Mapping the Spatiotemporal Distribution of Bovine Rabies in Colombia, 2005–2019
Abstract
1. Introduction
2. Materials and Methods
2.1. Type of Study
2.2. Data Source, GIS-Mapping and Statistical Analyses
2.3. Population and Sample
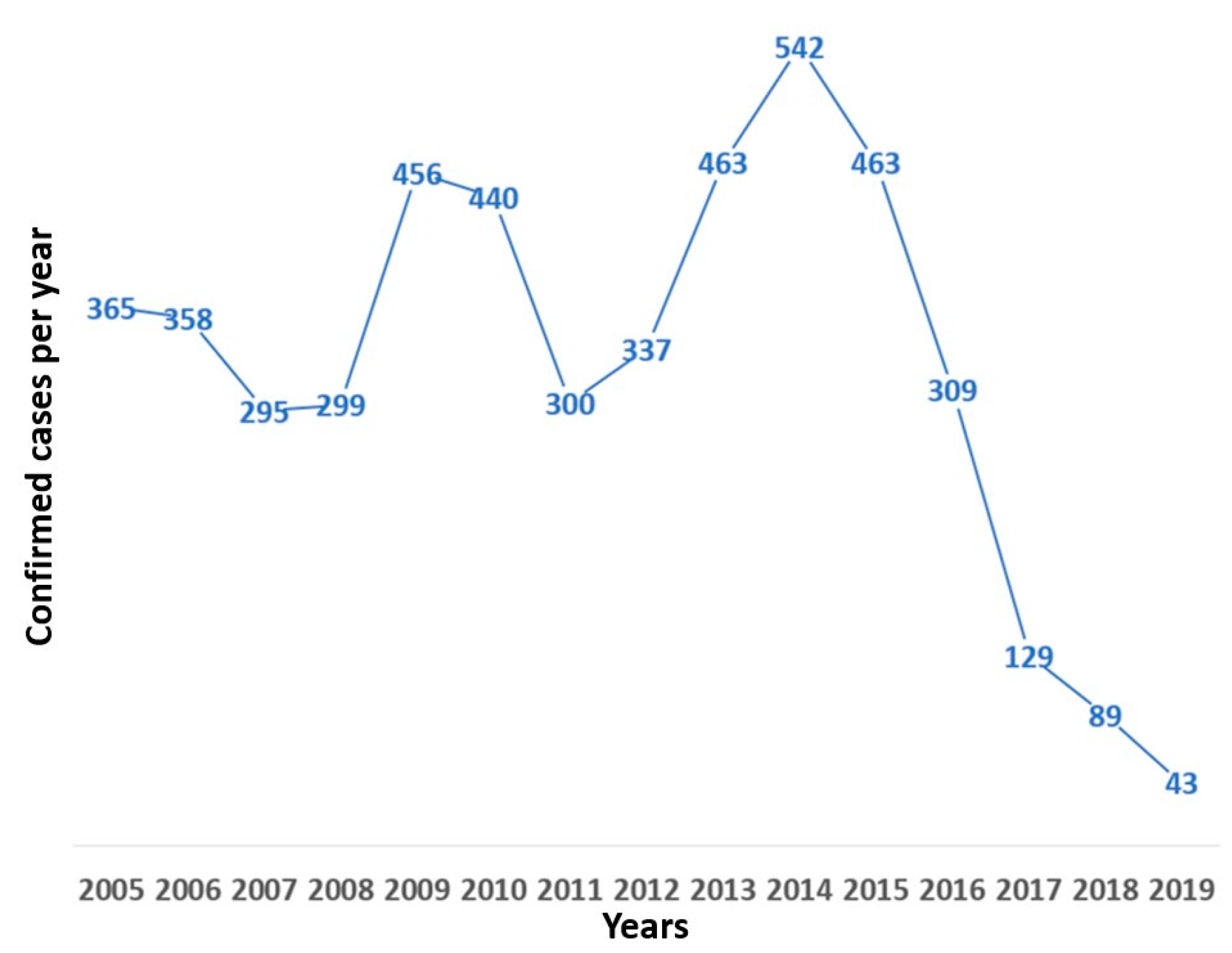
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Tsiodras, S.; Korou, L.M.; Tzani, M.; Tasioudi, K.E.; Kalachanis, K.; Mangana-Vougiouka, O.; Rigakos, G.; Dougas, G.; Seimenis, A.M.; Kontos, V. Rabies in Greece; historical perspectives in view of the current re-emergence in wild and domestic animals. Travel Med. Infect. Dis. 2014, 12, 628–635. [Google Scholar] [CrossRef] [PubMed]
- Megid, J.; Benavides Tala, J.A.; Belaz Silva, L.D.; Castro Castro, F.F.; Ribeiro, B.L.D.; Appolinário, C.M.; Katz, I.S.S.; Scheffer, K.C.; Silva, S.R.; Rosa, A.R.; et al. Serological Surveillance of Rabies in Free-Range and Captive Common Vampire Bats Desmodus rotundus. Front. Vet. Sci. 2021, 8, 681423. [Google Scholar] [CrossRef]
- Singh, R.; Singh, K.P.; Cherian, S.; Saminathan, M.; Kapoor, S.; Manjunatha Reddy, G.B.; Panda, S.; Dhama, K. Rabies—Epidemiology, pathogenesis, public health concerns and advances in diagnosis and control: A comprehensive review. Vet. Q. 2017, 37, 212–251. [Google Scholar] [CrossRef] [PubMed]
- Brito-Hoyos, D.M.; Sierra, E.B.; Álvarez, R.V. Distribución geográfica del riesgo de rabia de origen silvestre y evaluación de los factores asociados con su incidencia en Colombia, 1982–2010. Rev. Panam. Salud. Publica 2013, 33, 8–14. [Google Scholar] [CrossRef] [PubMed]
- Betancur Hurtado, C.; Calderón Rangel, A.; Rodríguez, V.C. Presencia de virus rábico en murciélagos hematófagos encolombia (ciénaga de oro y sahagún, córdoba). Biosalud 2016, 15, 17–24. [Google Scholar] [CrossRef]
- Ahmed, M.J.; Dirirsa, G.; Mengistu, D.A.; Demena, M.; Geremew, A. Rabies prevention and control practices and associated factors among dog owners in Chiro, West Hararghe, Ethiopia. J. Public Health Res. 2022, 11, 22799036221129373. [Google Scholar] [CrossRef]
- Acharya, K.P.; Chand, R.; Huettmann, F.; Ghimire, T.R. Rabies Elimination: Is It Feasible without Considering Wildlife? J. Trop. Med. 2022, 2022, 5942693. [Google Scholar] [CrossRef]
- Scheffer, K.C.; Iamamoto, K.; Asano, K.M.; Mori, E.; Estevez Garcia, A.I.; Achkar, S.M.; Fahl, W.O. Murciélagos hematófagos como reservorios de la rabia. Rev. Peru Med. Exp. Salud Publica 2014, 31, 302–309. [Google Scholar]
- Buenrostro, A.; Torre, M.; García-Grajales, J. Derriengue (Rabia paralítica bovina) y el murciélago hematófago. Cienc. Mar. 2019, XXIII, 87–96. [Google Scholar]
- Gilbert, A.T. Rabies virus vectors and reservoir species. Rev. Sci. Tech. (Int. Off. Epizoot.) 2018, 37, 371–384. [Google Scholar] [CrossRef]
- Mantovan, K.B.; Menozzi, B.D.; Paiz, L.M.; Sevá, A.P.; Brandão, P.E.; Langoni, H. Geographic Distribution of Common Vampire Bat Desmodus rotundus (Chiroptera: Phyllostomidae) Shelters: Implications for the Spread of Rabies Virus to Cattle in Southeastern Brazil. Pathogens 2022, 11, 942. [Google Scholar] [CrossRef] [PubMed]
- Gomez, C.; Black, J.; Koprowski, H. Rabies in cattle. III. Comparative studies on vaccination of cattle in Colombia with Flury virus and chloroform-inactivated vaccine. J. Am. Vet. Med. Assoc. 1955, 127, 360–363. [Google Scholar]
- Pereira, F.; Oliveira, A.R.; Soares-Neto, L.L.; Munhoz, L.; Fernandes, L.S.; Santos, D.O.D.; Carvalho, T.P.; Langoni, H.; Santos, R.L. Rabies in a Captive Lowland Tapir (Tapirus terrestris). J. Comp. Pathol. 2022, 198, 29–32. [Google Scholar] [CrossRef] [PubMed]
- Meza, D.K.; Mollentze, N.; Broos, A.; Tello, C.; Valderrama, W.; Recuenco, S.; Carrera, J.E.; Shiva, C.; Falcon, N.; Viana, M.; et al. Ecological determinants of rabies virus dynamics in vampire bats and spillover to livestock. Proc. Biol. Sci. 2022, 289, 20220860. [Google Scholar] [CrossRef] [PubMed]
- Taylor, E.; Aguilar-Ancori, E.G.; Banyard, A.C.; Abel, I.; Mantini-Briggs, C.; Briggs, C.L.; Carrillo, C.; Gavidia, C.M.; Castillo-Neyra, R.; Parola, A.D.; et al. The Amazonian Tropical Bites Research Initiative, a hope for resolving zoonotic neglected tropical diseases in the One Health era. Int. Health 2022, ihac048. [Google Scholar] [CrossRef]
- Everard, C.O.; Baer, G.M.; Alls, M.E.; Moore, S.A. Rabies serum neutralizing antibody in mongooses from Grenada. Trans. R. Soc. Trop. Med. Hyg. 1981, 75, 654–666. [Google Scholar] [CrossRef]
- Everard, C.O.; Everard, J.D. Mongoose rabies. Rev. Infect. Dis. 1988, 10 (Suppl. S4), S610–S614. [Google Scholar] [CrossRef] [PubMed]
- Johnson, N.; Arechiga-Ceballos, N.; Aguilar-Setien, A. Vampire bat rabies: Ecology, epidemiology and control. Viruses 2014, 6, 1911–1928. [Google Scholar] [CrossRef] [PubMed]
- Marín Álvarez, L.M.; Ruíz Sáenz, J.; Ruíz Buitrago, J.D. Analysis of a program for the prevention and control of wild rabies and its role in the number of cattle outbreaks during years 2001 to 2011. CES Med. Vet. Y Zootec. 2014, 9, 203–217. [Google Scholar]
- Loeb, J. Should all travelling dogs be tested for rabies? Vet. Rec. 2022, 191, 275. [Google Scholar] [CrossRef]
- Loeb, J. Rabies risk is very real. Vet. Rec. 2022, 191, 273. [Google Scholar] [CrossRef]
- Ballesteros, C.J.; Racero-Casarrubia, J. Murciélagos del área urbana en la ciudad de Montería, Córdoba—Colombia. Rev. MVZ Cordoba 2012, 17, 3. [Google Scholar] [CrossRef][Green Version]
- Instituto Colombiano Agropecuario (I.C.A.). Censo Pecuario Nacional. In Inst. Colomb. Agropecu. ICA; 2019. Available online: https://www.ica.gov.co/areas/pecuaria/servicios/epidemiologia-veterinaria/censos-2016/censo-2018 (accessed on 3 October 2022).
- Calderon, A.; Guzman, C.; Mattar, S.; Rodriguez, V.; Acosta, A.; Martinez, C. Frugivorous bats in the Colombian Caribbean region are reservoirs of the rabies virus. Ann. Clin. Microbiol. Antimicrob. 2019, 18, 11. [Google Scholar] [CrossRef]
- Páez, A.; Nũñez, C.; García, C.; Bóshell, J. Molecular epidemiology of rabies epizootics in Colombia: Evidence for human and dog rabies associated with bats. J. Gen. Virol. 2003, 84, 795–802. [Google Scholar] [CrossRef]
- Valderrama, J.; García, I.; Figueroa, G.; Rico, E.; Sanabria, J.; Rocha, N.; Parra, E.; Saad, C.; Páez, A. Outbreaks of human rabies transmitted by vampire bats in Alto Baudó and Bajo Baudó municipalities, department of chocó, Colombia, 2004–2005. Biomed. Rev. Inst. Nac. Salud 2006, 26, 387–396. [Google Scholar]
- Dulce Villarreal, Á.; Goenaga, N.; Zoonosis, R.; Transmisibles, G.E.; Salud, I.N.d.; Marta, S.D.d.S.d.S. Dos casos de encefalitis rábica en el distrito de Santa Marta, departamento de Magdalena. Minist. Protección Soc.-Inst. Nac. Salud 2007, 12, 17–28. [Google Scholar]
- Meske, M.; Fanelli, A.; Rocha, F.; Awada, L.; Soto, P.C.; Mapitse, N.; Tizzani, P. Evolution of Rabies in South America and Inter-Species Dynamics (2009–2018). Trop. Med. Infect. Dis. 2021, 6, 98. [Google Scholar] [CrossRef]
- Brito-Hoyos, D.M.; Brito Sierra, E.; Villalobos Alvarez, R. Geographic distribution of wild rabies risk and evaluation of the factors associated with its incidence in Colombia, 1982–2010. Rev. Panam. Salud Publica Pan Am. J. Public Health 2013, 33, 8–14. [Google Scholar] [CrossRef]
- Cediel, N.; de la Hoz, F.; Villamil, L.C.; Romero, J.; Díaz, A. The epidemiology of canine rabies in Colombia. Rev. Salud Publica (Bogota Colomb.) 2010, 12, 368–379. [Google Scholar] [CrossRef]
- Gómez-Sandoval, P.A.R.; Badillo, R.; Mantilla, J.C.; Pradilla, G. Human rabies encephalitis caused by bat bite in a urban area in Colombia. Biomedica 2010, 30, 152. [Google Scholar] [CrossRef]
- Mejía Angarita, J.F.; López Martínez, L. Reporte de casos de rabia humana. Santander, 2008–2010. Inf. Epidemiol. Santander 2010, 4, 1–12. [Google Scholar]
- Cediel Becerra, N.M.; Olaya Medellin, A.M.; Tomassone, L.; Chiesa, F.; De Meneghi, D. A Survey on One Health Approach in Colombia and Some Latin American Countries: From a Fragmented Health Organization to an Integrated Health Response to Global Challenges. Front. Public Health 2021, 9, 649240. [Google Scholar] [CrossRef]
- Arias Caicedo, M.R.; Xavier, D.A.; Arias Caicedo, C.A.; Andrade, E.; Abel, I. Epidemiological scenarios for human rabies exposure notified in Colombia during ten years: A challenge to implement surveillance actions with a differential approach on vulnerable populations. PLoS ONE 2019, 14, e0213120. [Google Scholar] [CrossRef]
- Valladales-Restrepo, L.F.; Vargas-Díaz, K.; Peña-Verjan, N.M.; Londoño-Cano, D.A.; Álvarez-Ayala, D.; Orrego-Giraldo, M.J.; Machado-Alba, J.E. Use of antibiotics in patients who were attacked by animals that can transmit rabies. Zoonoses Public Health 2022, 69, 978–986. [Google Scholar] [CrossRef]
- Tierradentro-García, L.O.; Cortés-Albornoz, M.C.; Talero-Gutiérrez, C. Of love and other demons: Depicting human rabies in Colombia. Heliyon 2022, 8, e09703. [Google Scholar] [CrossRef]
- Rifakis, P.M.; Benitez, J.A.; Rodriguez-Morales, A.J.; Dickson, S.M.; De-La-Paz-Pineda, J. Ecoepidemiological and Social Factors Related to Rabies Incidence in Venezuela during 2002–2004. Int. J. Biomed. Sci. IJBS 2006, 2, 1–6. [Google Scholar]
- Rodriguez-Morales, A.J.; Schlagenhauf, P. Zoonoses and travel medicine: "One world—one health". Travel Med. Infect. Dis. 2014, 12, 555–556. [Google Scholar] [CrossRef]
- Cabrera, M.; Leake, J.; Naranjo-Torres, J.; Valero, N.; Cabrera, J.C.; Rodríguez-Morales, A.J. Dengue Prediction in Latin America Using Machine Learning and the One Health Perspective: A Literature Review. Trop. Med. Infect. Dis. 2022, 7, 322. [Google Scholar] [CrossRef]
- Acharya, K.P.; Acharya, N.; Phuyal, S.; Upadhyaya, M.; Lasee, S. One-health approach: A best possible way to control rabies. One Health (Amst. Neth.) 2020, 10, 100161. [Google Scholar] [CrossRef]
- Beyene, T.J.; Fitzpatrick, M.C.; Galvani, A.P.; Mourits, M.C.M.; Revie, C.W.; Cernicchiaro, N.; Sanderson, M.W.; Hogeveen, H. Impact of One-Health framework on vaccination cost-effectiveness: A case study of rabies in Ethiopia. One Health (Amst. Neth.) 2019, 8, 100103. [Google Scholar] [CrossRef]
- Moore, S.M. Rabies prevention: The role of serology in parenteral vaccination of companion animals and livestock. Rev. Sci. Tech. (Int. Off. Epizoot.) 2018, 37, 461–472. [Google Scholar] [CrossRef]
- Yakobson, B.A.; King, R.; Sheichat, N.; Eventov, B.; David, D. Assessment of the efficacy of oral vaccination of livestock guardian dogs in the framework of oral rabies vaccination of wild canids in Israel. Dev. Biol. 2008, 131, 151–156. [Google Scholar]
- Rocha, F.; Dias, R.A. The common vampire bat Desmodus rotundus (Chiroptera: Phyllostomidae) and the transmission of the rabies virus to livestock: A contact network approach and recommendations for surveillance and control. Prev. Vet. Med. 2020, 174, 104809. [Google Scholar] [CrossRef]
- Mbilo, C.; Lechenne, M.; Mauti, S.; Chitnis, N.; Tschopp, R.; Zinsstag, J. Rabies in dogs, livestock and wildlife: A veterinary perspective. Rev. Sci. Tech. (Int. Off. Epizoot.) 2018, 37, 331–340. [Google Scholar] [CrossRef]
- Bonilla-Aldana, D.K.; Jimenez-Diaz, S.D.; Arango-Duque, J.S. Bats in ecosystems and their Wide spectrum of viral infectious potential threats: SARS-CoV-2 and other emerging viruses. Int. J. Infect. Dis. 2021, 102, 87–96. [Google Scholar] [CrossRef]





| Department | p-Value | r2 | Department | p-Value | r2 |
|---|---|---|---|---|---|
| Cesar | 0.0137 | 0.8151 | Caqueta | 0.4111 | - |
| Choco | 0.0189 | 0.7844 | Cordoba | 0.6017 | - |
| Norte de Santander | 0.0329 | 0.7192 | Tolima | 0.6803 | - |
| Magdalena | 0.0351 | 0.7105 | Boyaca | 0.8047 | - |
| La Guajira | 0.0595 | - | Quindio | 0.8047 | - |
| Casanare | 0.0708 | - | Valle del Cauca | 0.8047 | - |
| Arauca | 0.0752 | - | Meta | 0.8322 | - |
| Guaviare | 0.0963 | - | Cauca | 0.9443 | - |
| Sucre | 0.1405 | - | Amazonas | N/A | - |
| Bolivar | 0.1443 | - | Atlantico | N/A | - |
| Santander | 0.1583 | - | Caldas | N/A | - |
| Vaupes | 0.1583 | - | Cundinamarca | N/A | - |
| Vichada | 0.1894 | - | Guainia | N/A | - |
| Antioquia | 0.2857 | - | Huila | N/A | - |
| Nariño | 0.3528 | - | Risaralda | N/A | - |
| Putumayo | 0.3893 | - | San Andres | N/A | - |
| Cases per Year | Population per Year | Annual Incidence (Cases per 100,000 Animals) | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Department | 2016 | 2017 | 2018 | 2019 | 2016 | 2017 | 2018 | 2019 | 2016 | 2017 | 2018 | 2019 |
| Quindio | 48 | 0 | 0 | 0 | 81,788 | 76,814 | 83,425 | 306,918 | 58.688 | 0.000 | 0.000 | 0.000 |
| Cesar | 41 | 11 | 11 | 0 | 152,228 | 154,796 | 199,854 | 315,597 | 26.933 | 7.106 | 5.504 | 0.000 |
| Putumayo | 38 | 12 | 2 | 0 | 197,611 | 209,006 | 255,912 | 482,233 | 19.230 | 5.741 | 0.782 | 0.000 |
| Cordoba | 52 | 0 | 0 | 1 | 1,256,535 | 1,146,137 | 1,422,452 | 166,411 | 4.138 | 0.000 | 0.000 | 0.601 |
| Meta | 54 | 0 | 1 | 1 | 1,660,147 | 1,734,106 | 1,948,553 | 1,342,115 | 3.253 | 0.000 | 0.051 | 0.075 |
| Casanare | 7 | 11 | 11 | 2 | 273,663 | 276,891 | 298,135 | 1,897,555 | 2.558 | 3.973 | 3.690 | 0.105 |
| Nariño | 9 | 0 | 0 | 0 | 384,686 | 383,005 | 394,874 | 2,045,984 | 2.340 | 0.000 | 0.000 | 0.000 |
| Boyaca | 6 | 0 | 0 | 0 | 370,345 | 428,324 | 448,895 | 1,222,434 | 1.620 | 0.000 | 0.000 | 0.000 |
| Tolima | 8 | 0 | 0 | 0 | 547,647 | 583,166 | 685,274 | 733,644 | 1.461 | 0.000 | 0.000 | 0.000 |
| Norte De Santander | 5 | 13 | 0 | 0 | 389,694 | 455,711 | 467,782 | 407,143 | 1.283 | 2.853 | 0.000 | 0.000 |
| Caqueta | 22 | 6 | 3 | 3 | 1,845,226 | 1,861,776 | 1,992,767 | 451,994 | 1.192 | 0.322 | 0.151 | 0.664 |
| Arauca | 11 | 1 | 2 | 3 | 1,048,543 | 1,096,641 | 1,162,032 | 1,209,520 | 1.049 | 0.091 | 0.172 | 0.248 |
| Bolivar | 4 | 7 | 3 | 3 | 748,701 | 837,567 | 1,172,503 | 35,304 | 0.534 | 0.836 | 0.256 | 8.498 |
| Valle Del Cauca | 2 | 0 | 0 | 0 | 459,596 | 460,727 | 523,306 | 538,101 | 0.435 | 0.000 | 0.000 | 0.000 |
| Cauca | 1 | 8 | 2 | 0 | 1,357,512 | 1,305,984 | 1,415,681 | 2,134,723 | 0.074 | 0.613 | 0.141 | 0.000 |
| Choco | 1 | 3 | 0 | 0 | 1,942,770 | 2,052,151 | 2,140,312 | 1,482,922 | 0.051 | 0.146 | 0.000 | 0.000 |
| Amazonas | 0 | 0 | 0 | 0 | 1264 | 1552 | 823 | 854 | 0.000 | 0.000 | 0.000 | 0.000 |
| Antioquia | 0 | 8 | 8 | 2 | 2,632,125 | 2,760,633 | 3,038,779 | 3,090,631 | 0.000 | 0.290 | 0.263 | 0.065 |
| Atlantico | 0 | 0 | 0 | 0 | 224,978 | 217,003 | 238,413 | 723 | 0.000 | 0.000 | 0.000 | 0.000 |
| Caldas | 0 | 0 | 0 | 0 | 1,340,049 | 1,486,685 | 1,809,702 | 1,181,505 | 0.000 | 0.000 | 0.000 | 0.000 |
| Cundinamarca | 0 | 0 | 0 | 0 | 26,125 | 33,725 | 34,238 | 2,134,681 | 0.000 | 0.000 | 0.000 | 0.000 |
| Guainia | 0 | 0 | 0 | 0 | 4703 | 4703 | 4547 | 1,449,508 | 0.000 | 0.000 | 0.000 | 0.000 |
| Guaviare | 0 | 0 | 0 | 0 | 281,611 | 301,224 | 406,242 | 5711 | 0.000 | 0.000 | 0.000 | 0.000 |
| Huila | 0 | 0 | 0 | 0 | 415,246 | 370,884 | 411,720 | 443,633 | 0.000 | 0.000 | 0.000 | 0.000 |
| La Guajira | 0 | 0 | 1 | 0 | 285,298 | 242,137 | 286,962 | 432,587 | 0.000 | 0.000 | 0.348 | 0.000 |
| Magdalena | 0 | 24 | 12 | 2 | 1,207,764 | 1,205,715 | 1,359,672 | 302,483 | 0.000 | 1.991 | 0.883 | 0.661 |
| Risaralda | 0 | 0 | 0 | 0 | 109,117 | 117,209 | 109,334 | 83,221 | 0.000 | 0.000 | 0.000 | 0.000 |
| San Andres | 0 | 0 | 0 | 0 | 825 | 801 | 735 | 108,447 | 0.000 | 0.000 | 0.000 | 0.000 |
| Santander | 0 | 0 | 0 | 0 | 1,412,313 | 1,442,936 | 1,595,532 | 1,617,398 | 0.000 | 0.000 | 0.000 | 0.000 |
| Sucre | 0 | 20 | 25 | 21 | 862,008 | 992,238 | 1,076,327 | 1,103,675 | 0.000 | 2.016 | 2.323 | 1.903 |
| Vaupes | 0 | 0 | 0 | 0 | 1223 | 1074 | 1159 | 1205 | 0.000 | 0.000 | 0.000 | 0.000 |
| Vichada | 0 | 5 | 8 | 5 | 242,633 | 231,684 | 254,820 | 262,311 | 0.000 | 2.158 | 3.139 | 1.906 |
| Bogota D.C. | 0 | 0 | 0 | 0 | 925,446 | 1,002,017 | 1,172,465 | 242,856 | 0.000 | 0.000 | 0.000 | 0.000 |
| Total | 309 | 129 | 89 | 43 | 22,689,420 | 23,475,022 | 26,413,227 | 27,234,027 | 1.362 | 0.550 | 0.337 | 0.158 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bonilla-Aldana, D.K.; Jimenez-Diaz, S.D.; Barboza, J.J.; Rodriguez-Morales, A.J. Mapping the Spatiotemporal Distribution of Bovine Rabies in Colombia, 2005–2019. Trop. Med. Infect. Dis. 2022, 7, 406. https://doi.org/10.3390/tropicalmed7120406
Bonilla-Aldana DK, Jimenez-Diaz SD, Barboza JJ, Rodriguez-Morales AJ. Mapping the Spatiotemporal Distribution of Bovine Rabies in Colombia, 2005–2019. Tropical Medicine and Infectious Disease. 2022; 7(12):406. https://doi.org/10.3390/tropicalmed7120406
Chicago/Turabian StyleBonilla-Aldana, D. Katterine, S. Daniela Jimenez-Diaz, Joshuan J. Barboza, and Alfonso J. Rodriguez-Morales. 2022. "Mapping the Spatiotemporal Distribution of Bovine Rabies in Colombia, 2005–2019" Tropical Medicine and Infectious Disease 7, no. 12: 406. https://doi.org/10.3390/tropicalmed7120406
APA StyleBonilla-Aldana, D. K., Jimenez-Diaz, S. D., Barboza, J. J., & Rodriguez-Morales, A. J. (2022). Mapping the Spatiotemporal Distribution of Bovine Rabies in Colombia, 2005–2019. Tropical Medicine and Infectious Disease, 7(12), 406. https://doi.org/10.3390/tropicalmed7120406









