Elimination of Schistosomiasis Mekongi from Endemic Areas in Cambodia and the Lao People’s Democratic Republic: Current Status and Plans
Abstract
:1. Historical Background
1.1. Lao PDR
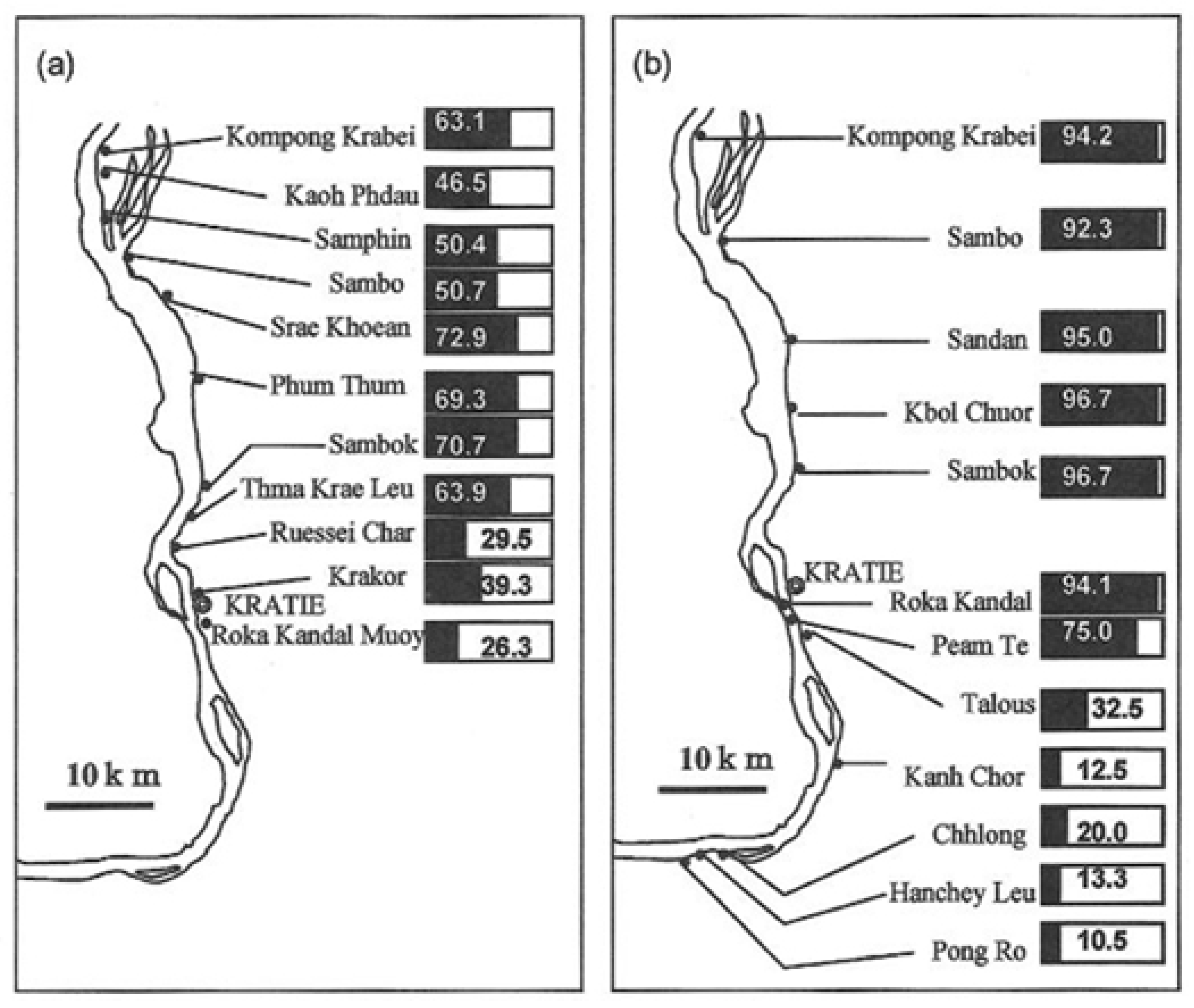
1.2. Cambodia
2. From Morbidity Control to Elimination
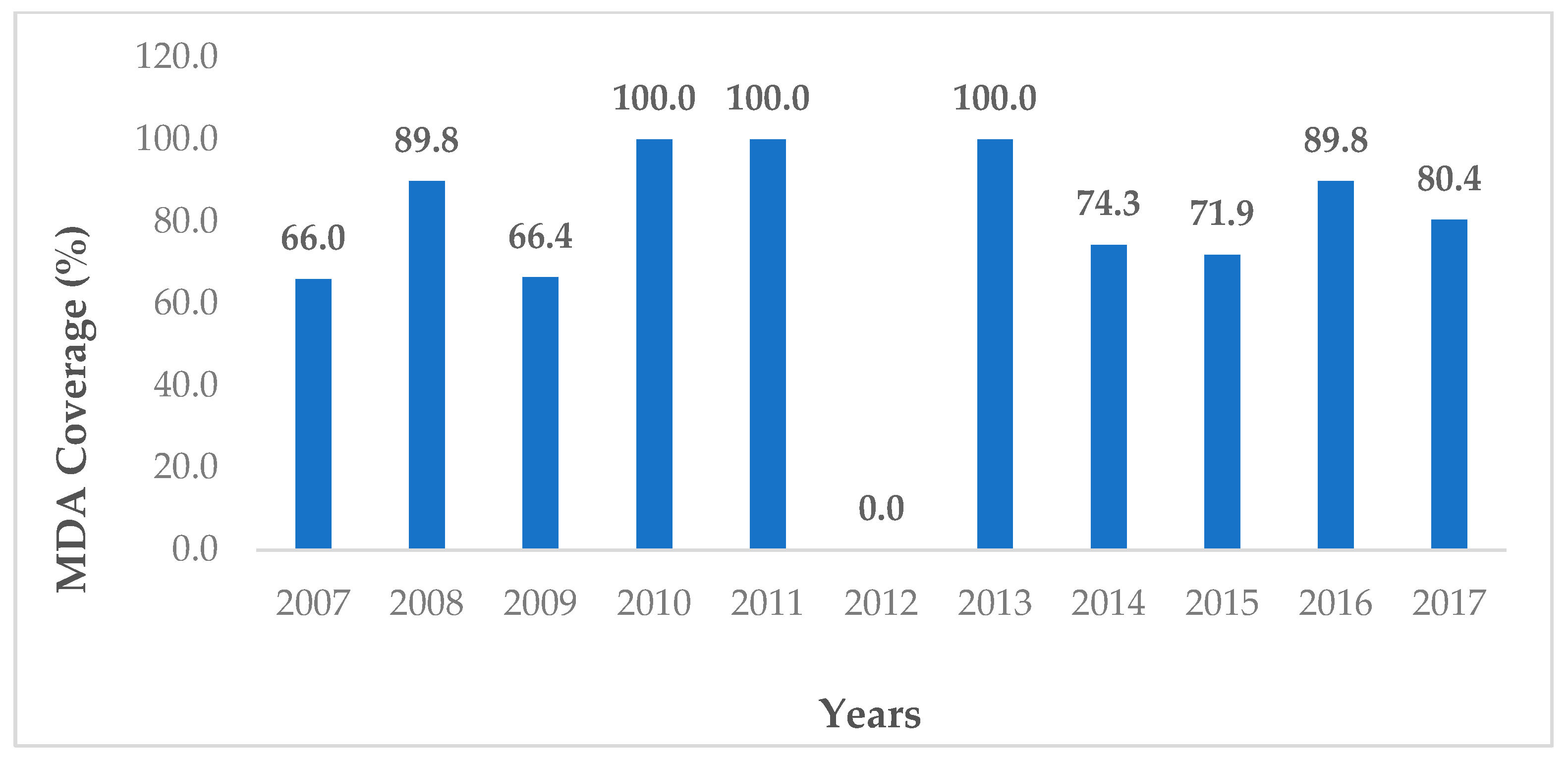
2.1. Lao PDR
2.1.1. Policy, Commitment, and Interdisciplinarity
2.1.2. Activities after the Millennium Shift
2.2. Cambodia
3. Achievements
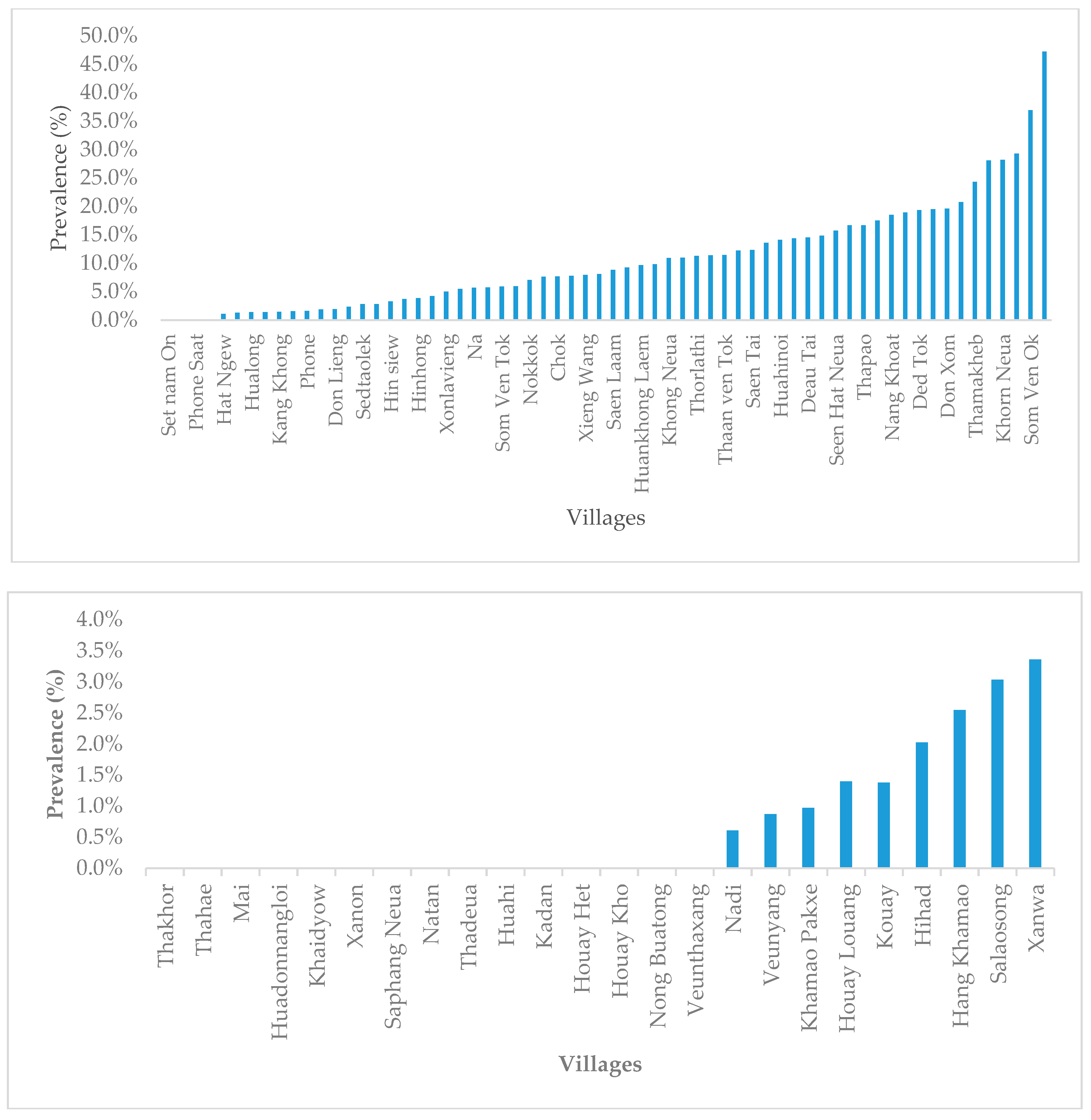
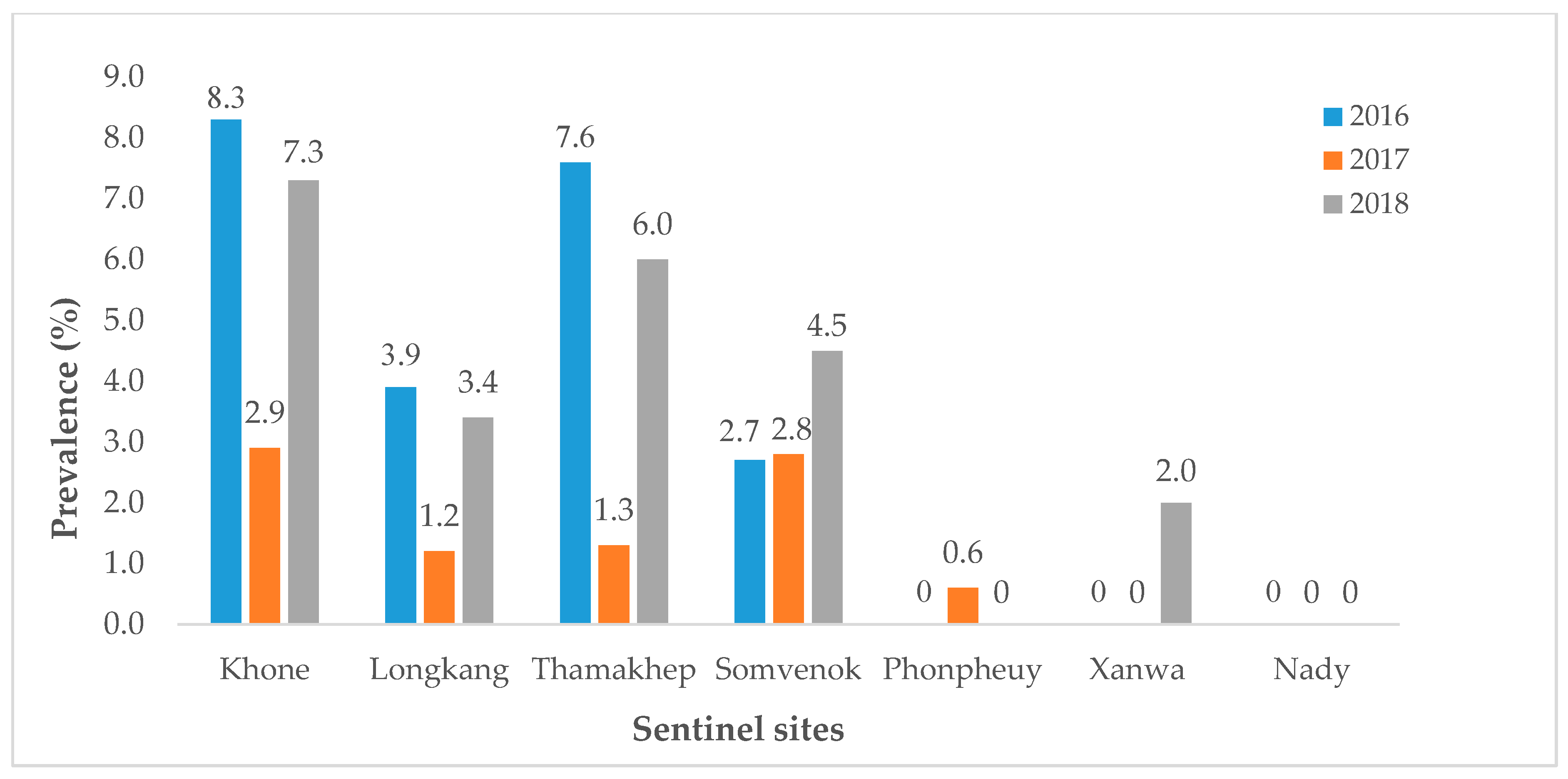
3.1. Lao PDR
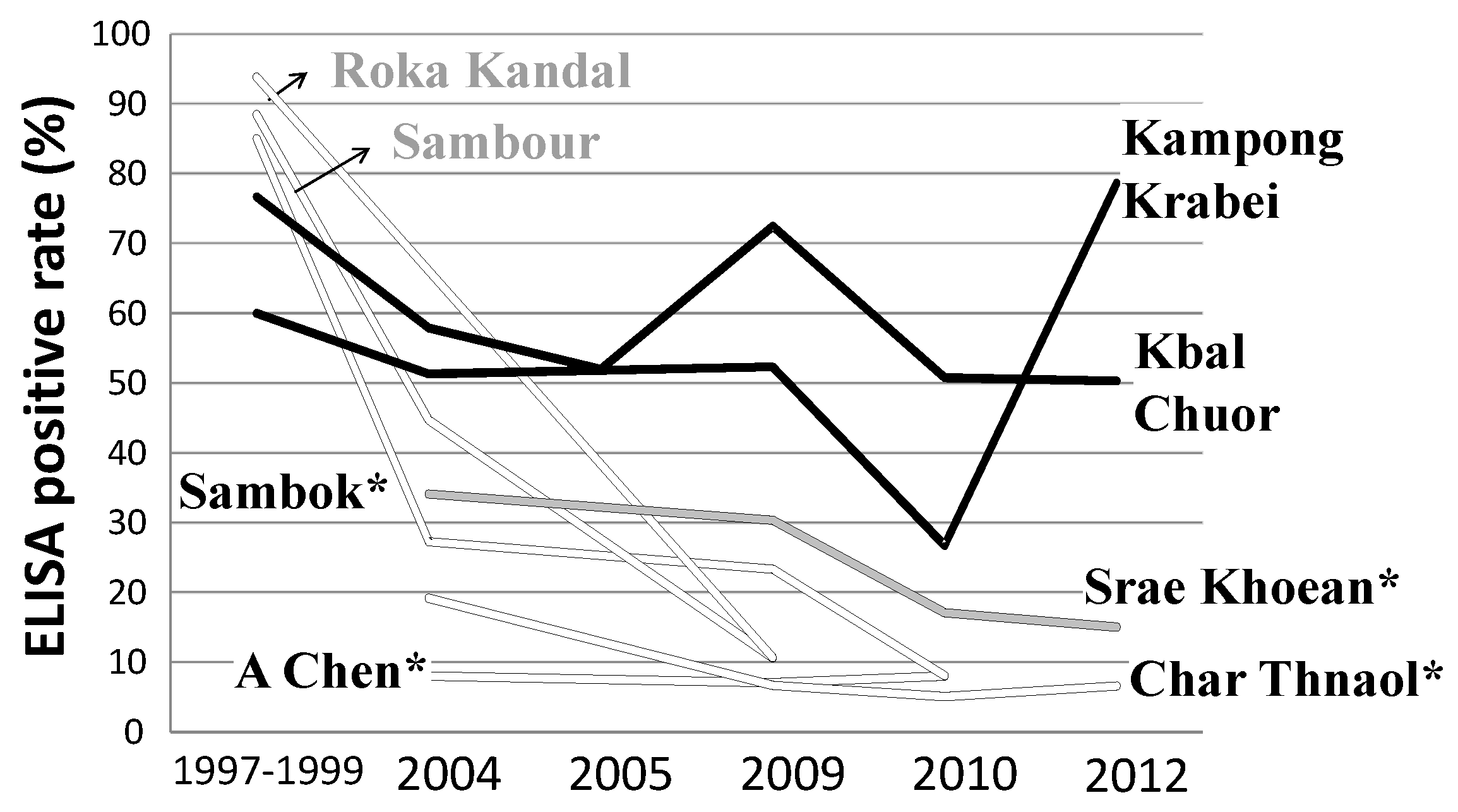
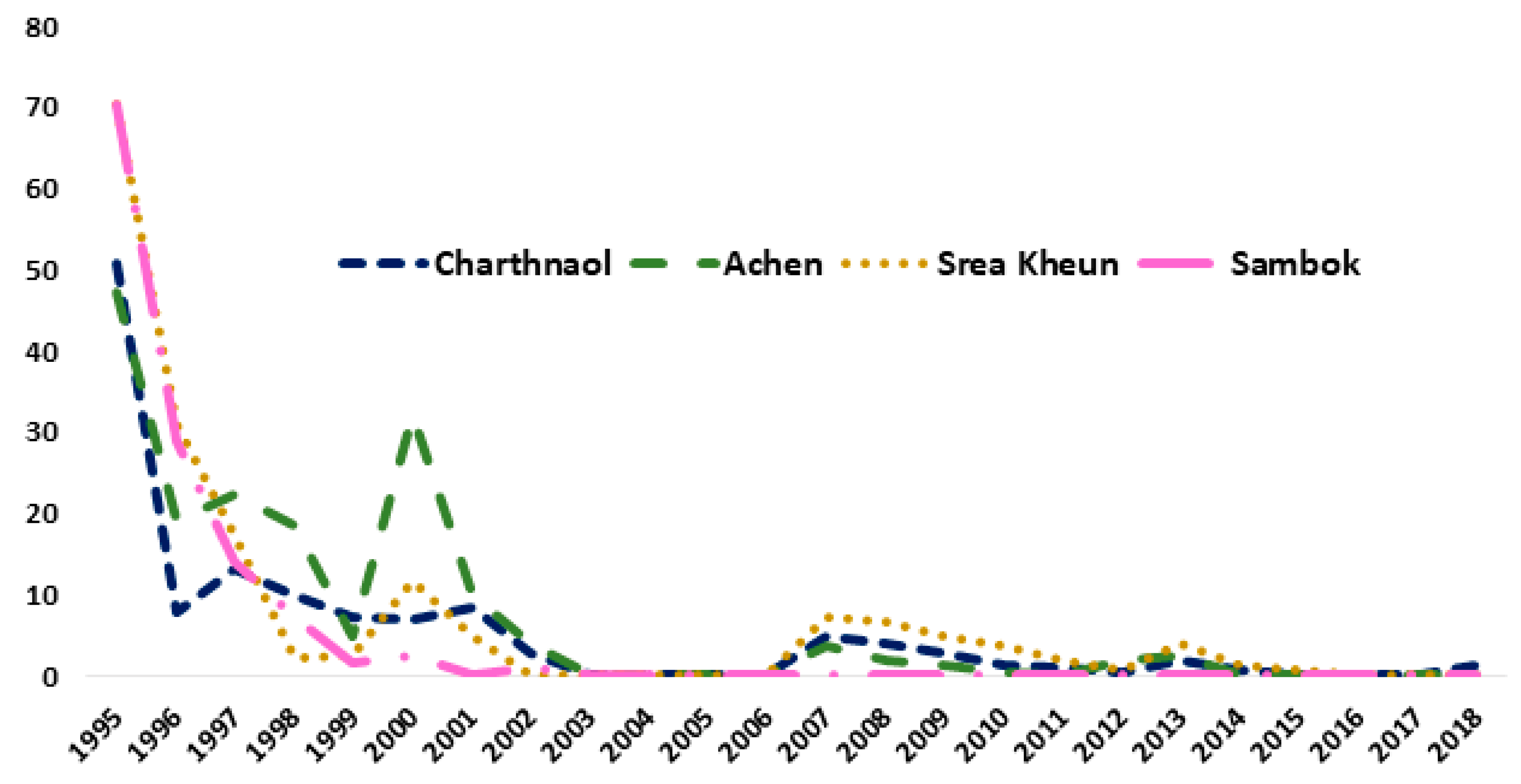
3.2. Cambodia
- Universal access to the One-Health intervention package consisting of preventive chemotherapy, CL-SWASH, and treatment of the animal reservoirs;
- Strengthening community members’ health literacy to prevent reinfection and interrupt transmission through a sustained change of sanitation and hygiene behaviour empowering people to act as drivers of schistosomiasis elimination; and
- Adoption of effective and sustained active and passive surveillance of schistosomiasis in human and reservoirs hosts.
4. Next Steps and Challenges
5. Conclusions
Funding
Acknowledgments
Conflicts of Interest
References
- Colley, D.G.; Bustinduy, A.L.; Secor, W.E.; King, C.H. Human schistosomiasis. Lancet 2014, 383, 2253–2264. [Google Scholar] [CrossRef]
- Steinmann, P.; Keiser, J.; Bos, R.; Tanner, M.; Utzinger, J. Schistosomiasis and water resources development: Systematic review, meta-analysis, and estimates of people at risk. Lancet Infect Dis. 2006, 6, 411–425. [Google Scholar] [CrossRef]
- Davis, A.; Wegner, D.H. Multicentre trials of praziquantel in human schistosomiasis: Design and techniques. Bull. World Health Organ. 1979, 57, 767–771. [Google Scholar] [PubMed]
- King, C.H. Parasites and poverty: The case of schistosomiasis. Acta Trop. 2010, 113, 95–104. [Google Scholar] [CrossRef] [PubMed]
- Dalys, G.B.D.; Collaborators, H. Global, regional, and national disability-adjusted life-years (DALYs) for 333 diseases and injuries and healthy life expectancy (HALE) for 195 countries and territories, 1990–2016: A systematic analysis for the Global Burden of Disease Study 2016. Lancet 2017, 390, 1260–1344. [Google Scholar] [CrossRef]
- Murray, C.J.; Vos, T.; Lozano, R.; Naghavi, M.; Flaxman, A.D.; Michaud, C.; Ezzati, M.; Shibuya, K.; Salomon, J.A.; Abdalla, S.; et al. Disability-adjusted life years (DALYs) for 291 diseases and injuries in 21 regions, 1990–2010: A systematic analysis for the Global Burden of Disease Study 2010. Lancet 2012, 380, 2197–2223. [Google Scholar] [CrossRef]
- Vic-Dupont, B.C.; Soubrane, J.; Halle, B.; Richir, C. Bilharziose à Schistosoma japonicum à forme hépato-spléenique révélée par une grande hématemese. Bull. Mem. Soc. Med. Hop. Paris 1957, 73, 933–941. [Google Scholar]
- Schneider, C.R.; Kitikoon, V.; Sornmani, S.; Thirachantra, S. Mekong schistosomiasis. III: A parasitological survey of domestic water buffalo (Bubalus bubalis) on Khong Island, Laos. Ann. Trop. Med. Parasitol. 1975, 69, 227–232. [Google Scholar] [CrossRef] [PubMed]
- Davis, G.M.; Kitikoon, V.; Temcharoen, P. Monograph on “Lithoglyphopsis” aperta, the snail host of Mekong River schistosomiasis. Malacologia 1976, 15, 241–287. [Google Scholar] [PubMed]
- Kitikoon, V.; Schneider, C.R. Notes on the aquatic ecology of Lithoglyphopsis aperta. Southeast Asian J. Trop. Med. Public Health 1976, 7, 238–243. [Google Scholar] [PubMed]
- Sornmani, S.; Kitikoon, V.; Schneider, C.R.; Harinasuta, C.; Pathammavong, O. Mekong schistosomiasis. 1. Life cycle of Schistosoma japonicum, Mekong strain in the laboratory. Southeast Asian J. Trop. Med. Public Health 1973, 4, 218–225. [Google Scholar] [PubMed]
- Voge, M.; Price, Z.; Bruckner, D.A. Changes in tegumental surface of Schistosoma mekongi Voge, Bruckner, and Bruce 1978, in the mammalian host. J. Parasitol. 1978, 64, 944–947. [Google Scholar] [CrossRef] [PubMed]
- Attwood, S.W.; Fatih, F.A.; Campbell, I.; Upatham, E.S. The distribution of Mekong schistosomiasis, past and future: Preliminary indications from an analysis of genetic variation in the intermediate host. Parasitol. Int. 2008, 57, 256–270. [Google Scholar] [CrossRef] [PubMed]
- Ohmae, H.; Sinuon, M.; Kirinoki, M.; Matsumoto, J.; Chigusa, Y.; Socheat, D.; Matsuda, H. Schistosomiasis mekongi: From discovery to control. Parasitol. Int. 2004, 53, 135–142. [Google Scholar] [CrossRef] [PubMed]
- Sinuon, M.; Tsuyuoka, R.; Socheat, D.; Odermatt, P.; Ohmae, H.; Matsuda, H.; Montresor, A.; Palmer, K. Control of Schistosoma mekongi in Cambodia: Results of eight years of control activities in the two endemic provinces. Trans. R. Soc. Trop. Med. Hyg. 2007, 101, 34–39. [Google Scholar] [CrossRef] [PubMed]
- Biays, S.; Stich, A.H.; Odermatt, P.; Long, C.; Yersin, C.; Men, C.; Saem, C.; Lormand, J.D. A foci of Schistosomiasis mekongi rediscovered in Northeast Cambodia: Cultural perception of the illness; description and clinical observation of 20 severe cases. Trop. Med. Int. Health 1999, 4, 662–673. [Google Scholar] [CrossRef] [PubMed]
- Stich, A.H.; Biays, S.; Odermatt, P.; Men, C.; Saem, C.; Sokha, K.; Ly, C.S.; Legros, P.; Philips, M.; Lormand, J.D.; et al. Foci of Schistosomiasis mekongi, Northern Cambodia: II. Distribution of infection and morbidity. Trop. Med. Int. Health 1999, 4, 674–685. [Google Scholar] [CrossRef] [PubMed]
- Barbier, M. Determination of aocus of arteriovenous bilharziosiin southern Laos (Sithadone Province). Bull. Soc. Pathol. Exot. Filiales 1966, 59, 4–83. [Google Scholar] [PubMed]
- Iijima, T.; Garcia, R.G. Rapport D’affectation (Enquête Préliminaire sur la Bilharziose au Laos Méridional); OMS, Bureau Rég. Pacif. Occid. Doc. WPRO-80, 23 Mai 1967 (11 Pages, 3 Cartes); WPRO: Manila, Philippines, 1967. [Google Scholar]
- Urbani, C.; Sinoun, M.; Socheat, D.; Pholsena, K.; Strandgaard, H.; Odermatt, P.; Hatz, C. Epidemiology and control of mekongi schistosomiasis. Acta Trop. 2002, 82, 157–168. [Google Scholar] [CrossRef]
- Audebaud, G.; Tournier-Lasserve, C.; Brumpt, V.; Jolly, M.; Mazaud, R.; Imbert, X.; Bazillio, R. 1st case of human schistosomiasis observed in Cambodia (Kratié area). Bull. Soc. Pathol. Exot. Filiales 1968, 61, 778–784. [Google Scholar] [PubMed]
- Tournier-Lasserve, C.; Audebaud, G.; Brumpt, V.; Jolly, M.; Calvez, F.; Mazaud, R.; Imbert, X.; Bazillio, R. Existence of a focus of human bilharziosis, in Cambodia in the Kratié area. I. Study of the 1st three clinical cases. Med. Trop. (Mars) 1970, 30, 451–461. [Google Scholar] [PubMed]
- Jolly, M.; Bazillio, R.; Audebaud, G.; Brumpt, V.; Sophinn, B. Existence of a focus of human bilharziosis, in Cambodia in Kratié area. II. Epidemiologic survey. Preliminary results. Med. Trop. (Mars) 1970, 30, 462–471. [Google Scholar] [PubMed]
- Jolly, M.; Bazillio, R.; Audebaud, G.; Brumpt, V.; Sophinn, B. First epidemiologic studies on a focus of human biharziasis in Cambodia, in the region of Kratié. Bull. Soc. Pathol. Exot. Filiales 1970, 63, 476–483. [Google Scholar] [PubMed]
- Sornmani, S.; Vivatanasesth, P.; Thirachantra, S. Clinical study of Mekong schistosomiasis at Khong Island, Southern Laos. Southeast Asian J. Trop. Med. Public Health 1976, 7, 270–281. [Google Scholar] [PubMed]
- Sornmani, S. Current status of schistosomiasis in Laos, Thailand and Malaysia. Southeast Asian J. Trop. Med. Public Health 1976, 7, 149–154. [Google Scholar] [PubMed]
- Hatz, C.; Odermatt, P.; Urbani, C. Preliminary Data on Morbidity Due to Schistosomiasis mekongi Infections among the Population of Sdau Village, Northestern Cambodia; Médecins Sans Frontières Suisse: Phnom Penh, Cambodia, 1997. [Google Scholar]
- MOH. National Policy and Strategy for Parasite Control 2009, Ministry of Health, Lao People Democratic Republic, Vientiane Lao PDR; MOH: Long Beach, CA, USA, 2009.
- Sayasone, S.; Mak, T.K.; Vanmany, M.; Rasphone, O.; Vounatsou, P.; Utzinger, J.; Akkhavong, K.; Odermatt, P. Helminth and intestinal protozoa infections, multiparasitism and risk factors in Champasack province, Lao People’s Democratic Republic. PLoS Negl. Trop. Dis. 2011, 5, e1037. [Google Scholar] [CrossRef] [PubMed]
- WHO. Report on Mass Drug Administration to Control of Schistosomiasis in Lao People’s Democratic Republic 2018. Available online: https://www.who.int/neglected_diseases/preventive_chemotherapy/sch/en/ (accessed on 19 January 2019).
- WHO. Report on Schistosoma mekongi Paratological Surveillance from Village Sentinel Sites (2016–2018); WHO: Geneva, Switzerland, 2018.
- Strandgaard, H.; Johansen, M.V.; Pholsena, K.; Teixayavong, K.; Christensen, N.O. The pig as a host for Schistosoma mekongi in Laos. J. Parasitol. 2001, 87, 708–709. [Google Scholar] [CrossRef]
- Matsuda, H.; Tanaka, H.; Blas, B.L.; Nosenas, J.S.; Tokawa, T.; Ohsawa, S. Evaluation of ELISA with ABTS, 2-2′-azino-di-(3-ethylbenzthiazoline sulfonic acid), as the substrate of peroxidase and its application to the diagnosis of schistosomiasis. Jpn. J. Exp. Med. 1984, 54, 131–138. [Google Scholar] [PubMed]
- Kirinoki, M.; Chigusa, Y.; Ohmae, H.; Sinuon, M.; Socheat, D.; Matsumoto, J.; Kitikoon, V.; Matsuda, H. Efficacy of sodium metaperiodate (SMP)-ELISA for the serodiagnosis of schistosomiasis mekongi. Southeast Asian J. Trop. Med. Public Health 2011, 42, 25–33. [Google Scholar] [PubMed]
- Hatz, C.F. The use of ultrasound in schistosomiasis. Adv. Parasitol. 2001, 48, 225–284. [Google Scholar] [PubMed]
- Keang, H.; Odermatt, P.; Odermatt-Biays, S.; Cheam, S.; Degremont, A.; Hatz, C. Liver morbidity due to Schistosoma mekongi in Cambodia after seven rounds of mass drug administration. Trans. R. Soc. Trop. Med. Hyg. 2007, 101, 759–765. [Google Scholar] [CrossRef] [PubMed]
- Katz, N.; Chaves, A.; Pellegrino, J. A simple device for quantitative stool thick-smear technique in schistosomiasis mansoni. Rev. Inst. Med. Trop. São Paulo 1972, 14, 397–400. [Google Scholar] [PubMed]
- Chigusa, Y.; Otake, H.; Kirinoki, M.; Ohmae, H.; Socheat, D.; Sinuon, M. Splenomegaly of Schistosoma mekongi infection in Kratie province Cambodia. Clin. Parasitol. 2001, 12, 63–65. [Google Scholar]
- CNM. National Policy and Guideline for Helminth Control in Cambodia by the National Task Force for the Control of Soil-Transmitted Helminthiasis, Schistosomiasis, and for the Elimination of Lymphatic Filariasis; CNM: Albuquerque, NM, USA, 2004. [Google Scholar]
- Matsumoto, J.; Muth, S.; Socheat, D.; Matsuda, H. The first reported cases of canine schistosomiasis mekongi in Cambodia. Southeast Asian, J. Trop. Med. Public Health 2002, 33, 458–461. [Google Scholar]
- Hisakane, N.; Kirinoki, M.; Chigusa, Y.; Sinuon, M.; Socheat, D.; Matsuda, H.; Ishikawa, H. The evaluation of control measures against Schistosoma mekongi in Cambodia by a mathematical model. Parasitol. Int. 2008, 57, 379–385. [Google Scholar] [CrossRef] [PubMed]
- WHO. Report on National Meeting on Neglected Tropical Disease Control Program; WHO: Geneva, Switzerland, 2018.
- WHO. Action Plan for Schistosomiasis Elimination 2016–2020; WHO: Geneva, Switzerland, 2016.
- WHO. Water Safety Planning Will Contribute to the Elimination of Schistosomiasis Worms and Improve Nutrition via a Community-Led Approach in the Province of Champassak. 2016. Available online: http://www.wpro.who.int/laos/mediacentre/releases/2016/20160524-lao-cl-swash-water-safety/en/ (accessed on 19 January 2019).
- WHO. Expert Consultation to Accelerate Elimination of Asian Schistosomiasis, Shanghai, China, 22–23 May 2017, Meeting Report; 2017 Report Series; RS/2017/GE/36(CHN); WHO: Geneva, Switzerland, 2017.
- WHO. Report on National Policy and Strategies on Neglected Tropical Diseases Prevention and Control, May 2018; WHO: Geneva, Switzerland, 2018.
- Elkins, D.B.; Sithithaworn, P.; Haswell-Elkins, M.; Kaewkes, S.; Awacharagan, P.; Wongratanacheewin, S. Opisthorchis viverrini: Relationships between egg counts, worms recovered and antibody levels within an endemic community in northeast Thailand. Parasitology 1991, 102, 283–288. [Google Scholar] [CrossRef] [PubMed]
- Kirinoki, M.; Ohmae, H.; Chigusa, Y.; Muth, S.; Khieu, V. Evaluation of the Status of Schistosomiasis Elimination as a Public Health Problem in Cambodia and Development of Monitoring Protocol and Indicators for Interruption of Transmission of Mekong Schistosomiasis; Technical Report to WPRO; WHO: Geneva, Switzerland, 2016.
- Bergquist, R.; Johansen, M.V.; Utzinger, J. Diagnostic dilemmas in helminthology: What tools to use and when? Trends Parasitol. 2009, 25, 151–156. [Google Scholar] [CrossRef] [PubMed]
- Lodh, N.; Mikita, K.; Bosompem, K.M.; Anyan, W.K.; Quartey, J.K.; Otchere, J.; Shiff, C.J. Point of care diagnosis of multiple schistosome parasites: Species-specific DNA detection in urine by loop-mediated isothermal amplification (LAMP). Acta Trop. 2017, 173, 125–129. [Google Scholar] [CrossRef] [PubMed]
- He, P.; Gordon, C.A.; Williams, G.M.; Li, Y.; Wang, Y.; Hu, J. Real-time PCR diagnosis of Schistosoma japonicum in low transmission areas of China. Infect. Dis. Poverty 2018, 7, 8. [Google Scholar] [CrossRef] [PubMed]
- Hamburger, J.; Abbasi, I.; Kariuki, C.; Wanjala, A.; Mzungu, E.; Mungai, P. Evaluation of loop-mediated isothermal amplification suitable for molecular monitoring of schistosome-infected snails in field laboratories. Am. J. Trop. Med. Hyg. 2013, 88, 344–351. [Google Scholar] [CrossRef] [PubMed]
- Qin, Z.Q.; Xu, J.; Feng, T.; Lv, S.; Qian, Y.J.; Zhang, L.J.; Li, Y.; Lv, C. Field Evaluation of a Loop-Mediated Isothermal Amplification (LAMP) Platform for the Detection of Schistosoma japonicum Infection in Oncomelania hupensis Snails. Trop. Med. Infect. Dis. 2018, 3, 124. [Google Scholar] [CrossRef] [PubMed]
- Bergquist, R.; van Dam, G.J.; Xu, J. Diagnostic tests for schistosomiasis. In Schistosoma: Biology, Pathology and Control; Jamieson, B.G.M., Ed.; CRC Press: Boca Raton, FL, USA, 2016; pp. 401–439. [Google Scholar]
- Colley, D.G.; Binder, S.; Campbell, C.; King, C.H.; Tchuem Tchuente, L.A.; N’Goran, E.K.; Erko, B.; Karanja, D.M.; Kabatereine, N.B.; van Lieshout, L.; et al. A five-country evaluation of a point-of-care circulating cathodic antigen urine assay for the prevalence of Schistosoma mansoni. Am. J. Trop. Med. Hyg. 2013, 88, 426–432. [Google Scholar] [CrossRef] [PubMed]
- Kittur, N.; Castleman, J.D.; Campbell, C.H., Jr.; King, C.H.; Colley, D.G. Comparison of Schistosoma mansoni Prevalence and Intensity of Infection, as Determined by the Circulating Cathodic Antigen Urine Assay or by the Kato-Katz Fecal Assay: A Systematic Review. Am. J. Trop. Med. Hyg. 2016, 94, 605–610. [Google Scholar] [CrossRef] [PubMed]
- Vonghachack, Y.; Sayasone, S.; Khieu, V.; Bergquist, R.; van Dam, G.J.; Hoekstra, P.T.; Corstjens, P.L.A.M.; Nickel, B.; Marti, H.; Utzinger, J.; et al. Comparison of novel and standard diagnostic tools for the detection of Schistosoma mekongi infection in Lao People’s Democratic Republic and Cambodia. Infect. Dis. Poverty 2017, 6, 127. [Google Scholar] [CrossRef] [PubMed]
- Malone, J.; Bergquist, R.; Rinaldi, L. Geospatial Surveillance and Repsonse Systems for Schistosomiasis. In Schistosoma: Biology, Pathology and Control; Jamieson, B.G.M., Ed.; CRC Press: Boca Raton, FL, USA, 2016; pp. 479–497. [Google Scholar]
- Malone, J.B.; Bergquist, R.; Luvall, J.C. Use of Geospatial Surveillance and Response Systems for Vector-borne Diseases in the Elimination Phase. Trop. Med. Infect. Dis. 2019, 18, 4. [Google Scholar] [CrossRef] [PubMed]
- Bergquist, R.; Yang, G.J.; Knopp, S.; Utzinger, J.; Tanner, M. Surveillance and response: Tools and approaches for the elimination stage of neglected tropical diseases. Acta Trop. 2015, 141 Pt B, 229–234. [Google Scholar] [CrossRef]
- Attwood, S.W.; Upatham, E.S. Observations on Neotricula aperta (Gastropoda: Pomatiopsidae) population densities in Thailand and central Laos: Implications for the spread of Mekong schistosomiasis. Parasit. Vectors 2012, 5, 126. [Google Scholar] [CrossRef] [PubMed]
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Khieu, V.; Sayasone, S.; Muth, S.; Kirinoki, M.; Laymanivong, S.; Ohmae, H.; Huy, R.; Chanthapaseuth, T.; Yajima, A.; Phetsouvanh, R.; et al. Elimination of Schistosomiasis Mekongi from Endemic Areas in Cambodia and the Lao People’s Democratic Republic: Current Status and Plans. Trop. Med. Infect. Dis. 2019, 4, 30. https://doi.org/10.3390/tropicalmed4010030
Khieu V, Sayasone S, Muth S, Kirinoki M, Laymanivong S, Ohmae H, Huy R, Chanthapaseuth T, Yajima A, Phetsouvanh R, et al. Elimination of Schistosomiasis Mekongi from Endemic Areas in Cambodia and the Lao People’s Democratic Republic: Current Status and Plans. Tropical Medicine and Infectious Disease. 2019; 4(1):30. https://doi.org/10.3390/tropicalmed4010030
Chicago/Turabian StyleKhieu, Virak, Somphou Sayasone, Sinuon Muth, Masashi Kirinoki, Sakhone Laymanivong, Hiroshi Ohmae, Rekol Huy, Thipphavanh Chanthapaseuth, Aya Yajima, Rattanaxay Phetsouvanh, and et al. 2019. "Elimination of Schistosomiasis Mekongi from Endemic Areas in Cambodia and the Lao People’s Democratic Republic: Current Status and Plans" Tropical Medicine and Infectious Disease 4, no. 1: 30. https://doi.org/10.3390/tropicalmed4010030
APA StyleKhieu, V., Sayasone, S., Muth, S., Kirinoki, M., Laymanivong, S., Ohmae, H., Huy, R., Chanthapaseuth, T., Yajima, A., Phetsouvanh, R., Bergquist, R., & Odermatt, P. (2019). Elimination of Schistosomiasis Mekongi from Endemic Areas in Cambodia and the Lao People’s Democratic Republic: Current Status and Plans. Tropical Medicine and Infectious Disease, 4(1), 30. https://doi.org/10.3390/tropicalmed4010030



