Prognosis and Personalized In Silico Prediction of Treatment Efficacy in Cardiovascular and Chronic Kidney Disease: A Proof-of-Concept Study
Abstract
:1. Introduction
2. Results
2.1. Clinical Characteristics of Population
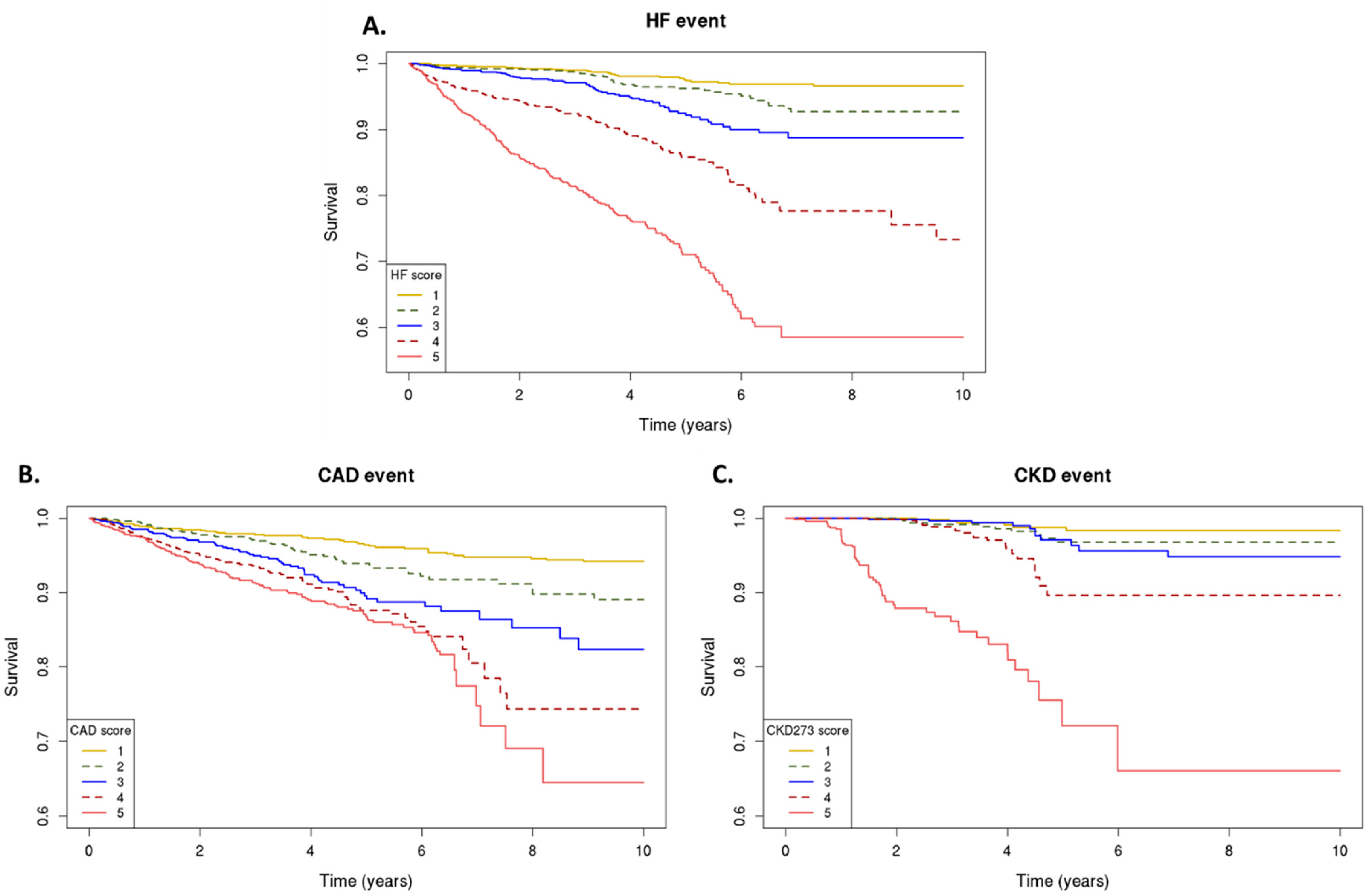
2.2. Peptide-Based Classifiers and Prediction of Events
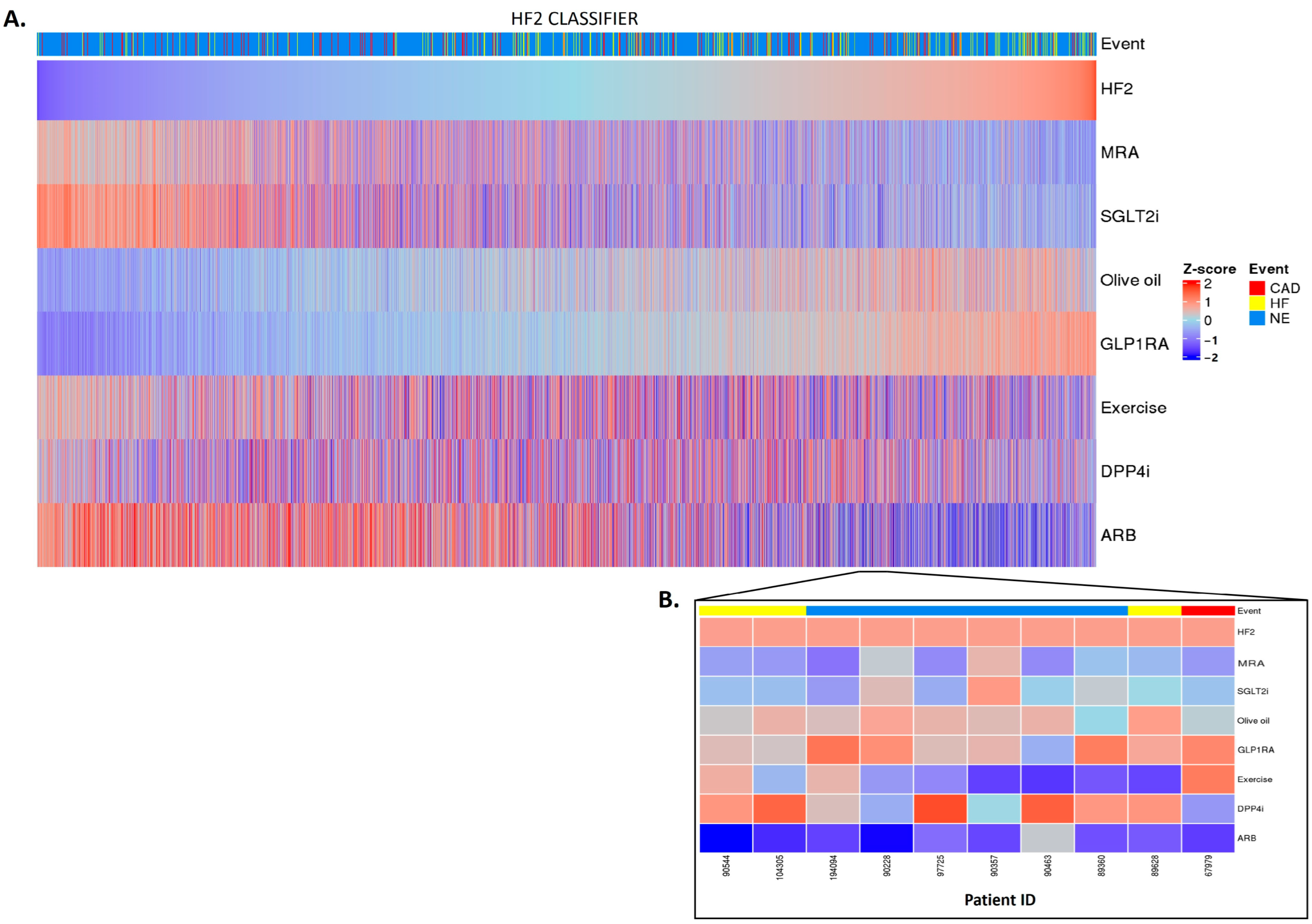
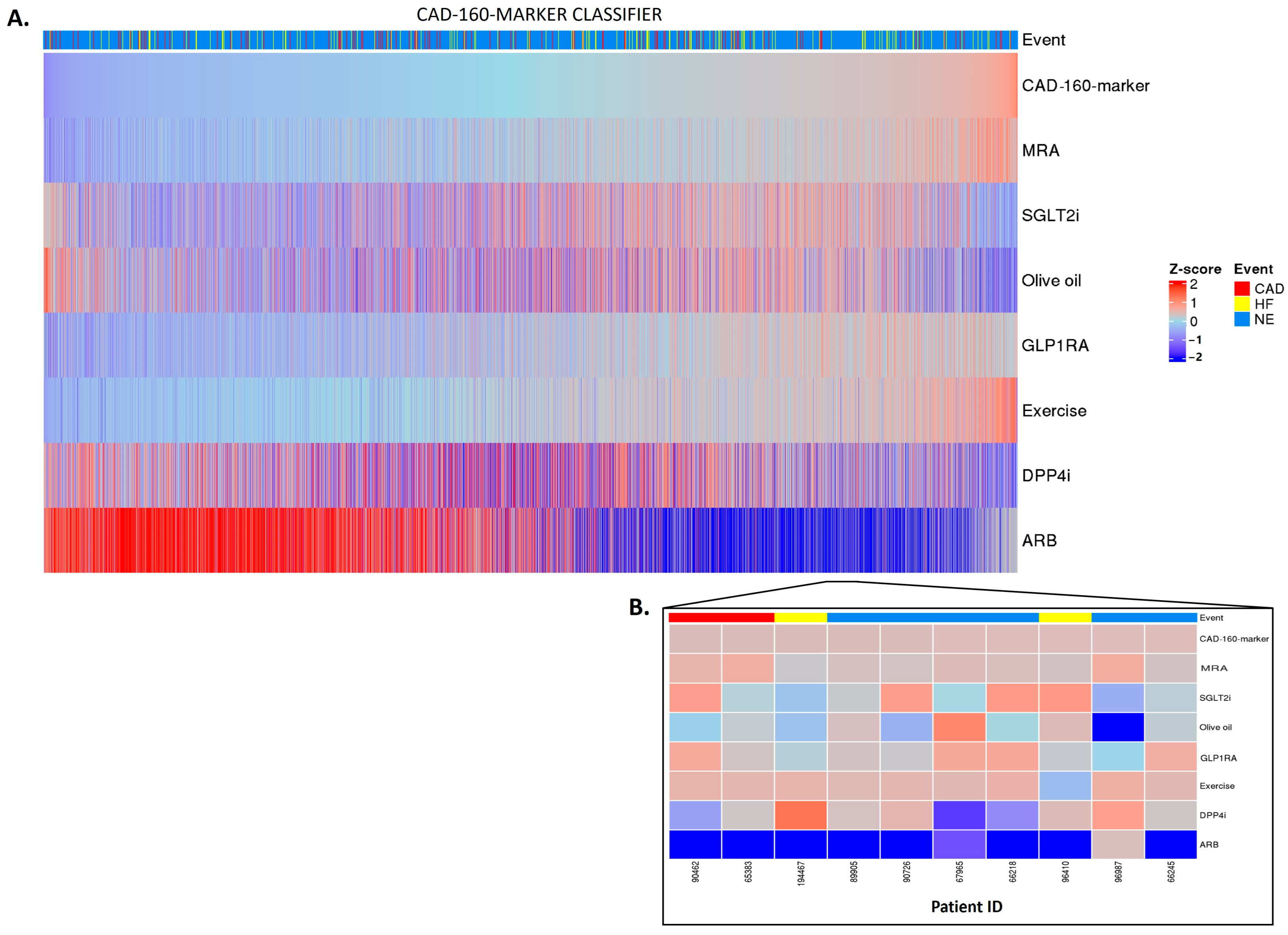
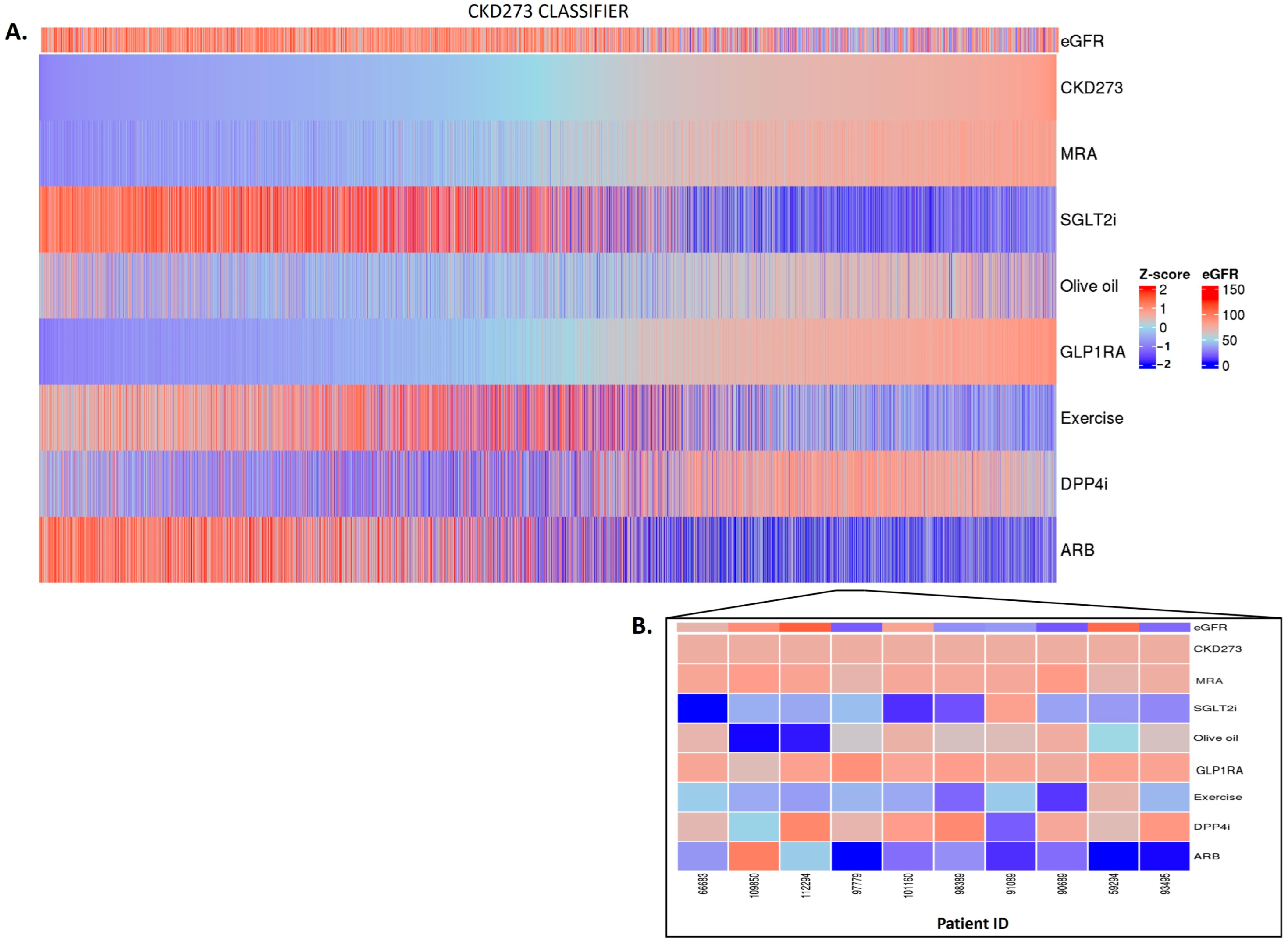
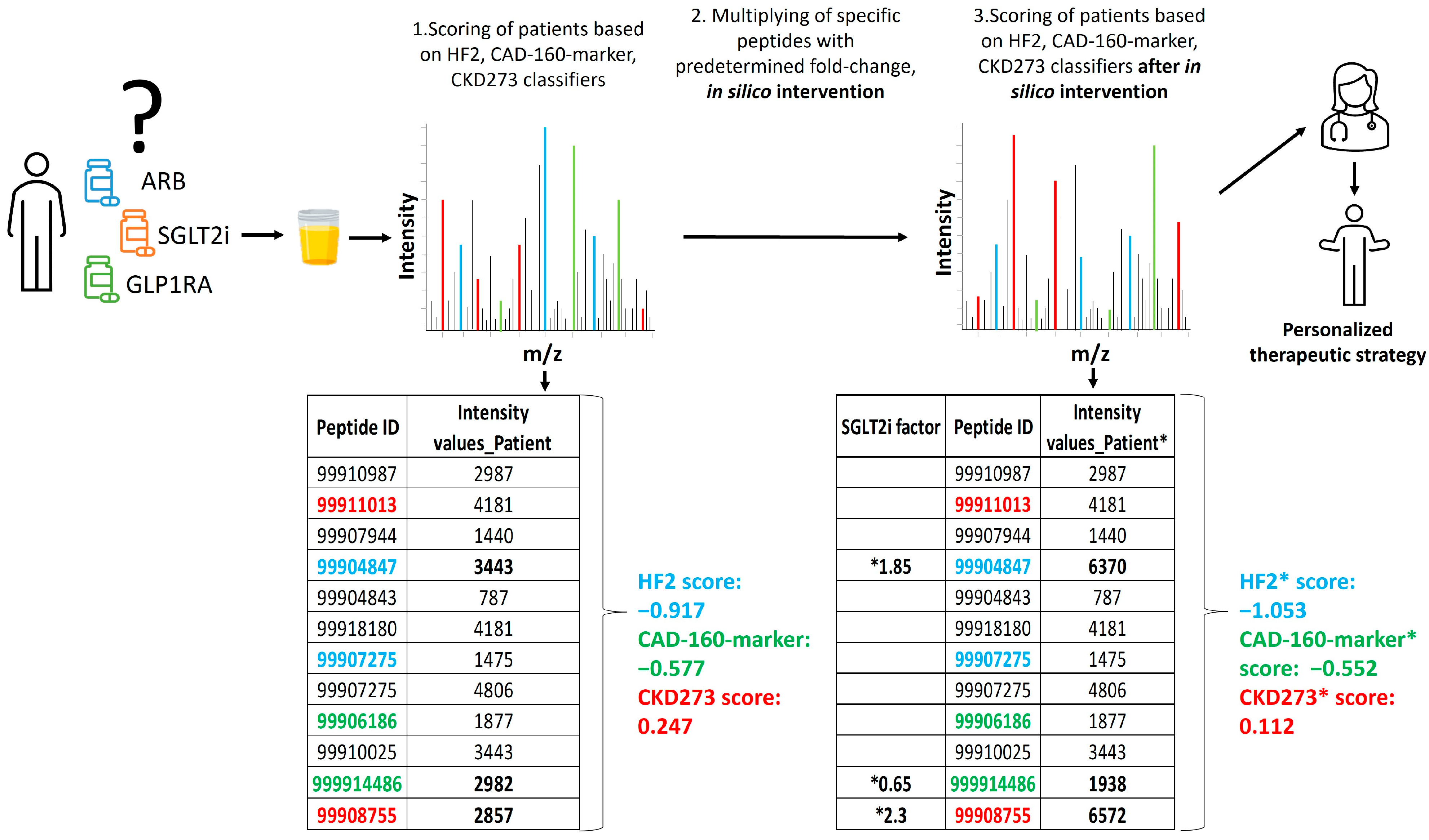
2.3. Personalized In Silico Prediction of Treatment Efficacy
3. Discussion
4. Materials and Methods
4.1. Study Participants and Study Design
4.2. Peptide-Based Classifiers and Prediction of Events
4.3. In Silico Impact of Treatments
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Ezzati, M.; Obermeyer, Z.; Tzoulaki, I.; Mayosi, B.M.; Elliott, P.; Leon, D.A. Contributions of Risk Factors and Medical Care to Cardiovascular Mortality Trends. Nat. Rev. Cardiol. 2015, 12, 508–530. [Google Scholar] [CrossRef] [PubMed]
- Webster, A.C.; Nagler, E.V.; Morton, R.L.; Masson, P. Chronic Kidney Disease. Lancet 2017, 389, 1238–1252. [Google Scholar] [CrossRef] [PubMed]
- Bays, H.E.; Taub, P.R.; Epstein, E.; Michos, E.D.; Ferraro, R.A.; Bailey, A.L.; Kelli, H.M.; Ferdinand, K.C.; Echols, M.R.; Weintraub, H.; et al. Ten Things to Know about Ten Cardiovascular Disease Risk Factors. Am. J. Prev. Cardiol. 2021, 5, 100149. [Google Scholar] [CrossRef] [PubMed]
- Tzoulaki, I.; Elliott, P.; Kontis, V.; Ezzati, M. Worldwide Exposures to Cardiovascular Risk Factors and Associated Health Effects: Current Knowledge and Data Gaps. Circulation 2016, 133, 2314–2333. [Google Scholar] [CrossRef]
- Chen, T.K.; Knicely, D.H.; Grams, M.E. Chronic Kidney Disease Diagnosis and Management: A Review. JAMA 2019, 322, 1294–1304. [Google Scholar] [CrossRef] [PubMed]
- Sinha, A.; Ning, H.; Carnethon, M.R.; Allen, N.B.; Wilkins, J.T.; Lloyd-Jones, D.M.; Khan, S.S. Race- and Sex-Specific Population Attributable Fractions of Incident Heart Failure. Circ. Heart Fail. 2021, 14, e008113. [Google Scholar] [CrossRef] [PubMed]
- Pontillo, C.; Zhang, Z.-Y.; Schanstra, J.P.; Jacobs, L.; Zürbig, P.; Thijs, L.; Ramírez-Torres, A.; Heerspink, H.J.L.; Lindhardt, M.; Klein, R.; et al. Prediction of Chronic Kidney Disease Stage 3 by CKD273, a Urinary Proteomic Biomarker. Kidney Int. Rep. 2017, 2, 1066–1075. [Google Scholar] [CrossRef] [PubMed]
- Vasan, R.S. Biomarkers of Cardiovascular Disease: Molecular Basis and Practical Considerations. Circulation 2006, 113, 2335–2362. [Google Scholar] [CrossRef]
- Hense, H.-W. Observations, Predictions and Decisions—Assessing Cardiovascular Risk Assessment. Int. J. Epidemiol. 2004, 33, 235–239. [Google Scholar] [CrossRef]
- Musunuru, K.; Hershberger, R.E.; Day, S.M.; Klinedinst, N.J.; Landstrom, A.P.; Parikh, V.N.; Prakash, S.; Semsarian, C.; Sturm, A.C. Genetic Testing for Inherited Cardiovascular Diseases: A Scientific Statement From the American Heart Association. Circ. Genomic Precis. Med. 2020, 13, E000067. [Google Scholar] [CrossRef]
- Tofte, N.; Lindhardt, M.; Adamova, K.; Bakker, S.J.L.; Beige, J.; Beulens, J.W.J.; Birkenfeld, A.L.; Currie, G.; Delles, C.; Dimos, I.; et al. Early Detection of Diabetic Kidney Disease by Urinary Proteomics and Subsequent Intervention with Spironolactone to Delay Progression (PRIORITY): A Prospective Observational Study and Embedded Randomised Placebo-Controlled Trial. Lancet Diabetes Endocrinol. 2020, 8, 301–312. [Google Scholar] [CrossRef] [PubMed]
- Ziff, O.J.; Samra, M.; Howard, J.P.; Bromage, D.I.; Ruschitzka, F.; Francis, D.P.; Kotecha, D. Beta-Blocker Efficacy across Different Cardiovascular Indications: An Umbrella Review and Meta-Analytic Assessment. BMC Med. 2020, 18, 103. [Google Scholar] [CrossRef] [PubMed]
- Andrikou, E.; Tsioufis, C.; Andrikou, I.; Leontsinis, I.; Tousoulis, D.; Papanas, N. GLP-1 Receptor Agonists and Cardiovascular Outcome Trials: An Update. Hell. J. Cardiol. 2019, 60, 347–351. [Google Scholar] [CrossRef] [PubMed]
- Ong, H.T.; Ong, L.M.; Ho, J.J. Angiotensin-Converting Enzyme Inhibitors (ACEIs) and Angiotensin-Receptor Blockers (ARBs) in Patients at High Risk of Cardiovascular Events: A Meta-Analysis of 10 Randomised Placebo-Controlled Trials. ISRN Cardiol. 2013, 2013, 478597. [Google Scholar] [CrossRef] [PubMed]
- Patoulias, D.I.; Boulmpou, A.; Teperikidis, E.; Katsimardou, A.; Siskos, F.; Doumas, M.; Papadopoulos, C.E.; Vassilikos, V. Cardiovascular Efficacy and Safety of Dipeptidyl Peptidase-4 Inhibitors: A Meta-Analysis of Cardiovascular Outcome Trials. World J. Cardiol. 2021, 13, 585–592. [Google Scholar] [CrossRef] [PubMed]
- Usman, M.S.; Siddiqi, T.J.; Memon, M.M.; Khan, M.S.; Rawasia, W.F.; Talha Ayub, M.; Sreenivasan, J.; Golzar, Y. Sodium-Glucose Co-Transporter 2 Inhibitors and Cardiovascular Outcomes: A Systematic Review and Meta-Analysis. Eur. J. Prev. Cardiol. 2018, 25, 495–502. [Google Scholar] [CrossRef] [PubMed]
- Yang, Y.; Gao, J.; Qin, Z.; Lu, Y.; Xu, Y.; Guo, J.; Cui, X.; Zhang, J.; Tang, J. The Present Clinical Treatment and Future Emerging Interdisciplinary for Heart Failure: Where We Are and What We Can Do. Intensive Care Res. 2023, 3, 3–11. [Google Scholar] [CrossRef]
- Shlipak, M.G.; Tummalapalli, S.L.; Boulware, L.E.; Grams, M.E.; Ix, J.H.; Jha, V.; Kengne, A.P.; Madero, M.; Mihaylova, B.; Tangri, N.; et al. The Case for Early Identification and Intervention of Chronic Kidney Disease: Conclusions from a Kidney Disease: Improving Global Outcomes (KDIGO) Controversies Conference. Kidney Int. 2021, 99, 34–47. [Google Scholar] [CrossRef]
- Wendt, R.; Thijs, L.; Kalbitz, S.; Mischak, H.; Siwy, J.; Raad, J.; Metzger, J.; Neuhaus, B.; von der Leyen, H.; Dudoignon, E.; et al. A Urinary Peptidomic Profile Predicts Outcome in SARS-CoV-2-Infected Patients. eClinicalMedicine 2021, 36, 6–7. [Google Scholar] [CrossRef]
- Argilés, À.; Siwy, J.; Duranton, F.; Gayrard, N.; Dakna, M.; Lundin, U.; Osaba, L.; Delles, C.; Mourad, G.; Weinberger, K.M.; et al. CKD273, a New Proteomics Classifier Assessing CKD and Its Prognosis. PLoS ONE 2013, 8, e62837. [Google Scholar] [CrossRef]
- Zhang, Z.-Y.; Nkuipou-Kenfack, E.; Staessen, J.A. Urinary Peptidomic Biomarker for Personalized Prevention and Treatment of Diastolic Left Ventricular Dysfunction. Proteomics. Clin. Appl. 2019, 13, e1800174. [Google Scholar] [CrossRef] [PubMed]
- Curovic, V.R.; Eickhoff, M.K.; Rönkkö, T.; Frimodt-Møller, M.; Hansen, T.W.; Mischak, H.; Rossing, P.; Ahluwalia, T.S.; Persson, F. Dapagliflozin Improves the Urinary Proteomic Kidney-Risk Classifier CKD273 in Type 2 Diabetes with Albuminuria: A Randomized Clinical Trial. Diabetes Care 2022, 45, 2662–2668. [Google Scholar] [CrossRef] [PubMed]
- Siwy, J.; Klein, T.; Rosler, M.; von Eynatten, M. Urinary Proteomics as a Tool to Identify Kidney Responders to Dipeptidyl Peptidase-4 Inhibition: A Hypothesis-Generating Analysis from the MARLINA-T2D Trial. Proteomics. Clin. Appl. 2019, 13, e1800144. [Google Scholar] [CrossRef] [PubMed]
- Shin, M.; Ahn, S.; Jung, J.; Hyung, S.; Kim, K.-M.; Kim, S.T.; Kang, W.K.; Lee, J. Impact of Programmed Death-Ligand 1 (PD-L1) Positivity on Clinical and Molecular Features of Patients with Metastatic Gastric Cancer. Cancer Med. 2023, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Soverini, S.; Mancini, M.; Bavaro, L.; Cavo, M.; Martinelli, G. Chronic Myeloid Leukemia: The Paradigm of Targeting Oncogenic Tyrosine Kinase Signaling and Counteracting Resistance for Successful Cancer Therapy. Mol. Cancer 2018, 17, 49. [Google Scholar] [CrossRef] [PubMed]
- André, F.; Ciruelos, E.; Rubovszky, G.; Campone, M.; Loibl, S.; Rugo, H.S.; Iwata, H.; Conte, P.; Mayer, I.A.; Kaufman, B.; et al. Alpelisib for PIK3CA-Mutated, Hormone Receptor-Positive Advanced Breast Cancer. N. Engl. J. Med. 2019, 380, 1929–1940. [Google Scholar] [CrossRef] [PubMed]
- Hyman, D.M.; Piha-Paul, S.A.; Won, H.; Rodon, J.; Saura, C.; Shapiro, G.I.; Juric, D.; Quinn, D.I.; Moreno, V.; Doger, B.; et al. HER Kinase Inhibition in Patients with HER2- and HER3-Mutant Cancers. Nature 2018, 554, 189–194. [Google Scholar] [CrossRef] [PubMed]
- Tye, S.C.; de Vries, S.T.; Wanner, C.; Denig, P.; Heerspink, H.J.L. Prediction of the Effects of Empagliflozin on Cardiovascular and Kidney Outcomes Based on Short-Term Changes in Multiple Risk Markers. Front. Pharmacol. 2021, 12, 786706. [Google Scholar] [CrossRef]
- Wei, D.; Melgare, J.D.; van Aelst, L.; Vanassche, T.; Verhamme, P.; Janssens, S.; Peter, K.; Zhang, Z.-Y. Prediction of Coronary Artery Disease Using Urinary Proteomics. J. Hypertens. 2023, 41, e95. [Google Scholar] [CrossRef]
- Zhang, Z.; Staessen, J.A.; Thijs, L.; Gu, Y.; Liu, Y.; Jacobs, L.; Koeck, T.; Zürbig, P.; Mischak, H.; Kuznetsova, T. Left Ventricular Diastolic Function in Relation to the Urinary Proteome: A Proof-of-Concept Study in a General Population. Int. J. Cardiol. 2014, 176, 158–165. [Google Scholar] [CrossRef]
- Lamb, J.; Crawford, E.D.; Peck, D.; Modell, J.W.; Blat, I.C.; Wrobel, M.J.; Lerner, J.; Brunet, J.-P.; Subramanian, A.; Ross, K.N.; et al. The Connectivity Map: Using Gene-Expression Signatures to Connect Small Molecules, Genes, and Disease. Science 2006, 313, 1929–1935. [Google Scholar] [CrossRef] [PubMed]
- Subramanian, A.; Narayan, R.; Corsello, S.M.; Peck, D.D.; Natoli, T.E.; Lu, X.; Gould, J.; Davis, J.F.; Tubelli, A.A.; Asiedu, J.K.; et al. A Next Generation Connectivity Map: L1000 Platform and the First 1,000,000 Profiles. Cell 2017, 171, 1437–1452.e17. [Google Scholar] [CrossRef] [PubMed]
- Mokou, M.; Lygirou, V.; Angelioudaki, I.; Paschalidis, N.; Stroggilos, R.; Frantzi, M.; Latosinska, A.; Bamias, A.; Hoffmann, M.J.; Mischak, H.; et al. A Novel Pipeline for Drug Repurposing for Bladder Cancer Based on Patients’ Omics Signatures. Cancers 2020, 12, 3519. [Google Scholar] [CrossRef] [PubMed]
- Mukoyama, M.; Kuwabara, T. Role of Renin-Angiotensin System Blockade in Advanced CKD: To Use or Not to Use? Hypertens. Res. 2022, 45, 1072–1075. [Google Scholar] [CrossRef] [PubMed]
- Heerspink, H.J.L.; Stefánsson, B.V.; Correa-Rotter, R.; Chertow, G.M.; Greene, T.; Hou, F.-F.; Mann, J.F.E.; McMurray, J.J.V.; Lindberg, M.; Rossing, P.; et al. Dapagliflozin in Patients with Chronic Kidney Disease. N. Engl. J. Med. 2020, 383, 1436–1446. [Google Scholar] [CrossRef] [PubMed]
- McMurray, J.J.V.; Solomon, S.D.; Inzucchi, S.E.; Køber, L.; Kosiborod, M.N.; Martinez, F.A.; Ponikowski, P.; Sabatine, M.S.; Anand, I.S.; Bělohlávek, J.; et al. Dapagliflozin in Patients with Heart Failure and Reduced Ejection Fraction. N. Engl. J. Med. 2019, 381, 1995–2008. [Google Scholar] [CrossRef] [PubMed]
- Herrington, W.G.; Staplin, N.; Wanner, C.; Green, J.B.; Hauske, S.J.; Emberson, J.R.; Preiss, D.; Judge, P.; Mayne, K.J.; Ng, S.Y.A.; et al. Empagliflozin in Patients with Chronic Kidney Disease. N. Engl. J. Med. 2023, 388, 117–127. [Google Scholar] [CrossRef] [PubMed]
- Zinman, B.; Wanner, C.; Lachin, J.M.; Fitchett, D.; Bluhmki, E.; Hantel, S.; Mattheus, M.; Devins, T.; Johansen, O.E.; Woerle, H.J.; et al. Empagliflozin, Cardiovascular Outcomes, and Mortality in Type 2 Diabetes. N. Engl. J. Med. 2015, 373, 2117–2128. [Google Scholar] [CrossRef]
- Messerli, F.H.; Bangalore, S. Angiotensin Receptor Blockers Reduce Cardiovascular Events, Including the Risk of Myocardial Infarction. Circulation 2017, 135, 2085–2087. [Google Scholar] [CrossRef]
- Hoang, V.; Alam, M.; Addison, D.; Macedo, F.; Virani, S.; Birnbaum, Y. Efficacy of Angiotensin-Converting Enzyme Inhibitors and Angiotensin-Receptor Blockers in Coronary Artery Disease without Heart Failure in the Modern Statin Era: A Meta-Analysis of Randomized-Controlled Trials. Cardiovasc. Drugs Ther. 2016, 30, 189–198. [Google Scholar] [CrossRef]
- Tsujimoto, T.; Kajio, H. Spironolactone Use and Improved Outcomes in Patients with Heart Failure with Preserved Ejection Fraction with Resistant Hypertension. J. Am. Heart Assoc. 2020, 9, e018827. [Google Scholar] [CrossRef] [PubMed]
- Ferreira, J.P.; Cleland, J.G.F.; Girerd, N.; Pellicori, P.; Hazebroek, M.R.; Verdonschot, J.; Collier, T.J.; Petutschnigg, J.; Clark, A.L.; Staessen, J.A.; et al. Early and Late Renal Function Changes with Spironolactone in Patients at Risk of Developing Heart Failure: Findings from the HOMAGE Trial. Clin. Res. Cardiol. 2023, 112, 330–332. [Google Scholar] [CrossRef] [PubMed]
- Mita, T.; Katakami, N.; Yoshii, H.; Onuma, T.; Kaneto, H.; Osonoi, T.; Shiraiwa, T.; Kosugi, K.; Umayahara, Y.; Yamamoto, T.; et al. Alogliptin, a Dipeptidyl Peptidase 4 Inhibitor, Prevents the Progression of Carotid Atherosclerosis in Patients with Type 2 Diabetes: The Study of Preventive Effects of Alogliptin on Diabetic Atherosclerosis (SPEAD-A). Diabetes Care 2016, 39, 139–148. [Google Scholar] [CrossRef] [PubMed]
- Ishikawa, S.; Shimano, M.; Watarai, M.; Koyasu, M.; Uchikawa, T.; Ishii, H.; Inden, Y.; Takemoto, K.; Murohara, T. Impact of Sitagliptin on Carotid Intima-Media Thickness in Patients with Coronary Artery Disease and Impaired Glucose Tolerance or Mild Diabetes Mellitus. Am. J. Cardiol. 2014, 114, 384–388. [Google Scholar] [CrossRef] [PubMed]
- Chen, S.Y.; Kong, X.Q.; Zhang, K.F.; Luo, S.; Wang, F.; Zhang, J.J. DPP4 as a Potential Candidate in Cardiovascular Disease. J. Inflamm. Res. 2022, 15, 5457–5469. [Google Scholar] [CrossRef] [PubMed]
- Baksh, S.; Wen, J.; Mansour, O.; Chang, H.Y.; McAdams-DeMarco, M.; Segal, J.B.; Ehrhardt, S.; Alexander, G.C. Dipeptidyl Peptidase-4 Inhibitor Cardiovascular Safety in Patients with Type 2 Diabetes, with Cardiovascular and Renal Disease: A Retrospective Cohort Study. Sci. Rep. 2021, 11, 16637. [Google Scholar] [CrossRef] [PubMed]
- Xia, C.; Goud, A.; D’Souza, J.; Dahagam, C.H.; Rao, X.; Rajagopalan, S.; Zhong, J. DPP4 Inhibitors and Cardiovascular Outcomes: Safety on Heart Failure. Heart Fail. Rev. 2017, 22, 299–304. [Google Scholar] [CrossRef]
- Yang, L.; Wu, X.; Wang, Y.; Wang, C.; Hu, R.; Wu, Y. Effects of Exercise Training on Proteinuria in Adult Patients with Chronic Kidney Disease: A Systematic Review and Meta-Analysis. BMC Nephrol. 2020, 21, 172. [Google Scholar] [CrossRef]
- Winzer, E.B.; Woitek, F.; Linke, A. Physical Activity in the Prevention and Treatment of Coronary Artery Disease. J. Am. Heart Assoc. 2018, 7, e007725. [Google Scholar] [CrossRef]
- Martínez-González, M.A.; Sayón-Orea, C.; Bullón-Vela, V.; Bes-Rastrollo, M.; Rodríguez-Artalejo, F.; Yusta-Boyo, M.J.; García-Solano, M. Effect of Olive Oil Consumption on Cardiovascular Disease, Cancer, Type 2 Diabetes, and All-Cause Mortality: A Systematic Review and Meta-Analysis. Clin. Nutr. 2022, 41, 2659–2682. [Google Scholar] [CrossRef]
- Marrone, G.; Urciuoli, S.; Di Lauro, M.; Ruzzolini, J.; Ieri, F.; Vignolini, P.; Di Daniele, F.; Guerriero, C.; Nediani, C.; Di Daniele, N.; et al. Extra Virgin Olive Oil and Cardiovascular Protection in Chronic Kidney Disease. Nutrients 2022, 14, 4265. [Google Scholar] [CrossRef]
- Thompson, S.; Wiebe, N.; Gyenes, G.; Davies, R.; Radhakrishnan, J.; Graham, M. Physical Activity in Renal Disease (PAIRED) and the Effect on Hypertension: Study Protocol for a Randomized Controlled Trial. Trials 2019, 20, 109. [Google Scholar] [CrossRef] [PubMed]
- Cattadori, G.; Segurini, C.; Picozzi, A.; Padeletti, L.; Anzà, C. Exercise and Heart Failure: An Update. ESC Heart Fail. 2018, 5, 222–232. [Google Scholar] [CrossRef] [PubMed]
- Snell-Bergeon, J.K.; Maahs, D.M.; Ogden, L.G.; Kinney, G.L.; Hokanson, J.E.; Schiffer, E.; Rewers, M.; Mischak, H. Evaluation of Urinary Biomarkers for Coronary Artery Disease, Diabetes, and Diabetic Kidney Disease. Diabetes Technol. Ther. 2009, 11, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Smith, B.H.; Campbell, A.; Linksted, P.; Fitzpatrick, B.; Jackson, C.; Kerr, S.M.; Deary, I.J.; Macintyre, D.J.; Campbell, H.; McGilchrist, M.; et al. Cohort Profile: Generation Scotland: Scottish Family Health Study (GS:SFHS). The Study, Its Participants and Their Potential for Genetic Research on Health and Illness. Int. J. Epidemiol. 2013, 42, 689–700. [Google Scholar] [CrossRef] [PubMed]
- He, T.; Mischak, M.; Clark, A.L.; Campbell, R.T.; Delles, C.; Díez, J.; Filippatos, G.; Mebazaa, A.; McMurray, J.J.V.; González, A.; et al. Urinary Peptides in Heart Failure: A Link to Molecular Pathophysiology. Eur. J. Heart Fail. 2021, 23, 1875–1887. [Google Scholar] [CrossRef] [PubMed]
- Futter, J.E.; Cleland, J.G.F.; Clark, A.L. Body Mass Indices and Outcome in Patients with Chronic Heart Failure. Eur. J. Heart Fail. 2011, 13, 207–213. [Google Scholar] [CrossRef] [PubMed]
- He, T.; Melgarejo, J.D.; Clark, A.L.; Yu, Y.-L.; Thijs, L.; Díez, J.; López, B.; González, A.; Cleland, J.G.; Schanstra, J.P.; et al. Serum and Urinary Biomarkers of Collagen Type-I Turnover Predict Prognosis in Patients with Heart Failure. Clin. Transl. Med. 2021, 11, e267. [Google Scholar] [CrossRef] [PubMed]
- Lindhardt, M.; Persson, F.; Zürbig, P.; Stalmach, A.; Mischak, H.; de Zeeuw, D.; Lambers Heerspink, H.; Klein, R.; Orchard, T.; Porta, M.; et al. Urinary Proteomics Predict Onset of Microalbuminuria in Normoalbuminuric Type 2 Diabetic Patients, a Sub-Study of the DIRECT-Protect 2 Study. Nephrol. Dial. Transplant. 2017, 32, 1866–1873. [Google Scholar] [CrossRef]
- Kuznetsova, T.; Mischak, H.; Mullen, W.; Staessen, J.A. Urinary Proteome Analysis in Hypertensive Patients with Left Ventricular Diastolic Dysfunction. Eur. Heart J. 2012, 33, 2342–2350. [Google Scholar] [CrossRef]
- Packham, D.K.; Wolfe, R.; Reutens, A.T.; Berl, T.; Heerspink, H.L.; Rohde, R.; Ivory, S.; Lewis, J.; Raz, I.; Wiegmann, T.B.; et al. Sulodexide Fails to Demonstrate Renoprotection in Overt Type 2 Diabetic Nephropathy. J. Am. Soc. Nephrol. 2012, 23, 123–130. [Google Scholar] [CrossRef]
- Verbeke, F.; Siwy, J.; Van Biesen, W.; Mischak, H.; Pletinck, A.; Schepers, E.; Neirynck, N.; Magalhães, P.; Pejchinovski, M.; Pontillo, C.; et al. The Urinary Proteomics Classifier Chronic Kidney Disease 273 Predicts Cardiovascular Outcome in Patients with Chronic Kidney Disease. Nephrol. Dial. Transplant. 2021, 36, 811–818. [Google Scholar] [CrossRef]
- Htun, N.M.; Magliano, D.J.; Zhang, Z.-Y.; Lyons, J.; Petit, T.; Nkuipou-Kenfack, E.; Ramirez-Torres, A.; von Zur Muhlen, C.; Maahs, D.; Schanstra, J.P.; et al. Prediction of Acute Coronary Syndromes by Urinary Proteome Analysis. PLoS ONE 2017, 12, e0172036. [Google Scholar] [CrossRef]
- Alkhalaf, A.; Zürbig, P.; Bakker, S.J.L.; Bilo, H.J.G.; Cerna, M.; Fischer, C.; Fuchs, S.; Janssen, B.; Medek, K.; Mischak, H.; et al. Multicentric Validation of Proteomic Biomarkers in Urine Specific for Diabetic Nephropathy. PLoS ONE 2010, 5, e13421. [Google Scholar] [CrossRef]
- Rossing, K.; Bosselmann, H.S.; Gustafsson, F.; Zhang, Z.-Y.; Gu, Y.-M.; Kuznetsova, T.; Nkuipou-Kenfack, E.; Mischak, H.; Staessen, J.A.; Koeck, T.; et al. Urinary Proteomics Pilot Study for Biomarker Discovery and Diagnosis in Heart Failure with Reduced Ejection Fraction. PLoS ONE 2016, 11, e0157167. [Google Scholar] [CrossRef]
| Characteristic | N = 5585 | Study Information |
|---|---|---|
| Study | ||
| CACTI | 19 (0.34%) | Adults with type 1 diabetes |
| CADPredictions | 147 (2.63%) | Adults with acute coronary syndromes |
| CardioRen | 116 (2.08%) | Adults with heart failure with reduced ejection fraction |
| DIRECT | 769 (13.77%) | Adults with type 2 diabetes with normoalbuminuria |
| EPOGH | 826 (14.79%) | Adults with type 2 diabetes treated with basal insulin |
| FLEMENGHO | 65 (1.16%) | General population |
| Generation Scotland | 450 (8.06%) | Adults with and without coronary artery disease (CAD) |
| HOMAGE | 354 (6.34%) | Adults with heart failure |
| Predictions Groningen_Prag | 38 (0.68%) | Adults with type 2 diabetes |
| PRIORITY | 1761 (31.53%) | Adults with type 2 diabetes, normal urinary albumin excretion, and preserved renal function |
| SUNmacro | 580 (10.38%) | Adults with type 2 diabetic nephropathy |
| UZ-Gent | 460 (8.24%) | Adults with chronic kidney disease |
| n (%) | ||
| Characteristic | N = 5585 |
|---|---|
| Duration of follow-up (years) | 3.75 (0.38, 7.11) |
| Clinic characteristics | |
| Age | 62 (28, 82.3) |
| Female | 3410 (61.06%) |
| sBP (mm Hg) | 133 (105, 171) |
| dBP (mm Hg) | 79 (58, 98) |
| Hypertension | 2381 (42.63%) |
| Diabetes | 3330 (59.62%) |
| eGFR (mL/min/1.73 m2) | 81.89 (23, 117) |
| BMI (kg/m2) | 28.4 (19.8, 41) |
| Urinary-peptide-based classifiers | |
| HF2 | −0.29 (−0.76, 0.21) |
| CAD 160-marker | −0.32 (−0.71, 0.03) |
| CKD273 | −0.50 (−0.84, −0.01) |
| Median (95% IC); n (%) | |
| Events/At Risk (%) | Model Unadjusted | Model (Adjusted for Age, BP, BMI, Sex, and eGFR) | |||
|---|---|---|---|---|---|
| HF2 | HF Events | HR (95% CI) | p-Value | HR (95% CI) | p-Value |
| Per 1-SD increment | 472/5200 (9.08) | 2.59 ± 0.047 | <2 × 10−16 | 1.64 ± 0.056 | 1.72 × 10−18 |
| Quintile 1 | 25/1041 (2.40) | Reference | Reference | Reference | Reference |
| Quintile 2 | 38/1040 (3.65) | 1.81 ± 0.26 | 0.02 | 1.15 ± 0.26 | 0.60 |
| Quintile 3 | 59/1040 (5.67) | 3.17 ± 0.24 | 1.42 × 10−6 | 1.51 ± 0.24 | 0.09 |
| Quintile 4 | 119/1040 (11.44) | 7.21 ± 0.22 | 4.60 × 10−19 | 2.53 ± 0.23 | 5.92 × 10−5 |
| Quintile 5 | 231/1039 (22.23) | 16.20 ± 0.21 | 3.15 × 10−39 | 3.84 ± 0.23 | 5.64 × 10−9 |
| CAD-160-marker | CAD events | ||||
| Per 1-SD increment | 384/5112 | 1.72 ± 0.050 | <2 × 10−16 | 1.33 ± 0.057 | 5.55 × 10−7 |
| Quintile 1 | 46/1024 (4.49) | Reference | Reference | Reference | Reference |
| Quintile 2 | 55/1020 (5.39) | 1.84 ± 0.20 | 2.45 × 10−3 | 1.39 ± 0.20 | 0.11 |
| Quintile 3 | 71/1026 (6.92) | 2.93 ± 0.19 | 2.77 × 10−8 | 2.13 ± 0.20 | 1.25 × 10−4 |
| Quintile 4 | 91/1019 (8.90) | 3.92 ± 0.19 | 2.65 × 10−13 | 2.53 ± 0.19 | 1.32 × 10−6 |
| Quintile 5 | 121/1023 (11.83) | 4.73 ± 0.18 | 4.93 × 10−18 | 2.82 ± 0.19 | 3.32 × 10−8 |
| CKD273 | CKD events | ||||
| Per 1-SD increment | 113/3635 (3.11) | 4.19 ± 0.094 | <2 × 10−16 | 3.18 ± 0.121 | 1.03 × 10−21 |
| Quintile 1 | 6/732 (0.82) | Reference | Reference | Reference | Reference |
| Quintile 2 | 11/722 (1.52) | 2.02 ± 0.51 | 0.17 | 1.96 ± 0.50 | 0.18 |
| Quintile 3 | 11/730 (1.51) | 2.13 ± 0.51 | 0.14 | 1.80 ± 0.51 | 0.25 |
| Quintile 4 | 22/724 (3.04) | 5.58 ± 0.46 | 2.07 × 10−4 | 5.33 ± 0.47 | 3.55 × 10−4 |
| Quintile 5 | 63/727 (8.67) | 35.47 ± 0.43 | 1.61 × 10−16 | 19.59 ± 0.47 | 7.32 × 10−11 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Jaimes Campos, M.A.; Andújar, I.; Keller, F.; Mayer, G.; Rossing, P.; Staessen, J.A.; Delles, C.; Beige, J.; Glorieux, G.; Clark, A.L.; et al. Prognosis and Personalized In Silico Prediction of Treatment Efficacy in Cardiovascular and Chronic Kidney Disease: A Proof-of-Concept Study. Pharmaceuticals 2023, 16, 1298. https://doi.org/10.3390/ph16091298
Jaimes Campos MA, Andújar I, Keller F, Mayer G, Rossing P, Staessen JA, Delles C, Beige J, Glorieux G, Clark AL, et al. Prognosis and Personalized In Silico Prediction of Treatment Efficacy in Cardiovascular and Chronic Kidney Disease: A Proof-of-Concept Study. Pharmaceuticals. 2023; 16(9):1298. https://doi.org/10.3390/ph16091298
Chicago/Turabian StyleJaimes Campos, Mayra Alejandra, Iván Andújar, Felix Keller, Gert Mayer, Peter Rossing, Jan A. Staessen, Christian Delles, Joachim Beige, Griet Glorieux, Andrew L. Clark, and et al. 2023. "Prognosis and Personalized In Silico Prediction of Treatment Efficacy in Cardiovascular and Chronic Kidney Disease: A Proof-of-Concept Study" Pharmaceuticals 16, no. 9: 1298. https://doi.org/10.3390/ph16091298
APA StyleJaimes Campos, M. A., Andújar, I., Keller, F., Mayer, G., Rossing, P., Staessen, J. A., Delles, C., Beige, J., Glorieux, G., Clark, A. L., Mullen, W., Schanstra, J. P., Vlahou, A., Rossing, K., Peter, K., Ortiz, A., Campbell, A., Persson, F., Latosinska, A., ... Jankowski, J. (2023). Prognosis and Personalized In Silico Prediction of Treatment Efficacy in Cardiovascular and Chronic Kidney Disease: A Proof-of-Concept Study. Pharmaceuticals, 16(9), 1298. https://doi.org/10.3390/ph16091298








