Screening, Vaccination Uptake and Linkage to Care for Hepatitis B Virus among Health Care Workers in Rural Sierra Leone
Abstract
1. Introduction
2. Materials and Methods
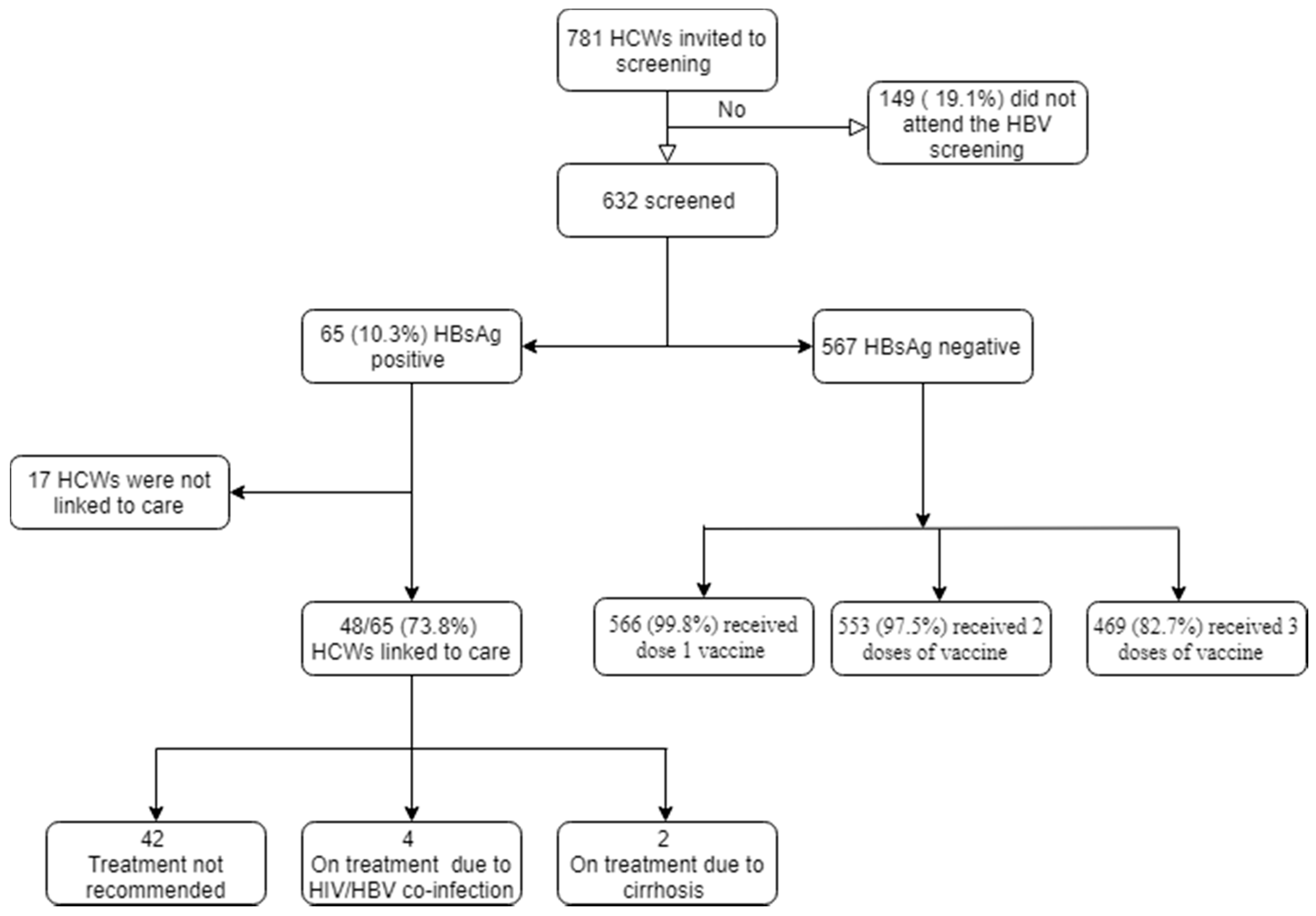
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- World Health Organization. Global Hepatitis Report, 2017; World Health Organization: Geneva, Switzerland, 2017. [Google Scholar]
- World Health Organization. Global Health Sector Strategy on Viral Hepatitis, 2016–2021; World Health Organization: Geneva, Switzerland, 2016. [Google Scholar]
- Stanaway, J.D.; Flaxman, A.D.; Naghavi, M.; Fitzmaurice, C.; Vos, T.; Abubakar, I.; Abu-Raddad, L.J.; Assadi, R.; Bhala, N.; Cowie, B.; et al. The global burden of viral hepatitis from 1990 to 2013: Findings from the Global Burden of Disease Study 2013. Lancet 2016, 388, 1081–1088. [Google Scholar] [CrossRef]
- World Health Organization. Prevention, Care and Treatment of Viral Hepatitis in the AFRICAN Region: Framework for Action, 2016–2020; World Health Organization: Geneva, Switzerland, 2017. [Google Scholar]
- Statistics Sierra Leone. 2015 Population and Housing Census; Summary of Final Results; Statistics Sierra Leone: Freetown, Sierra Leone, 2016. [Google Scholar]
- Word Health Organization. Sierra Leone, Hepatitis B Virus Country Profile; Word Health Organization: Geneva, Switzerland, 2015; Available online: http://whohbsagdashboard.com/# (accessed on 16 June 2020).
- Elduma, A.H.; Saeed, N.S. Hepatitis B virus infection among staff in three hospitals in Khartoum, Sudan, 2006–2007. East Mediterr Health J. 2011, 17, 474–478. [Google Scholar] [CrossRef] [PubMed]
- Auta, A.; Adewuyi, E.O.; Tor-Anyiin, A.; Aziz, D.; Ogbole, E.; Ogbonna, B.O.; Adeloye, D. Health-care workers’ occupational exposures to body fluids in 21 countries in Africa: Systematic review and meta-analysis. Bull World Health Organ. 2017, 95, 831–841f. [Google Scholar] [CrossRef] [PubMed]
- Ziraba, A.K.; Bwogi, J.; Namale, A.; Wainaina, C.W. Mayanja-Kizza H. Sero-prevalence and risk factors for hepatitis B virus infection among health care workers in a tertiary hospital in Uganda. BMC Infect. Dis. 2010, 10, 191. [Google Scholar] [CrossRef] [PubMed]
- Wiktor, S.Z.; Hutin, Y.J. The global burden of viral hepatitis: Better estimates to guide hepatitis elimination efforts. Lancet 2016, 388, 1030–1031. [Google Scholar] [CrossRef]
- Qin, Y.-L.; Li, B.; Zhou, Y.-S.; Zhang, X.; Li, L.; Song, B.; Liu, P.; Yuan, Y.; Zhao, Z.-P.; Jiao, J.; et al. Prevalence and associated knowledge of hepatitis B infection among healthcare workers in Freetown, Sierra Leone. BMC Infect. Dis. 2018, 18, 315. [Google Scholar] [CrossRef] [PubMed]
- Biotest. RightSign HBsAg Rapid Tests. Available online: http://en.biotests.com.cn/product/273120256 (accessed on 7 July 2020).
- Sierra Leone National Guideline for Treatment of Hepatitis B and C; Ministry of Health and Sanitation: Freetown, Sierra Leone, 2020.
- Massaquoi, T.A.; Burke, R.M.; Yang, G.; Lakoh, S.; Sevalie, S.; Li, B.; Jia, H.; Huang, L.; Deen, G.F.; Beynon, F.; et al. Cross sectional study of chronic hepatitis B prevalence among healthcare workers in an urban setting, Sierra Leone. PLoS ONE 2018, 13, e0201820. [Google Scholar] [CrossRef] [PubMed]
- Lemoine, M.; Shimakawa, Y.; Njie, R.; Taal, M.; Ndow, G.; Chemin, I.; Ghosh, S.; Njai, H.F.; Jeng, A.; Sow, A.; et al. Acceptability and feasibility of a screen-and-treat programme for hepatitis B virus infection in The Gambia: The Prevention of Liver Fibrosis and Cancer in Africa (PROLIFICA) study. Lancet Glob. Health 2016, 4, e559–567. [Google Scholar] [CrossRef]
- Burnett, R.J.; François, G.; Mphahlele, M.J.; Mureithi, J.G.; Africa, P.N.; Satekge, M.M.; Mokonoto, D.M.; Meheus, A.; Van Sprundel, M. Hepatitis B vaccination coverage in healthcare workers in Gauteng Province, South Africa. Vaccine 2011, 29, 4293–4297. [Google Scholar] [CrossRef] [PubMed]
- Ansa, G.A.; Ofori, K.N.A.; Houphouet, E.E.; Amoabeng, A.A.; Sifa, J.S.; Amenuveve, C.K.; Odame, G.H. Hepatitis B vaccine uptake among healthcare workers in a referral hospital, Accra. Pan. Afr. Med. J. 2019, 33, 96. [Google Scholar] [CrossRef] [PubMed]
- Puro, V.; De Carli, G.; Cicalini, S.; Soldani, F.; Balslev, U.; Begovac, J.; Boaventura, L.; Marti, M.C.; Navarrete, M.J.H.; Kammerlander, R.; et al. European recommendations for the management of healthcare workers occupationally exposed to hepatitis B virus and hepatitis C virus. Eurosurveillance 2005, 10, 11–12. [Google Scholar] [CrossRef] [PubMed]
- De Schryver, A.; Claesen, B.; Meheus, A.; van Sprundel, M.; François, G. European survey of hepatitis B vaccination policies for healthcare workers. Eur. J. Public Health 2011, 21, 338–343. [Google Scholar] [CrossRef] [PubMed]
- Lungosi, M.B.; Muzembo, B.A.; Mbendi, N.C.; Nkodila, N.A.; Ngatu, N.R.; Suzuki, T.; Wada, K.; Mbendi, N.S.; Ikeda, S.; Shunya, I. Assessing the prevalence of hepatitis B virus infection among health care workers in a referral hospital in Kisantu, Congo DR: A pilot study. Ind. Health 2019, 57, 621–626. [Google Scholar] [CrossRef] [PubMed]
- Ndongo, C.B.; Eteki, L.; Siedner, M.; Mbaye, R.; Chen, J.; Ntone, R.; Donfack, O.; Bongwong, B.; Evoue, R.E.; Zeh, F.; et al. Prevalence and vaccination coverage of Hepatitis B among healthcare workers in Cameroon: A national seroprevalence survey. J. Viral Hepat. 2018, 25, 1582–1587. [Google Scholar] [CrossRef] [PubMed]
- Elzouki, A.N.; Elgamay, S.M.; Zorgani, A.; Elahmer, O. Hepatitis B and C status among health care workers in the five main hospitals in eastern Libya. J. Infect. Public Health 2014, 7, 534–541. [Google Scholar] [CrossRef] [PubMed]
- Mosendane, T.; Kew, M.C.; Osih, R.; Mahomed, A. Nurses at risk for occupationally acquired blood-borne virus infection at a South African academic hospital. S. Afr. Med. J. 2012, 102, 153–156. [Google Scholar] [CrossRef] [PubMed]
- Braka, F.; Nanyunja, M.; Makumbi, I.; Mbabazi, W.; Kasasa, S.; Lewis, R.F. Hepatitis B infection among health workers in Uganda: Evidence of the need for health worker protection. Vaccine 2006, 24, 6930–6937. [Google Scholar] [CrossRef] [PubMed]
- Auta, A.; Adewuyi, E.O.; Kureh, G.T.; Onoviran, N.; Adeloye, D. Hepatitis B vaccination coverage among health-care workers in Africa: A systematic review and meta-analysis. Vaccine 2018, 36, 4851–4860. [Google Scholar] [CrossRef] [PubMed]
- Aaron, D.; Nagu, T.J.; Rwegasha, J.; Komba, E. Hepatitis B vaccination coverage among healthcare workers at national hospital in Tanzania: How much, who and why? BMC Infect. Dis. 2017, 17, 786. [Google Scholar] [CrossRef] [PubMed]
- Malewezi, B.; Omer, S.B.; Mwagomba, B.; Araru, T. Protecting health workers from nosocomial Hepatitis B infections: A review of strategies and challenges for implementation of Hepatitis B vaccination among health workers in Sub-Saharan Africa. J. Epidemiol. Glob. Health 2016, 6, 229–241. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization. Sierra Leone Confirms First Case of COVID-19; World Health Organization: Geneva, Switzerland, 2020; Available online: https://www.afro.who.int/news/sierra-leone-confirms-first-case-covid-19 (accessed on 1 July 2020).
| Total | HBsAg Negative (n,%) | HBsAg Positive (n,%) | |
|---|---|---|---|
| Age (median, IQR) | 32 (25–42) | 32 (18–43) | 29 (20–38) |
| Age category * | |||
| 30 years and above | 397 (62.8) | 365 (91.9) | 32 (8.1) |
| Less than 30 years | 235 (37.2) | 202 (86.0) | 33 (14.0) |
| Gender | |||
| Female | 304 (48.1) | 279 (91.8) | 25 (8.2) |
| Male | 328 (51.9) | 288 (87.8) | 40 (12.2) |
| Occupation | |||
| Nurses | 174(27.5) | 157 (90.2) | 17 (9.8) |
| CHWs | 135 (21.4) | 120 (88.9) | 15 (11.1) |
| Paramedical officers | 95 (15.0) | 83 (87.4) | 12 (12.6) |
| Equipment and facility maintenance officers | 91 (14.4) | 82 (90.1) | 9 (9.9) |
| Environmental and public health officers | 46 (7.3) | 39 (84.8) | 7 (15.2) |
| Others | 91 (14.4) | 86 (94.5) | 5 (5.5) |
| Years of work | |||
| Less than 1 | 78 (12.3) | 75 (96.1) | 3 (3.9) |
| 1–4 | 318 (50.3) | 279 (87.7) | 39 (12.3) |
| 5–9 | 166 (26.3) | 152 (91.6) | 14 (8.4) |
| More than 9 | 70 (11.1) | 61 (87.1) | 9 (12.9) |
| Education level | |||
| Primary | 52 (8.2) | 45 (86.5) | 7 (13.5) |
| Secondary | 243 (38.5) | 214 (88.1) | 29 ( 11.9) |
| Tertiary | 337 (53.3) | 308 (91.4) | 29 (8.6) |
| Never married | |||
| No | 410 (64.9) | 374 (91.2) | 36 (8.8) |
| Yes | 222 (35.1) | 193 (86.9) | 29 (13.1) |
| Have children | |||
| No | 92 (14.6) | 80 (87.0) | 12 (13) |
| Yes | 540 (85.4) | 487 (90.2) | 53 (9.8) |
| Previous exposure to vaccine | |||
| No | 619 (97.0) | 548 (89.4) | 65 (10.6) |
| Yes | 19 (3.0) | 19 (100) | 0 (0) |
| History of needle stick injury | |||
| No | 346 (54.8) | 317 (91.6) | 29 (8.4) |
| Yes | 286 (45.2) | 250 (87.4) | 36 (10.2) |
| Category | Univariate Analysis | Multivariate Analysis | ||||
|---|---|---|---|---|---|---|
| OR | CI | p-Value | aOR | CI | p-Value | |
| Age | ||||||
| Less than 30 years | 1.86 | 1.11–3.12 | 0.02 | 2.17 | 1.16–4.03 | 0.01 |
| More than 30 years | Ref | Ref | ||||
| Gender | ||||||
| Female | Ref | Ref | ||||
| Male | 1.55 | 0.92–2.62 | 0.1 | 2.0 | 1.06–3.78 | 0.03 |
| Occupation | ||||||
| Others | Ref | |||||
| Nurses | 1.86 | 0.66–5.22 | 0.24 | - | - | - |
| CHWs | 2.15 | 0.75–6.14 | 0.15 | 1.98 | 0.67–5.88 | 0.22 |
| Paramedical officers | 2.49 | 0.84–7.37 | 0.1 | 2.08 | 0.68–6.38 | 0.2 |
| Equipment and facility maintenance Officers | 1.89 | 0.61–5.87 | 0.27 | - | - | - |
| Environmental and public health officers | 3.09 | 0.92–10.34 | 0.07 | 2.48 | 0.71–8.72 | 0.16 |
| Education level | ||||||
| Primary | ref | |||||
| Secondary | 0.87 | 0.36–2.11 | 0.76 | - | - | - |
| Above secondary level | 0.61 | 0.25–1.46 | 0.27 | - | - | - |
| Never Married | ||||||
| No | ref | ref | ||||
| Yes | 1.56 | 0.93–2.62 | 0.09 | 1.37 | 0.75–2.51 | 0.31 |
| Have children | ||||||
| No | ref | |||||
| Yes | 0.73 | 0.37–1.42 | 0.35 | - | - | - |
| Years of work-years | ||||||
| Less than 1 | Ref | |||||
| 1–4 | 3.49 | 1.05–11.62 | 0.04 | 3.99 | 1.15–13.73 | 0.03 |
| 5–9 | 2.30 | 0.64–8.26 | 0.2 | - | - | - |
| More than 9 | 3.69 | 0.96–14.22 | 0.06 | 6.16 | 1.41–26.9 | 0.02 |
| History of Needle stick Injury | ||||||
| No | Ref | ref | ||||
| Yes | 1.57 | 0.94–2.64 | 0.09 | 1.51 | 0.86–2.66 | 0.15 |
| Variable | Not Linked to Care (n%) | Linked to Care (n/%) |
|---|---|---|
| Linkage | 17 (26.1) | 48 (73.9) |
| Age (median, IQR) | 29 (27–38) | 32. (27.5–43) |
| Age category | ||
| 30 years and above | 7 | 25 |
| Less than 30 years | 10 | 23 |
| Gender | ||
| Male | 7 | 18 |
| Female | 10 | 30 |
| Patient on treatment | 6 (12.5%) | |
| Cirrhosis | 2 | |
| HIV/HBV co-infection | 4 | |
| Follow-up and monitoring in the clinic | 42 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bangura, M.; Frühauf, A.; Mhango, M.; Lavallie, D.; Reed, V.; Rodriguez, M.P.; Smith, S.J.; Lakoh, S.; Ibrahim-Sayo, E.; Conteh, S.; et al. Screening, Vaccination Uptake and Linkage to Care for Hepatitis B Virus among Health Care Workers in Rural Sierra Leone. Trop. Med. Infect. Dis. 2021, 6, 65. https://doi.org/10.3390/tropicalmed6020065
Bangura M, Frühauf A, Mhango M, Lavallie D, Reed V, Rodriguez MP, Smith SJ, Lakoh S, Ibrahim-Sayo E, Conteh S, et al. Screening, Vaccination Uptake and Linkage to Care for Hepatitis B Virus among Health Care Workers in Rural Sierra Leone. Tropical Medicine and Infectious Disease. 2021; 6(2):65. https://doi.org/10.3390/tropicalmed6020065
Chicago/Turabian StyleBangura, Musa, Anna Frühauf, Michael Mhango, Daniel Lavallie, Vicky Reed, Marta Patiño Rodriguez, Samuel Juana Smith, Sulaiman Lakoh, Emmanuel Ibrahim-Sayo, Sorie Conteh, and et al. 2021. "Screening, Vaccination Uptake and Linkage to Care for Hepatitis B Virus among Health Care Workers in Rural Sierra Leone" Tropical Medicine and Infectious Disease 6, no. 2: 65. https://doi.org/10.3390/tropicalmed6020065
APA StyleBangura, M., Frühauf, A., Mhango, M., Lavallie, D., Reed, V., Rodriguez, M. P., Smith, S. J., Lakoh, S., Ibrahim-Sayo, E., Conteh, S., Lado, M., & Kachimanga, C. (2021). Screening, Vaccination Uptake and Linkage to Care for Hepatitis B Virus among Health Care Workers in Rural Sierra Leone. Tropical Medicine and Infectious Disease, 6(2), 65. https://doi.org/10.3390/tropicalmed6020065






