Japanese Encephalitis Virus in Australia: From Known Known to Known Unknown
Abstract
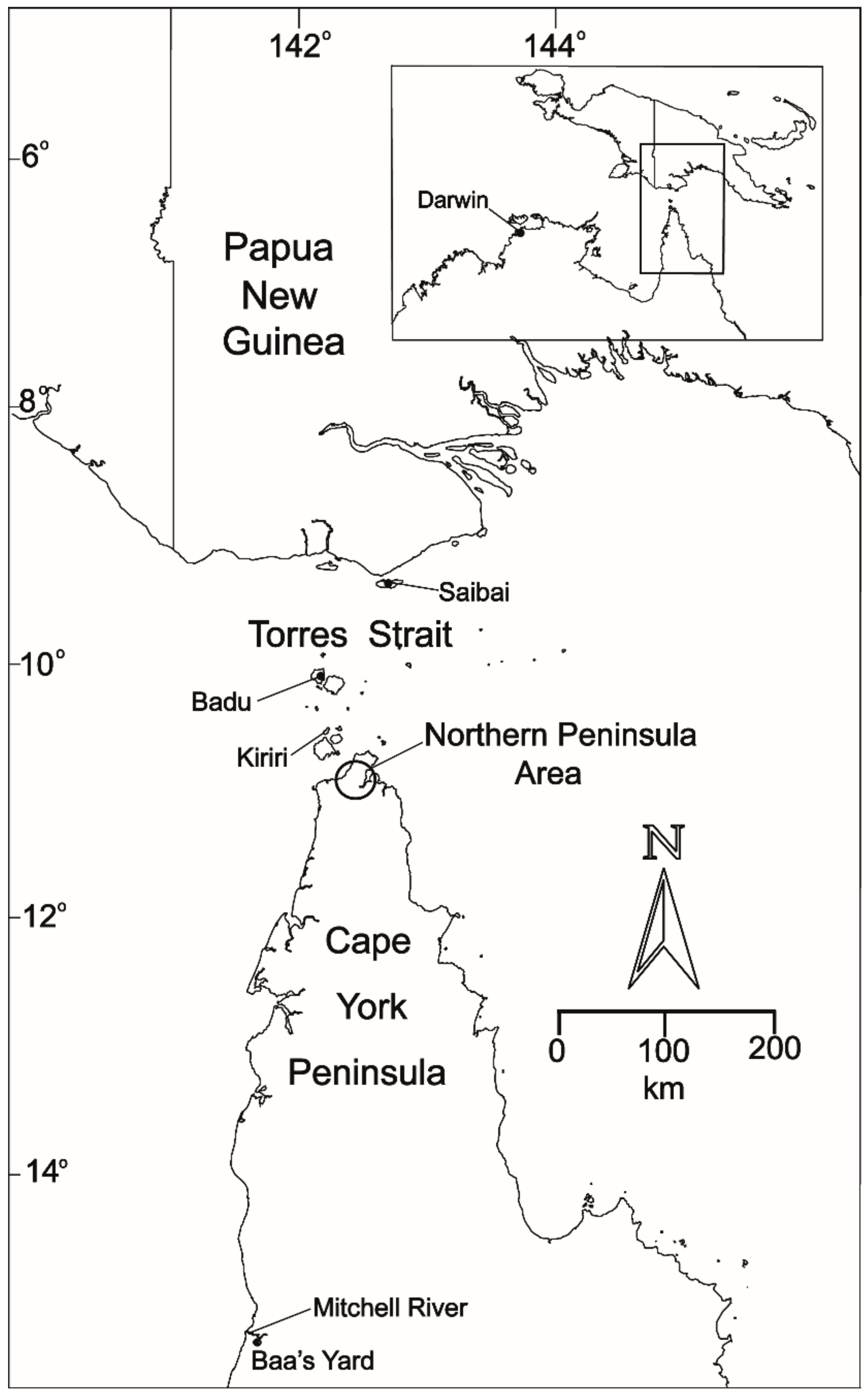
:1. Introduction
2. The Emergence of JEV in Northern Australia
3. Elucidating the Ecology of JEV Transmission Cycles in Australia
3.1. Vertebrate Host Studies
3.2. Incrimination of Mosquito Vectors
3.3. The Influence of Mosquito Host Feeding Patterns on JEV Transmission in Northern Australia
3.4. Development of Mosquito-Based JEV Surveillance Systems
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Campbell, G.L.; Hills, S.L.; Fischer, M.; Jacobson, J.A.; Hoke, C.H.; Hombach, J.M.; Marfin, A.A.; Solomon, T.; Tsai, T.F.; Tsu, V.D.; et al. Estimated global incidence of Japanese encephalitis: A systematic review. Bull. World Health Organ. 2011, 89, 766–774. [Google Scholar] [CrossRef]
- Mackenzie, J.S.; Williams, D.T.; Smith, D.W. Japanese encephalitis virus: The geographic distribution, incidence, and spread of a virus with a propensity to emerge in new areas. Perspect. Med. Virol. 2006, 16, 201–268. [Google Scholar] [CrossRef]
- Van den Hurk, A.F.; Ritchie, S.A.; Mackenzie, J.S. Ecology and geographical expansion of Japanese encephalitis virus. Ann. Rev. Entomol. 2009, 54, 17–35. [Google Scholar] [CrossRef] [PubMed]
- Ricklin, M.E.; García-Nicolás, O.; Brechbühl, D.; Python, S.; Zumkehr, B.; Nougairede, A.; Charrel, R.N.; Posthaus, H.; Oevermann, A.; Summerfield, A. Vector-free transmission and persistence of Japanese encephalitis virus in pigs. Nat. Commun. 2016, 7, 10832. [Google Scholar] [CrossRef] [PubMed]
- Impoinvil, D.E.; Baylis, M.; Solomon, T. Japanese encephalitis: On the One Health agenda. Curr. Top. Microbiol. Immunol. 2013, 365, 205–247. [Google Scholar] [CrossRef]
- Fleming, K. Japanese encephalitis in an Australian soldier returned from Vietnam. Med. J. Aust. 1975, 2, 19–23. [Google Scholar] [PubMed]
- Hanson, J.P.; Taylor, C.T.; Richards, A.R.; Smith, I.L.; Boutlis, C.S. Japanese encephalitis acquired near Port Moresby: Implications for residents and travellers to Papua New Guinea. Med. J. Aust. 2004, 181, 282. [Google Scholar]
- Macdonald, W.B.G.; Tink, A.R.; Ouvrier, R.A.; Menser, M.A.; de Silva, L.M.; Naim, H.; Hawkes, R.A. Japanese encephalitis after a two-week holiday in Bali. Med. J. Aust. 1989, 150, 334–339. [Google Scholar]
- Hanna, J.N.; Ritchie, S.A.; Phillips, D.A.; Shield, J.; Bailey, M.C.; Mackenzie, J.S.; Poidinger, M.; McCall, B.J.; Mills, P.J. An outbreak of Japanese encephalitis in the Torres Strait, Australia, 1995. Med. J. Aust. 1996, 165, 256–260. [Google Scholar]
- Ritchie, S.A.; Phillips, D.; Broom, A.; Mackenzie, J.; Poidinger, M.; van den Hurk, A. Isolation of Japanese encephalitis virus from Culex annulirostris in Australia. Am. J. Trop. Med. Hyg. 1997, 56, 80–84. [Google Scholar] [CrossRef]
- Ritchie, S.A.; van den Hurk, A.F.; Shield, J. The 1995 Japanese encephalitis outbreak: Why Badu? Arbovirus Res. Aust. 1997, 7, 224–227. [Google Scholar]
- Hanna, J.; Barnett, D.; Ewald, D. Vaccination against Japanese encephalitis in the Torres Strait. Comm. Dis. Intell. 1996, 19, 447. [Google Scholar]
- Shield, J.; Hanna, J.; Phillips, D. Reappearance of the Japanese encephalitis virus in the Torres Strait, 1996. Comm. Dis. Intell. 1996, 20, 191. [Google Scholar]
- Johansen, C.A.; van den Hurk, A.F.; Ritchie, S.A.; Zborowski, P.; Paru, R.; Bockari, M.J.; Drew, A.C.; Khromykh, T.I.; Mackenzie, J.S. Isolation of Japanese encephalitis virus from mosquitoes (Diptera: Culicidae) collected in the Western Province of Papua New Guinea, 1997–1998. Am. J. Trop. Med. Hyg. 2000, 62, 631–638. [Google Scholar] [CrossRef] [PubMed]
- Johansen, C.; Ritchie, S.; Hurk, A.v.d.; Bockarie, M.; Hanna, J.; Phillips, D.; Melrose, W.; Poidinger, M.; Scherret, J.; Hall, R.; et al. The Search for Japanese encephalitis virus in the Western Province of Papua New Guinea, 1996. Arbovirus Res. Aust. 1997, 7, 131–136. [Google Scholar]
- Ritchie, S.A.; Rochester, W. Wind-blown mosquitoes and introduction of Japanese encephalitis into Australia. Emerg. Infect. Dis. 2001, 7, 900–903. [Google Scholar] [CrossRef]
- Hanna, J.N.; Ritchie, S.A.; Phillips, D.A.; Lee, J.M.; Hills, S.; van den Hurk, A.F.; Pyke, A.; Johansen, C.A.; Mackenzie, J.S. Japanese encephalitis in north Queensland, Australia, 1998. Med. J. Aust. 1999, 170, 533–536. [Google Scholar] [CrossRef]
- Johansen, C.A.; van den Hurk, A.F.; Pyke, A.T.; Zborowski, P.; Phillips, D.A.; Mackenzie, J.S.; Ritchie, J.S. Entomological Investigations of an outbreak of Japanese encephalaitis virus in the Torres Strait, Australia, in 1998. J. Med. Entomol. 2001, 38, 581–588. [Google Scholar] [CrossRef]
- Van den Hurk, A.F.; Johansen, C.A.; Zborowski, P.; Phillips, D.A.; Pyke, A.T.; Mackenzie, J.S.; Ritchie, S.A. Flaviviruses isolated from mosquitoes collected during the first outbreak of Japanese encephalitis virus on Cape York Peninsula, Australia. Am. J. Trop. Med. Hyg. 2001, 64, 125–130. [Google Scholar] [CrossRef]
- Barr, J. Drought Assessment: The 1997-98 El Nino Drought in Papua New Guinea and the Solomon Islands. Aust. J. Emerg. Manag. 1999, 14, 31–37. [Google Scholar]
- Van den Hurk, A.F.; Nisbet, D.J.; Johansen, C.A.; Foley, P.N.; Ritchie, S.A.; Mackenzie, J.S. Japanese encephalitis on Badu Island, Australia: The first isolation of Japanese encephalitis virus from Culex gelidus in the Australasian region and the role of mosquito host-feeding patterns in virus transmission cycles. Trans. Royal. Soc. Trop. Med. Hyg. 2001, 95, 595–600. [Google Scholar] [CrossRef]
- Johansen, C.A.; Nisbet, D.J.; Foley, P.N.; van den Hurk, A.F.; Hall, R.A.; Mackenzie, J.S.; Ritchie, S.A. Flavivirus isolations from mosquitoes collected from Saibai Island in the Torres Strait, Australia, during an incursion of Japanese encephalitis virus. Med. Vet. Entomol. 2004, 18, 281–287. [Google Scholar] [CrossRef]
- Pyke, A.T.; Williams, D.T.; Nisbet, D.J.; van den Hurk, A.F.; Taylor, C.T.; Johansen, C.A.; Macdonald, J.; Hall, R.A.; Simmons, R.J.; Mason, R.J.V.; et al. The appearance of a second genotype of Japanese encephalitis virus in the Australasian region. Am. J. Trop. Med. Hyg. 2001, 65, 747–753. [Google Scholar] [CrossRef] [PubMed]
- Ritchie, S.A.; van den Hurk, A.F.; Zborowski, P.; Kerlin, T.J.; Banks, D.; Walker, J.A.; Lee, J.M.; Montgomery, B.L.; Smith, G.A.; Pyke, A.T.; et al. Operational trials of remote mosquito trap systems for Japanese encephalitis virus surveillance in the Torres Strait, Australia. Vector Borne Zoonotic Dis. 2007, 7, 497–506. [Google Scholar] [CrossRef] [PubMed]
- Van den Hurk, A.F.; Montgomery, B.L.; Northill, J.A.; Smith, I.L.; Zborowski, P.; Ritchie, S.A.; Mackenzie, J.S.; Smith, G.A. The first isolation of Japanese encephalitis virus from mosquitoes collected from mainland Australia. Am. J. Trop. Med. Hyg. 2006, 75, 21–25. [Google Scholar] [CrossRef]
- Johnson, B.J.; Kerlin, T.; Hall-Mendelin, S.; van den Hurk, A.F.; Cortis, G.; Doggett, S.L.; Toi, C.; Fall, K.; McMahon, J.L.; Townsend, M.; et al. Development and field evaluation of the sentinel mosquito arbovirus capture kit (SMACK). Parasit. Vectors 2015, 8, 509. [Google Scholar] [CrossRef]
- Boyle, D.B.; Dickerman, R.W.; Marshall, I.D. Primary viraemia responses of herons to experimental infection with Murray Valley encephalitis, Kunjin and Japanese encephalitis viruses. Aust. J. Exp. Biol. Med. Sci. 1983, 61, 655–664. [Google Scholar] [CrossRef]
- Mackenzie, J.S.; Johansen, C.A.; Ritchie, S.A.; van den Hurk, A.F.; Hall, R.A. The emergence and spread of Japanese encephalitis virus in Australasia. Curr. Top. Microbiol. Immunol. 2002, 267, 49–73. [Google Scholar] [CrossRef]
- Van den Hurk, A.F.; Smith, C.S.; Field, H.E.; Smith, I.L.; Northill, J.A.; Taylor, C.T.; Jansen, C.C.; Smith, G.A.; Mackenzie, J.S. Transmission of Japanese encephalitis virus from the black flying fox, Pteropus alecto, to Culex annulirostris mosquitoes, despite the absence of detectable viremia. Am. J. Trop. Med. Hyg. 2009, 81, 457–462. [Google Scholar] [CrossRef]
- Williams, D.T.; Daniels, P.W.; Lunt, R.A.; Wang, L.-F.; Newberry, K.M.; Mackenzie, J.S. Experimental infections of pigs with Japanese encephalitis virus and closely related Australian flaviviruses. Am. J. Trop. Med. Hyg. 2001, 65, 379–387. [Google Scholar] [CrossRef]
- Van den Hurk, A.F.; Jansen, C.C. Arboviruses of Oceania. In Neglected Tropical Diseases—Oceania; Loukas, A., Ed.; Springer Nature: Basel, Switzerland, 2016; pp. 193–235. [Google Scholar] [CrossRef]
- Van den Hurk, A.F.; Nisbet, D.J.; Hall, R.A.; Kay, B.H.; Mackenzie, J.S.; Ritchie, S.A. Vector competence of Australian mosquitoes (Diptera: Culicidae) for Japanese encephalitis virus. J. Med. Entomol. 2003, 40, 82–90. [Google Scholar] [CrossRef] [PubMed]
- Hemmerter, S.; Slapeta, J.; van den Hurk, A.F.; Cooper, R.D.; Whelan, P.I.; Russell, R.C.; Johansen, C.A.; Beebe, N.W. A curious coincidence: Mosquito biodiversity and the limits of the Japanese encephalitis virus in Australasia. BMC Evol. Biol. 2007, 7, 100. [Google Scholar] [CrossRef] [PubMed]
- Johnson, P.H.; Hall-Mendelin, S.; Whelan, P.I.; Frances, S.P.; Jansen, C.C.; Mackenzie, D.O.; Northill, J.A.; van den Hurk, A.F. Vector competence of Australian Culex gelidus Theobald (Diptera: Culicidae) for endemic and exotic arboviruses. Aust. J. Entomol. 2009, 48, 234–240. [Google Scholar] [CrossRef]
- Chapman, H.F.; Kay, B.H.; Ritchie, S.A.; van den Hurk, A.F.; Hughes, J.M. Definition of species in the Culex sitiens subgroup (Diptera: Culicidae) from Papua New Guinea and Australia. J. Med. Entomol. 2000, 37, 736–742. [Google Scholar] [CrossRef] [PubMed]
- Hall-Mendelin, S.; Jansen, C.C.; Cheah, W.Y.; Montgomery, B.L.; Hall, R.A.; Ritchie, S.A.; van den Hurk, A.F. Culex annulirostris (Diptera: Culicidae) host feeding patterns and Japanese encephalitis virus ecology in northern Australia. J. Med. Entomol. 2012, 49, 371–377. [Google Scholar] [CrossRef] [PubMed]
- Van den Hurk, A.F.; Johansen, C.A.; Zborowski, P.; Paru, R.; Foley, P.N.; Beebe, N.W.; Mackenzie, J.S.; Ritchie, S.A. Mosquito host-feeding patterns and implications for Japanese encephalitis virus transmission in northern Australia and Papua New Guinea. Med. Vet. Entomol. 2003, 17, 403–411. [Google Scholar] [CrossRef]
- Van den Hurk, A.F.; Smith, I.L.; Smith, G.A. Development and evaluation of real-time polymerase chain reaction assays to identify mosquito (Diptera: Culicidae) blood meals originating from native Australian mammals. J. Med. Entomol. 2007, 44, 85–92. [Google Scholar] [CrossRef]
- Van den Hurk, A.F.; Ritchie, S.A.; Johansen, C.A.; Mackenzie, J.S.; Smith, G.A. Domestic pigs and Japanese encephalitis virus infection, Australia. Emerg. Infect. Dis. 2008, 14, 1736–1738. [Google Scholar] [CrossRef]
- Bryan, J.H.; O’Donnell, M.S.; Berry, G.; Carvan, T. Dispersal of adult female Culex annulirostris in Griffith, New South Wales, Australia: A further study. J. Am. Mosq. Control. Assoc. 1992, 8, 398–403. [Google Scholar]
- Van den Hurk, A.F.; Johnson, P.H.; Hall-Mendelin, S.; Northill, J.A.; Simmons, R.J.; Jansen, C.C.; Frances, S.P.; Smith, G.A.; Ritchie, S.A. Expectoration of flaviviruses during sugar feeding by mosquitoes (Diptera: Culicidae). J. Med. Entomol. 2007, 44, 845–850. [Google Scholar] [CrossRef]
- Hall-Mendelin, S.; Ritchie, S.A.; Johansen, C.A.; Zborowski, P.; Cortis, G.; Dandridge, S.; Hall, R.A.; van den Hurk, A.F. Exploiting mosquito sugar feeding to detect mosquito-borne pathogens. Proc. Natl. Acad. Sci. USA 2010, 107, 11255–11259. [Google Scholar] [CrossRef] [PubMed]
- Ritchie, S.A.; Cortis, G.; Paton, C.; Townsend, M.; Shroyer, D.; Zborowski, P.; Hall-Mendelin, S.; van den Hurk, A.F. A simple non-powered passive trap for the collection of mosquitoes for arbovirus surveillance. J. Med. Entomol. 2013, 50, 185–194. [Google Scholar] [CrossRef] [PubMed]
- Huang, B.; Firth, C.; Watterson, D.; Allcock, R.; Colmant, A.M.; Hobson-Peters, J.; Kirkland, P.; Hewitson, G.; McMahon, J.; Hall-Mendelin, S.; et al. Genetic characterization of archived Bunyaviruses and their potential for emergence in Australia. Emerg. Infect. Dis. 2016, 22, 833–840. [Google Scholar] [CrossRef] [PubMed]
- Van den Hurk, A.F.; Hall-Mendelin, S.; Townsend, M.; Kurucz, N.; Edwards, J.; Ehlers, G.; Rodwell, C.; Moore, F.A.; McMahon, J.L.; Northill, J.A.; et al. Applications of a sugar-based surveillance system to track arboviruses in wild mosquito populations. Vector Borne Zoonotic Dis. 2014, 14, 66–73. [Google Scholar] [CrossRef] [PubMed]
- Ramirez, A.L.; Hall-Mendelin, S.; Doggett, S.L.; Hewitson, G.R.; McMahon, J.L.; Ritchie, S.A.; van den Hurk, A.F. Mosquito excreta: A sample type with many potential applications for the investigation of Ross River virus and West Nile virus ecology. PLoS Negl. Trop. Dis. 2018, 12, e0006771. [Google Scholar] [CrossRef] [PubMed]
- Fontaine, A.; Jiolle, D.; Moltini-Conclois, I.; Lequime, S.; Lambrechts, L. Excretion of dengue virus RNA by Aedes aegypti allows non-destructive monitoring of viral dissemination in individual mosquitoes. Sci. Rep. 2016, 6, 24885. [Google Scholar] [CrossRef]
- Australian Technical Advisory Group on Immunisation. Australian Immunisation Handbook; Australian Government Department of Health: Canberra, Australia, 2018. Available online: https://immunisationhandbook.health.gov.au/ (accessed on 18 February 2019).
- Australian Government Department of Health. Japanese Encephalitis Virus Infection Case Definition—V1.1. Available online: http://www.health.gov.au/internet/main/publishing.nsf/Content/cda-surveil-nndss-casedefs-cd_je.htm (accessed on 18 February 2019).
- Mackenzie, J.S.; Broom, A.K.; Hall, R.A.; Johansen, C.A.; Lindsay, M.D.; Phillips, D.A.; Ritchie, S.A.; Russell, R.C.; Smith, D.W. Arboviruses in the Australian region, 1990 to 1998. Comm. Dis. Intell. 1998, 22, 93–100. [Google Scholar]
- Mackenzie, J.S.; Gubler, D.J.; Petersen, L.R. Emerging flaviviruses: The spread and resurgence of Japanese encephalitis, West Nile and dengue viruses. Nature Med. 2004, 10, S98–S109. [Google Scholar] [CrossRef]
- Mackenzie, J.S.; Lindsay, M.D.A.; Smith, D.W.; Imrie, A. The ecology and epidemiology of Ross River and Murray Valley encephalitis viruses in Western Australia: Examples of One Health in action. Trans. R. Soc. Trop. Med. Hyg. 2017, 111, 248–254. [Google Scholar] [CrossRef]
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
van den Hurk, A.F.; Pyke, A.T.; Mackenzie, J.S.; Hall-Mendelin, S.; Ritchie, S.A. Japanese Encephalitis Virus in Australia: From Known Known to Known Unknown. Trop. Med. Infect. Dis. 2019, 4, 38. https://doi.org/10.3390/tropicalmed4010038
van den Hurk AF, Pyke AT, Mackenzie JS, Hall-Mendelin S, Ritchie SA. Japanese Encephalitis Virus in Australia: From Known Known to Known Unknown. Tropical Medicine and Infectious Disease. 2019; 4(1):38. https://doi.org/10.3390/tropicalmed4010038
Chicago/Turabian Stylevan den Hurk, Andrew F., Alyssa T. Pyke, John S. Mackenzie, Sonja Hall-Mendelin, and Scott A. Ritchie. 2019. "Japanese Encephalitis Virus in Australia: From Known Known to Known Unknown" Tropical Medicine and Infectious Disease 4, no. 1: 38. https://doi.org/10.3390/tropicalmed4010038
APA Stylevan den Hurk, A. F., Pyke, A. T., Mackenzie, J. S., Hall-Mendelin, S., & Ritchie, S. A. (2019). Japanese Encephalitis Virus in Australia: From Known Known to Known Unknown. Tropical Medicine and Infectious Disease, 4(1), 38. https://doi.org/10.3390/tropicalmed4010038





