Alcohol Consumption of Male Tuberculosis Index Cases and Tuberculosis Transmission Among Social Contacts in Puducherry, India: A Cross-Sectional Analytical Study
Abstract
1. Introduction
2. Materials and Methods
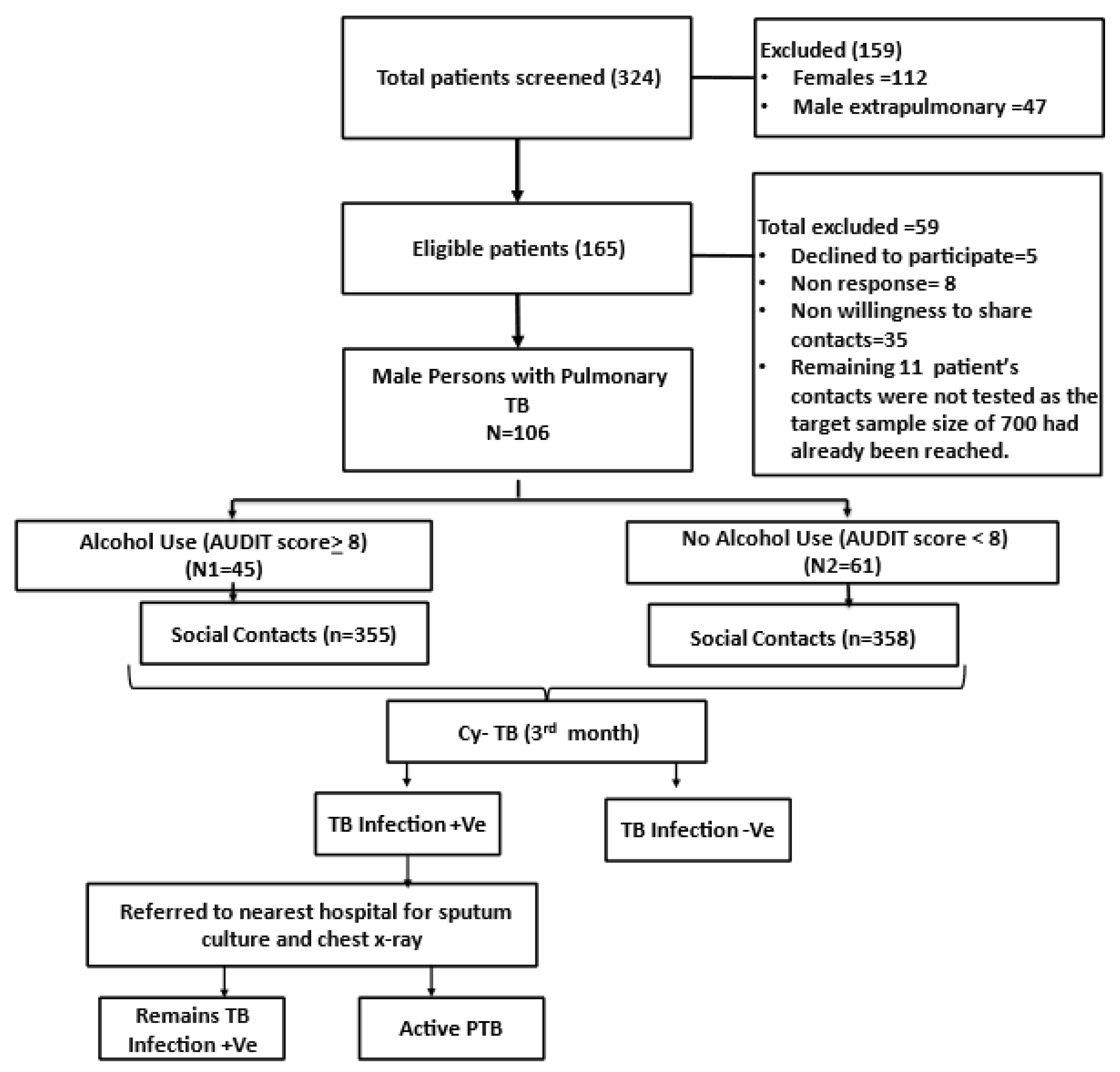
2.1. Study Design, Population and Setting
2.2. Data Collection Tools
2.3. Screening for TBI
2.4. Operational Definition
- Persons with pulmonary tuberculosis (PTB)/Index case (IC): Persons with confirmed tuberculosis by sputum smear microscopy/CBNAAT/Gene-Xpert.
- Alcohol use: Defined as study participants who scored ≥8 when screened using the Alcohol Use Disorder Identification Test (AUDIT).
- Social contact (SC): Non-household contact who shared an enclosed space (e.g., at social gatherings, workplaces, or other facilities) with the index case for at least three days per week, for two to four hours per day, in the three months preceding the index case’s current treatment episode.
- Casual and close contact: Based on the duration of time spent with the index case, social contacts were categorized as casual or close using a weighted score from three factors:
- Time spent with the index case (<4 weeks = 1, 4–8 weeks = 2, >8 weeks = 3);
- Frequency per week (3 or more times/week = 1, daily = 2);
- Hours per week (2–4 h = 1, 4+ h = 2, all day = 3).
- ○
- Those with a total score ≥ 6 was classified as close contacts, and those with a score <6 as casual contacts. A data-driven methodology for classification was used, were a score of 6 was determined based on the mean score obtained from the study population.
- Tuberculosis infection (TBI): A person who undergoes Cy-TB testing and develops an induration of 5 mm or more is considered to have TB infection.
2.5. Ethics
2.6. Sample Size
2.7. Study Participants
2.8. Statistical Analysis
3. Results
4. Discussion
5. Conclusions and Recommendations
6. Limitations
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| aOR | Adjusted Odds Ratio |
| AU | Alcohol use |
| AUD | Alcohol use disorder |
| BMI | Body Mass Index |
| IC | Index case |
| NAU | Non-alcohol use |
| PTB | Persons with Tuberculosis |
| TB | Tuberculosis |
| TBI | Tuberculosis Infection |
References
- National Strategic Plan [NSP] for TB Elimination 2017-25|Knowledge Base. Available online: https://ntep.in/node/364/CP-national-strategic-plan-nsp-tb-elimination-2017-25 (accessed on 18 June 2025).
- Press Release: Press Information Bureau. Available online: https://www.pib.gov.in/PressReleasePage.aspx?PRID=2114549 (accessed on 18 June 2025).
- Tuberculosis. Available online: https://www.who.int/news-room/fact-sheets/detail/tuberculosis (accessed on 12 February 2025).
- Kiazyk, S.; Ball, T.B. Latent tuberculosis infection: An overview. Can. Commun. Dis. Rep. 2017, 43, 62. [Google Scholar] [CrossRef]
- Yuen, C.M.; Amanullah, F.; Dharmadhikari, A.; Nardell, E.A.; Seddon, J.A.; Vasilyeva, I.; Zhao, Y.; Keshavjee, S.; Becerra, M.C. Turning off the tap: Stopping tuberculosis transmission through active case-finding and prompt effective treatment. Lancet 2015, 386, 2334–2343. [Google Scholar] [CrossRef]
- Jacobson, T.C.; Auld, K.R.; Sm, Y.; Staitieh, B.S. The Impact of Alcohol Use Disorder on Tuberculosis: A Review of the Epidemiology and Potential Immunologic Mechanisms. Front. Immunol. 2022, 13, 864817. [Google Scholar] [CrossRef]
- Sarkar, D.; Jung, K.M.; Wang, J.H.; Sarkar, D.K. Alcohol and the Immune System. Alcohol Res. 2015, 37, 153. Available online: https://pmc.ncbi.nlm.nih.gov/articles/PMC4590612/ (accessed on 12 February 2025).
- Thummar, P.D.; Rupani, M.P. Prevalence and predictors of hazardous alcohol use among tuberculosis patients: The need for a policy on joint tuberculosis-alcohol collaborative activities in India. Alcohol 2020, 86, 113–119. [Google Scholar] [CrossRef] [PubMed]
- Retnakumar, C.; Ezhumalai, K.; Raj, R.; Narasimhan, P.B.; Lakshminarayanan, S.; Horsburgh, C.; Hochberg, N.; Salgame, P.; Ellner, J.; Roy, G.; et al. Tuberculosis transmission in Households of Pulmonary Tuberculosis Patients with Alcohol use in the Puducherry Region. Med. Res. Arch. 2024, 12, 1–10. [Google Scholar] [CrossRef]
- Martinez, L.; Shen, Y.; Mupere, E.; Kizza, A.; Hill, P.C.; Whalen, C.C. Transmission of Mycobacterium Tuberculosis in Households and the Community: A Systematic Review and Meta-Analysis. Am. J. Epidemiol. 2017, 185, 1327–1339. [Google Scholar] [CrossRef] [PubMed]
- McCreesh, N.; White, R.G. An explanation for the low proportion of tuberculosis that results from transmission between household and known social contacts. Sci. Rep. 2018, 8, 5382. [Google Scholar] [CrossRef]
- Kakaire, R.; Kiwanuka, N.; Zalwango, S.; Sekandi, J.N.; Quach, T.H.T.; Castellanos, M.E.; Quinn, F.; Whalen, C.C. Excess Risk of Tuberculosis Infection Among Extra-household Contacts of Tuberculosis Cases in an African City. Clin. Infect. Dis. 2021, 73, E3438–E3445. [Google Scholar] [CrossRef]
- Singh, S.; Thulasingam, M.; Giriyappa, D.; Devasia, J.; Sarkar, S. Arrack shops as probable hot spots of tuberculosis transmission in urban Puducherry, South India: An exploratory study. Int. J. Adv. Med. Health Res. 2020, 7, 23–26. [Google Scholar] [CrossRef]
- Nagarajan, K.; Muniyandi, M.; Palani, B.; Sellappan, S. Tracing the potential extra-household contacts of TB patients: Findings from a personal social network survey in a high TB burden setting in India. Trans. R. Soc. Trop. Med. Hyg. 2022, 116, 190–192. [Google Scholar] [CrossRef]
- Godoy, P.; Alsedà, M.; Falguera, M.; Puig, T.; Bach, P.; Monrabà, M.; Manonelles, A. A highly transmissible tuberculosis outbreak: The importance of bars. Epidemiol. Infect. 2017, 145, 3497. [Google Scholar] [CrossRef]
- Castellanos, M.E.; Zalwango, S.; Quach, T.H.T.; Kakaire, R.; Martínez, L.; Ebell, M.H.; Dobbin, K.K.; Kiwanuka, N. Performance of a score to characterise adequate contact among the social network of persons with TB. IJTLD Open 2024, 1, 556–563. [Google Scholar] [CrossRef]
- Narasimhan, P.; Wood, J.; Macintyre, C.R.; Mathai, D. Risk Factors for Tuberculosis. Pulm. Med. 2013, 2013, 828939. [Google Scholar] [CrossRef]
- Global Tuberculosis Report 2024. Available online: https://www.who.int/teams/global-tuberculosis-programme/tb-reports/global-tuberculosis-report-2024 (accessed on 12 February 2025).
- Central TB Division. National TB Elimination Programme (NTEP). 100 Days Campaign NTEP. Available online: https://nhm.gov.in/pdf/2024/ntep.pdf (accessed on 19 February 2025).
- Gill, J.; Black, H.; O’May, F.; Rees, C. Follow-up study of people who misuse alcohol: Reflections on methodology. Nurse Res. 2016, 24, 10–17. [Google Scholar] [CrossRef][Green Version]
- Ngamvithayapong-Yanai, J.; Luangjina, S.; Thawthong, S.; Bupachat, S.; Imsangaun, W. Stigma against tuberculosis may hinder non-household contact investigation: A qualitative study in Thailand. Public Health Action 2019, 9, 15–23. [Google Scholar] [CrossRef]
- Nasrin, S.; Chowdhury, N.M.R.A. Dealing with tuberculosis: Factors of the tuberculosis medication adherence among marginalized communities: A scoping literature review. Int. J. Community Med. Public Health 2021, 8, 2544. [Google Scholar] [CrossRef]
- Veerakumar, A.M.; Sahu, S.K.; Sarkar, S.; Kattimani, S.; Govindarajan, S. Alcohol use disorders among pulmonary tuberculosis patients under RNTCP in urban Pondicherry, India. Indian J. Tuberc. 2015, 62, 171–177. [Google Scholar] [CrossRef]
- Nagarajan, K.; Palani, B.; Basha, J.; Jayabal, L.; Muniyandi, M. A social networks-driven approach to understand the unique alcohol mixing patterns of tuberculosis patients: Reporting methods and findings from a high TB-burden setting. Humanit. Soc. Sci. Commun. 2022, 9, 171. [Google Scholar] [CrossRef]
- AUDIT: The Alcohol Use Disorders Identification Test: Guidelines for Use in Primary Health Care. Available online: https://www.who.int/publications/i/item/WHO-MSD-MSB-01.6a (accessed on 21 July 2025).
- National TB Prevalence Survey in India 2019—2021—Central Tuberculosis Division. Available online: https://tbcindia.mohfw.gov.in/2023/06/06/national-tb-prevalence-survey-in-india-2019-2021/?utm_source=chatgpt.com (accessed on 21 July 2025).
- Krishnamoorthy, Y.; Ezhumalai, K.; Murali, S.; Rajaa, S.; Jose, M.; Sathishkumar, A.; Soundappan, G.; Horsburgh, C.; Hochberg, N.; Johnson, W.E.; et al. Prevalence and risk factors associated with latent tuberculosis infection among household contacts of smear positive pulmonary tuberculosis patients in South India. Trop. Med. Int. Health 2021, 26, 1645–1651. [Google Scholar] [CrossRef]
- Oxlade, O.; Murray, M. Tuberculosis and Poverty: Why Are the Poor at Greater Risk in India? PLoS ONE 2012, 7, e47533. [Google Scholar] [CrossRef] [PubMed]
- Rupani, M.P. Alcohol and Disease. In Handbook of Substance Misuse and Addictions; Springer: Cham, Switzerland, 2022; pp. 1–25. Available online: https://link.springer.com/referenceworkentry/10.1007/978-3-030-67928-6_54-1 (accessed on 18 April 2025).
- Verver, S.; Warren, R.M.; Munch, Z.; Richardson, M.; van der Spuy, G.D.; Borgdorff, M.W.; Behr, M.A.; Beyers, N.; van Helden, P.D. Proportion of tuberculosis transmission that takes place in households in a high-incidence area. Lancet 2004, 363, 212–214. [Google Scholar] [CrossRef]
- Imtiaz, S.; Shield, K.D.; Roerecke, M.; Samokhvalov, A.V.; Lönnroth, K.; Rehm, J. Alcohol consumption as a risk factor for tuberculosis: Meta-analyses and burden of disease. Eur. Respir. J. 2017, 50, 1700216. [Google Scholar] [CrossRef] [PubMed]
- Kulkarni, S.; Weber, S.E.; Buys, C.; Lambrechts, T.; Myers, B.; Drainoni, M.L.; Jacobson, K.R.; Theron, D.; Carney, T. Patient and provider perceptions of the relationship between alcohol use and TB and readiness for treatment: A qualitative study in South Africa. BMC Public Health 2024, 24, 2216. [Google Scholar] [CrossRef] [PubMed]
- Grodensky, C.A.; Golin, C.E.; Ochtera, R.D.; Turner, B.J. Systematic Review: Effect of Alcohol Intake on Adherence to Outpatient Medication Regimens for Chronic Diseases. J. Stud. Alcohol Drugs 2012, 73, 899–910. [Google Scholar] [CrossRef]
- Kim, S.J.; Kim, D.J. Alcoholism and Diabetes Mellitus. Diabetes Metab. J. 2012, 36, 108. [Google Scholar] [CrossRef]
- Salindri, A.D.; Auld, S.C.; Gujral, U.P.; Urbina, E.M.; Andrews, J.R.; Huaman, M.A.; Magee, M.J. Tuberculosis infection and hypertension: Prevalence estimates from the US National Health and Nutrition Examination Survey. BMJ Open 2024, 14, e075176. [Google Scholar] [CrossRef]
- Mandieka, E.; Saleh, D.; Chokshi, A.K.; Rivera, A.S.; Feinstein, M.J. Latent Tuberculosis Infection and Elevated Incidence of Hypertension. J. Am. Heart Assoc. Cardiovasc. Cerebrovasc. Dis. 2020, 9, e019144. [Google Scholar] [CrossRef]
- Poureslami, I.; Nimmon, L.; Rootman, I.; Fitzgerald, M.J. Health literacy and chronic disease management: Drawing from expert knowledge to set an agenda. Health Promot. Int. 2016, 32, 743–754. [Google Scholar] [CrossRef]
- Aibana, O.; Acharya, X.; Huang, C.C.; Becerra, M.C.; Galea, J.T.; Chiang, S.S.; Contreras, C.; Calderon, R.; Yataco, R.; Velasquez, G.E.; et al. Nutritional status and tuberculosis risk in adult and pediatric household contacts. PLoS ONE 2016, 11, e0166333. [Google Scholar] [CrossRef]
- Badawi, A.; Gregg, B.; Vasileva, D. Systematic analysis for the relationship between obesity and tuberculosis. Public Health 2020, 186, 246–256. [Google Scholar] [CrossRef]
- Yen, Y.F.; Hu, H.Y.; Lee, Y.L.; Ku, P.W.; Lin, I.F.; Chu, D.; Lai, Y.-J. Obesity/overweight reduces the risk of active tuberculosis: A nationwide population-based cohort study in Taiwan. Int. J. Obes. 2017, 41, 971–975. [Google Scholar] [CrossRef]
| Characteristics | IC with AU (N1 = 45) | IC without AU (N2 = 61) | Total IC (N = 106) | |||
|---|---|---|---|---|---|---|
| n | % | n | % | n | % | |
| Age groups (years) 19–30 31–45 46–60 >60 | 3 12 27 3 | 6.7 26.7 60 6.7 | 5 10 21 25 | 8.2 16.4 34.4 41 | 8 22 48 28 | 7.6 20.8 45.2 26.4 |
| Area of residence Rural Urban | 5 40 | 11.1 88.9 | 9 52 | 14.8 85.2 | 14 92 | 13.2 86.8 |
| Religion Hindu Christian Muslim | 41 3 1 | 91.1 6.7 2.2 | 55 2 4 | 90.2 3.3 6.6 | 96 5 5 | 90.6 4.7 4.7 |
| Education Illiterate Primary Secondary Higher secondary Graduate | 4 14 17 3 7 | 8.9 31.1 37.8 6.7 15.6 | 4 13 30 3 11 | 6.6 21.3 49.2 4.9 18 | 8 27 47 6 18 | 7.5 25.5 44.4 5.7 16.9 |
| Occupation Unskilled Unemployed Skilled Student Professional | 29 0 11 0 5 | 64.4 0 24.4 0 11.1 | 25 3 23 1 9 | 41 4.9 37.7 1.6 14.8 | 54 3 34 1 14 | 50.9 2.8 32.2 0.9 13.2 |
| SES APL BPL | 5 40 | 11.1 88.9 | 18 43 | 29.5 70.5 | 23 83 | 21.7 78.3 |
| Marital status Married Unmarried Separated/Widow | 39 5 1 | 86.7 11.1 2.2 | 52 90 | 85.2 14.80 | 91 14 1 | 85.8 13.2 0.94 |
| Type of TB New Recurrent | 40 5 | 88.9 11.1 | 55 6 | 90.2 9.8 | 95 11 | 89.6 10.4 |
| BCG Scar Yes | 43 | 95.6 | 54 | 88.5 | 97 | 91.5 |
| Smoking Yes | 7 | 15.6 | 2 | 3.3 | 9 | 8.5 |
| Chronic diseases Yes | 18 | 40 | 40 | 65.6 | 58 | 54.7 |
| Diabetes Yes | 15 | 33.3 | 34 | 55.7 | 49 | 46.2 |
| Hypertension Yes | 4 | 8.9 | 16 | 26.2 | 20 | 18.8 |
| Body mass index (kg/m2) Underweight (<18.5) Normal (18.5–22.9) Overweight (23–24.9) Obese (>25) | 25 15 4 1 | 55.6 33.3 8.9 2.2 | 25 29 6 1 | 41 47.5 9.8 1.6 | 50 44 10 2 | 47.2 41.5 9.4 1.9 |
| Contact History of TB Yes | 17 | 37.8 | 16 | 26.2 | 33 | 31.1 |
| Smear grade Scanty 1+ 2+ 3+ | 4 21 13 7 | 8.9 46.7 28.9 15.6 | 14 29 10 8 | 23 47.5 16.4 13.1 | 18 50 23 15 | 16.9 47.2 21.7 14.2 |
| Characteristics | Social Contacts of IC with AU (N = 358) | Social Contacts of IC without AU (N = 355) | Total Social Contacts (N = 713) | |||
|---|---|---|---|---|---|---|
| n | % | n | % | n | % | |
| Age ≤18 19–30 31–45 46–60 >60 | 27 79 103 117 32 | 7.5 22.1 28.8 32.7 8.9 | 20 58 121 92 64 | 5.6 16.3 34.2 25.9 18 | 47 137 224 209 96 | 6.6 19.2 31.4 29.3 13.5 |
| Area of residence Rural Urban | 60 298 | 16.8 83.2 | 61 294 | 17.2 82.8 | 121 592 | 16.9 83.1 |
| Gender Female Male | 156 202 | 43.6 56.4 | 170 185 | 47.9 52.1 | 326 387 | 45.7 54.3 |
| Religion Hindu Christian Muslim | 316 38 4 | 88.3 10.6 1.1 | 316 21 18 | 89 5.9 5.1 | 632 59 22 | 88.6 8.3 3.1 |
| Education No formal education Primary Secondary Higher secondary Graduate | 48 82 134 35 59 | 13.4 22.9 37.4 9.8 16.5 | 55 67 115 48 70 | 15.5 18.9 32.4 13.5 19.7 | 103 149 249 83 129 | 14.4 20.9 34.9 11.6 18.2 |
| Occupation Unskilled Unemployed Skilled Student Professional Retired | 153 81 75 28 20 1 | 42.7 22.6 20.9 7.8 5.7 0.3 | 125 93 76 29 23 9 | 35.2 26.2 21.4 8.2 6.5 2.5 | 278 174 151 57 43 10 | 38.9 24.4 21.2 7.9 6 1.6 |
| Socioeconomic Status APL BPL | 65 293 | 18.2 81.8 | 148 207 | 41.7 58.3 | 213 500 | 29.8 70.2 |
| Marital status Married Unmarried Separated/Widow | 278 79 1 | 77.6 22.1 0.3 | 288 60 7 | 81.1 16.9 2 | 566 139 8 | 79.4 19.5 1.1 |
| BCG Scar Yes | 320 | 89.4 | 318 | 89.6 | 638 | 89.5 |
| Smoking Status Yes | 75 | 20.9 | 31 | 8.7 | 106 | 14.9 |
| Alcohol use Yes | 138 | 38.5 | 58 | 16.3 | 196 | 27.5 |
| Spent night with index case Yes | 110 | 30.8 | 76 | 21.4 | 186 | 26.1 |
| Share food Yes | 147 | 41.2 | 154 | 43.4 | 301 | 42.2 |
| Presence of Chronic Diseases Yes | 122 | 34.1 | 103 | 29 | 225 | 31.6 |
| Diabetes Yes | 83 | 23.2 | 54 | 15.2 | 137 | 19.2 |
| Hypertension Yes | 82 | 22.9 | 77 | 21.7 | 159 | 22.3 |
| Body Mass Index Underweight (<18.5) Normal (18.5–22.9) Overweight (23–24.9) Obese (>25) | 50 131 44 133 | 14 36.6 12.2 37.2 | 34 128 45 148 | 9.6 36.1 12.6 41.7 | 84 259 89 281 | 11.8 36.3 12.4 39.5 |
| Characteristics | Social Contacts of IC with AU (N = 358) | Social Contacts of IC without AU (N = 355) | Total Social Contacts (N = 713) | |||
|---|---|---|---|---|---|---|
| n | % | n | % | n | % | |
| Relation Extended family Friend Neighbour Relative Workplace contact | 37 63 48 109 101 | 10.3 17.6 13.4 30.4 28.3 | 71 47 23 109 105 | 20 13.2 6.5 30.7 29.6 | 108 110 71 218 206 | 15.1 15.4 9.9 30.6 29 |
| Past TB history Yes | 2 | 0.6 | 3 | 0.8 | 5 | 0.7 |
| Knows TB patient other than index case Yes | 38 | 10.6 | 39 | 11 | 77 | 10.8 |
| Family history of TB Yes | 25 | 7 | 24 | 6.8 | 49 | 6.8 |
| Family history of death due to TB Yes | 3 | 0.8 | 7 | 2 | 10 | 1.4 |
| Type of contact Casual contact Close contact | 224 134 | 62.6 37.4 | 248 107 | 69.9 30.1 | 472 241 | 66.2 33.8 |
| Duration of knowing index case <12 years ≥12 years | 199 159 | 55.6 44.4 | 155 200 | 43.7 56.3 | 354 359 | 49.6 50.3 |
| Weeks spend with index case <4 weeks 4–8 weeks >8 weeks | 75 137 146 | 20.9 38.3 40.8 | 92 164 99 | 25.9 46.2 27.9 | 167 301 245 | 23.4 42.2 34.4 |
| Times in a week 3+ times/week Everyday/week | 219 139 | 61.2 38.8 | 224 131 | 63.1 36.9 | 443 270 | 62.1 37.9 |
| Hours in a week 2–4 h/week 4+ h/week All day | 172 153 33 | 48 42.8 9.2 | 201 132 22 | 56.6 37.2 6.2 | 373 285 55 | 52.3 39.9 7.8 |
| Spend night with index case Yes | 110 | 30.8 | 76 | 21.4 | 186 | 26.1 |
| Share food Yes | 147 | 41.2 | 154 | 43.4 | 301 | 42.2 |
| Variables | Social Contacts of IC with AU (N = 138) | Social Contacts of IC without AU (N = 58) | ||
|---|---|---|---|---|
| n | % | n | % | |
| AUDIT score Low risk (0–3) Risky (4–9) Harmful (10–13) Severe (14+) | 3 47 33 55 | 2.1 34.1 23.9 39.9 | 1 16 18 23 | 1.7 27.6 31 39.7 |
| Drink in arrack shop Yes No | 106 32 | 76.8 23.2 | 41 17 | 70.6 29.4 |
| Share alcohol with IC Yes | 75 | 54.3 | - | |
| Frequency of drink with IC 1–2 times/week 3+ times/week Everyday Less than once/week | 26 23 22 4 | 34.6 30.6 29.4 5.4 | - | |
| Share glass Yes No | 3 72 | 4 96 | - | |
| Variables | Total | TBI Positive | TBI Negative | Unadjusted Odds Ratio (95%CI) | Adjusted Odds Ratio (95%CI) | Adjusted p Value | ||
|---|---|---|---|---|---|---|---|---|
| N | n | % | n | % | ||||
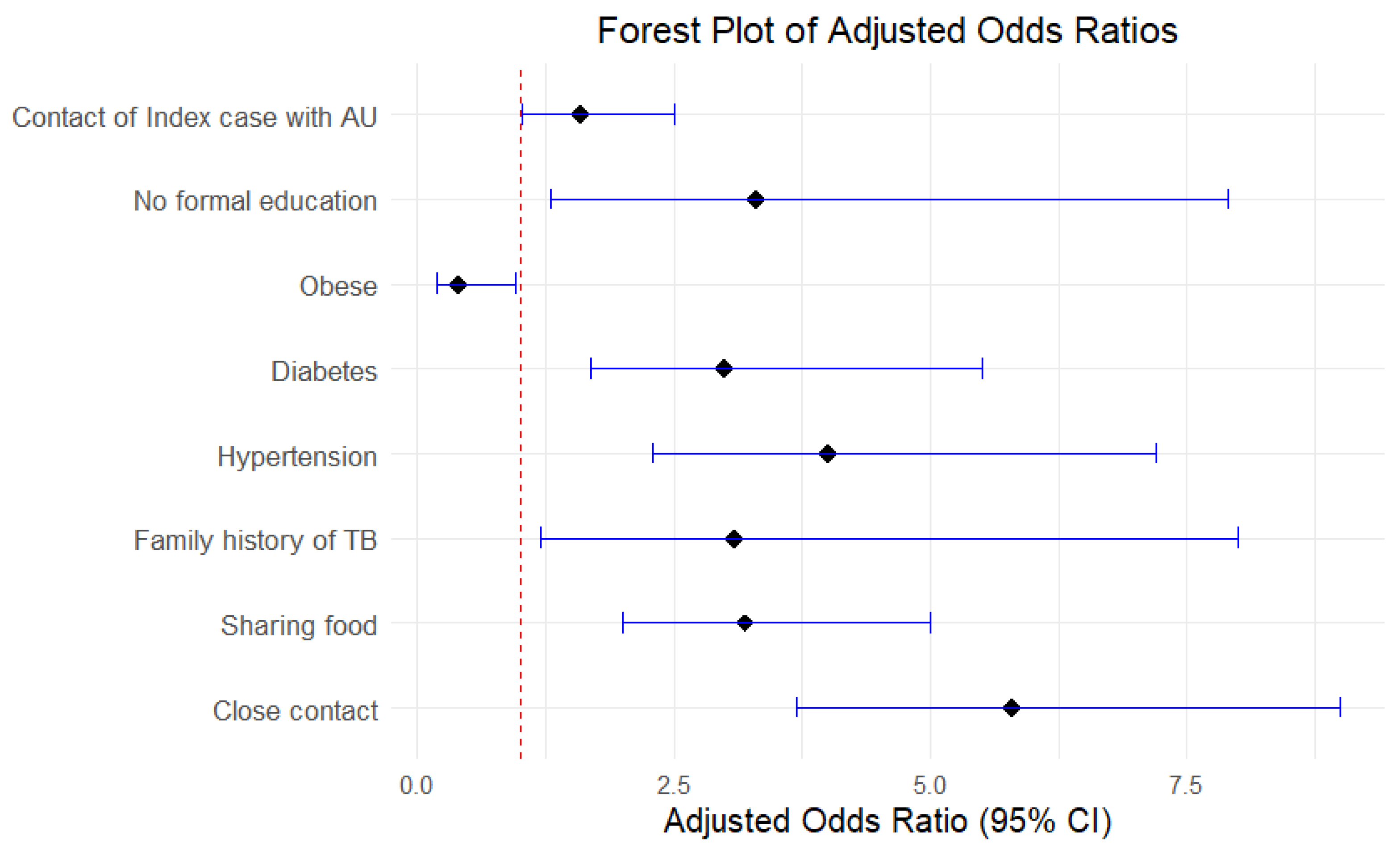
| Contact Type Index case with AU Index case without AU | 357 355 | 121 77 | 61.6 38.9 | 236 278 | 45.9 54.1 | 1.9 (1.3–2.5) (ref) | 1.6 * (1.02–2.5) (ref) | 0.04 |
| Age ≤18 19–30 31–45 46–60 >60 | 47 137 224 208 96 | 8 33 61 64 32 | 4 16.6 30.8 32.4 16.2 | 39 104 163 144 64 | 7.6 20.2 31.7 28 12.5 | (ref) 1.6 (0.6–3.6) 1.8 (0.8–4.1) 2.2 (0.9–4.8) 2.4 (1.02–5.8) | (ref) 1.3 (0.5–4) 1 (0.4–3) 0.5 (0.2–1.6) 0.5 (0.1–1.8) | 0.5 0.8 0.3 0.3 |
| Education No formal education School level Graduate level | 103 480 129 | 41 139 18 | 20.7 70.2 9.1 | 62 341 111 | 12 66.4 21.6 | 4 (2.2–7.7) 2.5 (1.4–4.3) (ref) | 3.3 (1.3–7.7) 1.8 (0.9–3.5) (ref) | 0.01 0.08 |
| Occupation Unskilled Unemployed Skilled Student Professional Retired | 278 173 151 57 43 10 | 93 48 42 5 9 1 | 47 24.2 21.2 2.5 4.6 0.5 | 185 125 109 52 34 9 | 36 24.3 21.2 10.1 6.6 1.8 | 1.9 (0.8–4.1) 1.5 (0.6–3.2) 1.5 (0.6–3.3) 0.4 (0.1–1.2) (ref) 0.4 (0.05–3.8) | Not included in model | |
| Area of residence Rural Urban | 120 592 | 39 159 | 19.7 80.3 | 81 433 | 15.8 84.2 | 1.3 (0.8–2) (ref) | Not included in model | |
| Gender Female Male | 325 387 | 84 114 | 42.4 57.6 | 241 273 | 46.9 53.1 | 0.8 (0.6–1.2) (ref) | Not included in model | |
| Religion Hindu Christian Muslim | 631 59 22 | 172 19 7 | 86.8 9.6 3.6 | 459 40 15 | 89.3 7.8 2.9 | (ref) 1.3 (0.7–2.2) 1.2 (0.5–3) | Not included in model | |
| Socioeconomic Status APL BPL | 213 499 | 37 161 | 18.7 81.3 | 176 338 | 34.2 65.8 | (ref) 2.3 (1.5–3.4) | Not included in model | |
| BMI Underweight Normal Overweight Obese | 84 258 89 281 | 39 68 30 61 | 19.7 34.3 15.2 30.8 | 45 190 59 220 | 8.8 36.9 11.5 42.8 | (ref) 0.4 (0.3–0.7) 0.6 (0.3–1.1) 0.3 (0.2–0.5) | (ref) 0.5 (0.3–1.1) 1 (0.4–2.4) 0.5 * (0.2–0.95) | 0.1 0.8 0.036 |
| Diabetes Yes No | 137 575 | 72 126 | 36.4 63.6 | 65 449 | 12.6 87.4 | 3.9 (2.7–5.8) (ref) | 3 * (1.7–5.5) (ref) | <0.001 |
| Hypertension Yes No | 159 553 | 81 117 | 40.9 59.1 | 77 437 | 15 85 | 3.9 (2.7–5.7) (ref) | 4 * (2.3–7) (ref) | <0.001 |
| Knowing person with TB other than index case Yes No | 77 635 | 41 157 | 20.7 79.3 | 36 478 | 7 93 | 3.5 (2.1–5.6) (ref) | 2.3 (0.9–5.1) (ref) | 0.05 |
| Family history of TB Yes No | 49 663 | 27 171 | 13.6 86.4 | 22 492 | 4.3 95.7 | 3.5 (1.9–6.4) (ref) | 3.1 * (1.2–8) (ref) | 0.02 |
| Smoking Yes No | 106 606 | 59 139 | 29.8 70.2 | 47 467 | 9.1 90.9 | 4.2 (2.7–6.5) (ref) | 1.8 (0.9–3.7) (ref) | 0.08 |
| Alcohol use Yes No | 196 516 | 91 107 | 46 54 | 105 409 | 20.4 79.6 | 3.3 (2.3–4.7) (ref) | 1.2 (0.7–2.2) (ref) | 0.5 |
| Sharing food Yes No | 301 412 | 131 67 | 66.2 33.8 | 170 344 | 33.1 66.9 | 3.9 (2.8–5.6) | 3.2 * (2–5) (ref) | <0.001 |
| Type of contact based on frequency of meeting Casual contact Close contact | 472 241 | 70 128 | 35.4 64.6 | 401 113 | 78 22 | (ref) 6.5 (4.5–9.3) | (ref) 5.8 * (3.7–9) | <0.001 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Retnakumar, C.; Chinnakali, P.; Bharadwaj, B.; Nagarajan, K.; Sarkar, S. Alcohol Consumption of Male Tuberculosis Index Cases and Tuberculosis Transmission Among Social Contacts in Puducherry, India: A Cross-Sectional Analytical Study. Trop. Med. Infect. Dis. 2025, 10, 248. https://doi.org/10.3390/tropicalmed10090248
Retnakumar C, Chinnakali P, Bharadwaj B, Nagarajan K, Sarkar S. Alcohol Consumption of Male Tuberculosis Index Cases and Tuberculosis Transmission Among Social Contacts in Puducherry, India: A Cross-Sectional Analytical Study. Tropical Medicine and Infectious Disease. 2025; 10(9):248. https://doi.org/10.3390/tropicalmed10090248
Chicago/Turabian StyleRetnakumar, Charutha, Palanivel Chinnakali, Balaji Bharadwaj, Karikalan Nagarajan, and Sonali Sarkar. 2025. "Alcohol Consumption of Male Tuberculosis Index Cases and Tuberculosis Transmission Among Social Contacts in Puducherry, India: A Cross-Sectional Analytical Study" Tropical Medicine and Infectious Disease 10, no. 9: 248. https://doi.org/10.3390/tropicalmed10090248
APA StyleRetnakumar, C., Chinnakali, P., Bharadwaj, B., Nagarajan, K., & Sarkar, S. (2025). Alcohol Consumption of Male Tuberculosis Index Cases and Tuberculosis Transmission Among Social Contacts in Puducherry, India: A Cross-Sectional Analytical Study. Tropical Medicine and Infectious Disease, 10(9), 248. https://doi.org/10.3390/tropicalmed10090248






