Analysis Technique by Laser-Induced Breakdown Spectroscopy of Powder Mineral and Slurry Samples †
Abstract
:1. Introduction
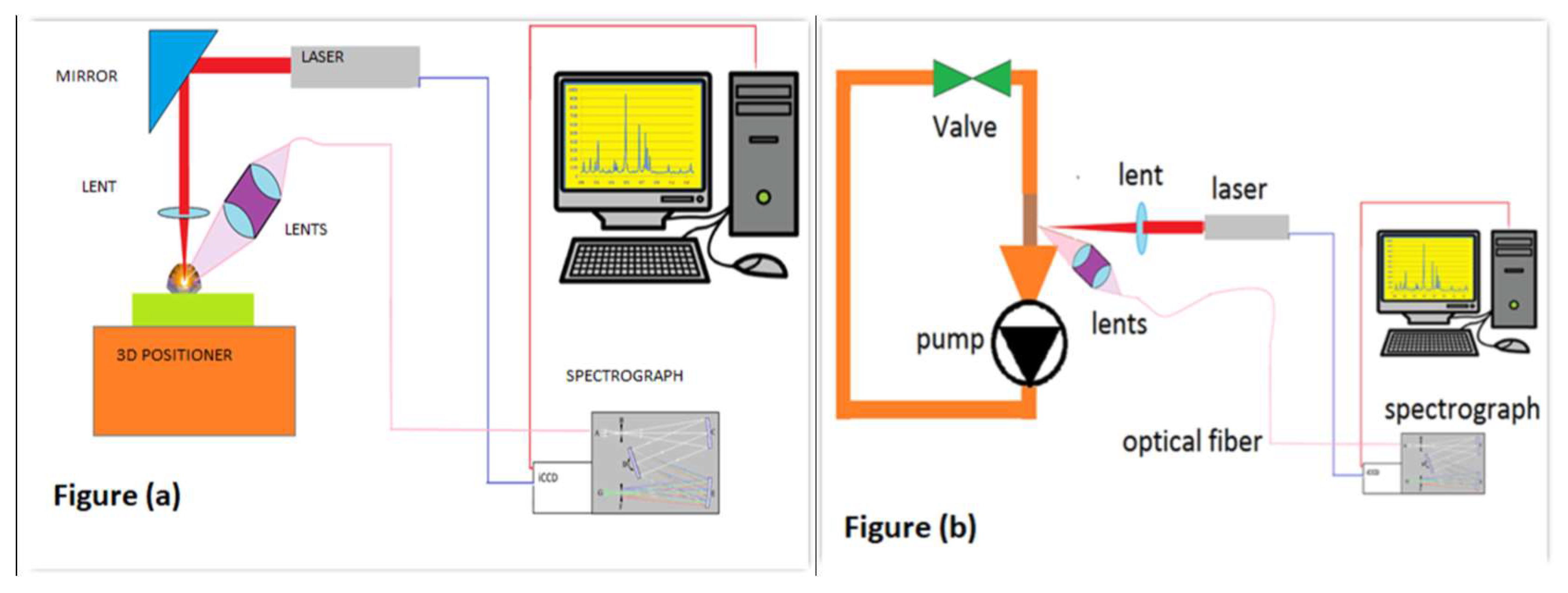
2. Materials and Methods
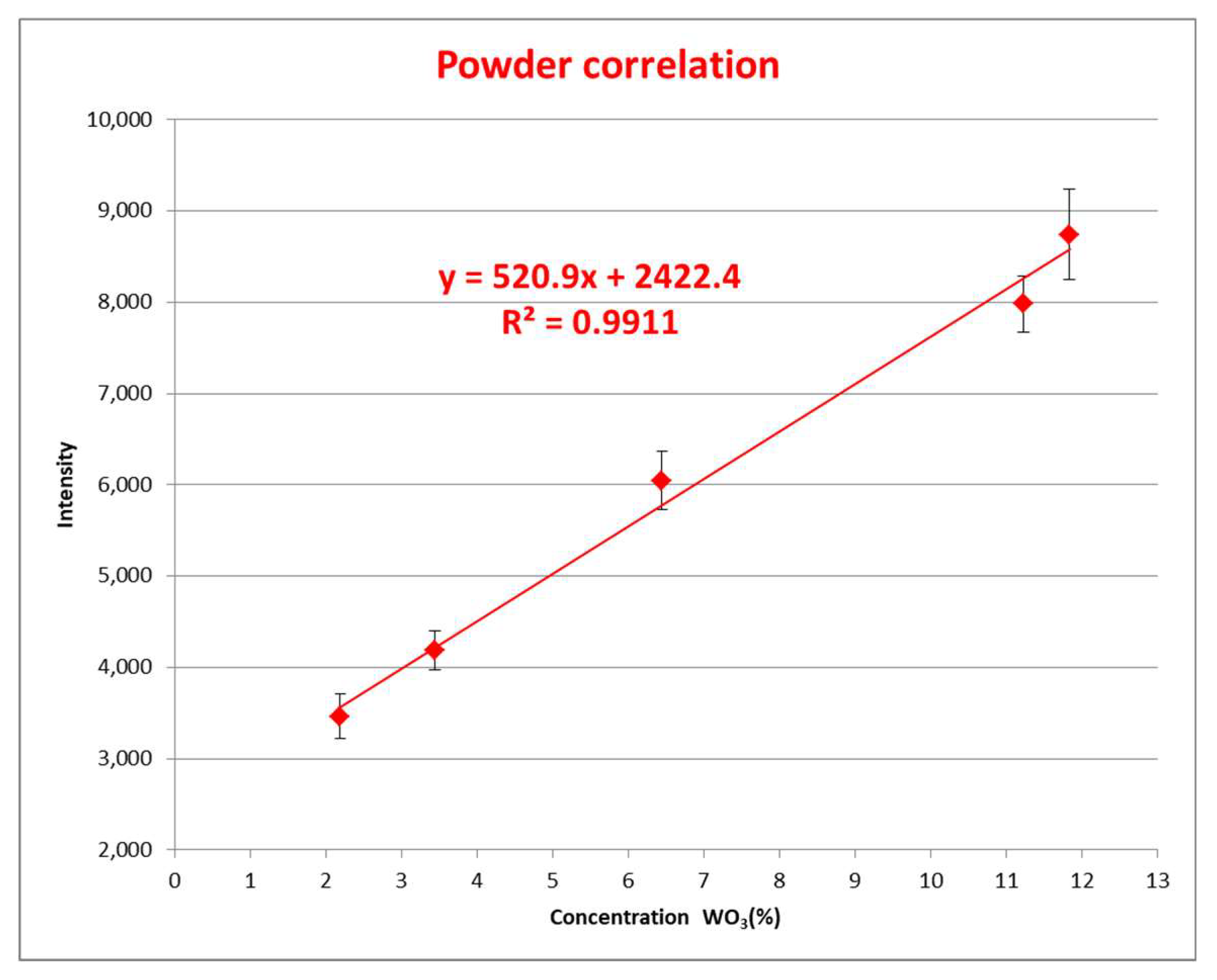
2.1. Methodology for Powder Samples
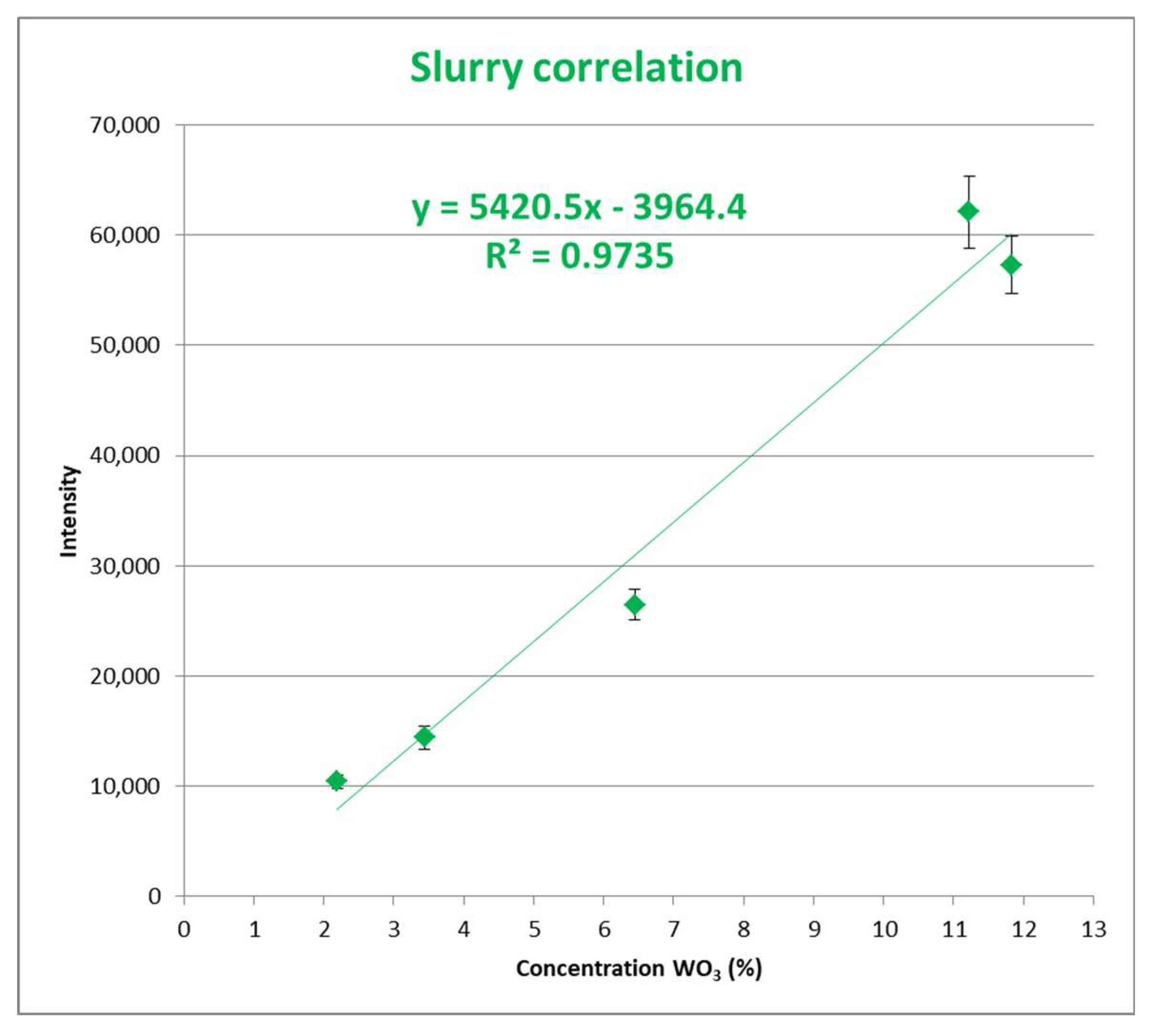
2.2. Methodology for Slurries Samples
3. Analysis of the Result
4. Conclusions
Acknowledgments
References
- Asquini, C.P. 19—Laser induced breakdown spectroscopy (LIBS). In Handbook of Solid-State Lasers; Denker, B., Shklovsky, E., Eds.; Woodhead Publishing: Sawston, UK, 2013; pp. 551–571. [Google Scholar]
- NIST: Atomic Spectra Database Lines Form. Available online: https://physics.nist.gov/PhysRefData/ASD/lines_form.html (accessed on 11 July 2018).
- Strong Lines of Tungsten (W). Available online: http://physics.nist.gov/PhysRefData/Handbook/Tables/tungstentable2.htm (accessed on 31 March 2017).
- Álvarez, C.; Pisonero, J.; Bordel, N. Quantification of fluorite mass-content in powdered ores using a Laser-Induced Breakdown Spectroscopy method based on the detection of minor elements and CaF molecular bands. Spectrochim. Acta Part B At. Spectrosc. 2014, 100, 123–128. [Google Scholar] [CrossRef]
- Khajehzadeh, N.; Haavisto, O.; Koresaar, L. On-stream and quantitative mineral identification of tailing slurries using LIBS technique. Miner. Eng. 2016, 98, 101–109. [Google Scholar] [CrossRef]
| Sample | Concentration WO3 (%) |
|---|---|
| Sample A | 11.829 |
| Sample B | 11.224 |
| Sample C | 6.444 |
| Sample D | 3.443 |
| Sample E | 2.194 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Marcos-Robredo, G.; Rey-Ronco, M.A.; Alonso-Sánchez, T. Analysis Technique by Laser-Induced Breakdown Spectroscopy of Powder Mineral and Slurry Samples. Proceedings 2018, 2, 1461. https://doi.org/10.3390/proceedings2231461
Marcos-Robredo G, Rey-Ronco MA, Alonso-Sánchez T. Analysis Technique by Laser-Induced Breakdown Spectroscopy of Powder Mineral and Slurry Samples. Proceedings. 2018; 2(23):1461. https://doi.org/10.3390/proceedings2231461
Chicago/Turabian StyleMarcos-Robredo, German, Miguel Angel Rey-Ronco, and Teresa Alonso-Sánchez. 2018. "Analysis Technique by Laser-Induced Breakdown Spectroscopy of Powder Mineral and Slurry Samples" Proceedings 2, no. 23: 1461. https://doi.org/10.3390/proceedings2231461
APA StyleMarcos-Robredo, G., Rey-Ronco, M. A., & Alonso-Sánchez, T. (2018). Analysis Technique by Laser-Induced Breakdown Spectroscopy of Powder Mineral and Slurry Samples. Proceedings, 2(23), 1461. https://doi.org/10.3390/proceedings2231461




