Melioidosis in South Asia (India, Nepal, Pakistan, Bhutan and Afghanistan)
Abstract
:1. Introduction
2. History
3. Review of Melioidosis Cases and Presence of B. pseudomallei (Animal/Human/Environment)
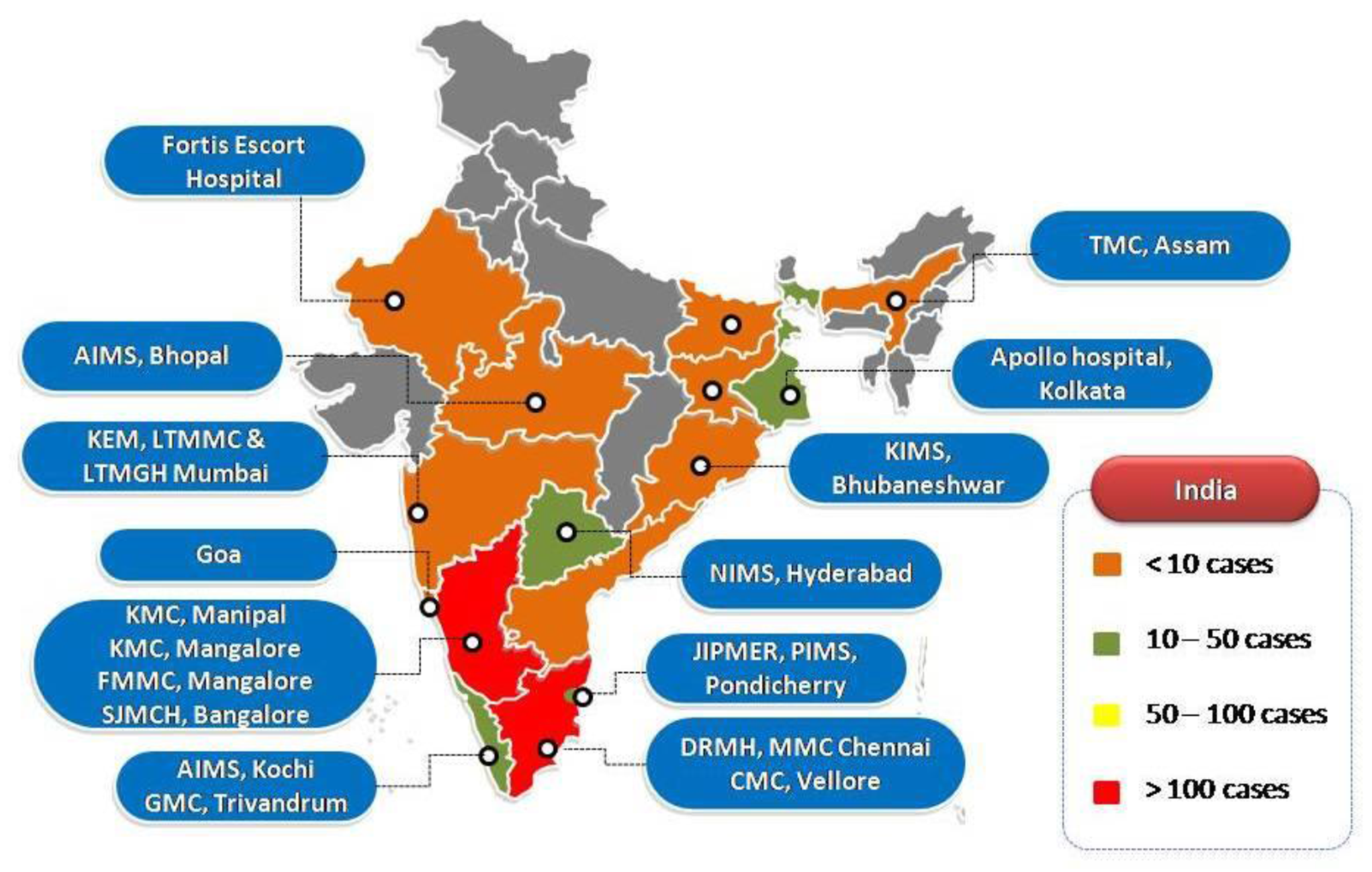
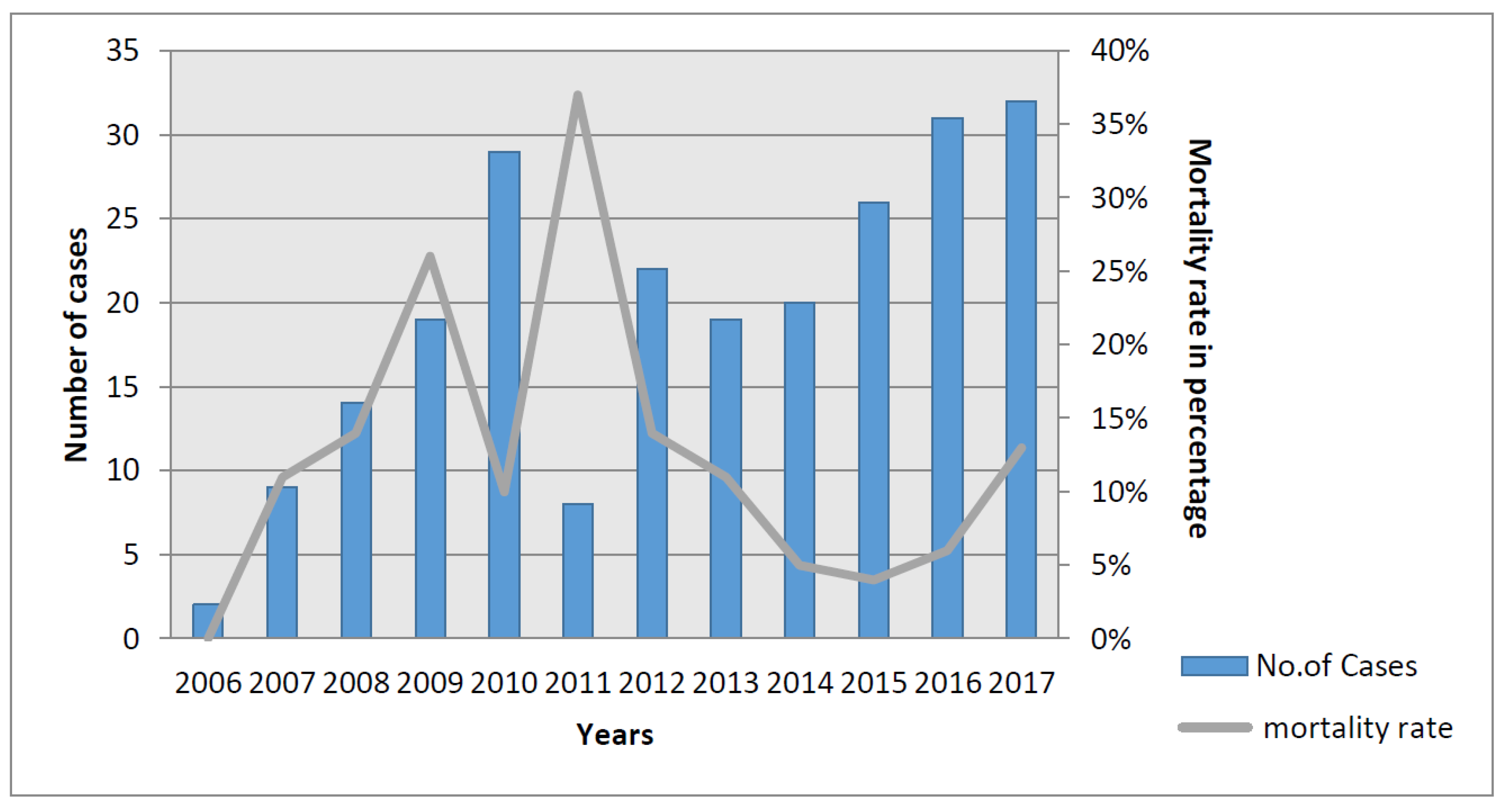
3.1. India
3.2. Manipal
3.3. Vellore
3.4. Other Neighboring Countries
3.5. Serosurveillance
3.6. Animals
3.7. Environmental Evidence
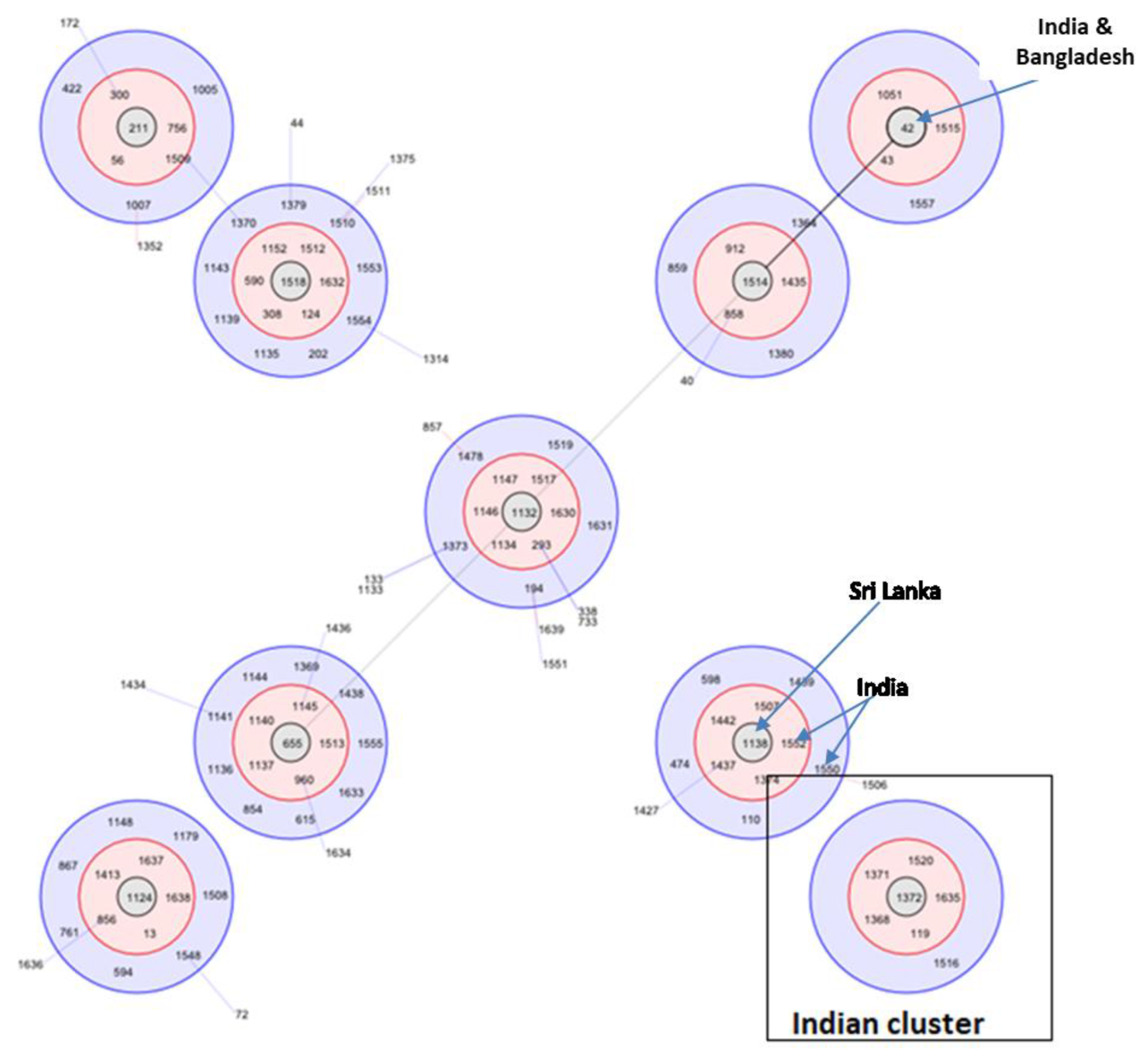
3.8. Molecular Epidemiology in South Asian Countries
4. Current Recommendations and Availability of Measures against Melioidosis
Surveillance Systems and Reporting
5. Diagnostic Facilities
6. Antibiotic Susceptibility and Treatment
7. Awareness of Melioidosis
8. Establishing a Melioidosis Network
9. Current and Future Challenges
- Collecting and collating better data on the number of cases of melioidosis occurring will require extensive communication with laboratories that are capable of identifying the organism.
- Training more physicians and laboratory staff to recognize the clinical features of the disease and the characteristics of its causative organism (for example, through the use of the three-antibiotic disc test mentioned above) will require concerted efforts through universities and colleges and professional associations but will be difficult because of the fragmented nature of healthcare in the region.
- Raising the profile of the disease with both policymakers and the general public at both local and national levels will inevitably have to be started locally, but, as the network of those with an interest grows, it should become easier to provide the necessary evidence that the morbidity and mortality of the disease warrant greater attention. One key objective would be to have melioidosis included in the list of diseases that are statutorily reportable by laboratories, which in turn would improve knowledge of the disease distribution and burden.
10. Conclusions
Supplementary Materials
Author Contributions
Conflicts of Interest
References
- Cheng, A.C.; Currie, B.J. Melioidosis: Epidemiology, pathophysiology, and management. Clin. Microbiol. Rev. 2005, 18, 383–416. [Google Scholar] [CrossRef] [PubMed]
- Limmathurotsakul, D.; Golding, N.; Dance, D.A.; Messina, J.P.; Pigott, D.M.; Moyes, C.L.; Rolim, D.B.; Bertherat, E.; Day, N.P.J.; Peacock, S.J.; et al. Predicted global distribution of Burkholderia pseudomallei and burden of melioidosis. Nat. Microbiol. 2016, 1, 15008. [Google Scholar] [CrossRef] [PubMed]
- Vidyalakshmi, K.; Chakrapani, M.; Shrikala, B.; Damodar, S.; Lipika, S.; Vishal, S. Tuberculosis mimicked by melioidosis. Int. J. Tuberc. Lung Dis. 2008, 12, 1209–1215. [Google Scholar] [PubMed]
- Lowe, P.; Engler, C.; Norton, R. Comparison of automated and nonautomated systems for identification of Burkholderia pseudomallei. J. Clin. Microbiol. 2002, 40, 4625–4627. [Google Scholar] [CrossRef] [PubMed]
- Ives, J.; Thomson, T. Chronic melioidosis: The first report of a case infected in central India. Glasgow Med. J. 1953, 34, 61–67. [Google Scholar] [PubMed]
- Thurnheer, U.; Novak, A.; Michel, M.; Ruchti, C.; Jutzi, H.; Weiss, M. Septic melioidosis following a visit to India. Schweizerische Medizinische Wochenschrift 1988, 118, 558–564. [Google Scholar] [PubMed]
- Raghavan, K.; Shenoi, R.; Zaer, F.; Aiyer, R.; Ramamoorthy, P.; Mehta, M. Melioidosis in India. Indian Pediatr. 1991, 28, 184–188. [Google Scholar] [PubMed]
- Bharadwaj, R.; Kagal, A.; Deshpandey, S.; Joshi, S.; Khare, P.; Junnarkar, A.; Phadke, M. Outbreak of plague-like illness caused by Pseudomonas pseudomallei in Maharashtra, India. Lancet 1994, 344, 1574. [Google Scholar] [CrossRef]
- Dance, D.A.B.; Sanders, D.; Pitt, T.L.; Speller, D.C. Burkholderia pseudomallei and Indian plague-like illness. Lancet 1995, 346, 904–905. [Google Scholar] [CrossRef]
- John, T.J.; Jesudason, M.V.; Lalitha, M.K.; Ganesh, A.; Mohandas, V.; Cherian, T.; Mathai, M.; Chandy, M.J. Melioidosis in India: The tip of the iceberg? Indian J. Med. Res. 1996, 103, 62–65. [Google Scholar] [PubMed]
- Cherian, T.; John, T.J.; Ramakrishna, B.; Lalitha, M.K.; Raghupathy, P. Disseminated melioidosis. Indian Pediatr. 1996, 33, 403–406. [Google Scholar] [PubMed]
- Murali, R.; Ganesh, A.; Jesudason, M.V.; Mathew, R.K.; Date, A. Surat ’94: Was it melioidosis? Interesting observation from the first case of imported melioidosis in India. J. Assoc. Phys. India 1996, 44, 218–219. [Google Scholar]
- Jesudason, M.V.; Shanthakumari, R.; John, T.J. Burkholderia pseudomallei—An emerging pathogen in India. Indian J. Med. Microbiol. 1997, 15, 1–2. [Google Scholar]
- Jesudasom, M.V.; Anbarasu, A.; John, T.J. Septicaemic melioidosis in a tertiary care hospital in South India. Indian J. Med. Res. 2003, 117, 119–121. [Google Scholar]
- Kang, G.; Rajan, D.P.; Ramakrishna, B.; Aucken, H.; Dance, D. Melioidosis in India. Lancet 1996, 347, 1565–1566. [Google Scholar] [CrossRef]
- Rao, P.S.; Dhawan, R.; Shivananda, P. Burkholderia pseudomallei infections. Trop. Dr. 2002, 32, 174–175. [Google Scholar] [CrossRef] [PubMed]
- Hinjoy, S.; Hantrakun, V.; Kongyu, S.; Kaewrakmuk, J.; Wangrangsimakul, T.; Jitsuronk, S.; Saengchun, W.; Bhengsri, S.; Akarachotpong, T.; Thamthitiwat, S.; et al. Melioidosis in Thailand: Present and future. Trop. Med. Infect. Dis. 2018, 3, 38. [Google Scholar] [CrossRef] [PubMed]
- Currie, B.J.; Fisher, D.A.; Howard, D.M.; Burrow, J.N.; Lo, D.; Selva-Nayagam, S.; Anstey, N.M.; Huffam, S.E.; Snelling, P.L.; Marks, P.J.; et al. Endemic melioidosis in tropical northern Australia: A 10-year prospective study and review of the literature. Clin. Infect. Dis. 2000, 31, 981–986. [Google Scholar] [CrossRef] [PubMed]
- Koshy, M.; Jagannati, M.; Ralph, R.; Punitha, J.V.; David, T.S.; Sathyendra, S.; Veeraraghavan, B.; Varghese, G.M. Clinical manifestations, susceptibility patterns and predictors of poor outcome of melioidosis in India. Emerg. Infect. Dis. 2018. under review. [Google Scholar]
- Shrestha, N.K.; Sharma, S.K.; Khanal, B.; Bhatta, N.; Dhakal, S.S. Melioidosis imported into Nepal. Scand. J. Infect. Dis. 2005, 37, 64–66. [Google Scholar] [CrossRef] [PubMed]
- Stokes, K.J.; McCarthy, S. Pseudomonas pseudomallei: Isolation from a chronic sternal abscess. J. Med. Lab. Technol. 1969, 26, 199–201. [Google Scholar] [PubMed]
- Sheppard, M.J.; Marriott, R.M.; Brown, T.J. Long latency pneumonia: A case of melioidosis. J. Infect. 1990, 20, 83–84. [Google Scholar] [CrossRef]
- Vandana, K.E.; Mukhopadhyay, C.; Tellapragada, C.; Kamath, A.; Tipre, M.; Bhat, V.; Sathiakumar, N. Seroprevalence of Burkholderia pseudomallei among adults in coastal areas in southwestern India. PLoS Negl. Trop. Dis. 2016, 10, e0004610. [Google Scholar] [CrossRef] [PubMed]
- Peacock, S.J.; Cheng, A.C.; Currie, B.J.; Dance, D.A. The use of positive serological tests as evidence of exposure to Burkholderia pseudomallei. Am. J. Trop. Med. Hyg. 2011, 84, 1021–1022. [Google Scholar] [CrossRef] [PubMed]
- Moore, T.D.; Allen, A.M.; Palmer, A.E. Melioidosis—Maryland. Morb. Mortal. Wkly. Rep. 1969, 18, 278–279. [Google Scholar]
- Das, A.M.; Paranjape, V.L. Pseudomonas pseudomallei associated with bovine abortion in India. Vet. Med. Rev. 1986, 2, 173–176. [Google Scholar]
- Prakash, A.; Thavaselvam, D.; Kumar, A.; Kumar, A.; Arora, S.; Tiwari, S.; Barua, A.; Sathyaseelan, K. Isolation, identification and characterization of Burkholderia pseudomallei from soil of coastal region of India. Springer Plus 2014, 3, 438. [Google Scholar] [CrossRef] [PubMed]
- Peddayelachagiri, B.V.; Paul, S.; Nagaraj, S.; Gogoi, M.; Sripathy, M.H.; Batra, H.V. Prevalence and identification of Burkholderia pseudomallei and near-neighbor species in the Malabar coastal region of India. PLoS Negl. Trop. Dis. 2016, 10, e0004956. [Google Scholar] [CrossRef] [PubMed]
- Shabbir, M.Z.; Jamil, T.; Ali, A.A.; Ahmad, A.; Naeem, M.; Chaudhary, M.H.; Bilal, M.; Ali, M.A.; Muhammad, K.; Yaqub, T.; et al. Prevalence and distribution of soil-borne zoonotic pathogens in Lahore district of Pakistan. Front. Microbiol. 2015, 6, 917. [Google Scholar] [CrossRef] [PubMed]
- Mukhopadhyay, C.; Kaestli, M.; Vandana, K.E.; Sushma, K.; Mayo, M.; Richardson, L.; Tuanyok, A.; Keim, P.; Godoy, D.; Spratt, B.G.; et al. Molecular characterization of clinical Burkholderia pseudomallei isolates from India. Am. J. Trop. Med. Hyg. 2011, 85, 121–123. [Google Scholar] [CrossRef] [PubMed]
- Tellapragada, C.; Kamthan, A.; Shaw, T.; Vandana, K.; Kumar, S.; Bhat, V.; Mukhopadhyay, C. Unravelling the molecular epidemiology and genetic diversity among Burkholderia pseudomallei isolates from South India using multi-locus sequence typing. PLoS ONE 2016, 11, e0168331. [Google Scholar] [CrossRef] [PubMed]
- De Smet, B.; Sarovich, D.S.; Price, E.P.; Mayo, M.; Theobald, V.; Kham, C.; Heng, S.; Thong, P.; Holden, M.T.; Parkhill, J.; et al. Whole-genome sequencing confirms that Burkholderia pseudomallei multilocus sequence types common to both Cambodia and Australia are due to homoplasy. J. Clin. Microbiol. 2015, 53, 323–326. [Google Scholar] [CrossRef] [PubMed]
- Limmathurotsakul, D.; Kanoksil, M.; Wuthiekanun, V.; Kitphati, R.; Day, N.P.; Peacock, S.J. Activities of daily living associated with acquisition of melioidosis in northeast Thailand: A matched case-control study. PLoS Negl. Trop. Dis. 2013, 7, e2072. [Google Scholar] [CrossRef] [PubMed]
- Suntornsut, P.; Wongsuwan, N.; Malasit, M.; Kitphati, R.; Michie, S.; Peacock, S.J.; Limmathurotsakul, D. Barriers and recommended interventions to prevent melioidosis in northeast Thailand: A focus group study using the behaviour change wheel. PLoS Negl. Trop. Dis. 2016, 10, e0004823. [Google Scholar] [CrossRef] [PubMed]
- Tellapragada, C.; Shaw, T.; D’Souza, A.; Eshwara, V.K.; Mukhopadhyay, C. Improved detection of Burkholderia pseudomallei from non-blood clinical specimens using enrichment culture and PCR: Narrowing diagnostic gap in resource constrained settings. Trop. Med. Int. Health 2017. [Google Scholar] [CrossRef] [PubMed]
- Tiangpitayakorn, C.; Songsivilai, S.; Piyasangthong, N.; Dharakul, T. Speed of detection of Burkholderia pseudomallei in blood cultures and its correlation with the clinical outcome. Am. J. Trop. Med. Hyg. 1997, 57, 96–99. [Google Scholar] [CrossRef] [PubMed]
- Mørch, K.; Manoharan, A.; Chandy, S.; Chacko, N.; Alvarez-Uria, G.; Patil, S.; Henry, A.; Nesaraj, J.; Kuriakose, C.; Singh, A.; et al. Acute undifferentiated fever in India: A multicentre study of aetiology and diagnostic accuracy. BMC Infect. Dis. 2017, 17, 665. [Google Scholar] [CrossRef] [PubMed]
- Khan, I.D.; Sahni, A.K.; Bharadwaj, R.; Lall, M.; Jindal, A.K.; Sashindran, V.K. Emerging organisms in a tertiary healthcare set up. Med. J. Armed Forces India 2014, 70, 120–128. [Google Scholar] [CrossRef] [PubMed]
- Gandra, S.; Mojica, N.; Klein, E.Y.; Ashok, A.; Nerurkar, V.; Kumari, M.; Ramesh, U.; Dey, S.; Vadwai, V.; Das, B.R.; et al. Trends in antibiotic resistance among major bacterial pathogens isolated from blood cultures tested at a large private laboratory network in India, 2008–2014. Int. J. Infect. Dis. 2016, 50, 75–82. [Google Scholar] [CrossRef] [PubMed]
- Trinh, T.T.; Hoang, T.S.; Tran, D.A.; Trinh, V.T.; Gohler, A.; Nguyen, T.T.; Hoang, S.N.; Krumkamp, R.; Nguyen, L.T.N.; May, J.; et al. A simple laboratory algorithm for diagnosis of melioidosis in resource-constrained areas: A study from north-central Vietnam. Clin. Microbiol. Infect. 2018, 24, e1–e4. [Google Scholar] [CrossRef] [PubMed]
- Houghton, R.L.; Reed, D.E.; Hubbard, M.A.; Dillon, M.J.; Chen, H.; Currie, B.J.; Mayo, M.; Sarovich, D.S.; Theobald, V.; Limmathurotsakul, D.; et al. Development of a prototype lateral flow immunoassay (LFI) for the rapid diagnosis of melioidosis. PLoS Negl. Trop. Dis. 2014, 8, e2727. [Google Scholar] [CrossRef] [PubMed]
- Shaw, T.; Tellapragada, C.; Ke, V.; Au Coin, D.P.; Mukhopadhyay, C. Performance evaluation of Active Melioidosis Detect-Lateral Flow Assay (AMD-LFA) for diagnosis of melioidosis in endemic settings with limited resources. PLoS ONE 2018, 13, e0194595. [Google Scholar] [CrossRef] [PubMed]
- Shaw, T.; Tellapragada, C.; Eshwara, V.K.; Bhat, H.V.; Mukhopadhyay, C. The antibiotics of choice for the treatment of melioidosis in Indian set up. Indian J. Med. Microbiol. 2016, 34, 353. [Google Scholar] [PubMed]
- Behera, B.; Prasad Babu, T.L.; Kamalesh, A.; Reddy, G. Ceftazidime resistance in Burkholderia pseudomallei: First report from India. Asian Pac. J. Trop. Med. 2012, 5, 329–330. [Google Scholar] [CrossRef]
- Pillai, M.P.; Faizal, B.P.; Urs, V.D. Emerging drug resistance in melioidosis. Ann. Trop. Med. Public Health 2014, 7, 263–265. [Google Scholar] [CrossRef]
- Chansrichavala, P.; Wongsuwan, N.; Suddee, S.; Malasit, M.; Hongsuwan, M.; Wannapinij, P.; Kitphati, R.; Day, N.P.J.; Michie, S.; Peacock, S.J.; et al. Public awareness of melioidosis in Thailand and potential use of video clips as educational tools. PLoS ONE 2015, 10, e0121311. [Google Scholar] [CrossRef] [PubMed]
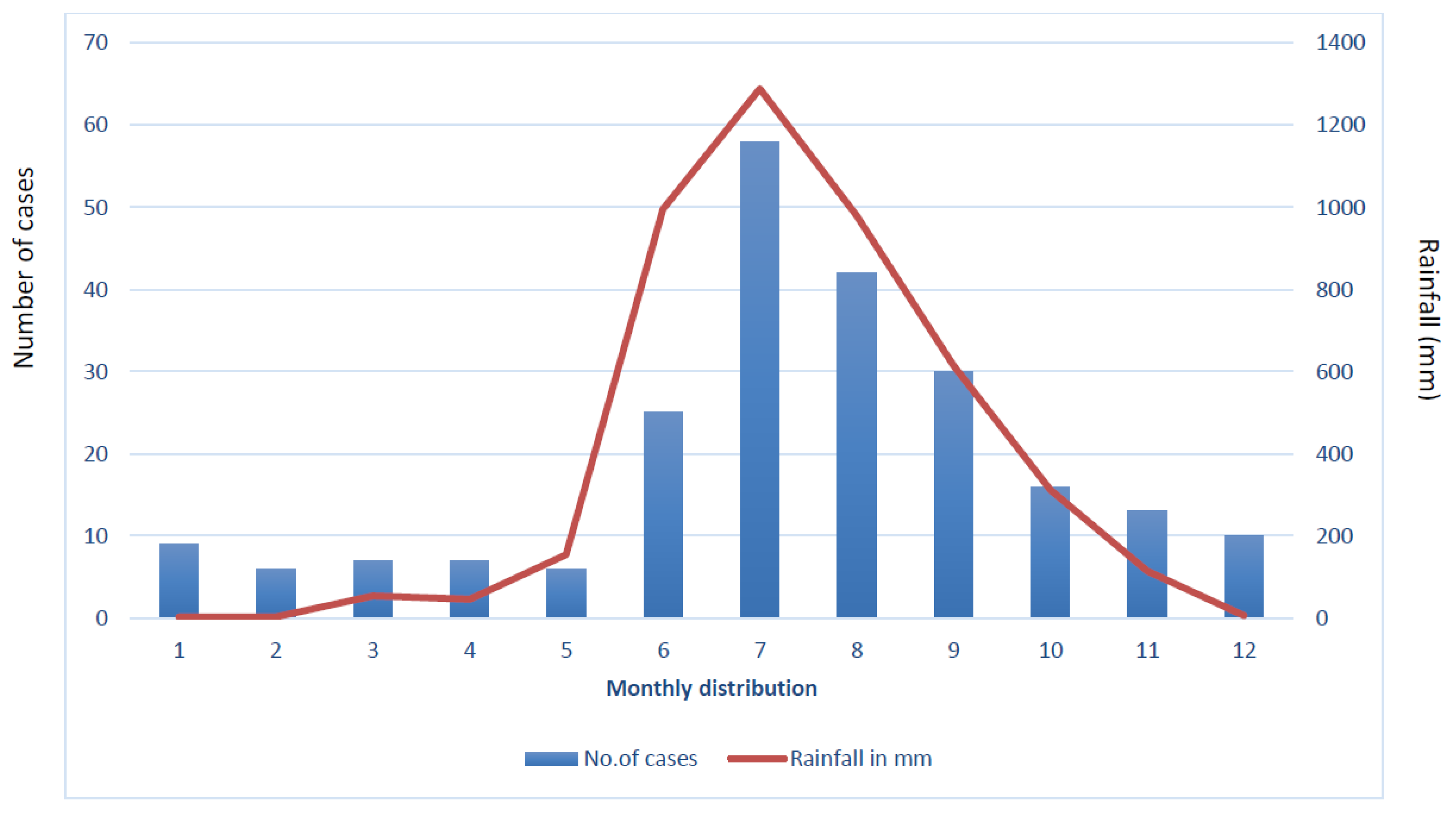
| Month | Season |
|---|---|
| October–February | Post-monsoon |
| March–May | Summer |
| June–September | Monsoon |

| State | Number of Cases | Age Range | Gender (Male: Female) | Diabetes n (%) | Intensive Treatment | Eradication Treatment | Mortality n (%) | ||
|---|---|---|---|---|---|---|---|---|---|
| MER | CAZ | SXT | Others | ||||||
| Karnataka | 306 | 0–84 | 3:1 | 241 (79) | 24 | 294 | 235 | 50 | 39 (13) |
| Tamil Nadu | 146 | 4–65 | 3:1 | 75 (50) | 3 | 82 | 80 | 2 | 37 (28) |
| Telangana | 35 | 30–66 | 2:1 | 27 (77) | 8 | 7 | 11 | 3 | 4 (11) |
| Kerala | 34 | 9–66 | 5:1 | 24 (77) | 3 | 15 | 14 | 2 | 5 (13) |
| Pondicherry | 14 | 0–58 | 3:1 | 3 (21) | 2 | 2 | - | 4 | 2 (14) |
| West Bengal | 11 | 29–71 | 11:0 | 9 (82) | 6 | 4 | 9 | 1 | 2 (18) |
| Maharashtra | 9 | 10–72 | 7:1 | 5 (50) | 0 | 4 | 4 | 0 | 4 (50) |
| Orissa | 8 | 47–51 | 7:0 | 5 (63) | 0 | 6 | 5 | 0 | 1 (12.5) |
| Assam | 6 | 0–57 | 2:1 | 3 (50) | 3 | 0 | 1 | 1 | 2 (33) |
| Goa | 5 | 34–53 | 5:0 | 5 (100) | 2 | 2 | 1 | 1 | 0 |
| Bihar | 4 | 50–65 | 4:0 | 4 (100) | 2 | 1 | 2 | 1 | 0 |
| Jharkhand | 2 | 32–33 | 2:0 | 1 (50) | 1 | 1 | 1 | 0 | 1 (50) |
| Rajasthan | 1 | 49 | 1:0 | 0 | 1 | 0 | 1 | 0 | 0 |
| Madhya Pradesh | 1 | 56 | 1:0 | 1 (100) | 0 | 1 | 0 | 0 | 0 |
| Andhra Pradesh | 1 | 23 | 1:0 | 1 (100) | 0 | 0 | 0 | 0 | 1 (100) |
| Institute (State) | Number of Isolates |
|---|---|
| Amritha Institute of Medical Sciences (Kerala) | 34 |
| Pondicherry Institute of Medical Sciences (Puducherry) | 20 |
| Nizams Institute of Medical Sciences (Hyderabad, Telangana) | 12 |
| Government Medical College (Tiruvanthapuram, Kerala) | 7 |
| Kalinga Institute of Medical Sciences (Orissa) | 4 |
| Government Medical College and Hospital (Mallapuram, Kerala) | 2 |
| Tripura Medical College (Assam) | 1 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mukhopadhyay, C.; Shaw, T.; Varghese, G.M.; Dance, D.A.B. Melioidosis in South Asia (India, Nepal, Pakistan, Bhutan and Afghanistan). Trop. Med. Infect. Dis. 2018, 3, 51. https://doi.org/10.3390/tropicalmed3020051
Mukhopadhyay C, Shaw T, Varghese GM, Dance DAB. Melioidosis in South Asia (India, Nepal, Pakistan, Bhutan and Afghanistan). Tropical Medicine and Infectious Disease. 2018; 3(2):51. https://doi.org/10.3390/tropicalmed3020051
Chicago/Turabian StyleMukhopadhyay, Chiranjay, Tushar Shaw, George M. Varghese, and David A. B. Dance. 2018. "Melioidosis in South Asia (India, Nepal, Pakistan, Bhutan and Afghanistan)" Tropical Medicine and Infectious Disease 3, no. 2: 51. https://doi.org/10.3390/tropicalmed3020051
APA StyleMukhopadhyay, C., Shaw, T., Varghese, G. M., & Dance, D. A. B. (2018). Melioidosis in South Asia (India, Nepal, Pakistan, Bhutan and Afghanistan). Tropical Medicine and Infectious Disease, 3(2), 51. https://doi.org/10.3390/tropicalmed3020051







