Abstract
Sugar beet fertilization is a very complex agrotechnical measure for farmers. The main reason is that technological quality is equally important as sugar beet yield, but the increment of the root yield does not follow the root quality. Technological quality implies the concentration of sucrose in the root and the possibility of its extraction in the production of white table sugar. The great variability of agroecological factors that directly affect root yield and quality are possible good agrotechnics, primarily by minimizing fertilization. It should be considered that for sugar beet, the status of a single plant available nutrient in the soil is more important than the total amounts of nutrients in the soil. Soil analysis will show us the amount of free nutrients, the degree of soil acidity and the status of individual elements in the soil so that farmers can make a compensation plan. An estimate of the mineralizing ability of the soil, the N min, is very important in determining the amount of mineral nitrogen that the plant can absorb for high root yield and good technological quality. The amount of N needed by the sugar beet crop to be grown is an important factor, and it will always will be in the focus for the producers, especially from the aspect of trying to reduce the N input in agricultural production to preserve soils and their biodiversity but also to establish high yields and quality.
1. Introduction
Sugar beet (Beta vulgaris L. subsp. vulgaris var. altissima Döll.) and sugar cane (Saccharum officinarum L.) are the plants that are mostly grown in the world as raw materials for processing into sugar. Sugar cane cultivation for sugar production began in India in 2000 BC, while sugar beet is a relatively young plant and has been grown since the late 18th century. Sugar beet is biologically adapted to the temperate climate zone and is grown in the area from 30° to 60° north latitude and from 25° to 35° south latitude. Nowadays, sugar beet accounts for 20% of world sugar production, while the remaining 80% of sugar is from sugar cane [1]. The most suitable geographical conditions for beet growing are found where the soil is deep, friable, well drained and contains a certain proportion of lime. Sugar beet is usually cultivated as a rotation crop. Even though there is a need for agrotechnical measures, it would be interesting to try the adoption of conservation tillage, which provides considerable environmental benefits while improving soil fertility [2,3,4,5,6]. Sugar beet yields under reduced tillage are similar to those of beets grown in plowed soil when the N application increases [7].
The sugar content in sugar beet root is usually 13–20% [8,9]. The total biomass of the sugar beet plant is divided into root and leaf dry matter. According to Hoffmann and Kenter [10], sugar beet root yield has increased in the past decades by 1.5% per year, but the sugar content has increased at the expense of the plant canopy (or leaf dry matter). More than 98% of total root sugar is sucrose, fructose and glucose in very small amounts [11].
Sugar beet fertilization significantly impacts yield formation and sugar beet root quality. Nitrogen is probably the most studied nutrient for sugar beet because N is the most limiting nutrient directly related to sugar beet yield and quality. Many researchers reported that N fertilization greatly influences excessive N fertilization, resulting in more lush development of the leaves and crowns, root maturity is slowed down, and the sugar content in the root is lower. In addition, root quality may be reduced due to the increased content of melas-forming elements [12,13,14,15].
As one of the main building blocks, nitrogen has a special significance in plants because it is an integral part of proteins from which protoplasm, cells and plant tissues are formed [16,17]. In addition, N is the main yielding element in plant production. The range of N concentration in the leaf blade of sugar beet is from 2.2 to 3.5% and in the leaf stalk from 1.0 to 1.5% [18]. Sugar beet needs a large amount of N as a nutrient. The amount of N applied to the soil should be performed according to the chemical and microbiological properties of the soil. Furthermore, weather conditions should be considered to avoid unused amounts of N by plants that will, due to extreme instability of this nutrient, move into the deeper layers of the soil and bring about groundwater eutrophication [19,20]. Moreover, excessive amounts of N fertilizers negatively affect the content of melas-forming substances and primarily alpha-amino [21,22,23].
2. Determination of N Rate: Importance of EUF and N Min Methods
Chemical analytical methods and techniques are used to make crop fertilization recommendations based on nutrient status in the soil [24]. The choice of analytical methods should consider that any analytical method is good if there is a well-designed system of interpretation of analytical values. The interpretation of the results uses tables of limit values, which give more indicative results because nutrient bioavailability is not a static value. It depends on a number of factors: biological, physical, hydrological, agrotechnical [25]. Classical methods of soil analysis are basically one-off extractions that estimate only the available nutrients in the soil and not the reserves of nutrients. Determining the available amount of nutrients in the soil is a common basis for recommending fertilization and application of fertilizers. Therefore, analytical and prognostic methods are required to provide insight into the amount of available nutrients status in the soil, the rate of change in the amount of identified nutrients during the growing season (e.g., for absorption, rinsing or fixation) and the amount of nutrients to be added for optimal plant nutrition. In some cases, the calculation also considers the need to increase the level of some nutrients in the soil to the target value.
The electro-ultrafiltration (EUF) method is the extraction of both the inorganic (EUF-NO3) and organic (EUF-Norg) EUF-N fractions, but it can also extract synthetic amino acids dissolved in water [26,27]. The plant nutrients determined are separated from solutions in the form of ions, cations (Ca2+, K+, Mg2+, NH4+, Mn2+, Fe3+) and anions (NO3−, SO42−, BO33−). By changing the temperature and voltage of the current in soil–water suspensions from the soil, weaker or more tightly bound nutrients are separated. The extraction medium is distilled water, and during an extraction under the influence of electric fields (direct current) from the suspension, differently charged ions and particles are detached from the anodes and cathodes [28]. This process first separates easily soluble nutrients and then bound nutrient fraction, which is gradually mobilized during the growing season from the soil. For the EUF method, the extracted inorganic N (main nitrate) and organic N are the basis for the N fertilizer recommendation [29], since sugar beet uses both N fractions (EUF-NO3 and EUF-Norg). The EUF method for soil analysis was introduced in Austria and Germany in 1974 and 1982 [30]. Since its introduction, more than 1.2 million soil samples have been analyzed by EUF [31]. Except for sugar beet, the method was used for other field crops, such as cereals, potatoes and vineyards.
For EUF calculation of the N fertilizer requirements, it is suggested that the sum of the EUF-extractable N amounts be used. For sugar beet grown as a spring crop in the climatic conditions of western and central Europe, the N supply during the summer months (May–September) is very important because it determines the yield and root quality. In Austria, Yugoslavia and Denmark, it was determined that over three years, the EUF-N value of 1 mg 100 g−1 soil in the period between June and September was equivalent to 40 kg ha−1 N [32]. Additionally, in addition to the EUF, mechanical analysis is often used, and in the spring planting, N status is often checked by the N min method.
With the N min method, the concentration and total amount of mineral N forms in the soil—nitrate (N-NO3), ammonia (N-NH4+) and soil moisture—were determined [33,34]. The N min method determines the amount of mineral forms of N (available N) for spring crops before sowing and before sowing and harvesting for winter cereals in the zone that is reached by the root system. Therefore, instead of an individual determination, the N min method represents a synthesis of several factors of N availability (previous crop residual N, harvest residues, soil and climatic conditions [35]). The N min calculator is intended for N fertilization and N starter fertilization of winter cereals, corn and sugar beet. Computer model fertilization recommendation based on the N min method considers all necessary parameters (the content of mineralized N in the soil, soil moisture, sampling time and soil texture) for estimating N doses. The disadvantages of the N min method are the impracticality of the short time between sampling and performing the analysis until the application of the recommendation for fertilization, and with the EUF method, a great variability of results, especially on clay soils [36]. For sugar beet as a spring crop, the soil samples for the N min method need to be sampled in the third week of February and in the first decade of March. A total of 20–25 individual samples were taken from an area of no more than 10–20 ha from the same pre-crop, the same type of soil and uniform terrain configuration and composited into a 0.5–1 kg sample. Individual samples were taken from a depth of 0–30 cm, 30–60 cm, 60–90 cm.
3. Nitrogen Fertilization Influence on Vegetative Growth
Malnou et al. [16] point out that it is desirable for sugar beet to reach 85% of the canopy, which covers the soil, as soon as possible to maximize the use of solar radiation, while Jaradat and Rinke [37] point out that newer genotypes can achieve up to 90% soil coverage. According to a two-year study in Germany (Göttingen), Hoffman and Kluge-Severin [38] state that under temperate climatic conditions, sugar beet as a spring crop absorbed only 24% of the incoming radiation in May and up to 83% in June.
The most intense leaf formation in European production area is from the beginning of June, when sugar beet leaves close the rows and last until mid-July [39]. After that, the plant forms leaves of smaller dimensions, and the leaves gradually die off by the end of the vegetation. The growth of sugar beet roots through vegetative stages has a linear trend, and the largest root growth occurs from mid-July to mid-August.
The leaf rosette of sugar beet reaches its maximum at the end of July and the beginning of August, while toward the end of the vegetation, it gradually decreases [6,40]. According to Kristek and Liović [41] and Jelić et al. [42], in agroecological conditions of southeast Europe, the largest daily increase in sugar beet leaves is from mid-June to mid-July.
Manderscheid et al. [43] state that a higher amount of N (126 and 156 kg ha−1 N) compared to a smaller one (63 and 78 kg ha−1 N) resulted in an increase in fresh leaf mass and an increase in individual leaves through the vegetation (11th, 16th, 26th and 31st leaf), while statistically significant differences were not found between different N fertilizations and the number of sugar beet leaves. Lüdecke [44] states that the development of sugar beet in conditions of reduced N supply at the beginning of vegetation is the same because beets have enough N available. The author emphasizes that sugar beet matures earlier in conditions of lower N supply, and phenotypically, the leaves are lighter in color, while the root lags behind in growth.
Newer genotypes have erect leaves that allow growth in a smaller vegetation area [45]. Vukadinović et al. [46] point out that hybrids with more upright leaves tolerate shading better, i.e., they can be grown in higher density. In addition to the above, in terms of photosynthesis, Müller-Linow et al. [47] emphasize the importance of the vertical distribution of leaves, i.e., the angle at which the leaves are located on the rosette of sugar beet.
In spring sowing, the dying of sugar beet leaves is more intense after July. Kenter et al. [48] point out that air temperature and radiation have a very significant impact (p < 0.001) on the growth of sugar beet leaves in the first 65 days after sowing (early June) by growing sugar beet at 27 locations in Germany.
In the phase of intensive leaf growth (mid-June to the end of July), the total leaf area of one plant is 2000 to 6000 cm2 [12,40,49], while the optimal leaf area index (LAI) of sugar beets in this period is from 3 to 4 m2 m−2 [12,50,51,52,53]. With the optimal leaf area index of sugar beet (3 to 4 m2 m−2), the outer leaves use almost all of the sun’s energy. If the LAI is higher than optimal, the photosynthesis of the lower leaves is reduced, the crop is not drought resistant and has a greater need for nutrients. Manderscheid et al. [43] emphasize the importance of N fertilization and state that depending on N fertilization, a different number of dry sugar beet leaves was found in August and September. Furthermore, the authors point out that sugar beet had an average of 1.58 dry leaves in late June and early July, 5.58 in August and 9.55 dry leaves in the second half of September.
Based on the analysis of eleven hybrids of the Institute of Field and Vegetable Crops in Novi Sad, Putnik-Delić [54] emphasizes the importance of the genotype itself on the formation of the leaf rosette. The author points out that in young beets in the 6–12 leaves phase, the leaf area per plant differs depending on the genotype (414.24 cm2 to 1099.89 cm2). According to Drachovská and Šandera [49], the differences in leaf area between genotypes in the phase of intensive root growth (11 July–11 August) were not so pronounced, which was confirmed by the example of leaf area of yield and sugar genotype (average 2741.5 cm2 and 2892.3 cm2). In the analysis of LAI, Tsialtas and Maslaris [52] again emphasize the importance of genotype. According to their research in Greece on 12 sugar beet genotypes, it was found that in the phase of the largest leaf area, in early July, LAI ranged from 1.78 m2 m−2 (hybrid Ramona) to 5.03 m2 m−2 (hybrid Dorothea).
Kosterj and Repka [55] obtained an LAI value from 0.38 m2 m−2 to 5.36 m2 m−2 during vegetative growth in the first year of vegetation. In 11 sampling dates (from June to October), Tsialtas and Maslaris [56] found very significant changes in LAI (hybrid Rizor). The authors also state that toward the end of the vegetation, the leaves become smaller in size (leaf area, length decreases, width and radius), and the leaf blade becomes more rounded.
Kristek and Halter [39] conclude that with a smaller number of plants per unit area, sugar beet cannot make full use of vegetation space, and due to a higher nutrient supply, the N uptake was higher, the root sugar content was reduced, and sugar yield was lower.
As with most field crops, N is an important nutrient in sugar beet. Sugar beet requires higher amounts of N to achieve maximum yields, so fertilization is one of the most important agronomic measures in sugar beet production. Excessive as well as scarce fertilization, especially N, can lead to loss of yield or reduced quality of sugar beet roots [57]. In conditions of scarce N supply, the sugar beet develops a smaller assimilation area and a smaller root with more sucrose, but the sugar yield per unit area is lower due to lower root yield [35].
Considering the needs of sugar beet for N, Pospišil [16] points out that sugar beet needs the most N in the phase of intensive leaf growth, which, in our agroecological conditions, most often occurs from early June, i.e., from the closing of rows, until mid-July. On the other hand, there is the least need for N in beets at the time of intensive accumulation of sugar in the root, so N’s availability in the soil should be lower to prevent the formation of undesirable N compounds that prevent sugar separation.
Malnou et al. [16] state that sugar beet needs 0.04 g cm−2 N leaf to develop 85% of the leaf rosette, which corresponds to 120 kg ha−1 N, so it is very important to supply the plant with enough N so that the leaf rosette can better harness solar energy.
Sugar beet also absorbs the ammonium form of N (NH4+) well, which, due to physiological processes within the root cells, can negatively affect the sugar content in the root, breaking it down or preventing the synthesis of sucrose. Therefore, in the spring, before sowing and in the feeding of sugar beet, fertilizers that have an ammonium form of N, such as urea (CO(NH2)2, 46% N) and UAN (urea + NH4NO3 + water, 30% N), should be avoided. This is confirmed by Brentrup et al. [58] who stated that the yield of sugar beet when applying 115 kg ha−1 N in the form of calcium ammonium nitrate (CAN) was 47.7 t ha−1, and the yield of sugar was 8.49 t ha−1, while with the same amount of N from urea, root yield was lower at 44.2 t ha−1 and sugar yield at 7.31 t ha−1.
After two years of research in Croatia, Kristek et al. [57] state that basic fertilization with 100 kg ha of urea increases root yield from an average of 57.6 t ha−1 (control) to 61.4 t ha−1, root sugar content from an average of 15.7% (control) to 15.8% and sugar yield from an average of 7.7 t ha−1 (control) to 8.3 t ha−1. Furthermore, the same authors state that top dressing leaves in phase 2–4 with 50 kg ha−1 CAN without basic fertilization in the autumn gave, on average, the best results: root yield 68.1 t ha−1, sugar content in root 15.9% and sugar yield 9.31 t ha−1.
Draycott [59] states that sugar beet root at harvest contains phosphorus in the amount of 0.8 kg t−1 of fresh matter, potassium 1.7 kg t−1 fresh matter, while the whole plant in the vegetation can take a total of 1.9 kg t−1 fresh matter phosphorus and potassium in the amount of 7.9 kg t−1 fresh matter.
4. Sugar Beet N Fertilization Management
In most soils in Croatia, sugar beet fertilization is 140–160 kg ha−1 N, 80–130 kg ha−1 P2O5 and 150–250 kg ha−1 K2O [17]. In sugar beet production, phosphorus and potassium are usually applied in the fall, while one-half to one-third of the total required N was added in the fall and the rest in the spring.
Märländer et al. [50] state that in Germany, in the last 20 years, the use of phosphorus fertilizers in sugar beet cultivation has been reduced from 65 to 20 kg ha−1 and potassium fertilizers from 200 to 60 kg ha−1. The authors point out that in order to achieve satisfactory yields, N was applied in the amount of 120 kg ha−1 N, and such practice is in use in most countries of the European Union. The main reason for that is that phosphorus fertilizer resources worldwide are very small in relation to nitrogen and potassium fertilizers [51,52,53,54]. Fertilizers should be used only if there is a proven lack in the soil to avoid overapplication and environmental pollution [55,56,57,58]. Soil analysis must be performed before use of fertilizers, and the dosage can be partly reduced by adding manure [59,60,61,62,63,64,65,66,67,68,69].
Draycott [70] points out that in many European countries, it is the practice to add all the N needed to achieve maximum sugar beet yields in the spring in the amount of 30 to 40 kg ha−1, which should be added before sowing, while the rest is added in top dressing in phase 2–4 leaves. In our conditions, 60–80 kg of ha−1 N is most often added before sowing [14,16,50].
The greatest need of sugar beet for nutrients is in the phase of intensive leaf growth (from early June to mid-July). Sugar yield is linearly related to dry matter yield and total N uptake. However, Last et al. [71] point out that increasing N above 200 kg ha−1 of N does not increase sugar yield, and the maximum sugar yield over six years of research was obtained by applying 125 kg ha−1 N or less.
Starke and Hoffmann [72] state that the differences in the share of root dry matter depending on N fertilization were not very pronounced, and according to the results of their research, the average share of root dry matter was 22.5%, with fertilization of 150 kg ha−1 N 23.6%, while at an even higher amount of N than 300 kg ha−1, dry matter was reduced to the level of the control treatment (22.5%). According to another study, Starke and Hoffmann [73] point out the positive effect of N on the dry matter yield of sugar beet leaves. Namely, the authors conclude that with higher fertilization, with as much as 300 kg ha−1 N, the yield of leaf dry matter increased to over 31 t ha−1, while without N fertilization, it was lower and amounted to 26 t ha−1.
Märländer et al. [50] also emphasize the importance of soil mineralization. Namely, although the average needs of sugar beet for N are about 200 to 250 kg ha−1, the authors state that approximately 100 to 150 kg ha−1 of N was mineralized in the soil through vegetation, which reduces the need for larger amounts of N fertilization. About 75% of the total area of sugar beet cultivation in Germany adds less than 120 kg ha−1 N by fertilization, while in the rest of the area, even less was applied, i.e., about 80 kg ha−1 N.
Jaćimović et al. [14], using a combination of different doses of NPK fertilizers in a two-year study, determined the highest yield of roots (98.86 t ha−1) and sugar (8.91 t ha−1) in the fertilization treatment of 100 kg ha−1 N, 150 kg ha−1 P2O5 and 150 kg ha−1 K2O.
The connection between N fertilization and N content in the plant is emphasized by Monreal et al. [74]. The authors found that N fertilization with 480 kg ha−1 N resulted in 569 kg ha−1 N in sugar beet plants, while unfertilized beets had 420 kg ha−1 N.
Vielemeyer et al. [75] state that increased N fertilization (0, 120 and 240 kg ha−1 N) decreased sugar content from 17.1% in the control treatment to 15.7%. However, the yield of crystallized sugar remained unchanged (average 5.7 t ha−1). Furthermore, the authors state that the increase in N fertilization to 240 kg ha−1 N led to an increase in leaf yield as well as crowns and root yield (82.8 t ha−1 and 42.9 t ha−1, respectively) compared to the control treatment of 42.7 t ha−1 and 38.2 t ha−1, respectively).
5. Nitrogen Use Efficiency (NUE) in Sugar Beet Production
Increasing nitrogen use efficiency can be achieved by using the right combination of nutrients, fertilizing at the right time and avoiding nutrient loss [76]. Nitrogen is a very mobile element and can be lost in several ways. Losses are often in the air volatile but also as leaching in the deeper layers of the soil by rainfall and groundwater. Both cases cause economic losses but also environmental problems. Nitrogen losses are affected by nitrogen form (nitrate, ammonium or urea), as well as soil properties (pH, texture, temperature, moisture, cation exchange ability, organic matter) and fertilizer management (time and dosage) [77,78].
Another important approach to sugar beet production is organic farming. Organic farming is one of the most important agricultural practices focused on food safety and biodiversity. This means producing food from a healthy environment of land, plants and animals. Organic fertilizers, genetically modified organisms, pesticides and other synthetic chemicals are not used in organic production. Organic agricultural production reduces human impact on the environment, improves soil quality in the long run and contributes to increasing biodiversity [79].
Therefore, a major challenge for breeders is developing sugar beet cultivars with higher NUE without creating nitrogen deficits and/or reducing yields to reduce environmental pollution and production costs. To create N-efficient cultivars, a selection can be carried out directly via N stress conditions or indirectly under optimal production conditions [80,81,82,83,84]. Direct selection in the target environment has the advantage to detect the interactions between the genotype and production system. Precision farming with the use of geographical information system (GIS) will also, in the future, have a great impact on improving NUE in sugar beet production [85,86,87].
6. Influence of Soil Type and N Fertilization on Sugar Beet Yield and Quality
The sugar beet production should always note that the land and weather conditions determine the framework and implement agrotechnical measures for the production level. Sugar beet can use large quantities of N, and the optimal amount moves within narrow limits. With an increased supply of N for the same root yield, beets can adopt up to 70% more N from the required amounts, negatively impacting root quality. Due to these facts, N fertilization should be done only to replenish stocks in the ground. As N in the soil occurs in the organic and mineral form, determining the stock of plants’ available N represents a problem, and because of the great variability in the soil, the problem is the time of soil sampling for analysis. Sugar beet depends on several factors, such as soil and weather, plant distribution in the field and number per unit area, a form of added N [88], as well as the fact that beets absorb uneven N throughout different growth phases in vegetation.
Today, we apply N based on the established stocks of N min in the soil before tillage and pre-sowing fertilization when fertilizing sugar beet. Early determination of N fertilization assumes the impossibility of accurately predicting N’s mobilization (subsequent delivery) from organic matter soil, since the period elapsed between the optimal fertilization period and harvest period is extremely long. This is important because the yield and quality of beets also depend on the availability of N during the second half of the vegetation. In practice, N fertilization is very often the cause of bad production results. In this research, the aim was to determine the effect of early N addition in the form of urea and increased fertilization through yield fertilization and root quality. Precipitation and air temperature over the season significantly affect N fertilization and achieved production results. Kristek et al. [68] point out that soil type and especially N application time significantly affect yield elements and sugar beet root quality. Sugar beet prefers the nitrate form of N(NO3−), so top dressing in production is usually performed in the phase 3–4 of leaves with mineral fertilizer calcium ammonium nitrate (CAN), which contains 27% N.
Last and Draycott [88] found in a three-year study that fertilization (0, 125, 250 kg ha−1 N) in carbonate loamy soils did not affect the increase in dry matter yield of roots. Icreasing the dry matter yield of roots requires a lower dose of fertilizer on clayey loamy soil than on sandy loamy soil. They further found that in the area with an average annual rainfall (around 600 mm for sugar beet), the loss of N by leaching was negligible but that the losses were significant during very humid springs, averaging 40 kg ha−1 N. Pospišil et al. [89] state that fertilization of 120 kg ha−1 N in soils of poor fertility (2 mg N-min/100 g soil, at a depth of 0–60 cm) and a more abundant rainfall during spring and summer significantly increase the leaf area of sugar beet. Marinković et al. [90] state that autumn fertilization of chernozem (Novi Sad, Serbia) with different doses of NPK (50, 100 and 150 kg ha−1 N, P, K) increased the average yield of root (58.5 t ha−1) in comparison with unfertilized plots (35.6 t ha−1). They further state that increasing the dose of N in the fertilizer of 100 kg ha−1 N significantly increases the sugar yield (8.69 t ha−1), while further increase in the amount of N of 150 kg ha−1 N reduces the sugar yield by 1.69 t ha−1. Hoffmann and Märländer [91] analyzed 57 genotypes from 22 locations in Germany (mainly luvisol and chernozem) with fertilization up to 160 kg N ha−1, stating that the concentration of total soluble N in beets ranges from 25 to 65 mmol kg−1 and amino N from 6 to 24 mmol kg−1. Malnou et al. [92], during a three-year experiment in Great Britain (Broom’s Barn) on sandy soil with favorable water–air relations, state that autumn fertilization of N (0 to 220 kg ha−1 N) with 160 kg ha−1 N significantly increases average sugar beet root yield (71.5 t ha−1) and sucrose content (17.4%), compared to the root yield and sucrosecontent without N fertilization, (55.7 t ha−1, 17.7% respectively). In the Republic of Croatia, according to Kristek et al. [68], after a large amount of precipitation in the winter period (in 2009/10), in soil with good physical properties in the spring, the amount of N min in the layer 0–60 cm was significantly smaller than in the previous year, when the amount of precipitation was almost half less and relative to soil with a differentiated profile. In fertile soil, urea application in the autumn with tillage was neither justified nor necessary, since the maximum root yield (74.9 t ha−1) and sugar yield (10.1 t ha−1) were achieved in the treatment without fertilization urea and top dressing (N-0 + 27 kg ha−1 N). Marinković and Crnobarac [93] determined the highest yield of roots (62.7 t ha−1) and sugar by fertilizing chernozem (Serbia, Rimski Šančevi) with 150, 100 and 50 kg N, P and K ha−1, respectively 41 t ha−1, with half of N (75 kg ha−1 N) applied in the autumn and the other half pre-sowing.
7. Cercospora Leaf Spot and N Fertilizers
In sugar beet production, the most important disease is the Cercospora leaf spot caused by the fungus Cercospora beticola Sacc., which occurs regularly in the production [94,95,96,97]. Several diseases are not so economically important, and they are caused by the fungus Ramularia beticola Futr (Ramularia leaf spot) [98,99], Alternaria tenuis Nees (Alternaria leaf spot) [100,101,102], Phoma betae Frank (zoned spot) [103,104], Uromyces betae Perset Lev. (sugar beet rust) and Pseudomonas syringae pathotype aptata Brown and Jamieson, Stevens (bacterial spot) [105].
Additionally, numerous authors’ results state that fertilization generally and high amounts of N fertilizers favor the development of the disease of the crops, affecting the plant’s disease risk either directly or indirectly through changes in the community structure or nitrogen [106,107,108,109].
The CLS is the most detrimental disease of sugar beet in temperate climates worldwide [110,111], which affects the above-ground parts of plants, including seeds. Symptoms are observed on older leaves in the form of gray spots inside and with a brown edge. The infected leaves become necrotic and eventually fall to the ground but remain attached to the root head. According to Kristek et al. [45], the damage caused by Cercospora beticola Sacc. reduces the photosynthesizing surface of the leaves. The yield of sugar beet root may be reduced up to 60% and sucrose content by 3–7%. These decrements in sugar beet root quality ultimately lead to a 42% reduction in technological sugar yield [112,113,114] up to 50% [115].
Ontario [116] suggests that the N application rate could reduce the need for fungicide applications to control Alternaria leaf blight and Cercospora leaf spot diseases in the field-grown carrots because lower N treatments resulted in fewer live leaves per plant at harvest. Makheti Mutebi and Atieno Ondede [117] found that for mulberry (Morus alba L.), plant fertilization amount of 200 kg ha−1 N is an effective approach for suppressing Cercospora leaf spot of mulberry and can be recommended to the farmers.
According to Kristek et al. [3], by applying non-symbiotic and associative N bacteria in sugar beet cultivation, there is a possible reduction in mineral N fertilizers.
8. Leaf N Content
According to Bergmann [118], the dry matter of a fully developed mature sugar beet leaf, 50 to 60 days after germination (June or July), optimally contains 4.0–6.0% N, 0.35–0.60% phosphorus, 3, 5–6.0% potassium and 0.30–0.70% magnesium. Similar values in the content of macro-elements in the dry matter of sugar beet leaves were shown by Stanaćev [40], according to which the N content is usually from 1.80 to 2.46%, phosphorus from 0.57 to 0.71%, potassium from 3.0 to 4.46%, sodium from 1.15 to 2.40% and magnesium from 0.37 to 0.75%.
The N concentration in the sugar beet plant depends on the amount of N in the soil. Draycott and Chirstenson [13] state that in the phase of fully expanded leaves, the N concentration in a sugar beet plant can range from 1.0 to 3.5%, in the root from 0.5 to 0.8%, in the leaf blade from 2.2 to 3.5% and in the stalk from 1.0 to 1.5%, but that in soils where beets are grown for a long time, there is N in the soil either in excessive or insufficient amounts and, therefore, N concentrations may be outside the specified limits.
Reviewing the results of several studies, Draycott and Chirstenson [13] state that the dry matter of sugar beet leaves at harvest contains about 0.3% phosphorus, 3% potassium, 2.5% sodium and 0.4% magnesium, while the dry matter of the root contains about 0.1 phosphorus, 0.8% potassium and 0.1% sodium. Furthermore, according to the experience in France and Great Britain, the authors state that at the time of the largest leaf area (July and August), the above-ground mass of sugar beet has the highest amount of phosphorus, from 20 to 25 kg ha−1 P2O5, while in the root, the amount of phosphorus increases during vegetation and at the time of extraction can be up to 40 kg ha−1 P2O5.
In recent breeding programs, the amino N concentration is the only selection criterion with regard to N components in the beet [119]. To achieve high-yield and high-quality sugar beet roots, sugar beet has high nutrient requirements, both macro and micronutrients. Fertilizing the soil with N is usually performed in the spring before sowing. Nitrates are present in the environment, i.e., in the air, food (mostly in fruits and vegetables) and water. Additionally, nitrates are used as fertilizers in agriculture. Nitrate or nitrite is always investigated for protecting the environment in water, agriculture and food. Nitrogen carries the offering but is also a nutrient that affects root quality. Both leaf blades and the sugar beet petioles leaf are important in determining the plants’ nutrient status [59].
Leaf blades and petioles of healthy sugar beet plants contain a large amount of N in the nitrate form. The lowest concentration of any nutrient can change a little with sugar beet variety, climate or soil conditions, moisture stress, disease, etc. Bilir and Saltalı [120] state that different N fertilization can greatly influence nitrate concentration in the sugar beet leaves, which needs to be analyzed if the leaves are used as animal fed.
Grzebisz et al. [121], according to the average of a two-year study in Poland, found that sugar beet leaf in the growth phase of six pairs of leaves (BBCH16) contains, on average, the most potassium (42.99 g−1 kg−1 dry matter), followed by N (33.59 g−1 kg−1 dry matter), magnesium (5.64 g−1 kg−1 dry matter) and the least phosphorus (3.79 g−1 kg−1 dry matter). Later, in the fully developed leaf rosette (BBCH43), the leaf dry matter contains the most N (42.53 g−1 kg−1 dry matter). At the same time, the content of other macro-elements, potassium, magnesium and phosphorus, was reduced (36.34 g−1 kg−1, 5.22 g−1 kg−1 and 3.09 g−1 kg−1 dry matter, respectively).
Sodium does not play a major role in the nutrition of sugar beet, but sodium affects the quality of sugar beet roots. If the root contains higher amounts of sodium (>0.65 mmol 100 g−1 beets), the yield of sugar from the root is reduced. Since sugar beet is a halophytic species, sodium can partially replace potassium in the nutrition of sugar beet [70,120]. For example, Hampe and Marschner [122] state that the cultivation of sugar beet when Na + ions were applied to the substrate instead of K + ions increased in dry matter, leaf area and the number of shoots, while net photosynthesis remained unchanged. Sugar beet requires nutrients throughout the growing season, and if there are not enough of them, the plant will lag behind in growth. Samples of plant material through the period of growth in weekly or monthly intervals can give a lot of information about the needs of plants for nutrients.
As a spring crop, sugar beet generally absorbs the most nutrients in July, when water needs are the highest. This is confirmed by Malnou et al. [92] who, on the example of N fertilization (0, 40, 80, 120 and 160 kg ha−1 N), state that the sugar beet crop in the phase of the highest leaf area index takes up from 102 to 147 kg ha−1 N in mid-July. Stanaćev [40] points out that the absorption of N from the soil slows down in sugar beet in the second half of July.
Draycott [70] considers that the amount of nutrients required for sugar beet is highly variable. Namely, the author points out that sugar beet absorbs the least Mg (23 kg ha−1), then P (5–100 kg ha−1) and Na (60–100 kg ha−1) and macronutrients N (100–220 kg ha−1), and the highest needs are for K (168–660 kg ha−1).
9. Sugar Beet Root Quality
Sugar beet fertilization is specific in relation to other field crops. As crop which have high yield, technological quality is also expected to be high. Proper fertilization management includes several essential questions, such as which fertilizer to apply, what amount of nutrients, in what ratio and when to apply them. With increasing rates of N, there is an increased content of α-amino N and another non-sugar matter at the root, which is unfavorable, reflecting sugar crystallization during processing.
Last et al. [88], during a six-year field study (1973–78), examined the impact of ammonium nitrate fertilization (0, 41, 82, 124, 166 and 207 kg N ha−1) and irrigation on the yield and quality of sugar beet. The authors stated that in the arable soil layer, the optimal concentration of N min was about 40 mg kg−1 N soil during May. Later in the growing season, N greatly increased dry matter, and root yield was also reflected in sugar yield. Sugar beet root yield linearly increase in relation to accumulation of dry matter. However, from the data of several experiments (Table 1), increasing in N resulted in higher root yield in most cases, but on the contrary, higher N rate even decreased sucrose content in the root.

Table 1.
Different amounts of N and their influence on final sugar beet yield.
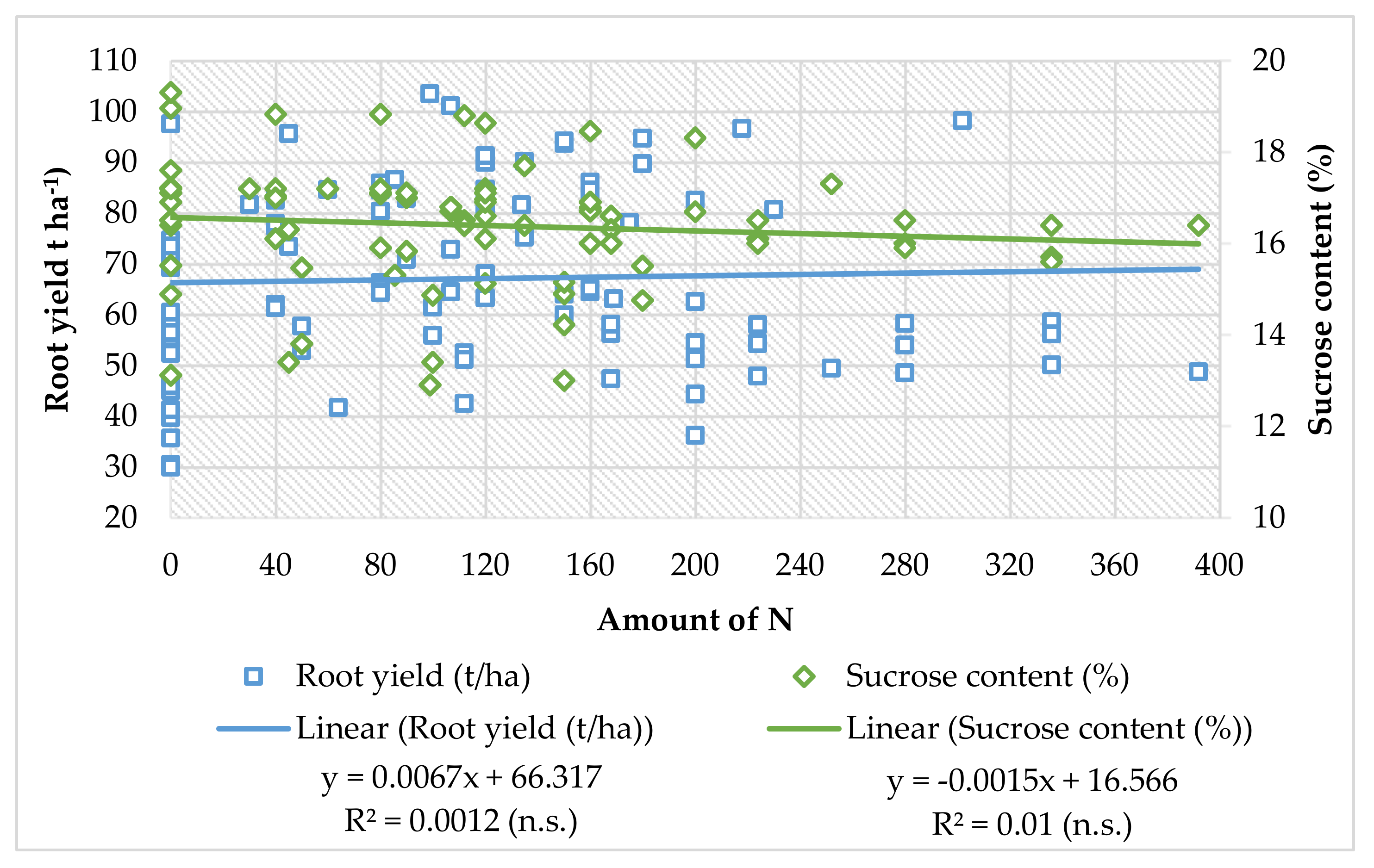
Kristek et al. [68] state that fertilization with urea in the year with insufficient rainfall (around 540 mm from April to October) was also not justified, as the highest root yield (61.53 t ha−1) and sugar (9.02 t ha−1) were achieved in the variant with top dressing N-0 + 13.5 (50 kg ha−1 CAN), while in a wet year (around 900 mm from April to October), in that soil, the highest root yield (75.13 t ha−1) and sugar (9.54 t ha−1) were achieved with fertilization urea (46% N) and without the addition of N in the vegetation. Based on the data from the literature, the regression analyses show that increment of N fertilizer amount can slightly increase the root yield, but for the sucrose content, the linear model has a negative trendline (Figure 1). There is evidence that sugar nitrogen fertilization has a great influence in increasing the biomass of the roots, but the increased amount of nitrogen has negative influence on sugar beet root quality and increases the amount of harmful nitrogen in the root and decreases the sucrose content at harvest. Even though the regression coefficient is not significant, the trendline shows negative influence on root quality, and there is still need for further experiments of nitrogen fertilization in sugar beet production.
Sugar beet fertilization is very specific because it needs to achieve high root yield, high sugar content, and small amounts of soluble non-sugars in the root. The deficit, as well as excessive amounts of nutrients, affect the final yield but also the quality of the roots; therefore, proper fertilization is crucial for a successful sugar beet production. The main reason for less sucrose content was an increased amount of brei impurities [116,117,131,132,133,134,135,136,137,138], especially the amount of amino N. In the case of N, it is impossible to build up long-term N reserves in the soil, so the excess or lack of available N during the year is likely. Sugar beet does have a mechanism to regulate N uptake, but the root can absorb as much N as is provided.
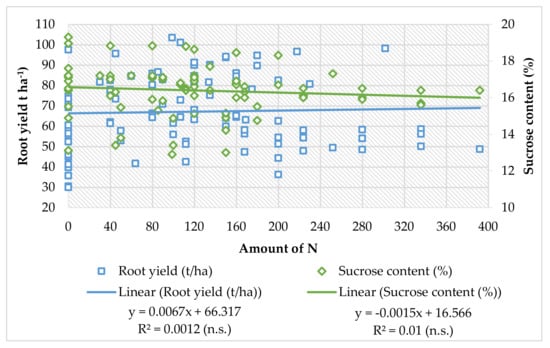
Figure 1.
Linear regression models of sugar beet root yield and sucrose content (data obtained from: Malnou et al. [16], Marinković et al. [90,93], Milić et al. [124], Pogłodziński et al. [130], Varga et al. [134], Barłóg et al. [85], Lentz and Lehrsch [137] Carter and Traveller [23], Tarkalson et al. [22], Lauer [138]).
Figure 1.
Linear regression models of sugar beet root yield and sucrose content (data obtained from: Malnou et al. [16], Marinković et al. [90,93], Milić et al. [124], Pogłodziński et al. [130], Varga et al. [134], Barłóg et al. [85], Lentz and Lehrsch [137] Carter and Traveller [23], Tarkalson et al. [22], Lauer [138]).
10. Conclusions
This study comprehensively describes N’s influence on sugar beet vegetative growth and root yield and quality. The main elements of nutrition N, phosphorus and potassium, have the greatest importance in plant nutrition and the greatest influence on the creation of yield and increased technological value of the sugar beet root. As a large consumer of plant assimilates, sugar beet will react strongly to enhanced nutrition with N, phosphorus and potassium used in the form of minerals nutrients. Even though the N needs of sugar beet have been the main focus for decades, studies with N fertilization of sugar beet are still very important due to the development of new genotypes with different accumulation capacities. In sugar beet production, the most important factors are proper and comprehensive plant nutrition and adoption of certain plant assimilates and their role in synthesizing organic matter in plant tissues and the accumulation of sugar in the roots. There is no unique plant nutrition system because the needs of sugar beet for certain elements depend on weather conditions, soil, cultivation methods (natural water regime, irrigation) but also on the genotypes. Increasing N increases the root yield of sugar beet but also the content of harmful N, which reduces the technological root value. Thus, soil analyses and fertilization recommendations represent one of the most important factors in successful production, especially in relation to sugar beet root quality.
Author Contributions
Conceptualization, I.V. and V.Z.; investigation, I.V., J.J., M.R. and M.A.; writing—original draft preparation, I.V., V.Z., J.J. and A.M.K.; writing—review and editing, I.V., A.M.K., Z.L. and D.I. All authors have read and agreed to the published version of the manuscript.
Funding
This research was part of project KK.01.1.1.04.0052: “Innovative production of organic fertilizers and substrates (INOPROFS)”, co-financed by the European Union from the European Regional Development Fund within the Operational programme Competitiveness and Cohesion 2014–2020 of the Republic of Croatia.
Institutional Review Board Statement
Not applicable.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Bruhns, J.; Baron, O.; Maier, K. Sugar Economy Europe 2004; No. Ed. 50; Verlag Dr. Albert Bartens KG: Berlin, Germany, 2003. [Google Scholar]
- Jug, D.; Jug, I.; Brozović, B.; Vukadinović, V.; Stipešević, B.; Đurđević, B. The role of conservation agriculture in mitigation and adaptation to climate change. Poljoprivreda 2018, 24, 35–44. [Google Scholar] [CrossRef]
- Kristek, S.; Brkić, S.; Jović, J.; Stanković, A.; Ćupurdija, B.; Brica, M.; Karalić, K. The application of nitrogen-fixing bacteria in order to reduce the mineral nitrogen fertilizers in sugar beet. Poljoprivreda 2020, 26, 65–71. [Google Scholar] [CrossRef]
- Stošić, M.; Brozović, B.; Vinković, T.; Ravnjak, B.; Kluz, M.; Zebec, V. Soil resistance and bulk density under different tillage system. Poljoprivreda 2020, 26, 17–24. [Google Scholar] [CrossRef]
- Rašovský, M.; Pačuta, V.; Černý, I.; Ernst, D.; Michalska-Klimczak, B.; Wyszyňski, Z. Monitoring of Influence of Biopreparates, Weather Conditions and Variety on Production Parameters of Sugar Beet. Listy Cukrov. A Řepařské 2021, 137, 154. [Google Scholar]
- Varga, I.; Lončarić, Z.; Pospišil, M.; Rastija, M.; Antunović, M. Dynamics of sugar beet root, crown and leaves mass with regard to plant densities and spring nitrogen fertilization. Poljoprivreda 2020, 26, 32–39. [Google Scholar] [CrossRef]
- Jurišić, M.; Radočaj, D.; Plaščak, I.; Rapčan, I. A Comparison of Precise Fertilization Prescription Rates to a Conventional Approach Based on the Open Source GIS Software. Poljoprivreda 2021, 27, 52–59. [Google Scholar] [CrossRef]
- Antunović, M.; Varga, I.; Stipešević, B.; Ranogajec, L. Analýza chorvatského cukrovarnického sektoru a produkce cukrové řepy. Listy Cukrov. A Řepařské 2021, 137, 383–386. [Google Scholar]
- Ernst, D.; Černý, I.; Pačuta, V.; Zapletalová, A.; Rašovský, M.; Skopal, J.; Vician, T.; Šulík, R.; Gažo, J. Yield and Sugar Content of Sugar Beet Depending on Different Soil Tillage Technologies. Listy Cukrov. A Řepařské 2021, 137, 319–324. [Google Scholar]
- Turesson, H.; Andersson, M.; Marttila, S.; Thulin, I.; Hofvander, P. Starch biosynthetic genes and enzymes are expressed and active in the absence of starch accumulation in sugar beet tap-root. BMC Plant Biol. 2014, 14, 104. [Google Scholar] [CrossRef]
- Hoffmann, C.M.; Kenter, C. Yield potential of Sugar beet—Have we hit the ceiling? Front. Plant Sci. 2018, 9, 289. [Google Scholar] [CrossRef]
- Franzen, D.W. Delineating nitrogen management zones in a sugarbeet rotation using remote sensing—A review. J. Sugar Beet Res. 2004, 41, 47–60. [Google Scholar] [CrossRef]
- Draycott, A.P.; Christenson, D.R. Nutrients for Sugar Beet Production: Soil-Plant Relationships; CABI: Wallingford, UK, 2003. [Google Scholar]
- Jaćimović, G.; Marinković, B.; Crnobarac, J.; Bogdanović, D.; Kovačev, L.; Danojević, D. Influence of fertilization and nitrate-nitrogen position in soil profile on the sugar beet root yield and quality. J. Agric. Sci. 2008, 53, 83–90. [Google Scholar]
- Kristek, S.; Kristek, A.; Evačić, M. Influence of nitrogen fertilization on sugar beet root yield and quality. Cereal Res. Commun. 2008, 36, 371–374. [Google Scholar]
- Malnou, C.S.; Jaggard, K.W.; Sparkes, D.L. A canopy approach to nitrogen fertilizer recommendations for the sugar beet crop. Eur. J. Agron. 2006, 25, 254–263. [Google Scholar] [CrossRef]
- Pospišil, M. Ratarstvo II. Dio—Industrijsko Bilje; Zrinski: Čakovec, Croatia, 2013. [Google Scholar]
- Lundegårdh, H. Plant Physiology; Oliver and Boyd: Edinburgh, UK; London, UK; T. & A. Constable Ltd.: Edinburgh, UK, 1966. [Google Scholar]
- Hoffmann, C.M.; Kluge-Severin, S. Growth analysis of autumn and spring sown sugar beet. Eur. J. Agron. 2011, 34, 1–9. [Google Scholar] [CrossRef]
- Pevalek-Kozlina, B. (Ed.) Fiziologija Bilja; Profil International: Zagreb, Croatia, 2003. [Google Scholar]
- Klotz, K.L.; Finger, F.L. Impact of temperature, length of storage and postharvest disease on sucrose catabolism in sugarbeet. Postharvest Biol. Technol. 2004, 34, 1–9. [Google Scholar] [CrossRef]
- Tarkalson, D.D.; Bjorneberg, D.L.; Camp, S.; Dean, G.; Elison, D.; Foote, P. Improving nitrogen management in Pacific Northwest sugarbeet production. J. Sugar Beet Res. 2016, 53, 14–36. [Google Scholar] [CrossRef]
- Carter, J.N.; Traveller, D.J. Effect of time and amount of nitrogen uptake on sugarbeet growth and yield. Agron. J. 1981, 73, 665–671. [Google Scholar] [CrossRef]
- Mary, B.; Recous, S. Measurement of nitrogen mineralization and immobilization fluxes in soil as a means of predicting net mineralization. Eur. J. Agron. 1994, 3, 291–300. [Google Scholar] [CrossRef]
- Mengel, K. Turnover of organic nitrogen in soils and its availability to crops. Plant Soil 1996, 181, 83–93. [Google Scholar] [CrossRef]
- Nemeth, K. Recent advances in EUF research (1980–1983). Plant Soil 1985, 83, 1–19. [Google Scholar] [CrossRef]
- Nemeth, K.; Bartels, H.; Vogel, M.; Mengel, K. Organic nitrogen compounds extracted from arable and forest soils by electro-ultrafiltration and recovery rates of amino acids. Biol. Fertil. Soils 1988, 5, 271–275. [Google Scholar] [CrossRef]
- Natesan, S.; Ranganathan, V.; Nemeth, K.; Krishnan, V. EUF-analysis of tea soils of Southern India and tea productivity. Plant Soil 1985, 83, 191–198. [Google Scholar] [CrossRef]
- Akinrinde, E.A.; Obigbesan, G.O.; Gaiser, T. Electro-ultrafiltration (EUF) technique in relation to conventional methods of soil testing for the determination of available P, Ca, Mg and NO3-N in some tropical soils. J. Agron. 2006, 5, 375–381. [Google Scholar] [CrossRef][Green Version]
- Wiedeman, H. Soil analysis and N-fertilizer recommendations for growing sugar beet in southern Germany and Austria. In Proceedings of the International Institute for Beet Research 57th Winter Congress, Bruxelles, Belgium, 16–17 February 1994. [Google Scholar]
- Horn, D.; Fürstenfeld, F. Nitrogen fertilizer recommendation for sugar beet according to the EUF soil testing system. In Plant Nutrition; Springer: Dordrecht, The Netherlands, 2001. [Google Scholar]
- Wiklicky, L. Application of the EUF procedure in sugar beet cultivation. Plant Soil 1982, 64, 115–127. [Google Scholar] [CrossRef]
- Scharpf, H.C.; Wehrmann, J. Bedeutung des Mineralstickstoffvorrates des Bodens zu Vegetationsbeginn fur die Bemessung der N Dungung zu Winterweizen. Landwirtschaftliche Forschung. Sonderheft 1976, 52, 109–126. [Google Scholar]
- Wehrmann, J.V.; Scharpf, H.C. Der Mineralstickstoffgehalt des Bodens als Maßstab für den Stickstoffdüngerbedarf (N min-Methode). Plant Soil 1979, 52, 109–126. [Google Scholar] [CrossRef]
- Vukadinović, V.; Lončarić, Z. Ishrana Bilja. Poljoprivredni Fakultet u Osijeku; Josip Juraj Strossmayer University of Osijek: Osijek, Croatia, 1997. [Google Scholar]
- Bertić, B.; Vukadinović, V. Filozofija Gnojidbe Sve Što Treba Znati o Gnojidbi; Studio HS Internet d.o.o. Osijek: Osijek, Croatia, 2013. [Google Scholar]
- Jaradat, A.A.; Rinke, J. Modeling sugar content of farmer-managed sugar beets (Beta vulgaris L.). Commun. Biometry Crop Sci. 2012, 7, 23–34. [Google Scholar]
- Hoffmann, C.M.; Kluge-Severin, S. Light absorption and radiation use efficiency of autumn and spring sown sugar beets. Field Crops Res. 2010, 119, 238–244. [Google Scholar] [CrossRef]
- Kristek, A.; Halter, J. Djelovanje vegetacijskog prostora na porast lišća šećerne repe i prinos korijena. Agron. Glas. 1988, 2–3, 79–94. [Google Scholar]
- Stanaćev, S. Šećerna Repa; Nolit: Beograd, Serbia, 1979. [Google Scholar]
- Kristek, A.; Liović, I. Ritam rasta šećerne repe u uvjetima 1987. godine. Poljopr. Aktualnosti 1988, 30, 173–185. [Google Scholar]
- Jelić, S.; Antunović, M.; Bukvić, G.; Varga, I.; Iljkić, D. Impact of plant density on growth, yield and quality of sugar beet. Listy Cukrov. A Řepařské 2019, 135, 107. [Google Scholar]
- Manderscheid, R.; Pacholski, A.; Weigel, H.J. Effect of free air carbon dioxide enrichment combined with two nitrogen levels on growth, yield and yield quality of sugar beet: Evidence for a sink limitation of beet growth under elevated CO2. Eur. J. Agron. 2010, 32, 228–239. [Google Scholar] [CrossRef]
- Lüdecke, H. Šećerna Repa; Poljoprivredni Nakladni Zavod Zagreb: Zagreb, Croatia, 1956. [Google Scholar]
- Kristek, A.; Kristek, S.; Varga, I.; Drmić, Z. Rezultati u proizvodnji šećerne repe u zavisnosti od izbora hibrida i broja tretiranja fungicida. Poljoprivreda 2015, 21, 15–22. [Google Scholar] [CrossRef]
- Vukadinović, V.; Jug, I.; Đurđević, B. Ekofiziologija Bilja. In Poljoprivredni Fakultet u Osijeku; Sveučilište Josipa Jurja Strossmayera u Osijeku Zebra: Vinkovci, Croatia, 2014. [Google Scholar]
- Müller-Linow, M.; Pinto-Espinosa, F.; Scharr, H.; Rascher, U. The leaf angle distribution of natural plant populations: Assessing the canopy with a novel software tool. Plant Methods 2015, 11, 11. [Google Scholar] [CrossRef] [PubMed]
- Kenter, C.; Hoffman, C.M. Seasonal patterns of sucrose concentration in relation to other quality parameters of sugar beet (Beta vulgaris L.). J. Sci. Food Agric. 2006, 86, 62–70. [Google Scholar] [CrossRef]
- Drachovská, M.; Sandera, K. Fysiologie Cukrovky; Nakladatelství Československé Akademie věd: Prague, Czech Republic, 1959. [Google Scholar]
- Märländer, B.; Hoffmann, C.M.; Koch, H.J.; Ladewig, E.; Merkes, R.; Petersen, J.; Stockfisch, N. Environmental Situation and Yield Performance of the Sugar Beet Crop in Germany: Heading for Sustainable Development. J. Agron. Crop Sci. 2003, 189, 201–226. [Google Scholar] [CrossRef]
- Tóth, M.; Pokrivčák, J.; Smutka, L.; Dvořák, M.; Pulkrábek, J. Economic aspects of sugar beet production and biodiversity: Effects of ban on neonicotinoids use. Listy Cukrov. Řepařské 2022, 138, 116–120. [Google Scholar]
- Pospišil, A.; Pospišil, M. The Effect of Organic Fertilizers on the Spelt Yield and the Yield of its Components. Poljoprivreda 2021, 27, 37–43. [Google Scholar] [CrossRef]
- Tan, Z.X.; Lal, R.; Wiebe, K.D. Global soil nutrient depletion and yield reduction. J. Sustain. Agric. 2005, 26, 123–146. [Google Scholar] [CrossRef]
- Pan, B.; Lam, S.K.; Mosier, A.; Luo, Y.; Chen, D. Ammonia volatilization from synthetic fertilizers and its mitigation strategies: A global synthesis. Agriculture. Ecosyst. Environ. 2016, 232, 283–289. [Google Scholar] [CrossRef]
- Cucina, M.; De Nisi, P.; Sordi, S.; Adani, F. Sewage Sludge as N-Fertilizers for Crop Production Enabling the Circular Bioeconomy in Agriculture: A Challenge for the New EU Regulation 1009/2019. Sustainability 2021, 13, 13165. [Google Scholar] [CrossRef]
- Lukas, V.; Neudert, L.; Širůček, P.; Novák, J.; Elbl, J. Effect of variable rate application of phosphorus and potassium fertilizers in sugar beet. Listy Cukrov. A Řepařské 2021, 137, 417–422. [Google Scholar]
- de Souza Braz, A.M.; da Costa, M.L.; Ramos, S.J.; Dall’Agnol, R.; Fernandes, A.R. Long Term Application of Fertilizers in Eastern Amazon and Effect on Uranium and Thorium Levels in Soils. Minerals 2021, 11, 994. [Google Scholar] [CrossRef]
- Issukindarsyah, I.; Sulistyaningsih, E.; Indradewa, D.; Putra, E.T.S. The Effect of Ammonium Nitrate Ratio and Support Types on the NPK Uptake and Growth of Black Pepper (Piper nigrum L.) in Field Conditions. Poljoprivreda 2021, 27, 25–33. [Google Scholar] [CrossRef]
- Varga, I.; Kerovec, D.; Engler, M.; Popović, B.; Lončarić, Z.; Iljkić, D.; Antunović, M. Determination N-NO3− in sugar beet leaves). Listy Cukrov. A Řepařské 2022, 138, 69–72. [Google Scholar]
- Trap, J.; Bonkowski, M.; Plassard, C.; Villenave, C.; Blanchart, E. Ecological importance of soil bacterivores for ecosystem functions. Plant Soil 2016, 398, 1–24. [Google Scholar] [CrossRef]
- Harrison, R.B.; Footen, P.W.; Strahm, B.D. Deep soil horizons: Contribution and importance to soil carbon pools and in assessing whole-ecosystem response to management and global change. For. Sci. 2011, 57, 67–76. [Google Scholar]
- Hoffmann, C.M.; Blomberg, M. Estimation of leaf area index of Beta vulgaris L. based on optical remote sensing data. J. Agron. Crop Sci. 2004, 190, 197–204. [Google Scholar] [CrossRef]
- Tsialtas, J.T.; Maslaris, N. Leaf allometry and prediction of specific leaf area (SLA) in sugar beet (Beta vulgaris L.) cultivar. Photosynthetica 2008, 46, 351–355. [Google Scholar] [CrossRef]
- Tsialtas, J.T.; Saulioti, E.; Maslaris, N.; Papakosta, D. Genotypic response to regrowth of defoliated sugar beets after re-watering in a water-limited environment: Effects on yield and quality. Int. J. Plant Prod. 2009, 3, 1–18. [Google Scholar]
- Putnik-Delić, M. Fiziološki i Molekularni Aspekti Tolerantnosti Šećerne Repe Prema Suši. Doctoral Dissertation, University of Novi Sad, Novi Sad, Serbia, 2013. [Google Scholar]
- Kosterj, A.; Repka, J. Quantitative indicators of growth, production process and yield-formation of sugar-beet. Rostl. Vyrob. 1993, 39, 1077–1086. [Google Scholar]
- Tsialtas, J.T.; Maslaris, N. Leaf physiological traits and its relation with sugar beet cultivar success in two contrasting environments. Int. J. Plant Prod. 2012, 6, 15–36. [Google Scholar]
- Kristek, A.; Kristek, S.; Antunović, M.; Varga, I.; Katušić, J.; Besek, Z. Utjecaj tipa tla i gnojidbe dušikom na prinos i kvalitetu korijena šećerne repe. Poljoprivreda 2011, 17, 16–22. [Google Scholar]
- Brentrup, F.; Küsters, J.; Kuhlmann, H.; Lammel, J. Application of the Life Cycle Assessment methodology to agricultural production: An example of sugar beet production with different forms of nitrogen fertilizers. Eur. J. Agron. 2001, 14, 221–233. [Google Scholar] [CrossRef]
- Draycott, A.P. Sugar Beet; Wiley-Blackwell: Hoboken, NJ, USA, 2008. [Google Scholar]
- Last, P.J.; Draycott, A.P.; Messem, A.B.; Webb, D.J. Effects of nitrogen fertilizer and irrigation on sugar beet at Broom’s Barn 1973–8. J. Agric. Sci. 1983, 101, 185–205. [Google Scholar] [CrossRef]
- Starke, P.; Hoffmann, C.M. Yield Parameters of Beta Beets as a Basis to Estimate the Biogas Yield; Nutzung von Zuckerrüben für die Biogaserzeugung–Definition der Qualität sowie ertragsrelevante Parameter von Rübe, Blatt und Schossern; Institut für Zuckerrübenforschung: Gottingen, Germany, 2014; Volume 42. [Google Scholar]
- Starke, P.; Hoffmann, C.M. Yield parameters of Beta beets as a basis to estimate the biogas yield. Sugar Ind. 2014, 139, 169–176. [Google Scholar] [CrossRef]
- Monreal, J.A.; Jiménez, E.T.; Remesal, E.; Morillo-Velarde, R.; García-Mauriñoa, S.; Echevarría, C. Proline content of sugar beet storage roots: Response to water deficit and nitrogen fertilization at field conditions. Environ. Exp. Bot. 2007, 60, 257–267. [Google Scholar] [CrossRef]
- Vielemeyer, H.-P.; Lux, H.; Weege, K.-H. Einfluß des zeitlichen N-Angebots auf den Ertragsbildungsprozeß der Zuckerrübe. Arch. Acker-Pflanzenbau Bodenkd. Berl. 1986, 30, 131–137. [Google Scholar]
- Yadav, M.R.; Kumar, R.; Parihar, C.M.; Yadav, R.K.; Jat, S.L.; Ram, H.; Meena, R.K.; Singh, M.; Birbal; Verma, A.P.; et al. Strategies for improving nitrogen use efficiency: A review. Agric. Rev. 2017, 38, 29–40. [Google Scholar] [CrossRef]
- Melino, V.J.; Tester, M.A.; Okamoto, M. Strategies for engineering improved nitrogen use efficiency in crop plants via redistribution and recycling of organic nitrogen. Curr. Opin. Biotechnol. 2022, 73, 263–269. [Google Scholar] [CrossRef] [PubMed]
- Stevanato, P.; Chiodi, C.; Broccanello, C.; Concheri, G.; Biancardi, E.; Pavli, O.; Skaracis, G. Sustainability of the sugar beet crop. Sugar Tech 2019, 21, 703–716. [Google Scholar] [CrossRef]
- Atlason, R.S.; Lehtinen, T.; Davíðsdóttir, B.; Gísladóttir, G.; Brocza, F.; Unnþórsson, R.; Ragnarsdóttir, K.V. Energy return on investment of Austrian sugar beet: A small-scale comparison between organic and conventional production. Biomass Bioenergy 2015, 75, 267–271. [Google Scholar] [CrossRef]
- Hoffmann, C.M. Changes in N composition of sugar beet varieties in response to increasing N supply. J. Agron. Crop Sci. 2005, 191, 138–145. [Google Scholar] [CrossRef]
- Laufer, D.; Nielsen, O.; Wilting, P.; Koch, H.J.; Märländer, B. Yield and nitrogen use efficiency of fodder and sugar beet (Beta vulgaris L.) in contrasting environments of northwestern Europe. Eur. J. Agron. 2016, 73, 124–132. [Google Scholar] [CrossRef]
- Monteiro, F.; Frese, L.; Castro, S.; Duarte, M.C.; Paulo, O.S.; Loureiro, J.; Romeiras, M.M. Genetic and genomic tools to asssist sugar beet improvement: The value of the crop wild relatives. Front. Plant Sci. 2018, 9, 74. [Google Scholar] [CrossRef]
- Barłóg, P.; Grzebisz, W.; Feć, M.; Łukowiak, R.; Szczepaniak, W. Row method of sugar beet (Beta vulgaris L.) fertilization with multicomponent fertilizer based on urea-ammonium nitrate solution as a way to increase nitrogen efficiency. J. Cent. Eur. Agric. 2010, 11, 225–234. [Google Scholar]
- Hergert, G.W. Sugar beet fertilization. Sugar Tech 2010, 12, 256–266. [Google Scholar] [CrossRef]
- Barłóg, P.; Nowacka, A.; Błaszyk, R. Effect of zinc band application on sugar beet yield, quality and nutrient uptake. Plant Soil Environ. 2016, 62, 30–35. [Google Scholar] [CrossRef]
- Bronson, K.F.; Scharf, P.C.; Kitchen, N.R. Use of GIS-Based Site-Specific Nitrogen Management for Improving Energy Efficiency; USDA-ARS; UNL Faculty: Lincoln, NE, USA, 2011; Volume 677, pp. 359–384.
- Clay, D.E.; Shanahan, J.F. GIS Applications in Agriculture, Volume Two: Nutrient Management for Energy Efficiency; CRC Press: Boca Raton, FL, USA, 2011. [Google Scholar]
- Last, P.J.; Draycott, A.P. Growth and yield of sugar beet on contrasting soils in relation to nitrogen supply: II. Growth, uptake and leaching of nitrogen. J. Agric. Sci. 1975, 85, 27–37. [Google Scholar] [CrossRef]
- Pospišil, M.; Pospišil, A.; Rastija, M. Effect of plant density and nitrogen rates upon the leaf area of seed sugar beet on seed yield and quality. Eur. J. Agron. 2000, 12, 69–78. [Google Scholar] [CrossRef]
- Marinković, B.; Crnobarac, J.; Jaćimović, G.; Rajić, M.; Latković, D.; Aćin, V. Sugar yield and technological quality of sugar beet at different levels of nitrogen fertilization. Res. J. Agric. Sci. 2010, 42, 162–167. [Google Scholar]
- Hoffmann, C.M.; Märländer, B. Composition of harmful nitrogen in sugar beet (Beta vulgaris L.)—Amino acids, betaine, nitrate—As affected by genotype and environment. Eur. J. Agron. 2005, 22, 255–265. [Google Scholar] [CrossRef]
- Malnou, C.S.; Jaggard, K.W.; Sparkes, D.L. Nitrogen fertilizer and the efficiency of the sugar beet crop in late summer. Eur. J. Agron. 2008, 28, 47–56. [Google Scholar] [CrossRef]
- Marinković, B.J.; Crnobarac, J.Ž. Dependence of sugarbeet quality and yield on the application of NPK nutrients. Acta Period. Technol. 2000, 31, 345–350. [Google Scholar]
- Bolton, M.D.; Birla, K.; Rivera-Varas, V.; Rudolph, K.D.; Secor, G.A. Characterization of CbCyp51 from field isolates of Cercospora beticola. Phytopathology 2012, 102, 298–305. [Google Scholar] [CrossRef] [PubMed]
- Kristek, S.; Jović, J.; Zmaić, K.; Kraljičak, Ž.; Kišpal, H.; Bešlo, D.; Horvat, D.; Stjepanović, B.; Rašić, B. Problem of Development of Resistance to Some Fungicide Active Sub stances Intended for Suppressing of Cercospora beticola Sacc. Listy Cukrov. A Řepařské 2017, 133, 222–226. [Google Scholar]
- Vogel, J.; Kenter, C.; Holst, C.; Märländer, B. New generation of resistant sugar beet varieties for advanced integrated management of Cercospora leaf spot in central Europe. Front. Plant Sci. 2018, 9, 222. [Google Scholar] [CrossRef] [PubMed]
- Ereš, H.; Dujković, A.; Vrandečić, K. Cercospora leaf spot. Glas. Zaštite Bilja 2021, 44, 52–54. [Google Scholar] [CrossRef]
- Byford, W.J. Ramularia beticola in sugar-beet seed crops in England. J. Agric. Sci. 1975, 85, 369–375. [Google Scholar] [CrossRef]
- Wieczorek, T.M.; Jørgensen, L.N.; Hansen, A.L.; Munk, L.; Justesen, A.F. Early detection of sugar beet pathogen Ramularia beticola in leaf and air samples using qPCR. Eur. J. Plant Pathol. 2014, 138, 775–785. [Google Scholar] [CrossRef][Green Version]
- Mcfarlane, J.S.; Bardin, R.; Snyder, W.C. An Alternaria leaf spot of the sugar beet. Proc. Am. Soc. Sugarbeet Technol. 1954, 8, 241–245. [Google Scholar]
- Rosenzweig, N.; Hanson, L.E.; Mambetova, S.; Jiang, Q.W.; Guza, C.; Stewart, J.; Somohano, P. Fungicide sensitivity monitoring of Alternaria spp. causing leaf spot of sugarbeet (Beta vulgaris) in the Upper Great Lakes. Plant Dis. 2019, 103, 2263–2270. [Google Scholar] [CrossRef] [PubMed]
- Khan, M.F.R.; Haque, M.E.; Bloomquist, M.; Bhuiyan, M.Z.R.; Brueggeman, R.; Zhong, S.; Sharma Poudel, R.; Gross, T.; Hakk, P.; Leng, Y.; et al. First Report of Alternaria Leaf Spot Caused by Alternaria tenuissima on Sugar Beet (Beta vulgaris) in Minnesota, USA. Plant Dis. 2020, 104, 580. [Google Scholar] [CrossRef]
- Koenick, L.B.; Vaghefi, N.; Knight, N.L.; du Toit, L.J.; Pethybridge, S.J. Genetic diversity and differentiation in Phoma betae populations on table beet in New York and Washington States. Plant Dis. 2019, 103, 1487–1497. [Google Scholar] [CrossRef] [PubMed]
- Chand, N.; Jones, E.E.; Casonato, S. Pathogenicity of Phoma betae isolates from red beet (Beta vulgaris) at seed farms in Canterbury, New Zealand. Plant Prot. 2019, 72, 21–26. [Google Scholar] [CrossRef][Green Version]
- Agarwal, P.C.; Dev, U.; Rani, I.; Khetarpal, R.K. Seed-borne fungi detected in sugar beet seeds imported into India during last three decades. Plant Health Prog. 2006, 7, 2. [Google Scholar] [CrossRef]
- Veresoglou, S.D.; Barto, E.K.; Menexes, G.; Rillig, M.C. Fertilization affects severity of disease caused by fungal plant pathogens. Plant Pathol. 2013, 62, 961–969. [Google Scholar] [CrossRef]
- Huber, D.M.; Haneklaus, S. Managing nutrition to control plant disease. Landbauforsch. Volkenrode 2007, 57, 313. [Google Scholar]
- Long, D.H.; Lee, F.N.; TeBeest, D.O. Effect of nitrogen fertilization on disease progress of rice blast on susceptible and resistant cultivars. Plant Dis. 2000, 84, 403–409. [Google Scholar] [CrossRef]
- Liu, X.; Lyu, S.; Sun, D.; Bradshaw, C.J.; Zhou, S. Species decline under nitrogen fertilization increases community-level competence of fungal diseases. Proc. R. Soc. B Biol. Sci. 2017, 284, 20162621. [Google Scholar] [CrossRef] [PubMed]
- Skaracis, G.N.; Pavli, O.I.; Biancardi, E. Cercospora leaf spot disease of sugar beet. Sugar Tech 2010, 12, 220–228. [Google Scholar] [CrossRef]
- Schmittgen, S. Effects of Cercospora Leaf Spot Disease on Sugar Beet Geno-Types with Contrasting Disease Susceptibility. Ph.D. Thesis, Universitäts-und Landesbibliothek der Heinrich-Heine-Universität Düsseldorf, Düsseldorf, Germany, 2015. [Google Scholar]
- Vereijssen, J. Cercospora Leaf Spot in Sugar Beet. Epidemiology, Life Cycle Components and Disease Management. Ph.D. Thesis, Wageningen University, Wageningen, The Netherlands, 2004. [Google Scholar]
- Vereijssen, J.; Schneider, J.H.; Termorshuizen, A.J. Root infection of sugar beet by Cercospora beticola in a climate chamber and in the field. Eur. J. Plant Pathol. 2005, 112, 201–210. [Google Scholar] [CrossRef]
- Vukčević, R. Nova primena aviometode na zaštiti šećerne repe od cerkospore. Agron. Glas. 1964, 14, 245–256. [Google Scholar]
- Westerveld, S.M.; McKeown, A.W.; McDonald, M.R. Relationship between nitrogen fertilization and Cercospora leaf spot and Alternaria leaf blight of carrot. HortScience 2008, 43, 1522–1527. [Google Scholar] [CrossRef]
- Makheti Mutebi, C.; Atieno Ondede, D. Effect of nitrogen nutrition on the intensity of Cercospora leaf spot of Mulberry. Int. J. Hortic. Sci. Technol. 2021, 8, 335–342. [Google Scholar]
- Bergmann, W. Nutritional Disorders of Plants: Visual and Analytical Diagnosis; Jena Gustav Fischer Verlag: New York, NY, USA, 1992. [Google Scholar]
- Mäck, G.; Hoffmann, C.M.; Märländer, B. Nitrogen compounds in organs of two sugar beet genotypes (Beta vulgaris L.) during the season. Field Crops Res. 2007, 102, 210–218. [Google Scholar] [CrossRef]
- Bilir, B.; Saltalı, K. The Effect of Nitrogen-Boron Application and Time on the Nitrate Content of Sugar Beet Leaves Used as Animal Feed. Turk. J. Agric. Food Sci. Technol. 2021, 9, 395–400. [Google Scholar]
- Grzebisz, W.; Przygocka-Cyna, K.; Łukowiak, R.; Biber, M. An evaluation of macronutrient nutritional status of sugar beets in critical stages of growth in response to foliar application of multi-micronutrient fertilizers. J. Elem. 2010, 15, 493–507. [Google Scholar] [CrossRef][Green Version]
- Pi, Z.; Stevanato, P.; Sun, F.; Yang, Y.; Sun, X.; Zhao, H.; Geng, G.; Yu, L. Proteomic changes induced by potassium deficiency and potassium substitution by sodium in sugar beet. J. Plant Res. 2016, 129, 527–538. [Google Scholar] [CrossRef]
- Hampe, T.; Marschner, H. Effect of sodium on morphology, water relations and net photosynthesis of sugar beet leaves. Z. Pflanzenphysiol. 1982, 108, 151–162. [Google Scholar] [CrossRef]
- Marinković, B.; Crnobarac, J. Zavisnost kvaliteta i prinosa šećerne repe od primene NPK hraniva. Acta Period. Technol. 2000, 31, 345–350. [Google Scholar]
- Milić, S.; Pejić, B.; Maksimović, L. Uticaj navodnjavanja i đubrenja na sadržaj šećera i prinos korena šećerne repe. J. Sci. Agric. Res. 2006, 67, 5–12. [Google Scholar]
- Hergert, G.W.; Nielsen, R.A. Comparison of strip tillage versus broadcast N application for sugar beets. In Proceedings of the 2009 Biennial Meeting, Denver, CO, USA, 2 April 2009. [Google Scholar]
- Barłóg, P.; Grzebisz, W.; Peplinski, K.; Szczepaniak, W. Sugar beet response to balanced nitrogen fertilization with phosphorus and potassium. Part II. Dynamics of beet quality. Bulg. J. Agric. Sci 2013, 19, 1311–1318. [Google Scholar]
- Abdelaal, K.A.; Sahar, F.T. Response of sugar beet plant (Beta vulgaris L.) to mineral nitrogen fertilization and bio-fertilizers. Int. J. Curr. Microbiol. Appl. Sci. 2015, 4, 677–688. [Google Scholar]
- Pulkrábek, J.; Brinar, J.; Javor, T.; Dvořák, P.; Bečková, L.; Kuchtová, P.; Hubáčková, J. Experience with variable fertilization of sugar beet. Listy Cukrov. A Reparske 2021, 137, 184–193. [Google Scholar]
- Leilah, A.A.; Khan, N. Interactive Effects of Gibberellic Acid and Nitrogen Fertilization on the Growth, Yield, and Quality of Sugar Beet. Agronomy 2021, 11, 137. [Google Scholar] [CrossRef]
- Pogłodziński, R.; Barłóg, P.; Grzbisz, W. Effect of nitrogen and magnesium sulfate application on sugar beet yield and quality. Plant Soil Environ. 2021, 67, 507–513. [Google Scholar] [CrossRef]
- Varga, I.; Lončarić, Z.; Pospišil, M.; Rastija, M.; Antunović, M. Changes of Nitrate Nitrogen in Sugar Beet Petioles Fresh Tissue during Season with Regard to Nitrogen Fertilization and Plant Population. Listy Cukrov. A Řepařské 2020, 136, 198–204. [Google Scholar]
- Last, P.J.; Tinker, P.B.H. Nitrate nitrogen in leaves and petioles of sugar beet in relation to yield of sugar and juice purity. J. Agric. Sci. 1968, 71, 383–392. [Google Scholar] [CrossRef]
- Steinke, K.; Bauer, C. Enhanced Efficiency Fertilizer Effects in Michigan Sugarbeet Production. J. Sugar Beet Res. 2017, 54, 2–19. [Google Scholar] [CrossRef]
- Varga, I.; Lončarić, Z.; Kristek, S.; Kulundžić, A.M.; Rebekić, A.; Antunović, M. Sugar Beet Root Yield and Quality with Leaf Seasonal Dynamics in Relation to Planting Densities and Nitrogen Fertilization. Agriculture 2021, 11, 407. [Google Scholar] [CrossRef]
- Idris, M.; Baha, E.; Wael, A.M.; Abubaker Haroun, M.A. Effect of Nitrogen Fertilizer and Plant Spacing on Vegetative Growth of Sugar Beet (Beta vulgaris). J. Agron. Res. 2021, 4, 6–13. [Google Scholar] [CrossRef]
- Barłóg, P.; Grzebisz, W.; Szczepaniak, W.; Peplinski, K. Sugar beet response to balanced nitrogen fertilization with phosphorus and potassium. Part II. Dynamics of beet quality. Bulg. J. Agric. Sci. 2014, 20, 1326–1333. [Google Scholar]
- Lentz, R.D.; Lehrsch, G.A. Nitrogen availability and uptake by sugarbeet in years following a manure application. Int. J. Agron. 2012, 2012, 120429. [Google Scholar] [CrossRef]
- Lauer, J.G. Plant density and nitrogen rate effects on sugar beet yield and quality early in harvest. Agron. J. 1995, 87, 586–591. [Google Scholar] [CrossRef]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
