Distribution of Dissolved Nitrogen Compounds in the Water Column of a Meromictic Subarctic Lake
Abstract
:1. Introduction
2. Study Site and Methods
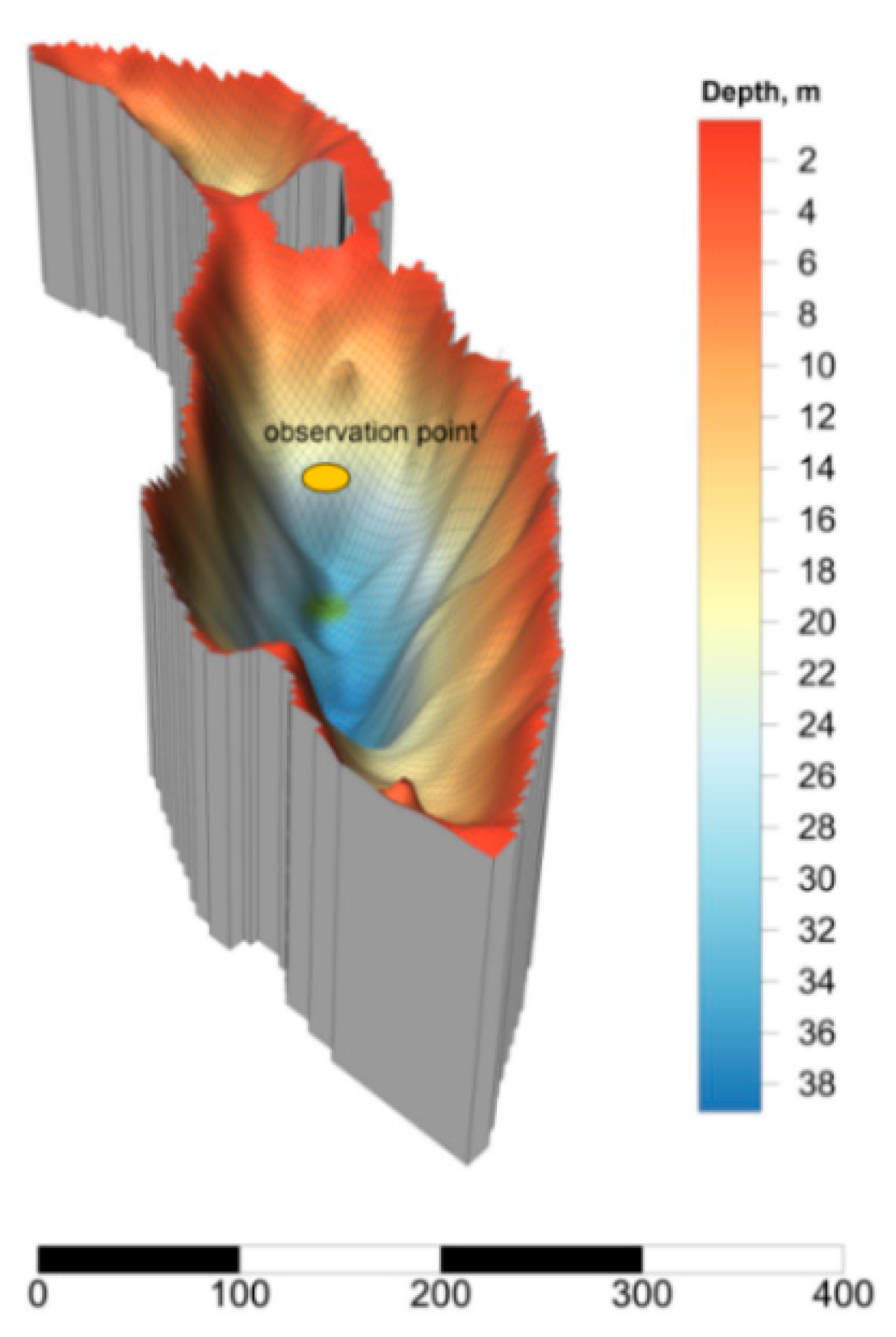
2.1. Site Description
2.2. Sampling and Analyses
3. Results
3.1. Main Hydrochemical Characteristics
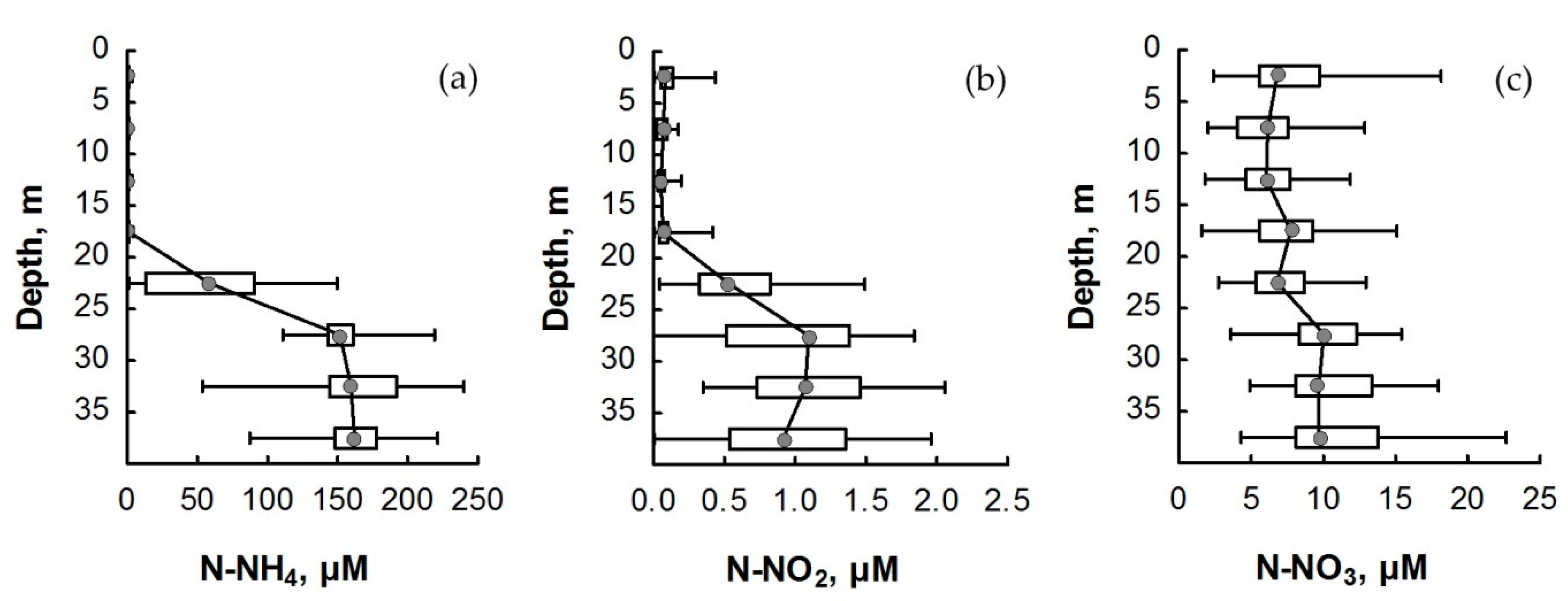
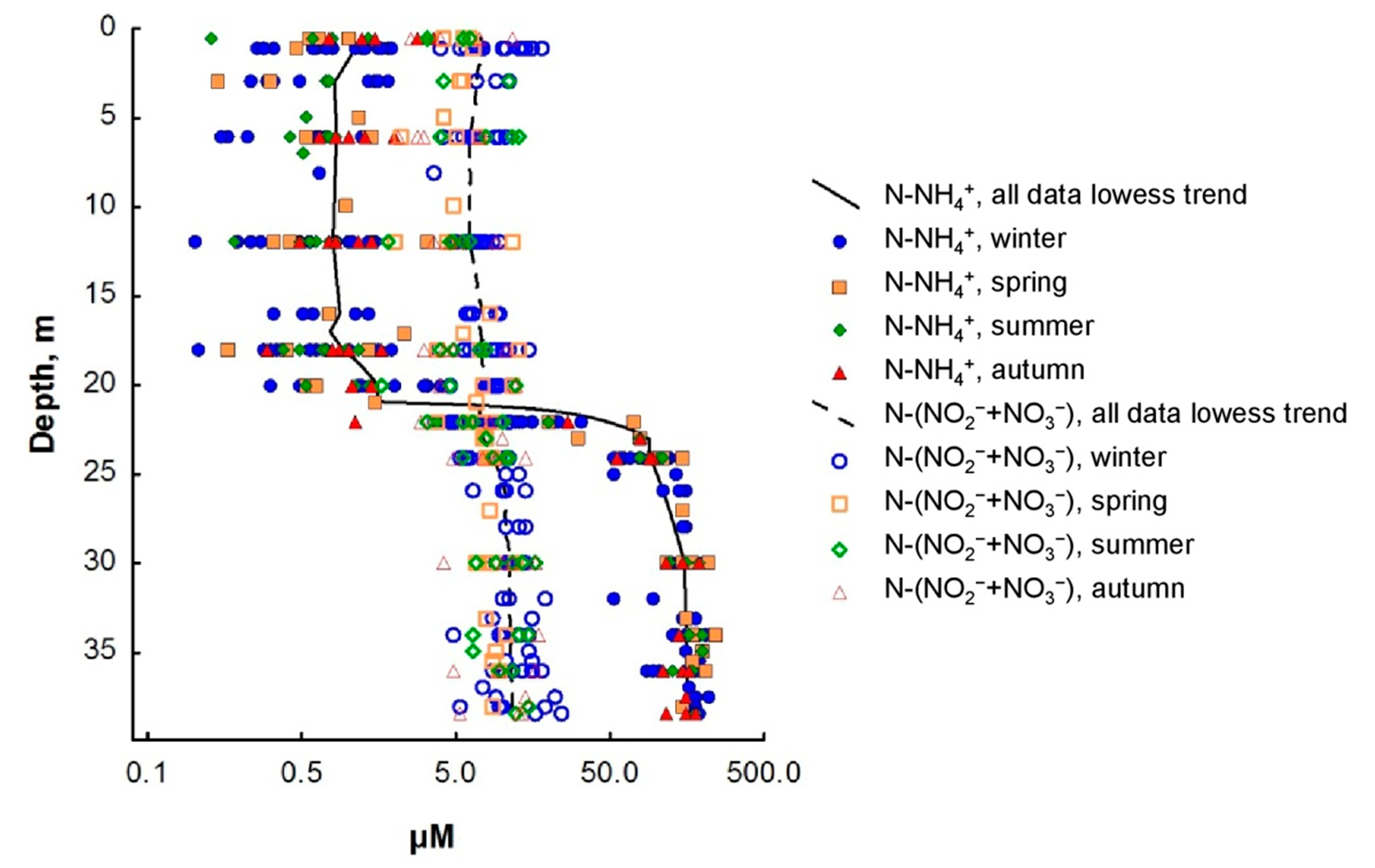
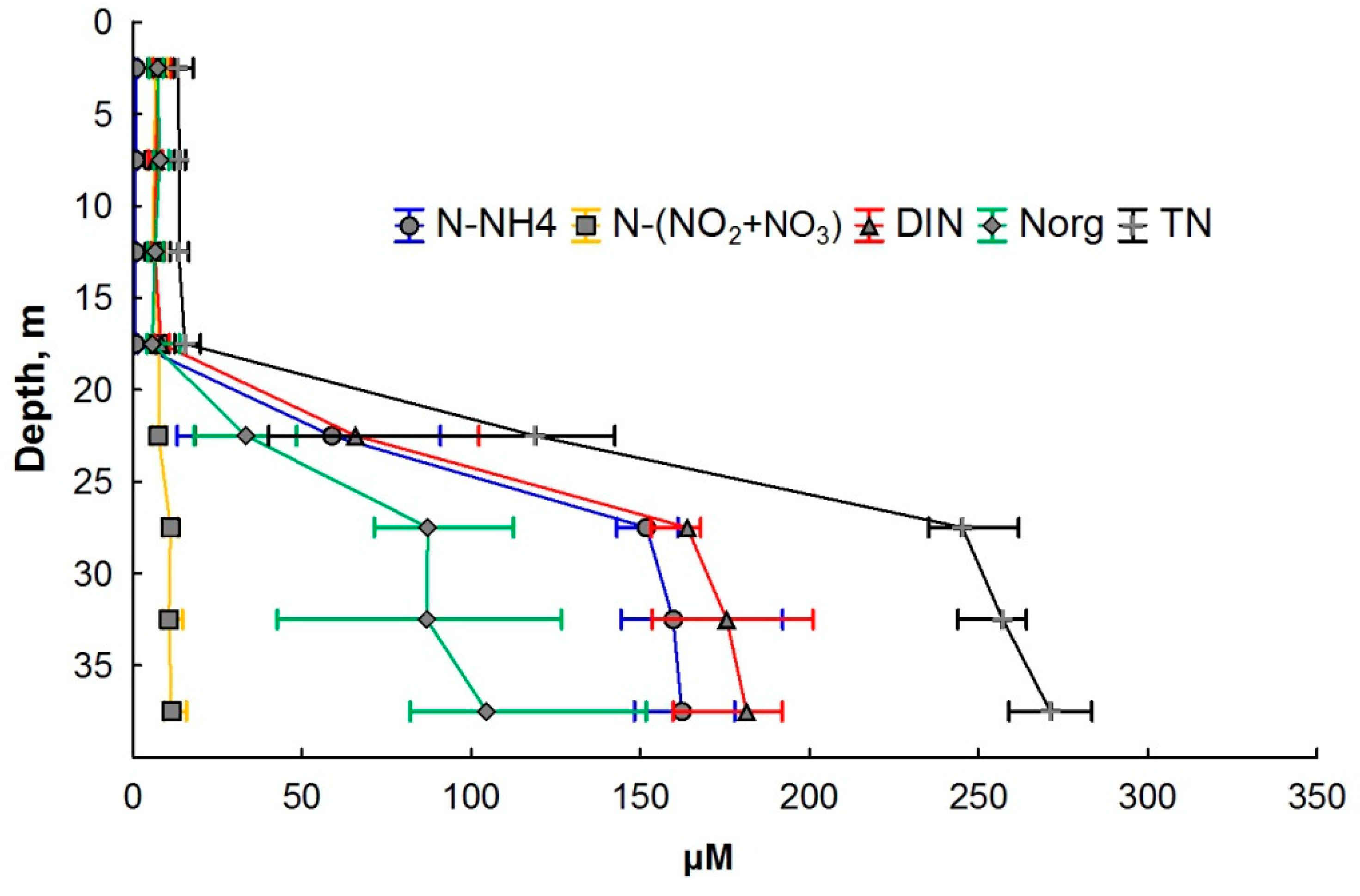
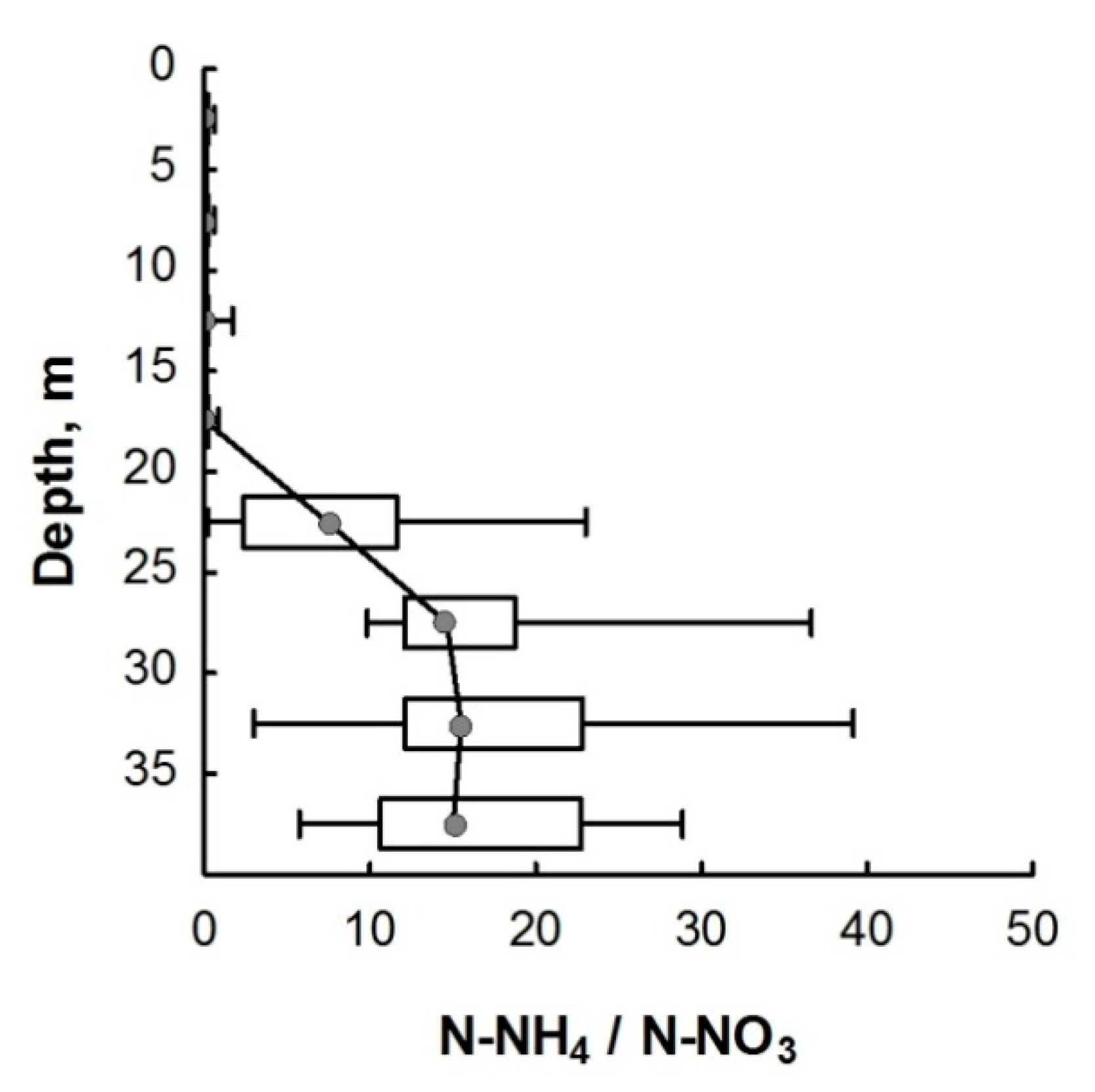
3.2. Nitrogen Compounds
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Campbell, N.A. Biology, 2nd ed.; The Benjamin/Cummings Publishing Company: Redwood City, CA, USA, 1990. [Google Scholar]
- Bashkin, V.N. Biogeochemistry; Scientific World: Moscow, Russia, 2004; p. 584. (In Russian) [Google Scholar]
- Downing, J.; McCauley, E. The nitrogen: Phosphorus relationship in lakes. Limnol. Oceanogr. 1992, 37, 936–945. [Google Scholar] [CrossRef] [Green Version]
- Burt, T.P. Nitrogen Cycle. In Encyclopedia of Ecology, 2nd ed.; Fath, B., Ed.; Elsevier: Amsterdam, The Netherlands, 2013; Volume 4, pp. 135–142. [Google Scholar] [CrossRef]
- Gruber, N.; Galloway, J. An Earth-system perspective of the global nitrogen cycle. Nature 2008, 451, 293–296. [Google Scholar] [CrossRef] [PubMed]
- Mackenzie, F.; Ver, L.M.; Sabine, C.; Lane, M.; Lerman, A. C, N, P, S global biogeochemical cycles and modeling of global change. In Interactions of C, N, P and S Biogeochemical Cycles and Global Change; NATO ASI Series (Series I: Global Environmental Change); Wollast, R., Mackenzie, F.T., Chou, L., Eds.; Springer: Berlin/Heidelberg, Germany, 1993; Volume 4, pp. 1–61. [Google Scholar] [CrossRef]
- Kuznetsov, S.I.; Dubinina, G.A. Methods for Studying Aquatic Microorganisms; Science: Moscow, Russia, 1989; p. 285. (In Russian) [Google Scholar]
- Cabello, P.; Roldán, M.D.; Castillo, F.; Moreno-Vivián, C. Nitrogen Cycle. In Encyclopedia of Microbiology; Schaechter, M., Ed.; Elsevier Science Ltd.: Amsterdam, The Netherlands, 2009; pp. 299–321. [Google Scholar] [CrossRef]
- Kuypers, M.; Marchant, H.; Kartal, B. The microbial nitrogen-cycling network. Nat. Rev. Microbiol. 2018, 16, 263–276. [Google Scholar] [CrossRef] [PubMed]
- Pajares, S.; Ramos, R. Processes and Microorganisms Involved in the Marine Nitrogen Cycle: Knowledge and Gaps. Front. Mar. Sci. 2019, 6, 739. [Google Scholar] [CrossRef]
- Hayatsu, M.; Tago, K.; Saito, M. Various players in the nitrogen cycle: Diversity and functions of the microorganisms involved in nitrification and denitrification. Soil Sci. Plant Nutr. 2008, 54, 33–45. [Google Scholar] [CrossRef]
- Stein, L.Y.; Klotz, M.G. The nitrogen cycle. Curr. Biol. 2016, 26, R94–R98. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zehr, J.P.; Ward, B.B. Nitrogen cycling in the ocean: New perspectives on processes and paradigms. Appl. Environ. Microbiol. 2002, 68, 1015–1024. [Google Scholar] [CrossRef] [Green Version]
- Kuznetsov, S.I.; Saralov, A.I.; Nazina, T.N. Microbiological Processes of Carbon and Nitrogen Circulation in Lakes; Science: Moscow, Russia, 1985; p. 216. (In Russian) [Google Scholar]
- Gorlenko, V.M.; Dubinina, G.A.; Kuznetsov, S.I. Ecology of Aquatic Microorganisms; Science: Moscow, Russia, 1977; p. 289. (In Russian) [Google Scholar]
- Camargo, J.A.; Alonso, Á. Ecological and toxicological effects of inorganic nitrogen pollution in aquatic ecosystems: A global assessment. Environ. Int. 2006, 32, 831–849. [Google Scholar] [CrossRef]
- Stewart, K.M.; Walker, K.F.; Likens, G.E. Meromictic Lakes. In Encyclopedia of Inland Waters; Likens, G.E., Ed.; Academic Press (Elsevier): Amsterdam, the Netherlands, 2009; pp. 589–602. [Google Scholar] [CrossRef]
- Gulati, R.D.; Zadereev, E.S.; Degermendzhi, A.G. (Eds.) Ecology of Meromictic Lakes; Springer International Publishing: Cham, Switzerland, 2017. [Google Scholar] [CrossRef]
- Lam, P.; Kuypers, M.M. Microbial nitrogen cycling processes in oxygen minimum zones. Ann. Rev. Mar. Sci. 2011, 3, 317–345. [Google Scholar] [CrossRef]
- Ward, B.B.; Glover, H.E.; Lipschultz, F. Chemoautotrophic activity and nitrification in the oxygen minimum zone off Peru. Deep. Sea Res. Part A. Oceanogr. Res. Pap. 1989, 36, 1031–1051. [Google Scholar] [CrossRef]
- Crowe, S.A.; Katsev, S.; Leslie, K.; Sturm, A.; Magen, C.; Nomosatryo, S.; Pack, M.A.; Kessler, J.D.; Reeburgh, W.S.; Roberts, J.A.; et al. The methane cycle in ferruginous Lake Matano. Geobiology 2011, 9, 61–78. [Google Scholar] [CrossRef] [PubMed]
- Oshkin, I.Y.; Beck, D.A.; Lamb, A.E.; Tchesnokova, V.; Benuska, G.; McTaggart, T.L.; Kalyuzhnaya, M.G.; Dedysh, S.N.; Lidstrom, M.E.; Chistoserdova, L. Methane-fed microbial microcosms show differential community dynamics and pinpoint taxa involved in communal response. ISME J. 2015, 9, 1119–1129. [Google Scholar] [CrossRef] [Green Version]
- Chistoserdova, L. Methylotrophs in natural habitats: Current insights through metagenomics. Appl. Microbiol. Biotechnol. 2015, 99, 5763–5779. [Google Scholar] [CrossRef]
- Rissanen, A.J.; Saarenheimo, J.; Tiirola, M.; Peura, S.; Aalto, S.L.; Karvinen, A.; Nykanen, H. Gammaproteobacterial methanotrophs dominate methanotrophy in aerobic and anaerobic layers of boreal lake waters. Aquat. Microb. Ecol. 2018, 81, 257–276. [Google Scholar] [CrossRef] [Green Version]
- He, Z.; Zhang, Q.; Feng, Y.; Luo, H.; Pan, X.; Gadd, G.M. Microbiological and environmental significance of metal-dependent anaerobic oxidation of methane. Sci. Total Environ. 2018, 610, 759–768. [Google Scholar] [CrossRef]
- Cabrol, L.; Thalasso, F.; Gandois, L.; Sepulveda-Jauregui, A.; Martinez-Cruz, K.; Teisserenc, R.; Tananaev, N.; Tveit, A.; Svenning, M.M.; Barret, M. Anaerobic oxidation of methane and associated microbiome in anoxic water of Northwestern Siberian lakes. Sci. Total Environ. 2020, 736, 139588. [Google Scholar] [CrossRef]
- Knowles, R. Denitrifiers associated with methanotrophs and their potential impact on the nitrogen cycle. Ecol. Eng. 2005, 24, 441–446. [Google Scholar] [CrossRef]
- Megmw, S.R.; Knowles, R. Active methanotrophs suppress nitrification in a humisol. Biol. Fert. Soils 1987, 4, 205–212. [Google Scholar] [CrossRef]
- Raghoebarsing, A.A.; Pol, A.; van de Pas-Schoonen, K.T.; Smolders, A.J.P.; Ettwig, K.F.; Rijpstra, W.I.C.; Schouten, S.; Damsté, J.S.S.; Op den Camp, H.J.M.; Jetten, M.S.M.; et al. A microbial consortium couples anaerobic methane oxidation to denitrification. Nature 2006, 440, 918–921. [Google Scholar] [CrossRef]
- Guggenheim, C.; Freimann, R.; Mayr, M.J.; Beck, K.; Wehrli, B.; Bürgmann, H. Environmental and Microbial Interactions Shape Methane-Oxidizing Bacterial Communities in a Stratified Lake. Front. Microbiol. 2020, 11, 579427. [Google Scholar] [CrossRef]
- Einsiedl, F.; Wunderlich, A.; Sebilo, M.; Coskun, Ö.K.; Orsi, W.D.; Mayer, B. Biogeochemical evidence of anaerobic methane oxidation and anaerobic ammonium oxidation in a stratified lake using stable isotopes. Biogeosciences 2020, 17, 5149–5161. [Google Scholar] [CrossRef]
- Kits, K.D.; Klotz, M.G.; Stein, L.Y. Methane oxidation coupled to nitrate reduction under hypoxia by the Gammaproteobacterium Methylomonas denitrificans, sp. nov. type strain FJG1: Denitrifying metabolism in M. denitrificans FJG1. Environ. Microbiol. 2015, 17, 3219–3232. [Google Scholar] [CrossRef] [PubMed]
- Zabelina, S.A.; Kompantseva, E.I.; Chupakov, A.V.; Ershova, A.A. Seasonal dynamics of photosynthetic pigments in a freshwater meromictic lake. In Biodiagnostics of the State of Natural and Natural-Technogenic SYSTEMS, Proceedings of the XII All-Russian scientific-Practical Conference with International Participation, Kirov, Russia, 2–3 December 2014; Publishing House of LLC “Vesi”: Kirov, Russia, 2014; pp. 119–123. (In Russian) [Google Scholar]
- Kokryatskaya, N.M.; Chupakov, A.V.; Titova, K.; Chupakova, A.А.; Zabelina, S.A.; Moreva, O.Yu.; Neverova, N.V.; Zhibareva, T.A. Hydrological and hydrochemical characteristics of the iron-manganese meromictic freshwater Lake Svetloe (Arkhangelsk Region). J. Sib. Fed. Univ. Biol. 2019, 12, 147–159. [Google Scholar] [CrossRef] [Green Version]
- Kallistova, A.; Kadnikov, V.; Rusanov, I.; Kokryatskaya, N.; Beletsky, A.; Mardanov, A.; Savvichev, A.; Ravin, N.; Pimenov, N. Microbial communities involved in aerobic and anaerobic methane cycling in a meromictic ferruginous subarctic lake. Aquat. Microb. Ecol. 2019, 82, 1–18. [Google Scholar] [CrossRef]
- Savvichev, A.S.; Rusanov, I.I.; Zakharova, E.E.; Veslopolova, E.F.; Lunina, O.N.; Sigalevich, P.A.; Pimenov, N.V.; Gorlenko, V.M.; Kokryatskaya, N.M.; Zabelina, S.A.; et al. Microbial processes of the carbon and sulfur cycles in an ice-covered, iron-rich meromictic lake Svetloe (Arkhangelsk region, Russia). Environ. Microbiol. 2017, 19, 659–672. [Google Scholar] [CrossRef] [PubMed]
- Kadnikov, V.V.; Mardanov, A.V.; Beletsky, A.V.; Ravin, N.V.; Savvichev, A.S.; Merkel, A.Y.; Pimenov, N.V. Microbial communities involved in the methane cycle in the near-bottom water layer and sediments of the meromictic subarctic Lake Svetloe. Antonie Leeuwenhoek 2019, 112, 1801–1814. [Google Scholar] [CrossRef]
- Ershova, A.A.; Vorobyeva, T.Ya.; Moreva, O.Yu.; Chupakov, A.V.; Zabelina, S.A.; Neverova, N.V. Hydrochemical and microbiological research of a nitrogen cycle in freshwater meromictic lake Svetloe (the Arkhangelsk region). Reg Environ. 2015, 5, 44–50. (In Russian) [Google Scholar]
- Chupakov, A.V.; Pokrovsky, O.S.; Shirokova, L.S.; Vorobyova, T.Ya.; Zabelina, S.A.; Kokryatskaya, N.M.; Moreva, O.Yu.; Ershova, A.A.; Shorina, N.V.; Klimov, S.I. Hydrochemical characteristics of meromictic freshwater Lake Svetloe (Arkhangelsk region). Vestn. North. Arct. Fed. University. Ser. Nat. Sci. 2013, 1, 20–31. (In Russian) [Google Scholar]
- Chupakov, A.V.; Chupakova, A.A.; Moreva, O.Yu.; Shirokova, L.S.; Zabelina, S.A.; Vorobieva, T.Y.; Klimov, S.I.; Brovko, O.S.; Pokrovsky, O.S. Allochthonous and autochthonous carbon in deep, organic rich and organic-poor lakes of the European Russian subarctic. Boreal Environ. Res. 2017, 22, 213–230. [Google Scholar]
- Canfield, D.E.; Rosing, M.T.; Bjerrum, C. Early anaerobic metabolisms. Philos. Trans. R. Soc. Lond. B Biol. Sci. 2006, 361, 1819–1836. [Google Scholar] [CrossRef] [Green Version]
- Scanlan, D.J.; West, N.J. Molecular ecology of the marine cyanobacterial genera Prochlorococcus and Synechococcus. FEMS Microbiol. Ecol. 2002, 40, 1–12. [Google Scholar] [CrossRef]
- Partensky, F.; Blanchot, J.; Vaulot, D. Differential distribution and ecology of Prochlorococcus and Synechococcus in oceanic waters. In Reviews in Marine Cyanobacteria. No. NS 19. Bulletin de l’Institut Oceanographique Monaco; Charpy, L., Larkum, A.W.D., Eds.; Musee Oceanographique: Monaco, 1999; Volume 19, pp. 457–475. [Google Scholar]
- Willey, J.M.; Waterbury, J.B. Chemotaxis toward Nitrogenous Compounds by Swimming Strains of Marine Synechococcus spp. Appl. Environ. Microbiol. 1989, 55, 1888–1894. [Google Scholar] [CrossRef] [Green Version]
- Steunou, A.S.; Jensen, S.I.; Brecht, E.; Becraft, E.D.; Bateson, M.M.; Kilian, O.; Bhaya, D.; Ward, D.M.; Peters, J.W.; Grossman, A.R.; et al. Regulation of nif gene expression and the energetics of N2 fixation over the diel cycle in a hot spring microbial mat. ISME J. 2008, 2, 364–378. [Google Scholar] [CrossRef] [Green Version]
- Koroleff, F. Total and organic nitrogen. In Methods for Seawater Analysis; Grasshoff, K., Ehrhardt, M., Kremling, K., Eds.; Verlag Chemie: Weinheim, Germany, 1983; pp. 162–168. [Google Scholar]
- Lozovik, P.A.; Efremenko, N.A. (Eds.) Analytical, Kinetic and Computational Methods in Hydrochemical Practice; Nestor-History: S-Pb, Russia, 2017; p. 272. (In Russian) [Google Scholar]
- Golterman, H.L. Methods for Chemical Analysis of Fresh Waters; Blackwell Scientific: Qxford, UK, 1971; p. 166. [Google Scholar]
- Biderre-Petit, C.; Taib, N.; Gardon, H.; Hochart, C.; Debroas, D. New insights into the pelagic microorganisms involved in the methane cycle in the meromictic Lake Pavin through metagenomics. FEMS Microbiol. Ecol. 2019, 95, fiy183. [Google Scholar] [CrossRef]
- Oswald, K.; Milucka, J.; Brand, A.; Hach, P.; Littmann, S.; Wehrli, B.; Kuypers, M.M.M.; Schubert, C.J. Aerobic gammaproteobacterial methanotrophs mitigate methane emissions from oxic and anoxic lake waters. Limnol. Oceanogr. 2016, 61, S101–S118. [Google Scholar] [CrossRef] [Green Version]
- Rodrigo, M.; Miracle, M.; Vicente, E. The meromictic Lake La Cruz (Central Spain). Patterns of stratification. Aquat. Sci. 2001, 63, 406–416. [Google Scholar] [CrossRef]
- Llirós, M.; Garciá-Armisen, T.; Darchambeau, F.; Morana, C.; Triadó-Margarit, X.; Inceoğlu, Ö.; Borrego, C.M.; Bouillon, S.; Servais, P.; Borges, A.V.; et al. Pelagic photoferrotrophy and iron cycling in a modern ferruginous basin. Sci. Rep. 2015, 5, 13803. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Umanskaya, M.V.; Tarasova, N.G.; Gorbunov, M.Y. Phototrophic plankton of siderotrophic meromictic Lake Kuznechikha (Republic Mari El, Russia). Inland Water Biol. 2017, 10, 158–167. [Google Scholar] [CrossRef]
- Hove, P.V.; Belzile, C.; Gibson, J.A.E.; Vincent, W.F. Coupled landscape–lake evolution in High Arctic Canada. Can. J. Earth Sci. 2006, 43, 533–546. [Google Scholar] [CrossRef]
- Stepanova, A.B.; Sharafutdinova, G.F.; Voyakina, E.Y. Hydrochemical features of Valaam Island small lakes. Sci. Notes RSHU 2010, 12, 97–110. (In Russian) [Google Scholar]
- Culver, D.A. Biogenic meromixis and stability in a soft-water lake. Limnol. Oceanogr. 1977, 22, 667–686. [Google Scholar] [CrossRef]
- Newton, R.J.; Jones, S.E.; Eiler, A.; McMahon, K.D.; Bertilsson, S. A guide to the natural history of freshwater lake bacteria. Microbiol. Mol. Biol. Rev. 2011, 75, 14–49. [Google Scholar] [CrossRef] [Green Version]
- Melack, J.M.; Jellison, R. Limnological conditions in Mono Lake: Contrasting monomixis and meromixis in the 1990s. Hydrobiologia 1998, 384, 21–39. [Google Scholar] [CrossRef]
- Zabelina, S.A.; Pokrovsky, O.S.; Klimov, S.I.; Chupakov, A.V.; Kokryatskaya, N.M.; Vorobyova, T.Y. The first results of the methane concentration in the stratified lakes of the River Svetlaya (the catchment area of the White Sea). In Geology of Seas and Oceans, Proceedings of the XX International Scientific Conference (School) on Marine Geology, Moscow, Russia, 18–23 November 2013; Lisitzin, A.P., Politova, N.V., Shevchenko, V.P., Eds.; GEOS: Moscow, Russia, 2013; Volume III, pp. 156–160. (In Russian) [Google Scholar]
- Brochier-Armanet, C.; Boussau, B.; Gribaldo, S.; Forterre, P. Mesophilic crenarchaeota: Proposal for a third archaeal phylum, the Thaumarchaeota. Nat. Rev. Microbiol. 2008, 6, 245–252. [Google Scholar] [CrossRef] [PubMed]
- Hugoni, M.; Etien, S.; Bourges, A.; Lepère, C.; Domaizon, I.; Mallet, C.; Bronner, G.; Debroas, D.; Mary, I. Dynamics of ammonia-oxidizing Archaea and Bacteria in contrasted freshwater ecosystems. Res. Microbiol. 2013, 164, 360–370. [Google Scholar] [CrossRef] [PubMed]
- Lliros, M.; Gich, F.; Plasencia, A.; Auguet, J.C.; Darchambeau, F.; Casamayor, E.O.; Descy, J.P.; Borrego, C. Vertical distribution of ammonia-oxidizing crenarchaeota and methanogens in the epipelagic waters of Lake Kivu (Rwanda-Democratic Republic of the Congo). Appl. Environ. Microbiol. 2010, 76, 6853–6863. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Pouliot, J.; Galand, P.E.; Lovejoy, C.; Vincent, W.F. Vertical structure of archaeal communities and the distribution of ammonia monooxygenase A gene variants in two meromictic High Arctic lakes. Environ. Microbiol. 2009, 11, 687–699. [Google Scholar] [CrossRef] [PubMed]
- Hatzenpichler, R.; Lebedeva, A.; Spieck, E.; Stoecker, K.; Richter, A.; Daims, H.; Wagner, M. A moderately thermophilic ammoniaoxidizing crenarchaeote from a hot spring. Proc. Natl. Acad. Sci. USA 2008, 105, 2134–2139. [Google Scholar] [CrossRef] [Green Version]
- Park, H.D.; Wells, G.F.; Bae, H.; Criddle, C.S.; Francis, C.A. Occurrence of ammonia-oxidizing archaea in wastewater treatment plant bioreactors. Appl. Environ. Microb. 2006, 72, 5643–5647. [Google Scholar] [CrossRef] [Green Version]
- Erguder, T.H.; Boon, N.; Wittebolle, L.; Marzorati, M.; Verstraete, W. Environmental factors shaping the ecological niches of ammonia-oxidizing archaea. FEMS Microbiol. Rev. 2009, 33, 855–869. [Google Scholar] [CrossRef] [Green Version]
- Lam, P.; Jensen, M.M.; Lavik, G.; McGinnis, D.F.; Muller, B.; Schubert, C.J.; Amann, R.; Thamdrup, B.; Kuypers, M.M.M. Linking crenarchaeal and bacterial nitrification to anammox in the Black Sea. Proc. Natl. Acad. Sci. USA 2007, 104, 7104–7109. [Google Scholar] [CrossRef] [Green Version]
- Beman, J.M.; Popp, B.N.; Francis, C.A. Molecular and biogeochemical evidence for ammonia oxidation by marine Crenarchaeota in the Gulf of California. ISME J. 2008, 2, 429–441. [Google Scholar] [CrossRef] [Green Version]
- Molina, V.; Belmar, L.; Ulloa, O. High diversity of ammonia-oxidizing archaea in permanent and seasonal oxygen-deficient waters of the eastern South Pacific. Environ. Microbiol. 2010, 12, 2450–2465. [Google Scholar] [CrossRef] [PubMed]
- Simon, J. Enzymology and bioenergetics of respiratory nitrite ammonification. FEMS Microbiol. Rev. 2002, 26, 285–309. [Google Scholar] [CrossRef]
- Kartal, B.; Kuypers, M.M.M.; Lavik, G.; Schalk, J.; Op den Camp, H.J.M.; Jetten, M.S.; Strous, M. Anammox bacteria disguised as denitrifiers: Nitrate reduction to dinitrogen gas via nitrite and ammonium. Environ. Microbiol. 2007, 9, 635–642. [Google Scholar] [CrossRef]
- Flores, E.; Ramos, J.L.; Herrero, A.; Guerrero, M.G. Nitrate assimilation by cyanobacteria. In Photosynthetic Prokaryotes: Cell Differentiation and Function; Papageorgiou, G.C., Packer, L., Eds.; Elsevier: New York, NY, USA, 1983; pp. 363–387. [Google Scholar]
- Martínez-Espinosa, R.M.; Marhuenda-Egea, F.C.; Bonete, M.J. Assimilatory nitrate reductase from the haloarchaeon Haloferax mediterranei: Purification and characterisation. FEMS Microbiol. Lett. 2001, 204, 381–385. [Google Scholar] [CrossRef]
- Martínez-Espinosa, R.M.; Lledó, B.; Marhuenda-Egea, F.C.; Bonete, M.J. The effect of ammonium on assimilatory nitrate reduction in the haloarchaeon Haloferax mediterranei. Extremophiles 2007, 11, 759–767. [Google Scholar] [CrossRef] [PubMed]
- Martínez-Espinosa, R.M. Microorganisms and their metabolic capabilities in the context of the biogeochemical nitrogen cycle at extreme environments. Int. J. Mol. Sci. 2020, 21, 4228. [Google Scholar] [CrossRef]
- Moreno-Vivián, C.; Cabello, P.; Martínez-Luque, M.; Blasco, R.; Castillo, F. Prokaryotic nitrate reduction: Molecular properties and functional distinction among bacterial nitrate reductases. J. Bacteriol. 1999, 181, 6573–6584. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Richardson, D. Introduction: Nitrate reduction and the nitrogen cycle. Cell. Mol. Life Sci. 2001, 58, 163–164. [Google Scholar] [CrossRef] [PubMed]
- Daims, H.; Lebedeva, E.V.; Pjevac, P.; Han, P.; Herbold, C.; Albertsen, M.; Jehmlich, N.; Palatinszky, M.; Vierheilig, J.; Bulaev, A.; et al. Complete nitrification by Nitrospira bacteria. Nature 2015, 528, 504–509. [Google Scholar] [CrossRef]
- Kits, K.D.; Sedlacek, C.J.; Lebedeva, E.V.; Han, P.; Bulaev, A.; Pjevac, P.; Daebeler, A.; Romano, S.; Albertsen, M. Kinetic analysis of a complete nitrifier reveals an oligotrophic lifestyle. Nature 2017, 549, 269–272. [Google Scholar] [CrossRef] [Green Version]
- Froelich, P.N.; Klinkhammer, G.P.; Bender, M.L.; Luedtke, N.A.; Heath, G.R.; Cullen, D.; Dauphin, P.; Hammond, D.; Hartman, B.; Maynard, V. Early oxidation of organic matter in pelagic sediments of eastern equatorial Atlantic: Suboxic diagenesis. Geochim. Cosmochim. Acta 1979, 43, 1075–1090. [Google Scholar] [CrossRef]
- Luther, G.W.; Sundby, B.; Lewis, B.L.; Brendel, P.J.; Silverberg, N. Interactions of manganese with the nitrogen cycle: Alternative pathways to dinitrogen. Geochim. Cosmochim. Acta 1997, 61, 4043–4052. [Google Scholar] [CrossRef]
- Hulth, S.; Aller, R.C.; Gilbert, F. Coupled anoxic nitrification/manganese reduction in marine sediments. Geochim. Cosmochim. Acta 1999, 63, 49–66. [Google Scholar] [CrossRef] [Green Version]
- Jones, C.; Crowe, S.A.; Sturm, A.; Leslie, K.L.; MacLean, L.C.W.; Katsev, S.; Henny, C.; Fowle, D.A.; Canfield, D.E. Biogeochemistry of manganese in ferruginous Lake Matano, Indonesia. Biogeosciences 2011, 8, 2977–2991. [Google Scholar] [CrossRef] [Green Version]
- Bratina, B.J.; Stevenson, B.S.; Green, W.J.; Schmidt, T.M. Manganese reduction by microbes from oxic regions of the Lake Vanda (Antarctica) water column. Appl. Environ. Microbiol. 1998, 64, 3791–3797. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Canfield, D.; Kristensen, E.; Thamdrup, B. Aquatic Geomicrobiology; Advances Marine Biology Series; Southward, A.J., Tyler, P.A., Young, C.M., Fuiman, L.A., Eds.; Elsevier: San Diego, CA, USA, 2005; p. 640. [Google Scholar]
- Konneke, M.; Bernhard, A.E.; de la Torre, J.R.; Walker, C.B.; Waterbury, J.B.; Stahl, D.A. Isolation of an autotrophic ammonia-oxidizing marine archaeon. Nature 2005, 437, 543–546. [Google Scholar] [CrossRef] [PubMed]
- Walker, C.B.; de la Torre, J.R.; Klotz, M.G.; Urakawa, H.; Pinel, N.; Arp, D.J.; Brochier-Armanet, C.; Chain, P.S.J.; Chan, P.P.; Gollabgir, A.; et al. Nitrosopumilus maritimus genome reveals unique mechanisms for nitrification and autotrophy in globally distributed marine crenarchaea. Proc. Natl. Acad. Sci. USA 2010, 107, 8818–8823. [Google Scholar] [CrossRef] [Green Version]
- Mulder, A.; van de Graaf, A.A.; Robertson, L.A.; Kuenen, J.G. Anaerobic ammonium oxidation discovered in a denitrifying fluidized bed reactor. FEMS Microbiol. Ecol. 1995, 16, 177–184. [Google Scholar] [CrossRef]
- Jensen, M.M.; Thamdrup, B.; Dalsgaard, T. Effects of specific inhibitors on anammox and denitrification in marine sediments. Appl. Environ. Microbiol. 2007, 73, 3151–3158. [Google Scholar] [CrossRef] [Green Version]
- Hu, B.-I.; Shen, L.-D.; Xu, X.-Y.; Zheng, P. Anaerobic ammonium oxidation (anammox) in different natural ecosystems. Biochem. Soc. Trans. 2011, 39, 1811–1816. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Schubert, C.J.; Durisch-Kaiser, E.; Wehrli, B.; Thamdrup, B.; Lam, P.; Kuypers, M.M.M. Anaerobic ammonium oxidation in a tropical freshwater system (Lake Tangayika). Environ. Microbiol. 2006, 8, 1857–1863. [Google Scholar] [CrossRef] [PubMed]
- Hamersley, M.R.; Woebken, D.; Boehrer, B.; Schultze, M.; Lavik, G.; Kuypers, M.M.M. Water column anammox and denitrification in a temperate permanently stratified lake (Lake Rassnitzer, Germany). Syst. Appl. Microbiol. 2009, 32, 571–582. [Google Scholar] [CrossRef] [PubMed]
- Wenk, C.B.; Zopfi, J.; Blees, J.; Veronesi, M.; Niemann, H.; Lehmann, M.F. Community N and O isotope fractionation by sulphide-dependent denitrification and anammox in a stratified lacustrine water column. Geochim. Cosmochim. Acta 2014, 125, 551–563. [Google Scholar] [CrossRef]
- Strous, M.; Pelletier, E.; Mangenot, S.; Rattei, T.; Lehner, A.; Taylor, M.; Horn, M.; Daims, H.; Bartol-Mavel, D.; Wincker, P.; et al. Deciphering the evolution and metabolism of an anammox bacterium from a community genome. Nature 2006, 440, 790–794. [Google Scholar] [CrossRef]
- Anthony, C. The Biochemistry of Methylotrophs; Academic Press: London, UK, 1982. [Google Scholar]
| Lake | N-NH4 | N-NO2 | N-NO3 | Reference | ||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Mix | Chem | Monim | Mix | Chem | Monim | Mix | Chem | Monim | ||
| Lake Svetloe | 0.5–1.2 | 13–91 | 144–177 | 0–0.07 | 0.3–0.8 | 0.6–1.4 | 5–9 | 5–8 | 9–13 | This Research |
| Lake Matano | up to 20 | 20–300 | 200–300 | <0.1 | all layers < 0.1 only 90–100 m 0.1–0.2 | [21] | ||||
| Lake Pavin | – | – | max 389 | – | – | – | max 32 | – | – | [49] |
| Lake La Cruz | 1.1–12.8 | 122.2 | up to 3000 | 0.04–0.11 | 0 | 0 | 5–20 | 0 | 0 | [18,51] |
| Kabuno Bay | – | – | – | N-(NO2 + NO3) | [52] | |||||
| up to 1 | Up to 0.5 in dry season up to 1.5 | up to 0.5 | ||||||||
| Lake Kuznechikha | 1.4–3.5 | 17.8–50 | 83–121 | – | – | – | – | – | – | [53] |
| Char lake | – | – | – | – | – | – | <0.7 | – | <0.7 | [54] |
| Lake Chernoe | – | – | up to 265 | – | – | – | – | – | – | [55] |
| Hall Lake | 0 | – | 305 | – | – | – | 13.3 | – | 4.4 | [18,56] |
| Lake Zug | 10 | – | – | – | 20–25 | 17–18 | 1–5 | [50] | ||
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Vorobyeva, T.Y.; Chupakova, A.A.; Chupakov, A.V.; Zabelina, S.A.; Moreva, O.Y.; Pokrovsky, O.S. Distribution of Dissolved Nitrogen Compounds in the Water Column of a Meromictic Subarctic Lake. Nitrogen 2021, 2, 428-443. https://doi.org/10.3390/nitrogen2040029
Vorobyeva TY, Chupakova AA, Chupakov AV, Zabelina SA, Moreva OY, Pokrovsky OS. Distribution of Dissolved Nitrogen Compounds in the Water Column of a Meromictic Subarctic Lake. Nitrogen. 2021; 2(4):428-443. https://doi.org/10.3390/nitrogen2040029
Chicago/Turabian StyleVorobyeva, Taisiya Ya., Anna A. Chupakova, Artem V. Chupakov, Svetlana A. Zabelina, Olga Y. Moreva, and Oleg S. Pokrovsky. 2021. "Distribution of Dissolved Nitrogen Compounds in the Water Column of a Meromictic Subarctic Lake" Nitrogen 2, no. 4: 428-443. https://doi.org/10.3390/nitrogen2040029
APA StyleVorobyeva, T. Y., Chupakova, A. A., Chupakov, A. V., Zabelina, S. A., Moreva, O. Y., & Pokrovsky, O. S. (2021). Distribution of Dissolved Nitrogen Compounds in the Water Column of a Meromictic Subarctic Lake. Nitrogen, 2(4), 428-443. https://doi.org/10.3390/nitrogen2040029







