cultures of nutrient agar medium were used for culture of bacteria. the medium was prepared by dissolving 14 g, and culture of fungi’s 32.5 g of powder in 500 mL of sterile distilled water, Then the medium was sterilized by autoclaving at 121 °C for15 min.
3. Result and Discussion
3.1. General Properties
The color of ligand was change from white color of the free ligand to several different colors according to the type metal, this change mainly due to the effect the linkage between the ligand and the different electrons in 3d orbital’s, where the attracting electrons between the ligand and the metal in d orbitals where the high and the less in energy, the magnetic frequency beam is proportion to the different in energy between the two states energy in atom. Some electrons rise into energy high level.
The E.C values in the most of the studied complexes are low and ranged between (0.17–1.86 µS), as shown in Table 1. The conductivity of the complexes depended mainly on the free electrons which non conjugation in the last orbital’s, where the conductivity become less when conjugating occur between the metal and the ligand, this mean these electrons are bounded [16].

Table 1.
Physical analysis data of Adenine Metal complexes.
3.2. Infrared Spectral Analysis
The IR spectra of adenine and its metal complexes have been characterized as it has been shown in Table 2, where NH2 bands of adenine at 3291 and 3322 and 3287 cm−1 are slightly shifted on complication. The NH2 mode of the free adenine at 1668 cm−1 undergoes shifts as large as those corresponding to N-bonded complexes of this ligand. So, the NH2 group (exocylic NH2 nitrogen), is coordinated to the metal ion in the complexes. Adenine presumably coordinates through ring nitrogen with appreciable shifts and occasional splitting of νC = C, νC = N and ring vibrations of the ligand (1638–1698 cm−1). The NH region, 3114–3183 cm−1, has considerable changes in the most complexes resulting of two weak maxima in this region. The 1250 cm−1 of adenine due to νC–NH2 or N–H ring mode shifts to lower wavelength upon complication. The bands at 1023 and 938 cm−1 in most complexes (Table 2), respectively, are due to NH2. This facilitates that adenine is binding exclusively through ring nitrogen [17].

Table 2.
Infra-Red spectroscopy values of the Adenine metal Complexes.
3.3. Thermal Decomposition
The thermogravimeteric analysis curves allowed us to establish the temperature below which the characterized compound has a constant weight and which begins to decompose and how far the decomposition reaction can proceed [18]. The observed and stoichiometric decreases in weight of the substance make it possible to estimate an intermediate product which is formed during decomposition and at what temperature occasionally the temperature rang where this intermediate has constant weight.
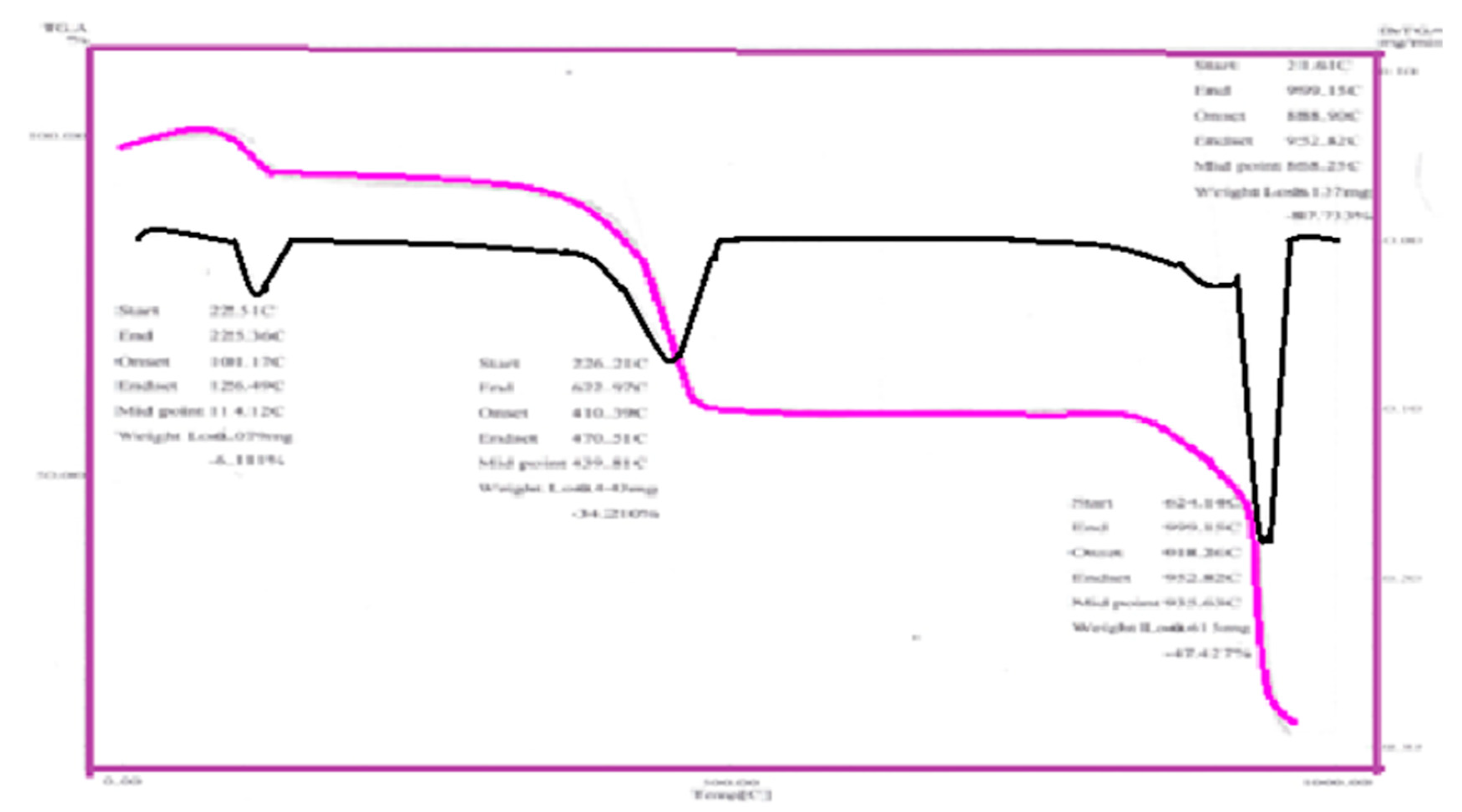
The TGA analysis of the prepared complexes exhibit mostly similar steps of decomposition Figure 3. The thermogravimetric analysis data for adenine complexes are given in Table 3. The weigh-loss of Fe(Ad), Cd(Ade), Mn(Ad), Zn(Ad), Cu(Ad), Ni(Ad) and Co(Ad), mainly corresponding to loss of hydrated water molecules at the temperature range of (100 to 350 °C), and the loss of coordination molecules were occur at the temperature range (400 to 500 °C) While the residual of metals oxides and/or (M-N) bands of (700 °C) appeared at temperature for Fe(Ad), Cd(Ad), Zn(Ad), Cu(Ad), Ni(Ad), Co(Ad) and Mn(Ad), respectively. The TGA curve displays the observed weight loss 7.5% in the temperature range (478.6–651 °C), which is probably due to replacement of HCl molecule. TGA curves depicted that as the HCl is removed, the complex is decomposed. The third strong endothermic peak in the temperature range (606–829 °C) could be argued to the first step of the decomposition. This is confirmed by the observed TG weight loss equivalent to 3N2, 2CH2=C=CH2 and H2O. The fourth endothermic peak in the temperature range is due to the second decomposition step, as evident from the observed TG weight loss equivalent to 2C=N. On the other hand, the fifth endothermic peak in the temperature range (811.1–937.7 °C).
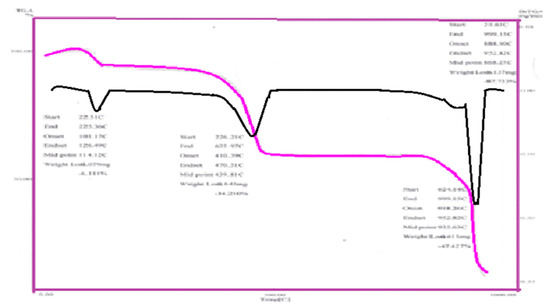
Figure 3.
TGA steps curve of Ad-Cu complex.

Table 3.
Thermo gravimetric analysis data for Adenine metal complexes.
3.4. Effect of Solvents on the Electronic Spectra of the Adenine
The solvent effects on the electronic absorption spectra are used to study the chemical properties of the excited state and to identify the electronic transitions in a molecule [19]. The solvent polarity tends to move the absorption maximum towards lower energy, due to the stabilization of the excited state by the induced dipole interaction between the transition moment and the solvent molecule. Also, the frequency shift of the spectra bands from the vapor state to solution could be related to salvation stabilization energy of the excited and ground states depending on the various types of intermolecular interaction.
In polar or hydrogen bonding solvents with permanent dipole moment for polar solute, blue shift occurs of λ max with increasing solvent polarity with the presence of “frank Condon” phenomena. If the excited state—dipole moment is less than that of the ground state, blue shift of λ max occurs with increasing solvent polarity. This explains the shift of n―π* transition on hydrogen bonding, relative to hydrocarbon solvents. In general, the functional groups with high bond moments are involved in H-bond formation. The non-polar solute in non-polar solvents leading approximate equal salvation energies of both ground and excited state is due to dispersion depending mainly on the solvent refractive index, similar situation was found for the non-polar solute in polar solvents but with more H-bonding molecule with the increase of solvent cage molecules, the behavior of the polar solute in the non-polar or polar solvent was found to depend on the dipole moment of the solute (decreased or increased during excitation). The process of the reaction in the first case leads to a blue shift of the maximum absorption and in the second case a red shift occurs. It is expected that in the presence of polar solvents, accumulation of H-bonding forces occurs depending on many factors:
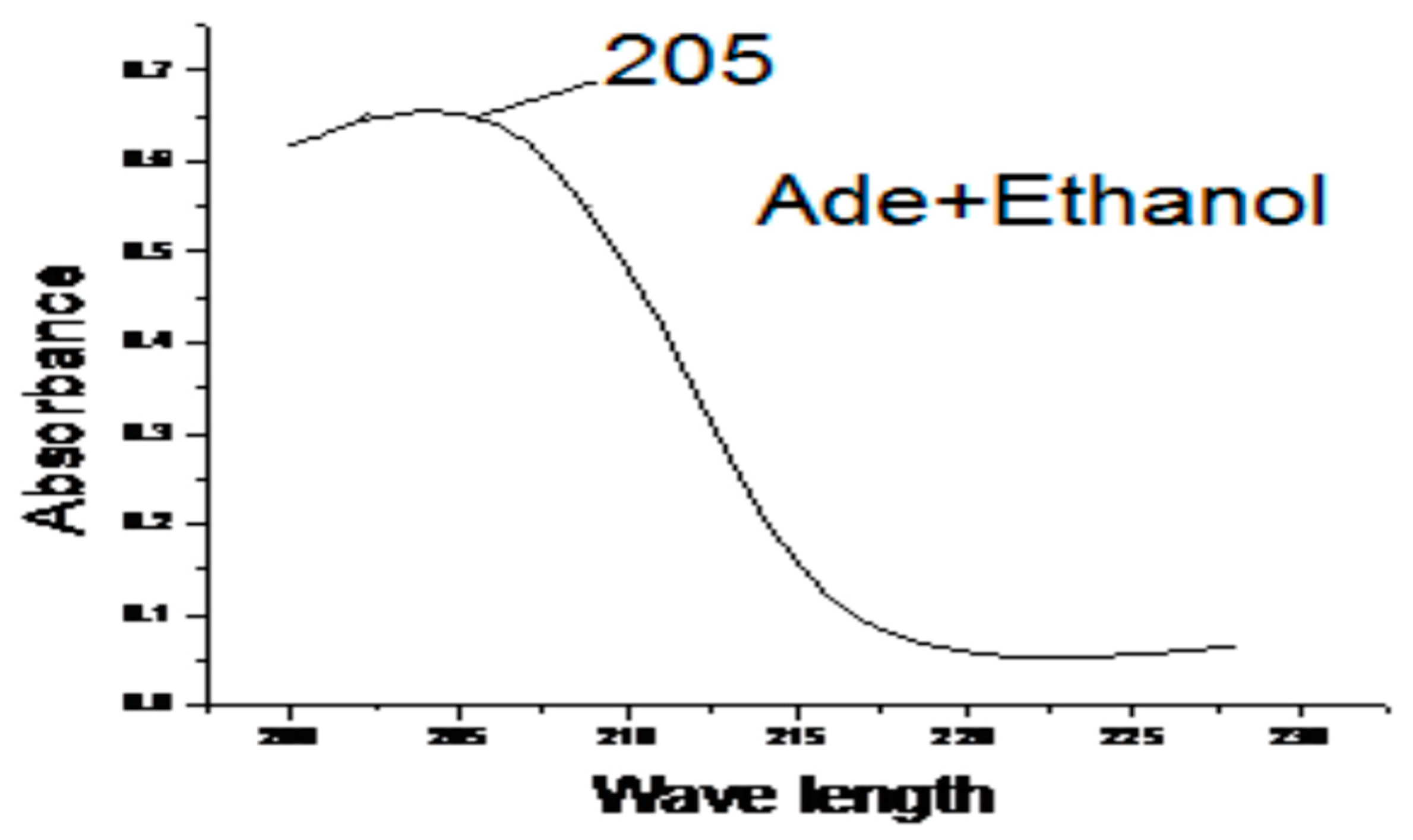
The magnitude of the charge in dipole moment during the electronic transitions, the solvent dipole moment value, and the size of solvent and solute. Many empirical single-solvent polarity parameters have been introduced and have had varying degrees of success correlating solvent-dependent data. The Adenine ligand in solvent (H2O, DMF, DMSO, Ethanol and Methanol) the values of λ max are collected in Table 4. Electronic spectra of Adenine ligand in presence of Ethanol solvents as an example is illustrated in Figure 4. Adenine in Ethanol, and DMF gave bands at 205 nm this bands is due to n―π* electronic transition, this band is blue shifted in presence of water to be at 202 nm, and appear strongly red shifted to be at 256, 218 nm in presence DMSO, and Methanol respectively.

Table 4.
The maximum absorbance of the ligand Adenine at the different solvents.
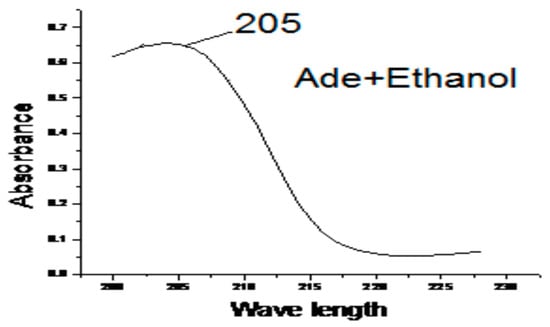
Figure 4.
The effect of Ethanol on the electronic spectra of Adenine.
3.5. Molar Absorbance
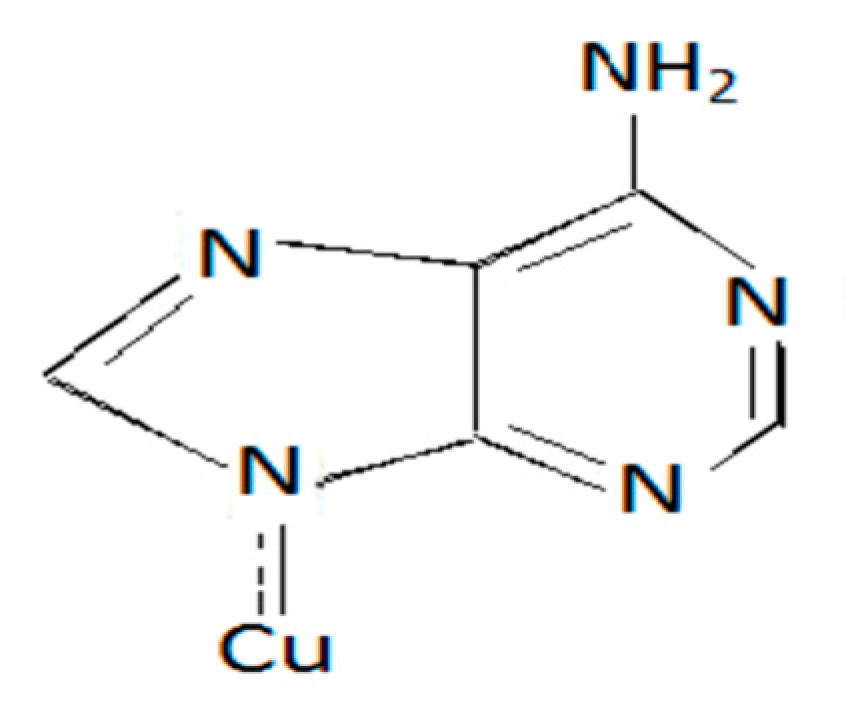
A series of metal–ligand aqueous solutions were prepared with different [L]/[M] ratios. The absorptions of these solutions were measured using UV spectrophotometer at λ max of the expected complex MLx. The proposal structures of the linkage between ligand and metals can be given as in Figure 5.
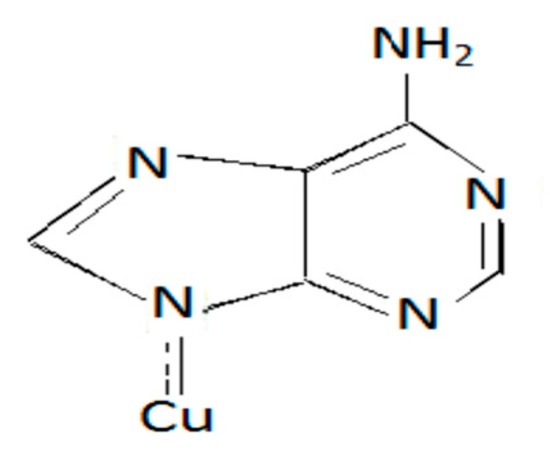
Figure 5.
Cu-Adenine.
It was observed that the absorption increase linearly as the ligand concentration increase, because of the formation of the complex, until the solution reaches the actual molar ratio of the investigated complex. At this point, all of the added materials were completely reacted, and the absorbance observed is the absorption of the investigated complex alone.
After this point, the excess amount of the added ligand causes an inflection in the straight line, that is because the ligand has an absorptance value differ from that of the complex at λ max of the complex. [L]/[M] ratio corresponding to the inflection point in (ABS—[L]/[M] curve) indicates to the actual [L]/[M] ratio of the investigated complex,
Referring to the Figure 5, The data showed that of the studied complexes in this investigation are able to be stable in the form ML, and ML2 were geometric isomerism.
3.6. Antimicrobial Activity
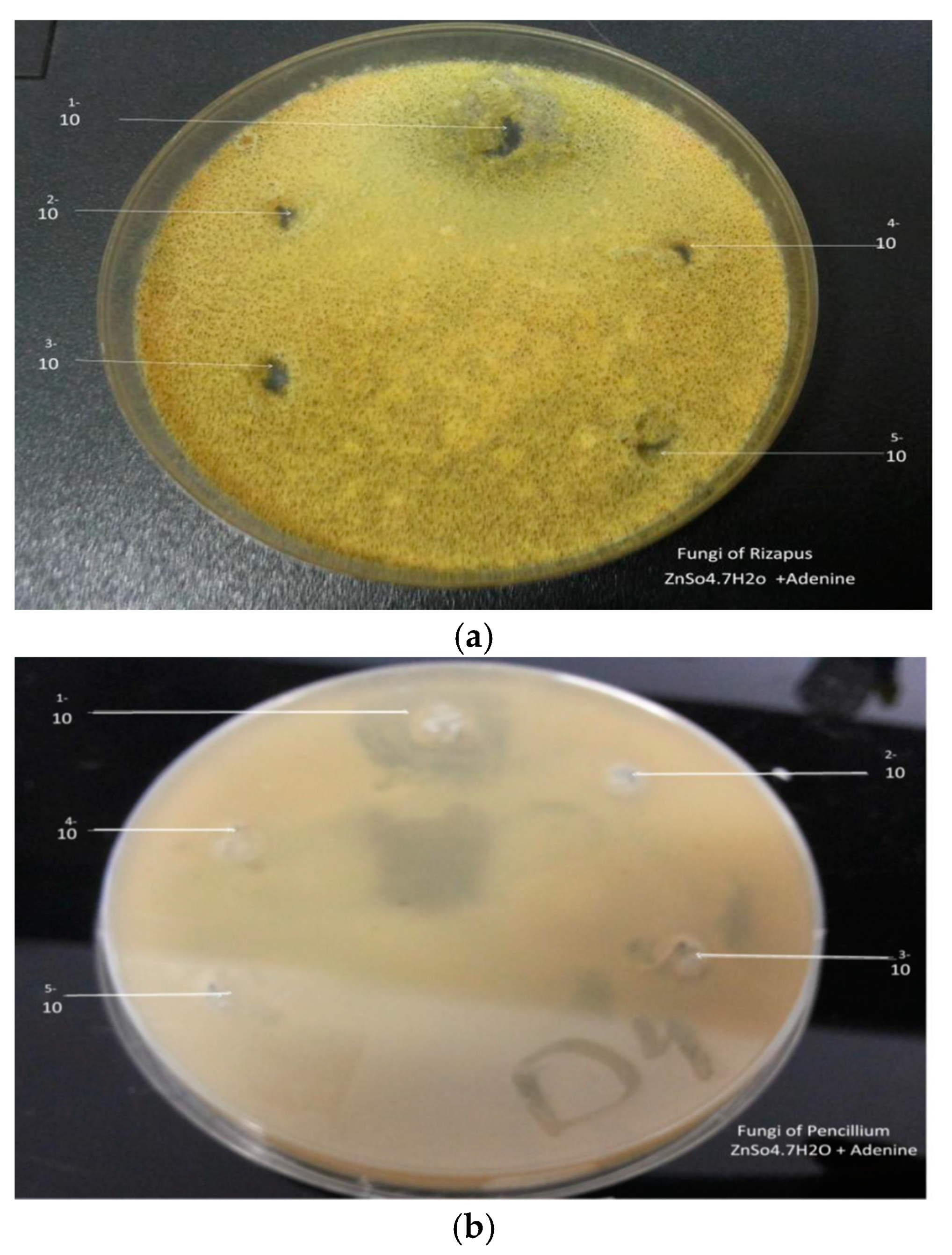
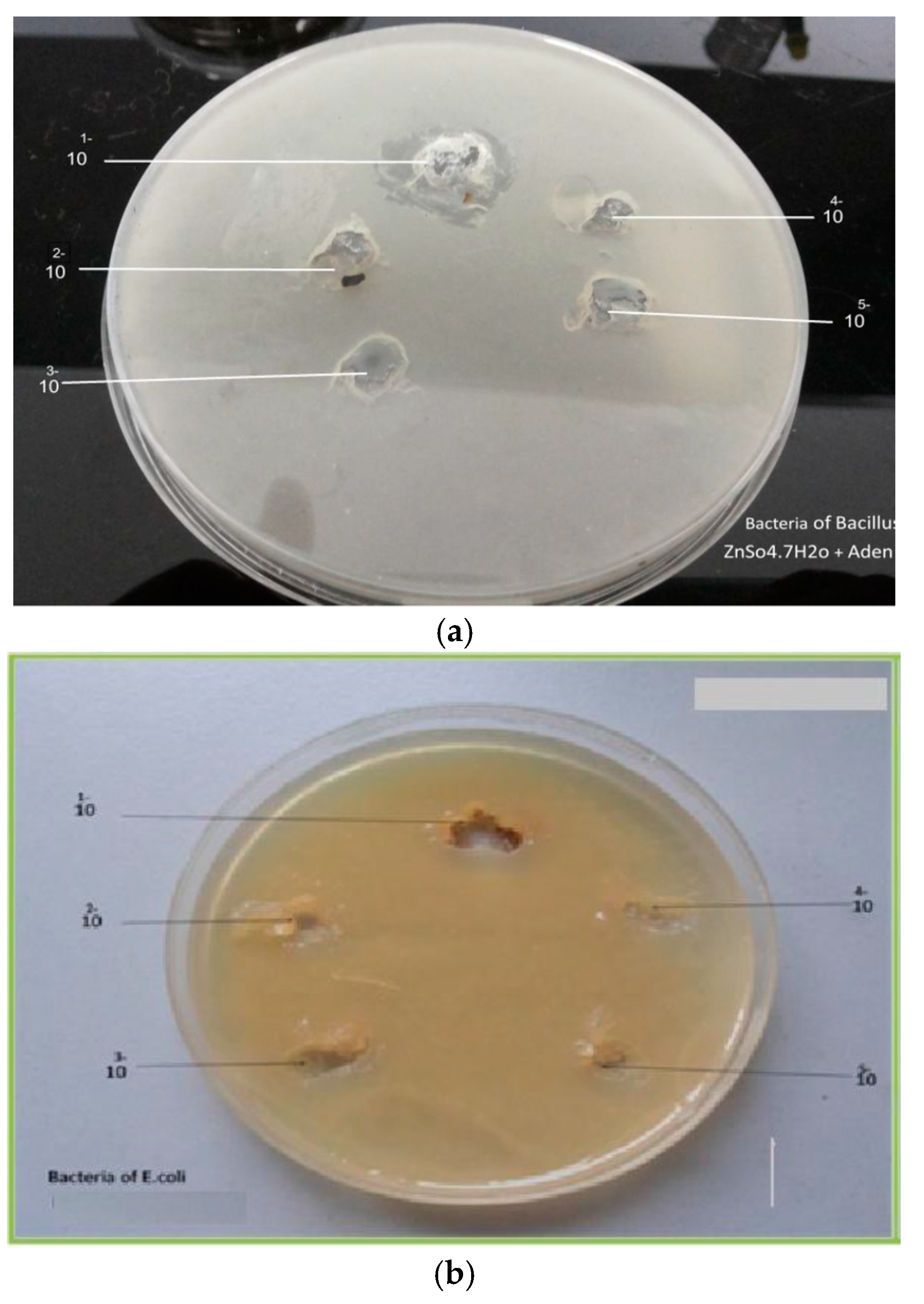
The antimicrobial activity of the seventh prepared complexes were tested against Gram-positive bacteria (Bacillus), and Gram-negative bacteria (Escherichia coli) as in table 5, The products (1g) were dissolved in sterile distilled water at concentration of 10 mL then take (1 mL) of tube (1) and transported into tube (2) and continue the dilution into reach (5) tubes of concentration for each product. The plates were incubated inverted at 37 °C for 24 h in case of bacteria and at 28 °C for 48 h in case of fungi. After incubation, the inhibition zones were recorded in mm. Diameter less than 10 mm indicates no effect. Products (Cd(NO3)·2.4H2O + Ad) and (ZnSO4·7H2O + Ad) each were used as appositive control, only 5(Cd(II)Adenine), (Mn(II)Adenine), (Zn(II)Adenine), (Ni(II)Adenine), (Co(II)Adenine) of the tested products revealed significant antimicrobial effect against the fungal (Rizapus, Pencillium), while the products (Fe(III)Adenine), (Cu(II)Adenine), not revealed significant effect against the fungal (Table 5). In general, the antimicrobial activity for the tested products were higher on Gram-negative (Escherichia coli) than Gram positive(Bacillus) bacteria. For Gram-negative bacteria, E.coli showed higher sensitivity to the tested products (MIC ranged between 12–25 mm), With respect to Gram-positive bacteria (Bacillus) the (MIC level between12–30 mm) the products (Cd(II)Adenine), and (Zn(II)Adenine) more potent against Gram-positive bacteria (Bacillus), Gram-negative[20] (Table 6).

Table 5.
Antimicrobial (Fungal) activity of the tested products.

Table 6.
Antimicrobial (bacteria) activity of tested products.
(Escherichia coli), and fungal (Rizapus, Pencillium), The observed results revealed that, the tested compounds were very active as antimicrobial, Figure 6 and Figure 7) and not gave activity on the applied fungal species.
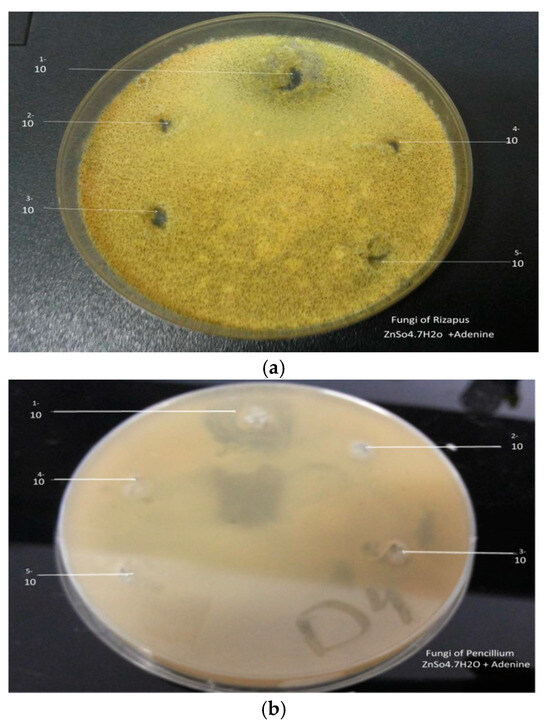
Figure 6.
(a) Effect Zn(II)Ad on Rizapus, (b) Effect Zn(II)Ad on pencillium.
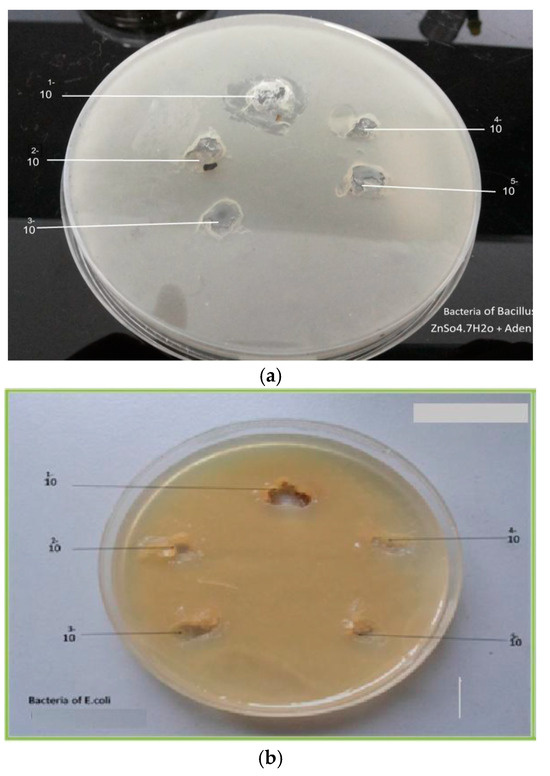
Figure 7.
(a) Effect of Zn(II)Ad on Bacillus, (b) Effect of Zn(II)Ad on E. coli.
4. Conclusions
The spectroscopy methods gave good results to an identification of the metal complexes. The molar ratio calculations gave (ML2) ratio. Generally the study composed simple method to prepare adenine complexes. From the obtained results in this study, the adenine complexes with some metals including Mn2+, Fe3+, Co2+, Ni2+, Cu2+, Zn2+, and Cd2+, gave indication that those complexes have significant effect on some types of bacteria and negative significant against fungi. The methods which used during the reactions between adenine and the selected transition metals showed a quick and simple way.
Funding
This research received no external funding.
References
- Dilip, C.S.; Thangaraj, V.; Raj, A.P. Synthesis, spectroscopic characterization, biological and DNA cleavage properties of complexes of nicotinamide. Arab. J. Chem. 2016, 9, 5731–5742. [Google Scholar]
- Zaworotko, M.J.; Hammud, H.H.; Kabbani, A.; McManus, G.J.; Ghannoum, A.M.; Masoud, M.S. Mamdouh, Synthesis and Characterization of Some Transition Metal Complexes with Mixed Adenine and Acetylacetonate Ligands: Crystal Structures of Solvated Complex {[Cu(acac)2(adenine)]_EtOH} and {[Cu(acac)2(adenine)]_DMF_H2O}. J. Chem. Crystallogr. 2009, 39, 853–863. [Google Scholar] [CrossRef]
- El Azzouzi1, N.; El Fadli, Z.; Metni, R.M. Synthesis and chemical characterization of some transition metal complexes with a 6-acetyl-1,3,7-trimetyllumazine ligand. J. Mater. Environ. Sci. 2017, 12, 4323–4328. [Google Scholar] [CrossRef]
- Köse, D.A.; Necefoglu, H. SYNTHESIS AND CHARACTERIZATION OF BIS(NICOTINAMIDE) m-HYDROXYBENZOATE COMPLEXES OF Co(II), Ni(II), Cu(II) AND Zn(II). J. Therm. Anal. Calorim. 2008, 93, 509–514. [Google Scholar] [CrossRef]
- Lawal1, A.; Ayanwale, A.P.; Obaleye, A.J.; Rajee, O.A.; Babamale, F.H.; Lawal, M. Synthesis, Characterization and Biological Studies of Mixed Ligands Nicotinamide-Trimethoprim Complexes. Int. J. Chem. Mater. Environ. Res. 2017, 4, 97–101. [Google Scholar]
- García-Terán, J.P.; Castillo, O.; Luque, A.; García-Couceiro, U.; Román, P.; Lloret, F. One-Dimensional Oxalato-Bridged Cu(II), Co(II), and Zn(II) Complexes with Purine and Adenine as Terminal Ligands. Inorg. Chem. 2004, 43, 5761–5770. [Google Scholar] [CrossRef] [PubMed]
- Marlin, D.S.; Olmstead, M.M.; Mascharak, P.K. Structure−Spectroscopy Correlation in Distorted Five-Coordinate Cu(II) Complexes: A Case Study with a Set of Closely Related Copper Complexes of Pyridine-2,6-dicarboxamide Ligands. Am. Chem. Soc. 2001, 40, 7003–7008. [Google Scholar] [CrossRef] [PubMed]
- Poltronieri, P.; Čerekovic, N. Roles of Nicotinamide Adenine Dinucleotide (NAD+) in Biological Systems. Challenges 2018, 9, 3. [Google Scholar] [CrossRef]
- Ghose, R. Interaction of carbonylchlorohydridotris (triphenylphosphine) ruthenium (II) with purine, adenine, cytosine and cytidine. J. Chem. Res. 2004, 2004, 257–260. [Google Scholar] [CrossRef]
- Kremplova, M.; Fialova, D.; Nejdl, L.; Hynek, D.; Trnkova, L.; Hubalek, J.; Kizek, R.; Adam, V. Influence of Magnetic Microparticles Isolation on Adenine Homonucleotides Structure. Materials 2014, 7, 1455–1472. [Google Scholar] [CrossRef] [PubMed]
- Di Stefano, M.; Conforti, L. Diversification of NAD biological role: The importance of location. FEBS J. 2013, 280, 4711–4728. [Google Scholar] [CrossRef] [PubMed]
- Onori, G.; Blidaru, D. Optical Spectroscopic Study of the Cu(II) Complexes with Adenosine and Adenine Nueleotides. IL Nuova Cimento D 1985, 5, 339–347. [Google Scholar] [CrossRef]
- Lomozik, L.; Gasowska, A.; Basinski, K.; Bregier-Jarzebowska, R.; Jastrzab, R. Potentiometric and spectral studies of complex formation in the Cu(II), 3′,5′-cyclic adenosine monophosphate, and tetramine systems. J. Coord. Chem. 2013, 66, 261–273. [Google Scholar] [CrossRef]
- Shaker, S.A.; Farina, Y.; Mahmmod, S. Synthesis and Characterization of Mixed Ligand Complexes of Caffeine, Adenine and Thiocyanate with Some Transition Metal Ions. Sains Malays. 2010, 39, 957–962. [Google Scholar]
- Clinical & Laboratory Standards Institute (CLSI). M07-A8 Method for Dilution Antimicrobial Susceptibility Tests for Bacteria that Grow Aerobically, 8th ed.; Clinical & Laboratory Standards Institute (CLSI): Wayne, PA, USA, 2009; ISBN 1-56238-689-1. [Google Scholar]
- Ilavarasi, R.; Muthulakshmi, L. Synthesis and Characterization of Platinum(II) Mixed Ligand Complexes of Purines And N-Glycylglycine. Int. J. ChemTech Res. 2014, 6, 1499–1514. [Google Scholar]
- Cui, Y.; Gong, Y.J.; Su, L.; Wu, C.K.; Pang, S.B.; Feng, S.L. Spectroscopy studies of sandwich-type complex of silver(I) coordinated to nuclear fast red and adenine and its analytical applications. Chem. Pap. 2015, 69, 510–517. [Google Scholar] [CrossRef]
- Masoud, M.; El-Merghany, A.; Ramadan, A.; Abd El-Kaway, M. Thermal studies of some purine compounds and their metal complexes. J. Therm. Anal. Calorim. 2010, 101, 839–847. [Google Scholar] [CrossRef]
- Irani, S.; Atyabi, S.M.; Mivehchi, H.; Siadat, S.D.; Aghasadeghi, M.R.; Farhangi, A. Solvent effects on structural and thermochemical properties of p53 tumor-suppressor gene: A molecular modeling approach in drug design. Int. J. Nanomed. 2011, 6, 2063–2069. [Google Scholar]
- De La Rubia, T.; Gonzalez-Lopez, J.; Moreno, J.; Martinez-Toledo, M.V.; Ramos-Cormenzana, A. Adenine nucleotide content and energy charge of Bacillus megaterium during batch growth in low-phosphate medium. FEMS Microbiol. Lett. 1986, 35, 5–9. [Google Scholar]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).