Carriage Prevalence of Extended-Spectrum β-Lactamase Producing Enterobacterales in Outpatients Attending Community Health Centers in Blantyre, Malawi
Abstract
1. Introduction
2. Materials and Methods
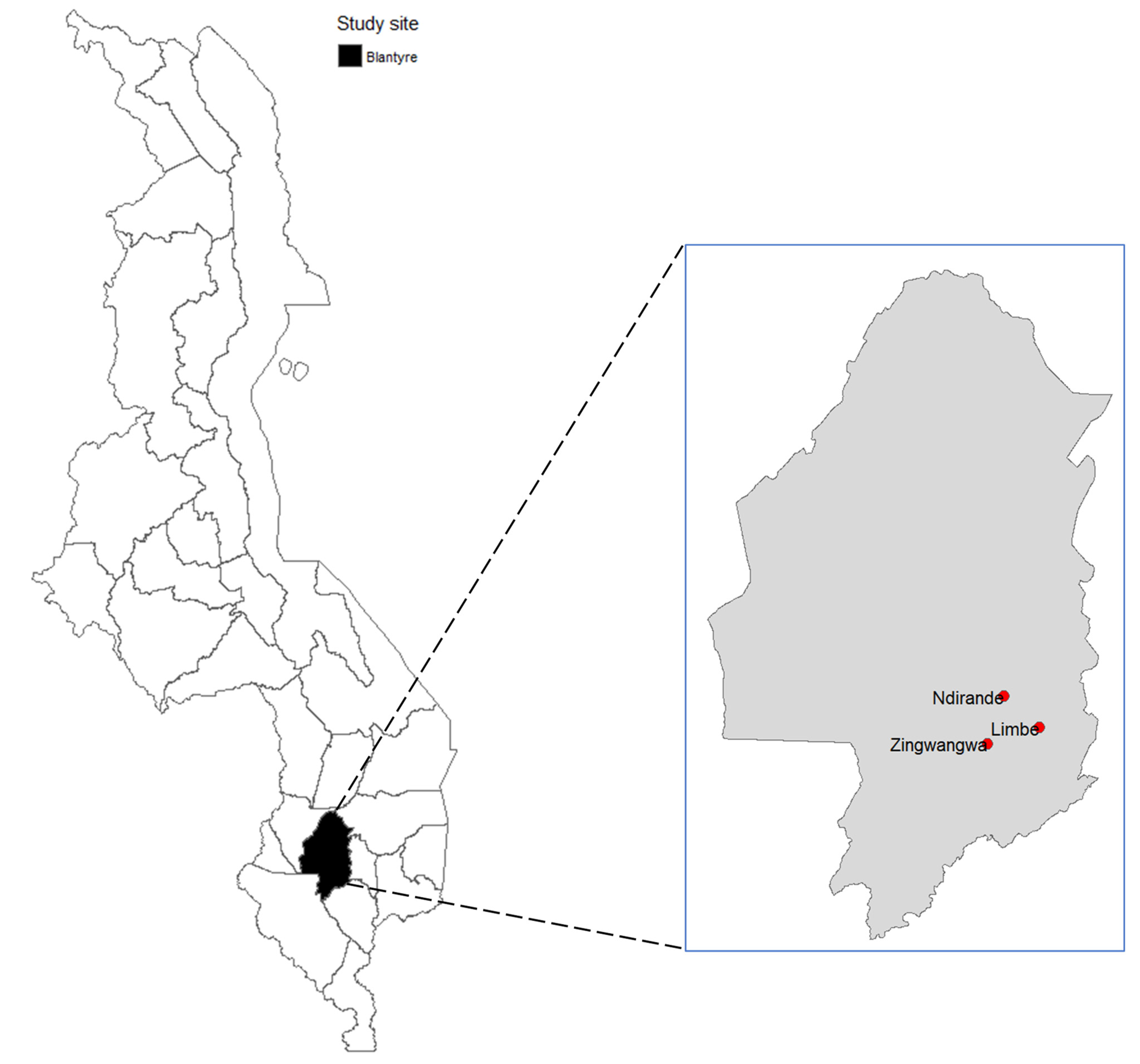
2.1. Study Design, Duration and Location
2.2. Study Population, Sample Size and Sampling
2.3. Specimen Collection
2.4. Screening for ESBL-E
2.5. Identification of Enterobacterales Species
2.6. Phenotypic Confirmation of ESBL Production by Enterobacterales
2.7. Antimicrobial Susceptibility Testing (AST)
2.8. Quality Control
2.9. Statistical Analysis
3. Results
3.1. Social Demographic and Clinical Characteristics of the Study Population
3.2. Prevalence of ESBL-E in Community Patients in Blantyre
3.3. Antimicrobial Resistance Profiles of Commonly Isolated ESBL-E
3.4. Univariate Analysis
4. Discussion
5. Limitations of the Study
6. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Jacoby, G.A.; Medeiros, A.A. More extended-spectrum β-lactamases. Antimicrob. Agents Chemother. 1991, 35, 1697–1704. [Google Scholar] [CrossRef] [PubMed]
- Knothe, H.; Shah, P.; Krcmery, V.; Antal, M.; Mitsuhashi, S. Transferable resistance to cefotaxime, cefoxitin, cefamandole and cefuroxime in clinical isolates of Klebsiella pneumoniae and Serratia marcescens. Infection 1983, 11, 315–317. [Google Scholar] [CrossRef] [PubMed]
- Tacconelli, E.; Carrara, E.; Savoldi, A.; Harbarth, S.; Mendelson, M.; Monnet, D.L.; Pulcini, C.; Kahlmeter, G.; Kluytmans, J.; Carmeli, Y.; et al. Discovery, research, and development of new antibiotics: The WHO priority list of antibiotic-resistant bacteria and tuberculosis. Lancet Infect. Dis. 2018, 18, 318–327. [Google Scholar] [CrossRef]
- Rossolini, G.M.; Mantengoli, E. Antimicrobial resistance in Europe and its potential impact on empirical therapy. In Clinical Microbiology and Infection; Blackwell Publishing Ltd.: Hoboken, NJ, USA; pp. 2–8.
- Woerther, P.-L.; Burdet, C.; Chachaty, E.; Andremont, A. Trends in human fecal carriage of extended-spectrum β-lactamases in the community: Toward the globalization of CTX-M. Clin. Microbiol. Rev. 2013, 26, 744–758. [Google Scholar] [CrossRef] [PubMed]
- Sasaki, T.; Hirai, I.; Niki, M.; Nakamura, T.; Komalamisra, C.; Maipanich, W.; Kusolsuk, T.; Sa-Nguankiat, S.; Pubampen, S.; Yamamoto, Y. High prevalence of CTX-M b-lactamase-producing Enterobacteriaceae in stool specimens obtained from healthy individuals in Thailand. J. Antimicrob. Chemother. 2010, 65, 666–668. [Google Scholar] [CrossRef]
- Ayukekbong, J.A.; Ntemgwa, M.; Atabe, A.N. The threat of antimicrobial resistance in developing countries: Causes and control strategies. Antimicrob. Resist. Infect. Control. 2017, 6, 47. [Google Scholar] [CrossRef] [PubMed]
- Friedman, N.D.; Temkin, E.; Carmeli, Y. The negative impact of antibiotic resistance. Clin. Microbiol. Infect. 2016, 22, 416–422. [Google Scholar] [CrossRef] [PubMed]
- Prestinaci, F.; Pezzotti, P.; Pantosti, A. Antimicrobial resistance: A global multifaceted phenomenon. Pathog. Glob. Health 2015, 109, 309–318. [Google Scholar] [CrossRef]
- Musicha, P.; Msefula, C.L.; Mather, A.E.; Chaguza, C.; Cain, A.K.; Peno, C.; Kallonen, T.; Khonga, M.; Denis, B.; Gray, K.J.; et al. Genomic analysis of Klebsiella pneumoniae isolates from Malawi reveals acquisition of multiple ESBL determinants across diverse lineages. J. Antimicrob. Chemother. 2019, 74, 1223–1232. [Google Scholar] [CrossRef]
- Gray, K.J.; Wilson, L.K.; Phiri, A.; Corkill, J.E.; French, N.; Hart, C.A. Identification and characterization of ceftriaxone resistance and extended-spectrum β-lactamases in Malawian bacteraemic Enterobacteriaceae. J. Antimicrob. Chemother. 2006, 57, 661–665. [Google Scholar] [CrossRef][Green Version]
- Musicha, P.; Cornick, J.E.; Bar-Zeev, N.; French, N.; Masesa, C.; Denis, B.; Kennedy, N.; Mallewa, J.; Gordon, M.A.; Msefula, C.L.; et al. Trends in antimicrobial resistance in bloodstream infection isolates at a large urban hospital in Malawi (1998–2016): A surveillance study. Lancet Infect. Dis. 2017, 17, 1042–1052. [Google Scholar] [CrossRef]
- Lewis, J.M.; Lester, R.; Mphasa, M.; Banda, R.; Edwards, T.; Thomson, N.R.; Feasey, N. Emergence of carbapenemase-producing Enterobacteriaceae in Malawi. J. Glob. Antimicrob. Resist. 2020, 20, 225–227. [Google Scholar] [CrossRef] [PubMed]
- Lwanga, S.; Lemeshow, S. Sample size determination in health studies: A practical manual. World Health Organ. Geneva 1991, 88, 1–22. [Google Scholar]
- Phillips, I. Cowan and Steel’s manual for the identification of medical bacteria. J. Clin. Pathol. 1993, 46, 975. [Google Scholar] [CrossRef]
- Wayne, P. M100 Performance Standards for Antimicrobial Susceptibility Testing A CLSI Supplement for Global Application, 30th ed.; Clinical and Laboratory Standards Institute: Wayne, PA, USA, 2020; Available online: www.clsi.org (accessed on 12 June 2021).
- M’Zali, F.H.; Chanawong, A.; Kerr, K.G.; Birkenhead, D.; Hawkey, P.M. Detection of extended-spectrum β-lactamases in members of the family Enterobacteriaceae: Comparison of the MAST DD test, the double disc and the Etest ESBL. J. Antimicrob. Chemother. 2000, 45, 881–885. [Google Scholar] [CrossRef]
- Henson, S.P.; Boinett, C.J.; Ellington, M.J.; Kagia, N.; Mwarumba, S.; Nyongesa, S.; Mturi, N.; Kariuki, S.; Scott, J.; Thomson, N.R. Molecular epidemiology of Klebsiella pneumoniae invasive infections over a decade at Kilifi County Hospital in Kenya. Int. J. Med. Microbiol. 2017, 307, 422–429. [Google Scholar] [CrossRef] [PubMed]
- Moses, A.; Bwanga, F.; Boum, Y.; Bazira, J. Prevalence and genotypic characterization of extended-spectrum beta-lactamases produced by gram negative bacilli at a Tertiary Care Hospital in Rural South Western Uganda. Br. Microbiol. Res. J. 2014, 4, 1541. [Google Scholar] [CrossRef] [PubMed]
- Irenge, L.M.; Kabego, L.; Vandenberg, O.; Chirimwami, R.B.; Gala, J.L. Antimicrobial resistance in urinary isolates from inpatients and outpatients at a tertiary care hospital in South-Kivu Province (Democratic Republic of Congo). BMC Res. Notes 2014, 7, 1–6. [Google Scholar] [CrossRef]
- Janatova, M.; Albrechtova, K.; Petrzelkova, K.J.; Dolejska, M.; Papousek, I.; Masarikova, M.; Cizek, A.; Todd, A.; Shutt, K.; Kalousova, B.; et al. Antimicrobial-resistant enterobacteriaceae from humans and wildlife in Dzanga-Sangha Protected Area, Central African Republic. Vet. Microbiol. 2014, 171, 422–431. [Google Scholar] [CrossRef]
- Kiiru, J.; Kariuki, S.; Goddeeris, B.M.; Butaye, P. Analysis of -lactamase phenotypes and carriage of selected -lactamase genes among Escherichia coli strains obtained from Kenyan patients during an 18-year period. BMC Microbiol. 2012, 12, 1471–2180. [Google Scholar] [CrossRef]
- Maina, D.; Revathi, G.; Kariuki, S.; Ozwara, H. Genotypes and cephalosporin susceptibility in extended-spectrum beta-lactamase producing enterobacteriaceae in the community. J. Infect. Dev. Ctries 2011, 6, 470–477. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Almagor, J.; Temkin, E.; Benenson, I.; Fallach, N.; Carmeli, Y. DRIVE-AB consortium. The impact of antibiotic use on transmission of resistant bacteria in hospitals: Insights from an agent-based model. PLoS ONE 2018, 13, e0197111. [Google Scholar] [CrossRef] [PubMed]
- Aslam, B.; Wang, W.; Arshad, M.I.; Khurshid, M.; Muzammil, S.; Rasool, M.H.; Nisar, M.A.; Alvi, R.F.; Aslam, M.A.; Qamar, M.U.; et al. Antibiotic resistance: A rundown of a global crisis. Infect. Drug Resist. 2018, 11, 1645–1658. [Google Scholar] [CrossRef]
- Shah, A.A.; Hasan, F.; Ahmed, S.; Hameed, A. Extended-spectrum β-lactamases in enterobacteriaceae: Related to age and gender. New Microbiol. 2002, 25, 363–366. [Google Scholar]
- Shashwati, N.; Kiran, T.; Dhanvijay, A. Study of extended spectrum β-lactamase producing enterobacteriaceae and antibiotic coresistance in a tertiary care teaching hospital. J. Nat. Sci. Biol. Med. 2014, 5, 30. [Google Scholar] [CrossRef]
- Tellevik, M.G.; Blomberg, B.; Kommedal, Ø.; Maselle, S.Y.; Langeland, N.; Moyo, S.J. High prevalence of faecal carriage of ESBL-producing enterobacteriaceae among children in Dar es Salaam, Tanzania. PLoS ONE 2016, 11, e0168024. [Google Scholar] [CrossRef]
- Moyo, S.J.; Aboud, S.; Kasubi, M.; Lyamuya, E.F.; Maselle, S.Y. Antimicrobial resistance among producers and non-producers of extended spectrum beta-lactamases in urinary isolates at a tertiary Hospital in Tanzania. BMC Res. Notes 2010, 3, 348. [Google Scholar] [CrossRef] [PubMed]
- Kateregga, J.N.; Kantume, R.; Atuhaire, C.; Lubowa, M.N.; Ndukui, J.G. Phenotypic expression and prevalence of ESBL-producing Enterobacteriaceae in samples collected from patients in various wards of Mulago Hospital, Uganda. BMC Pharmacol. Toxicol. 2015, 16, 1–6. [Google Scholar] [CrossRef]
- Williams, P.C.M.; Isaacs, D.; Berkley, J.A. Antimicrobial resistance among children in sub-Saharan Africa. Lancet Infect. Dis. 2018, 18, e33–e44. [Google Scholar] [CrossRef]
- Teklu, D.S.; Negeri, A.A.; Legese, M.H.; Bedada, T.L.; Woldemariam, H.K.; Tullu, K.D. Extended-spectrum beta-lactamase production and multi-drug resistance among enterobacteriaceae isolated in Addis Ababa, Ethiopia. Antimicrob. Resist. Infect. Control. 2019, 8, 39. [Google Scholar] [CrossRef] [PubMed]
- Ouedraogo, A.S.; Sanou, M.; Kissou, A.; Sanou, S.; Solaré, H.; Kaboré, F.; Poda, A.; Aberkane, S.; Bouzinbi, N.; Sano, I.; et al. High prevalence of extended-spectrum β-lactamase producing enterobacteriaceae among clinical isolates in Burkina Faso. BMC Infect. Dis. 2016, 11, 326. [Google Scholar] [CrossRef] [PubMed]
- Van den Bunt, G.; van Pelt, W.; Hidalgo, L.; Scharringa, J.; de Greeff, S.C.; Schürch, A.C.; Mughini-Gras, L.; Bonten, M.; Fluit, A.C. Prevalence, risk factors and genetic characterisation of extended-spectrum beta-lactamase and carbapenemase-producing Enterobacteriaceae (ESBL-E and CPE): A community-based cross-sectional study, the Netherlands, 2014 to 2016. Eurosurveillance 2019, 24, 1–11. [Google Scholar] [CrossRef]
- Najjuka, C.F.; Kateete, D.P.; Kajumbula, H.M.; Joloba, M.L.; Essack, S.Y. Antimicrobial susceptibility profiles of Escherichia coli and Klebsiella pneumoniae isolated from outpatients in urban and rural districts of Uganda. BMC Res. Notes 2016, 9, 235. [Google Scholar] [CrossRef] [PubMed]
- Rao, S.P.; Rama, P.S.; Gurushanthappa, V.; Manipura, R.; Srinivasan, K. Extended-spectrum beta-lactamases producing Escherichia coli and Klebsiella pneumoniaei: A multi-centric study across Karnataka. J. Lab. Physicians 2014, 6, 007–013. [Google Scholar] [CrossRef]
- Mahamat, O.O.; Lounnas, M.; Hide, M.; Dumont, Y.; Tidjani, A.; Kamougam, K.; Abderrahmane, M.; Benavides, J.; Solassol, J.; Bañuls, A.-L.; et al. High prevalence and characterization of extended-spectrum β-lactamase producing Enterobacteriaceae in Chadian hospitals. BMC Infect. Dis. 2019, 19, 1–7. [Google Scholar]
- Brink, A.; Moolman, G.J.J.; Cruz da Silva, M.; Botha, M. National Antibiotic Surveillance Forum. Antimicrobial susceptibility profile of selected bacteraemic pathogens from private institutions in South Africa. S. Afr. Med. J. 2007, 97, 273–279. [Google Scholar]
- Sanneh, B.; Kebbeh, A.; Jallow, H.S.; Camara, Y.; Mwamakamba, L.W.; Ceesay, I.F.; Barrow, E.; Sowe, F.O.; Sambou, S.M.; Baldeh, I.; et al. Prevalence and risk factors for faecal carriage of extended spectrum β-lactamase producing enterobacteriaceae among food handlers in lower basic schools in West Coast Region of The Gambia. PLoS ONE 2018, 13, e0200894. [Google Scholar] [CrossRef] [PubMed]
- Aworh, M.K.; Abiodun-Adewusi, O.; Mba, N.; Helwigh, B.; Hendriksen, R.S. Prevalence and risk factors for faecal carriage of multidrug resistant Escherichia coli among slaughterhouse workers. Sci. Rep. 2021, 11, 13362. [Google Scholar] [CrossRef]
- Lonchel, C.M.; Meex, C.; Gangoué-Piéboji, J.; Boreux, R.; Assoumou, M.C.; Melin, P.; De Mol, P. Proportion of extended-spectrum ß-lactamase-producing Enterobacteriaceae in community setting in Ngaoundere, Cameroon. BMC Infect. Dis. 2012, 12, 53. [Google Scholar] [CrossRef] [PubMed]
- Paltansing, S.; Vlot, J.A.; Kraakman, M.E.M.; Mesman, R.; Bruijning, M.L.; Bernards, A.T.; Visser, L.G.; Veldkamp, K.E. Extended-spectrum β-lactamase-producing enterobacteriaceae among travelers from the Netherlands. Emerg. Infect. Dis. 2013, 19, 1206–1213. [Google Scholar] [CrossRef]
- Kantele, A.; Laaveri, T.; Mero, S.; Vilkman, K.; Pakkanen, S.H.; Ollgren, J.; Antikainen, J.; Kirveskari, J. Antimicrobials increase travelers’ risk of colonization by extended-spectrum betalactamase-producing enterobacteriaceae. Clin. Infect. Dis. 2015, 60, 837–846. [Google Scholar] [CrossRef] [PubMed]
- Peirano, G.; Laupland, K.B.; Gregson, D.B.; Pitout, J.D. Colonization of returning travelers with CTX-M-producing Escherichia coli. J. Travel Med. 2011, 18, 299–303. [Google Scholar] [CrossRef] [PubMed]
- Onduru, O.G.; Mkakosya, R.S.; Aboud, S.; Rumisha, S.F. Genetic determinants of resistance among ESBL-producing enterobacteriaceae in community and hospital settings in east, central, and Southern Africa: A systematic review and meta-analysis of prevalence. Can. J. Infect. Dis. Med. Microbiol. 2021, 2021, 1–9. [Google Scholar] [CrossRef]
- Moremi, N.; Claus, H.; Rutta, L.; Frosch, M.; Vogel, U.; Mshana, S.E. High carriage rate of extended-spectrum beta-lactamase-producing Enterobacteriaceae among patients admitted for surgery in Tanzanian hospitals with a low rate of endogenous surgical site infections. J. Hosp. Infect. 2018, 100, 47–53. [Google Scholar] [CrossRef]
- Schaumburg, F.; Alabi, A.; Kokou, C.; Grobusch, M.P.; Köck, R.; Kaba, H.; Becker, K.; Adegnika, A.A.; Kremsner, P.G.; Peters, G.; et al. High burden of extended-spectrum β-lactamase-producing enterobacteriaceae in Gabon. J. Antimicrob. Chemother. 2013, 68, 2140–2143. [Google Scholar] [CrossRef] [PubMed]

| Variables | Frequency (%) | ESBL Phenotype | p-Value | |
|---|---|---|---|---|
| Positive, n (%) | Negative, n (%) | |||
| Age, years | 0.63 | |||
| 18–27 | 125 (41.67) | 20 (6.67) | 105 (35.00) | |
| 28–37 | 95 (31.67) | 13 (4.34) | 82 (27.33) | |
| 38–47 | 43 (14.33) | 10 (3.33) | 33 (11.00) | |
| 48–57 | 20 (6.67) | 3 (1.00) | 17 (5.67) | |
| ≥58 | 17 (5.67) | 4 (1.33) | 13 (4.34) | |
| Sex | 0.11 | |||
| Male | 137 (45.67) | 28 (9.33) | 109 (36.33) | |
| Female | 163 (54.33) | 22 (7.34) | 141 (47.00) | |
| Marital status | 0.05 | |||
| Separated, divorced or widowed | 23 (7.67) | 8 (2.67) | 15 (5.00) | |
| Single (never married) | 93 (31.00) | 14 (4.67) | 79 (26.33) | |
| Married or cohabiting | 184 (61.33) | 28 (9.33) | 156 (52.00) | |
| Education | 0.54 | |||
| Primary | 133 (44.33) | 23 (7.67) | 110 (36.67) | |
| Secondary | 115 (38.33) | 16 (5.33) | 99 (33.00) | |
| College/University | 6 (2.00) | 2 (0.67) | 4 (1.33) | |
| Did not attend any school | 46 (15.33) | 9 (3.00) | 37 (12.33) | |
| Occupation | 0.29 | |||
| Unemployed | 138 (46.00) | 24 (8.00) | 114 (38.01) | |
| Self-employed or business | 57 (19.00) | 12 (4.00) | 45 (15.00) | |
| Employed | 78 (26.00) | 8 (2.67) | 70 (23.33) | |
| Student | 27 (9.00) | 6 (2.00) | 21 (7.00) | |
| History of prior antibiotic use in the previous three months | 0.71 | |||
| Yes | 66 (22.00) | 10 (3.33) | 56 (18.67) | |
| No | 234 (78.00) | 40 (13.33) | 194 (64.67) | |
| History of surgery in the previous three months | 0.54 | |||
| Yes | 29 (9.67) | 6 (2.00) | 23 (7.67) | |
| No | 271 (90.33) | 44 (14.67) | 227 (75.66) | |
| History of hospital admission in the previous three months | 0.23 | |||
| Yes | 7 (2.33) | 0 (0) | 7 (2.33) | |
| No | 293 (97.67) | 50 (16.67) | 243 (81.00) | |
| Outpatient health center | 0.20 | |||
| Limbe | 99 (33.00) | 13 (4.33) | 86 (28.67) | |
| Ndirande | 100 (33.33) | 22 (7.33) | 78 (26.00) | |
| Zingwangwa | 101 (33.67) | 15 (5.00) | 86 (28.67) | |
| ESBL Enterobacterales | Rectal Swab | Urine | Total n (%) |
|---|---|---|---|
| Acinetobacter baumannii | 1 | 0 | 1 (2) |
| Acinetobacter lwoffii | 2 | 0 | 2 (4) |
| Enterobacter aerogens | 1 | 0 | 1 (2) |
| Enterobacter agglomerans | 1 | 1 | 2 (4) |
| Escherichia coli | 29 | 4 | 33 (66) |
| Klebsiella oxytoca | 2 | 0 | 2 (4) |
| Klebsiella pneumoniae | 2 | 0 | 2 (4) |
| Providencia rettgeri | 0 | 1 | 1 (2) |
| Serratia liquefaciens | 0 | 1 | 1 (2) |
| Serratia rubidaea | 0 | 1 | 1 (2) |
| Shigella sonnei | 1 | 0 | 1 (2) |
| Yersinia enterocolitica | 3 | 0 | 3 (6) |
| Total, n (%) | 42 (84) | 8 (16) | 50 (100) |
| Antimicrobial Agents | ESBL-Enterobacterales (% Resistant) | |||||
|---|---|---|---|---|---|---|
| Acinetobacter spp. | Enterobacter spp. | E.coli | Klebsiella spp. | Serratia spp. | Yersinia spp. | |
| Amikacin | 0 | 0 | 15 | 25 | 0 | 0 |
| Amoxicillin | 67 | 67 | 79 | 75 | 50 | 33 |
| Cefepime | 33 | 67 | 21 | 50 | 0 | 33 |
| Ceftriaxone | 100 | 67 | 87 | 75 | 50 | 100 |
| Ciprofloxacin | 67 | 33 | 70 | 75 | 50 | 67 |
| Doxycycline | 67 | 67 | 82 | 75 | 50 | 67 |
| Gentamicin | 67 | 67 | 85 | 50 | 100 | 67 |
| Imipenem | 0 | 0 | 0 | 0 | 0 | 0 |
| Meropenem | 33 | 0 | 0 | 0 | 0 | 33 |
| Nitrofurantoin | 67 | 0 | 27 | 75 | 0 | 100 |
| Trimethoprim-Sulfamethoxazole | 100 | 100 | 91 | 100 | 100 | 100 |
| Associated Factor | Odds Ratio (95% CI) | p-Value |
|---|---|---|
| Age, years | ||
| 18–27 | 0.62 (0.18–2.09) | 0.44 |
| 28–37 | 0.52 (0.15–1.82) | 0.30 |
| 38–47 | 0.98 (0.26–3.71) | 0.98 |
| 48–57 | 0.57 (0.12–3.02) | 0.51 |
| Sex | ||
| Male | 1.65 (0.89–3.04) | 0.11 |
| Marital status | ||
| Married or cohabiting | 0.77 (0.41–1.42) | 0.34 |
| Education | ||
| Primary | 0.86 (0.36–2.02) | 0.71 |
| Secondary | 0.66 (0.27–1.63) | 0.37 |
| College/University | 2.06 (0.32–13.03) | 0.45 |
| Occupation | ||
| Unemployed | 0.72 (0.12–1.28) | 0.12 |
| Self-employed or business | 0.93 (0.31–2.83) | 0.90 |
| Employed | 0.4 (0.12–1.28) | 0.12 |
| History of surgery in the previous three months (Yes) | 1.35 (0.52–3.49) | 0.54 |
| History of antibiotic use in the last three months (Yes) | 0.87 (0.41–1.84) | 0.71 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Onduru, O.G.; Mkakosya, R.S.; Rumisha, S.F.; Aboud, S. Carriage Prevalence of Extended-Spectrum β-Lactamase Producing Enterobacterales in Outpatients Attending Community Health Centers in Blantyre, Malawi. Trop. Med. Infect. Dis. 2021, 6, 179. https://doi.org/10.3390/tropicalmed6040179
Onduru OG, Mkakosya RS, Rumisha SF, Aboud S. Carriage Prevalence of Extended-Spectrum β-Lactamase Producing Enterobacterales in Outpatients Attending Community Health Centers in Blantyre, Malawi. Tropical Medicine and Infectious Disease. 2021; 6(4):179. https://doi.org/10.3390/tropicalmed6040179
Chicago/Turabian StyleOnduru, Onduru Gervas, Rajhab Sawasawa Mkakosya, Susan Fred Rumisha, and Said Aboud. 2021. "Carriage Prevalence of Extended-Spectrum β-Lactamase Producing Enterobacterales in Outpatients Attending Community Health Centers in Blantyre, Malawi" Tropical Medicine and Infectious Disease 6, no. 4: 179. https://doi.org/10.3390/tropicalmed6040179
APA StyleOnduru, O. G., Mkakosya, R. S., Rumisha, S. F., & Aboud, S. (2021). Carriage Prevalence of Extended-Spectrum β-Lactamase Producing Enterobacterales in Outpatients Attending Community Health Centers in Blantyre, Malawi. Tropical Medicine and Infectious Disease, 6(4), 179. https://doi.org/10.3390/tropicalmed6040179






