A New Integrated Multi-Pathogen In Situ Detection Platform Using Electrochemical Impedance Spectroscopy †
Abstract
:1. Introduction
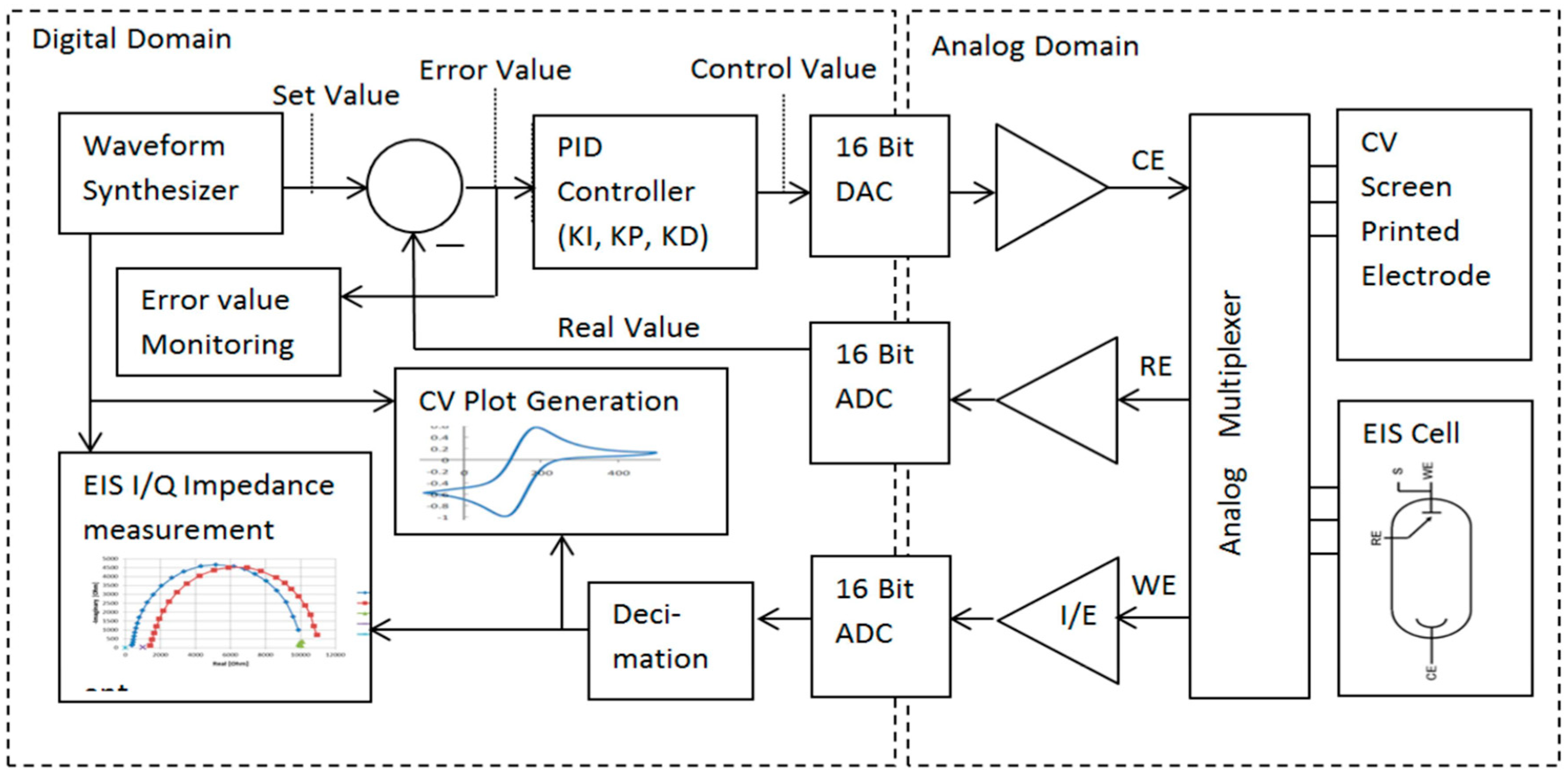
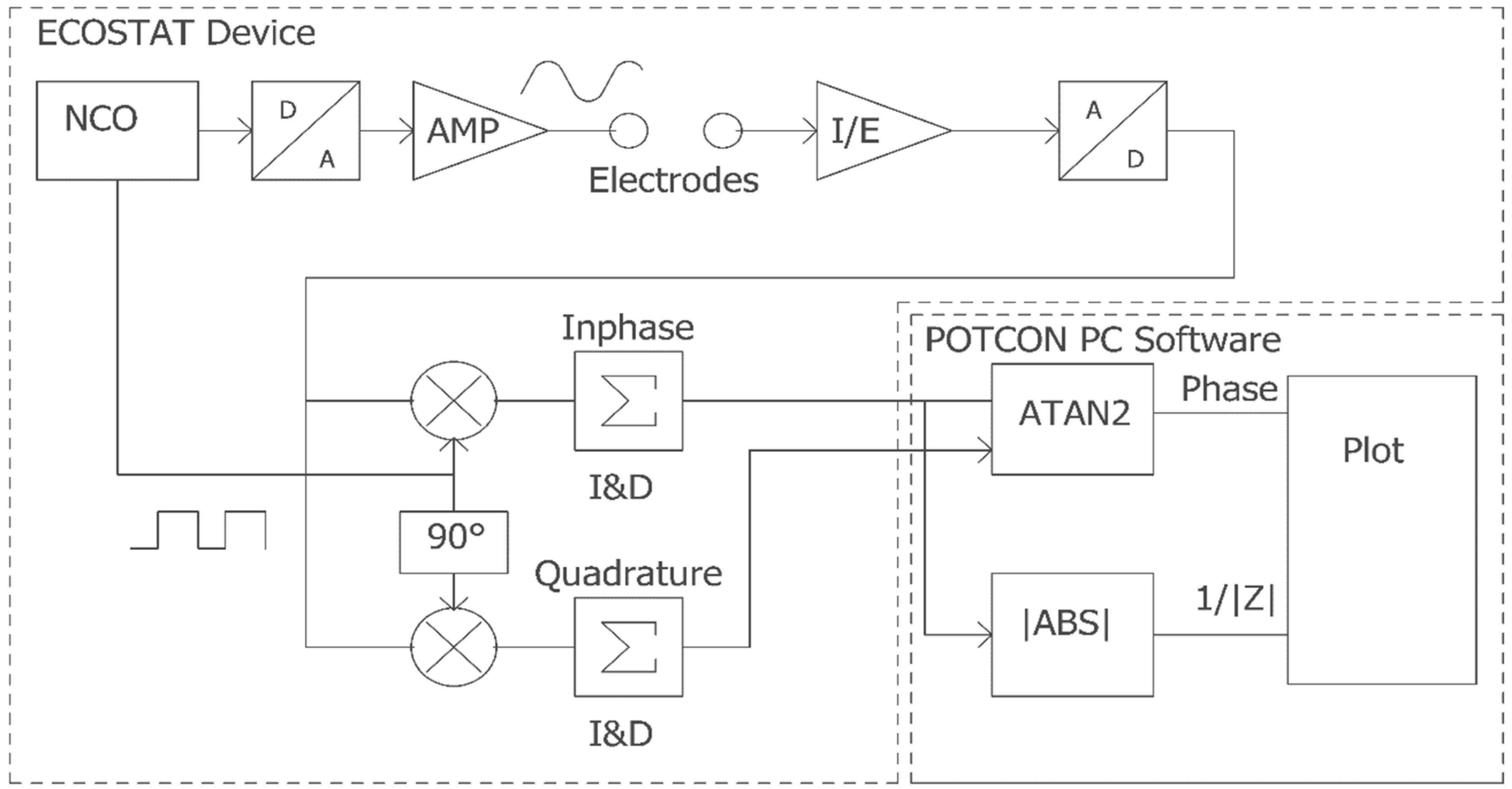
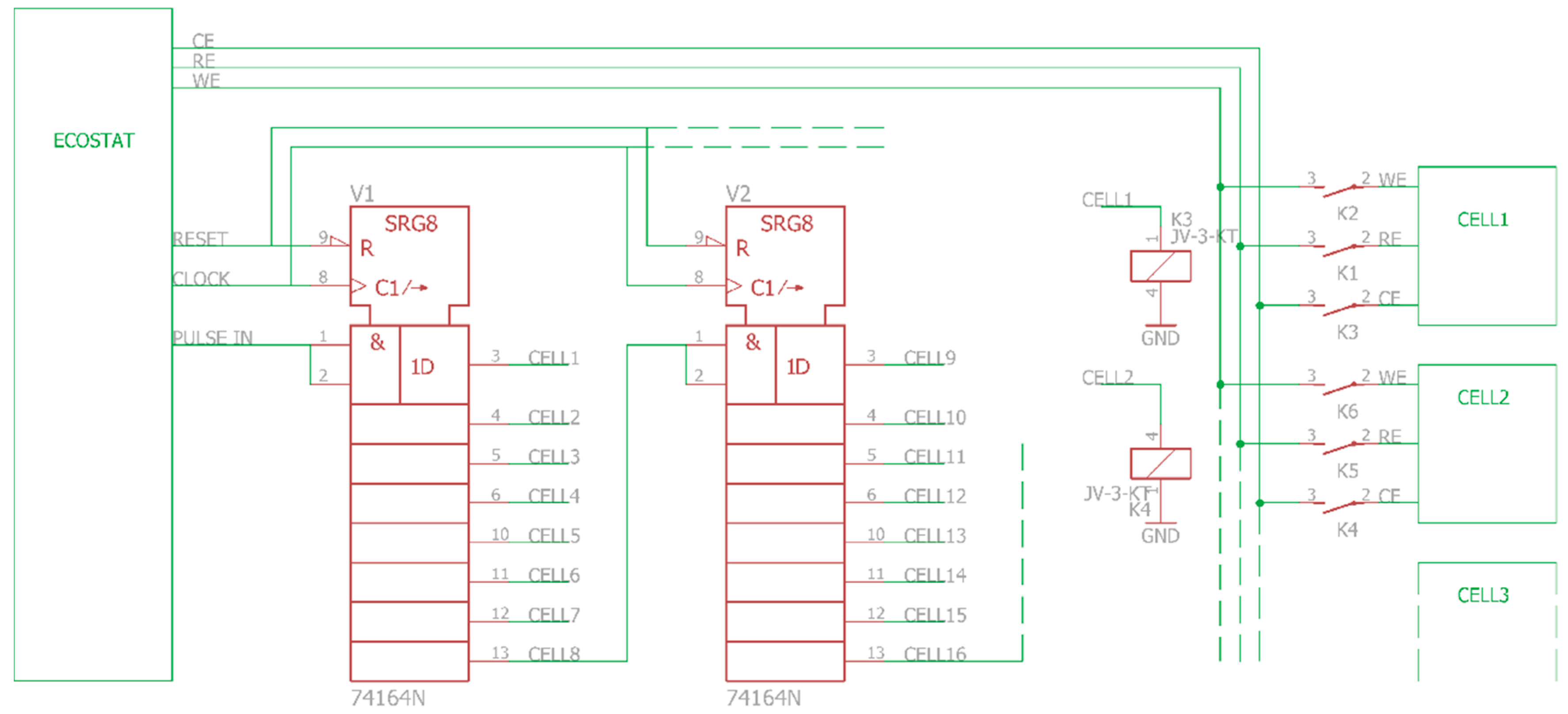
2. Principle of Operation
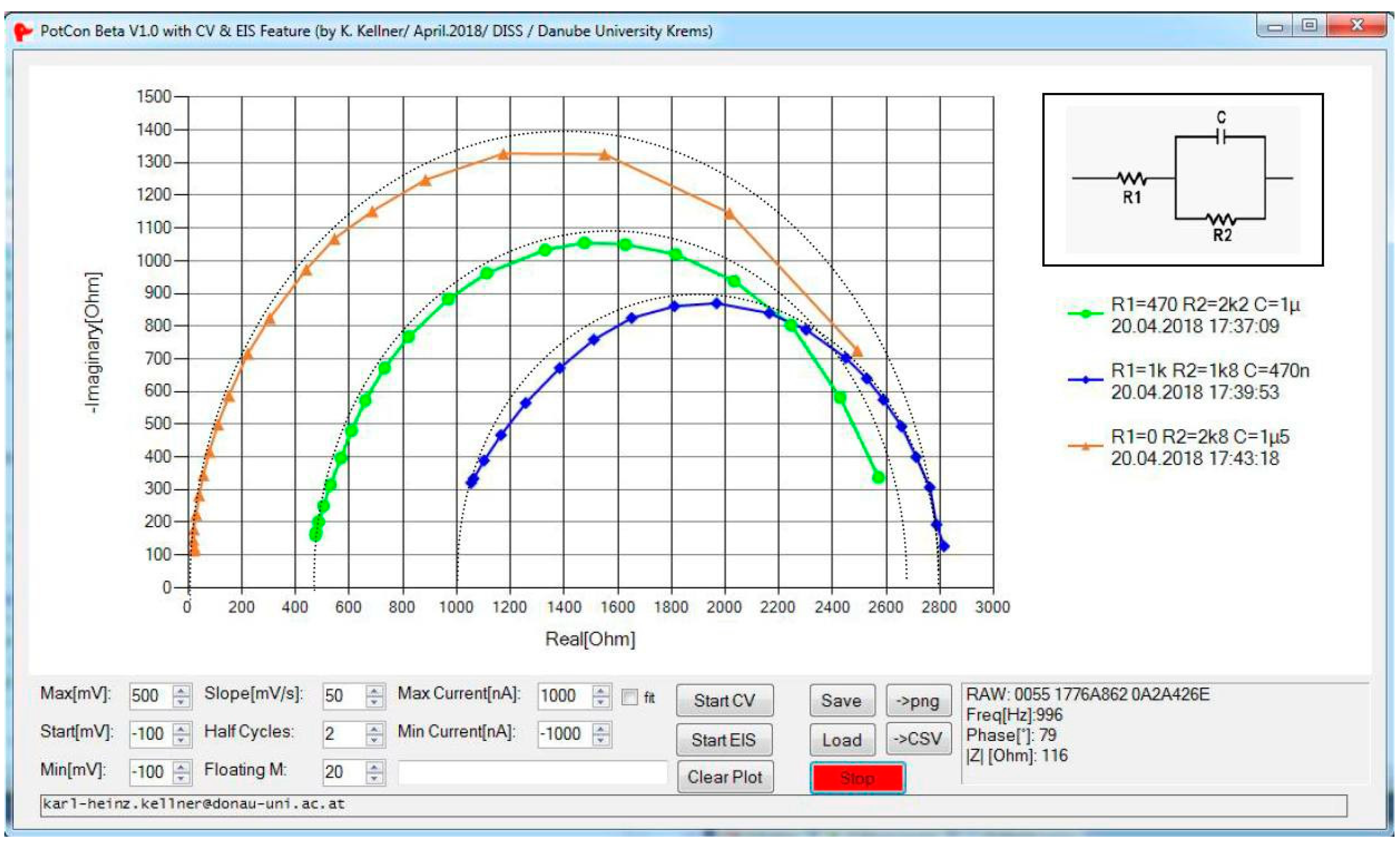
3. Results
4. Conclusions and Outlook
Acknowledgments
Conflicts of Interest
References
- Hayat, A.; Marty, J.L. Disposable screen printed electrochemical sensors: Tools for environmental monitoring. Sensors 2014, 14, 10432–10453. [Google Scholar] [CrossRef] [PubMed]
- Kellner, K.; Posnicek, T.; Ettenauer, J.; Zuser, K.; Brandl, M. A new, low-cost potentiostat for environmental measurements with an easy-to-use PC interface. Procedia Eng. 2015, 120, 956–960. [Google Scholar] [CrossRef]
- Kellner, K.; Ettenauer, J.; Zuser, K.; Posnicek, T.; Brandl, M. An automated, robotic biosensor for the electrochemical detection of E. coli in water. Procedia Eng. 2016, 168, 594–597. [Google Scholar] [CrossRef]
- Randles, J.E.B. Kinetics of rapid electrode reactions. Discuss. Faraday Soc. 1947, 1, 11–19. [Google Scholar] [CrossRef]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kellner, K.; Ettenauer, J.; Brandl, M. A New Integrated Multi-Pathogen In Situ Detection Platform Using Electrochemical Impedance Spectroscopy. Proceedings 2018, 2, 1506. https://doi.org/10.3390/proceedings2131506
Kellner K, Ettenauer J, Brandl M. A New Integrated Multi-Pathogen In Situ Detection Platform Using Electrochemical Impedance Spectroscopy. Proceedings. 2018; 2(13):1506. https://doi.org/10.3390/proceedings2131506
Chicago/Turabian StyleKellner, Karlheinz, Jörg Ettenauer, and Martin Brandl. 2018. "A New Integrated Multi-Pathogen In Situ Detection Platform Using Electrochemical Impedance Spectroscopy" Proceedings 2, no. 13: 1506. https://doi.org/10.3390/proceedings2131506
APA StyleKellner, K., Ettenauer, J., & Brandl, M. (2018). A New Integrated Multi-Pathogen In Situ Detection Platform Using Electrochemical Impedance Spectroscopy. Proceedings, 2(13), 1506. https://doi.org/10.3390/proceedings2131506




