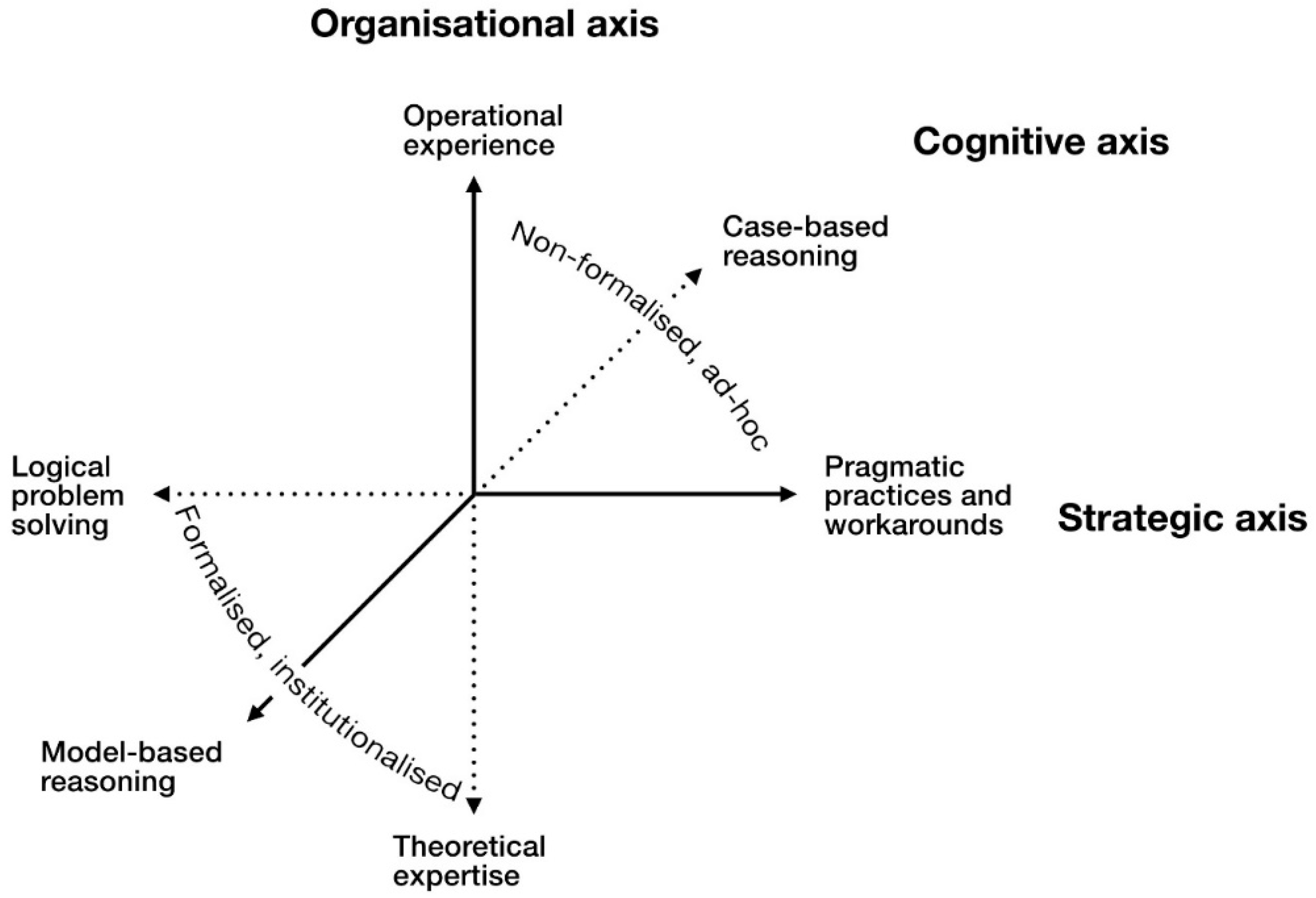
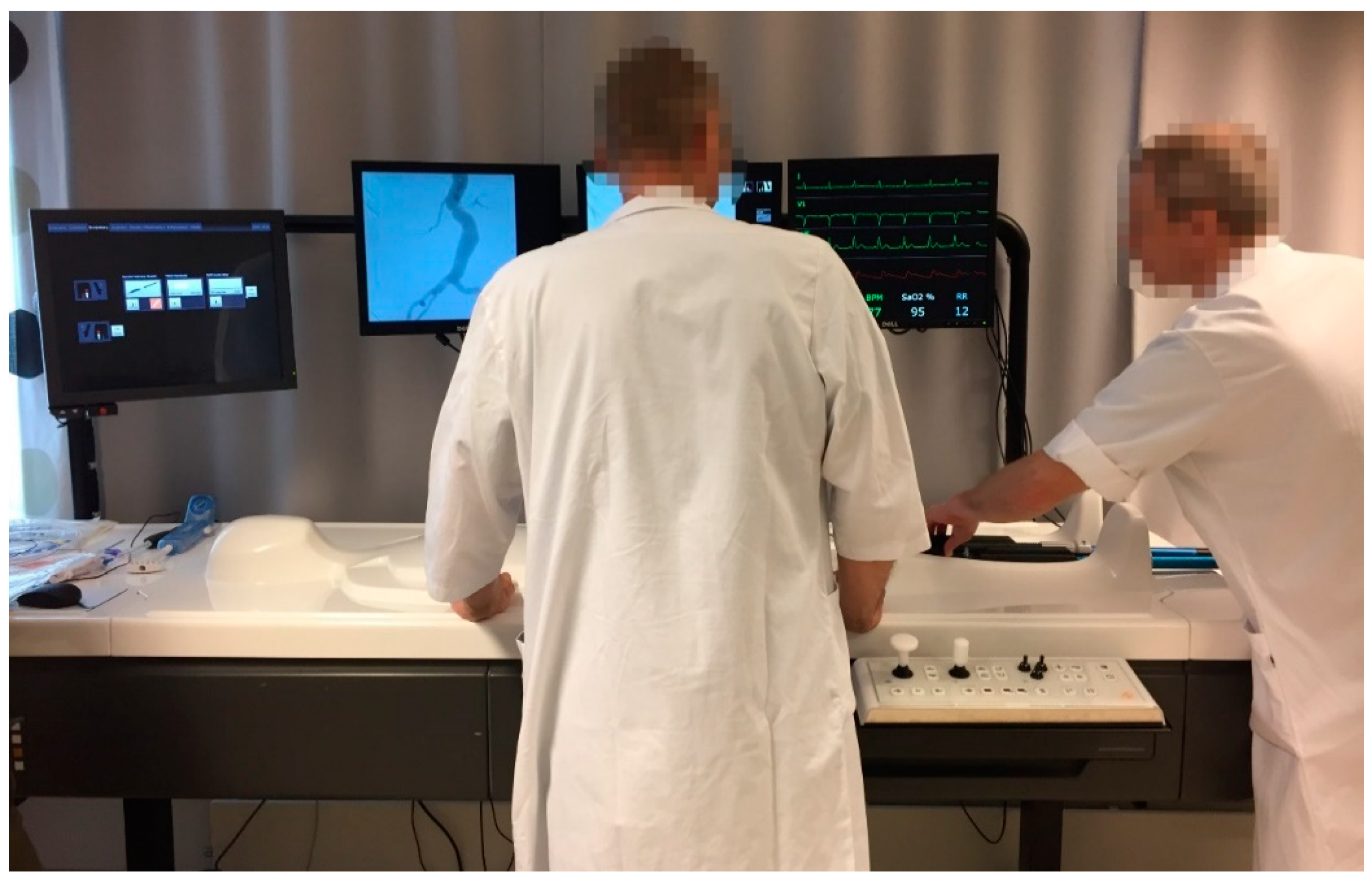
1. Introduction
With ever more tools and technologies being introduced to, and acquiring integral roles in, surgical practice, the nature of surgical work is gradually developing [
1,
2,
3,
4]. For aneurysm surgery, this development is evident in the role of imaging technologies and simulation, and in the inclusion of both surgical and radiological disciplines in the operating teams. Although the boundary between the tasks allocated to surgery and radiology is becoming increasingly blurred, the historical development and trademarks of the professions are still evident both in the literature and in practice. While surgery both in etymological terms and by reputation is particularly associated with hands (the term surgery is derived from the Greek word kheirourgia which is composed by kheir, meaning “working or done by hand”, and ergon, meaning “work” (etymonline.com/index.php?term=surgery)) and craftsmanship, radiology is associated with the eyes and the practice of seeing [
5]. In this article we explore the changing relationship between surgery and radiology as simulators are introduced into the working environment of endovascular aneurysm repair.
1.1. Treatment of Abdominal Aortic Aneurysms
An aneurysm is a medical condition of a permanent and irreversible dilatation of a blood vessel. When this condition appears in the abdominal aorta, the condition is known as abdominal aortic aneurysm (AAA). AAAs are often asymptomatic but with increased risk of life-threatening ruptures. To avoid ruptures a preventive repair of large AAA are recommended [
6]. A trade-off between the risk of the procedure and the risk of a ruptured AAA is weighted with recommended cut-off values for when to treat and when to observe [
7].
There are two main methods for treatment of aneurysms: open surgery and endovascular repair. With open surgery, the abdomen is physically opened with a long (or wide, if transverse) incision, whereafter a graft is fitted around the aneurysm. Alternatively, one may use the more recent method—introduced by Parodi in 1991 [
8]—of guiding the stent graft in place, through the arteries from two small incision points in the groin, using X-rays as guidance, placing it at the inside of the aneurysm. The stent graft consists of a self-expanding metal frame (the stent) that is covered with a material (the graft) forming a tunnel that makes the blood flow through the stent graft instead of the aneurysm. This method is called endovascular aneurysm repair (EVAR) [
7].
The developments over the last few decades have transformed aneurysm repair dramatically in terms of technology, methods and the organization of work [
9]. The minimal invasive endovascular approach changed its nature, making aneurysm repair available for larger patient groups [
9]. EVAR is regarded a complex procedure where expertise and operator preparedness are factors that might influence the results of the procedure [
7]. Among the advantages over open surgery is that it is done with smaller incisions, it promises faster patient recovery, and it is found to have lower operative mortality rates than open surgery. EVAR although requires more follow-up than open surgery. According to guidelines EVAR is usually recommended before open surgery. AAA repair is constantly evolving and less intrusive methods such as EVAR with the percutaneous approach as opposed to EVAR with the surgical approach, give the operators more alternatives to consider, all with potential controversies when they decide whether to operate or not, and if yes with what technique. In some countries screening is used to detect large AAAs; in others, large AAAs are detected due to patient complaints or by chance on patients’ CT images taken for other reasons [
7].
1.2. Simulators for Surgical Training
With the introduction of minimally invasive surgery in the 1990’s followed narratives regarding the need to train outside of the operating room (OR) using simulators [
2,
10]. The new techniques initially resulted in more adverse events which triggered concerns regarding patient safety and a wish to create systematic external control of surgical skills [
10,
11]. Simulation technology did also evolve in these years and was proposed as a solution for these issues in surgical education in general [
2,
12,
13,
14,
15] and with regards to EVAR procedures [
16,
17,
18,
19,
20,
21,
22,
23,
24]. Simulators are designed to support learning and training of several procedures and are assumed useful for training of basic [
25] and procedural [
26] skills. Simulators are often shaped to bear some resemblance to a human being, but the physical shape of the simulator in principle does no more than provide the context for computer software to provide the feel and experience of real procedures.
While endovascular virtual reality (VR) simulators are designed to imitate the physical body of the patient as the instruments are guided inside the vessels, this is done without physically imitating the bodily structures but by providing a simulated haptic feedback based upon a 3D model of the vascular structure built from the CT images and actuators that apply more or less force on the inserted instruments depending on what they are “touching” in the virtual world. While this can imitate, today’s technology is still far from duplicating the complex and highly individual haptic feedback from instruments and bodily structures as perceived by the operator (simulating haptic feedback has been found technically challenging, and operators easily perceives the simulated haptic feedback as not realistic. Simulated haptic feedback has also been found to change operators’ performance on the simulator. See [
27] for operators’ appraisal of simulated haptic feedback in laparoscopy) [
28].
While education and training of surgeons and interventional radiologists have traditionally been in a context of novices working under the supervision of experts, this mode of acquiring competences could, with simulators, be forwarded in a more forgiving environment; Prentice [
2] explores the practices of surgical training as virtual reality simulators are proposed as substitutes to the traditional apprenticeship-based training in the OR. She reflects upon differences between training in high-stakes environments, the OR, and training in low-stakes environments, the simulator skills lab, and suggests that we need to restate what matters as knowledge in a wide sense in surgical training.
The possibility to perform simulated patient-specific rehearsals is a recent innovation in the world of virtual reality simulation [
23] and as such there was great interest in, at the hospital that we investigated, whether a rehearsal on a simulated patient before operating on the “real patient” afterwards would improve operative outcomes and operation efficiencies (time, x-ray exposure etc.) [
29].
Research studies of the introduction of simulators into the EVAR environment have mainly been concerned with the effect of the simulator on technical metrics aspects such as operating time, fluoroscopy angle (this relates to the number of iterations necessary for producing good intraoperative X-ray images, with consequences for radiation exposure) [
24,
29], tool selection [
22], technical and non-technical skills [
16], and with the realism of the simulation [
24,
28].
The introduction of a simulator into the EVAR environment is one of numerous digital technological contributions to the medical field where technology development and medical application co-develops in an experimental, non-deterministic fashion, and where the role to be found by the technology is not clear before work processes are stabilized—something that may take considerable time. This following study covering the initial phase of the EVAR simulation project explored the issues at stake—related to the nature of EVAR procedures and the organization of work—when a simulator is being introduced to the clinic. Hence, the research approach is not oriented towards measuring the impact of the simulator in terms of parameters such as operation time and the usage of contrast liquids, but to identify sociotechnical themes and topics that are made relevant by the introduction of new technology, but that lie outside the immediate scope of the technology.
1.3. Operation Planning and New Professional Relationships
Another effect of endovascular surgery and EVAR was a gradual change in the relation between the fields of surgery and radiology. This included a new distribution of skills and knowledge, new division of labor and new forms of collaboration [
9]. This is not unique for endovascular surgery; changing professional relations—structures, tasks and roles of work—in an increasingly technified, heterogeneous health domain is also a broader development trend that can be witnessed for example in anesthesia [
30]. In the context of representational work in the health sector, Barley [
1] studied the effect of new medical imaging devices on the role relations among radiologists and radiological technologists. While the introduction of identical CT scanners in two radiological departments occasioned similar structuring effects both places, the resulting organizational forms differed. In other words, technology may have a shaping effect on organization, but this effect cannot be understood in isolation from organizational and social context.
In the traditional method of open surgery, the job of the radiologists was concluded before the operation started, having produced the images necessary for planning the operation. Interventional radiologists came to play an important role with the development of EVAR and the actual execution of the procedure. In order to bring the stent graft through the blood vessels from the femoral artery to the aneurysm and position it accurately, continuous production of X-ray images is necessary. X-ray images traditionally belonged to the domain of radiology. However, with the clear aspect of intervention, endovascular procedures could be interpreted as either surgical, thus belonging to the surgical domain, or belonging to the domain of the radiologist as they were guided by X-ray images. Much of the pioneering work within endovascular procedures were done by radiologists, and a new group of radiologists emerged: interventional radiologists. However, it was still open to debate who were to be responsible for endovascular procedures. Should the radiologists learn to operate, the surgeon’s main competence, or should the surgeons learn image interpretation, the radiologist’s main competence. Endovascular procedures, and in this case, EVAR occasioned new competences, new relations and new forms of collaboration between vascular surgeons and interventional radiologists. Who performs EVAR procedures at hospitals today varies from a team of only vascular surgeons, a team of only interventional radiologists or a team of both vascular surgeons and interventional radiologists. We studied the latter configuration. Exactly why this came to be the configuration at our study hospital would be interesting to explore, but that was outside the scope of this study.
It is relations and collaborations and how they are affected when a virtual reality simulator is introduced to the working environment that is at the heart of our attention in this article: how do the politics of a simulator work? How do the users enact it and how does its socio-materiality influence relations and division of competence and labor between the actors? Because politics is not limited to the social sphere—objects too have politics [
31]—working relations, professional identity and division of labor do not result from management strategies, professional considerations and social interaction alone; they can also be traced back to material contexts and developments, although that was not necessarily the intention of the designers or those that introduced the equipment. Accounting for the politics of the virtual reality simulator is a first step towards a more holistic understanding of the intimate relationship between technological innovation, professional identity and organizational development.
4. Results
4.1. Adapting to the Simulator
The simulator provides the opportunity for a practical patient-specific rehearsal, which can take place as a preparation for a planned “real” procedure. Although realistic, there are also necessary considerations that need to be taken. In one of the training sessions we observed, an error message popped up on the simulator monitor when the participants proceeded as they would usually do in a real situation, guiding the main body (stent graft) into the simulator, but before choosing the right instrument on the simulator interface. “Now you’ve done something…”, the radiographer said. “Yes, we inserted the main body”, one of the operators answered. Whereafter the radiographer made clear that with the simulator one has to do things in the right sequence, sometimes deviating from how it is done in reality.
The simulator does not only come with certain requirements for work processes—it also recruits new stakeholders; in the simulator project that constituted our research case, the simulator was accompanied with the social researchers (authors 1 and 2) and the medical technologist (author 2) of the project, several technicians from the producer and vendor side, new file formats from patient-specific CT images that must be created according to particular procedures in order to fit into the simulator, new software for producing the image file conversions and new procedures for doing so, to mention some of the central ones. As a result, it was quite crowded and hectic at some simulator training sessions and a few subsequent procedures in the OR during our fieldwork—at one point there were twelve persons in the OR. With the reservation that this had to do with the simulator introduction as a time limited project: from a fairly concentrated gathering of actors and practices before the introduction of the simulator (it should be said that the simulator is only the last branch of an ongoing development leading to an already fairly populated environment, as already mentioned), the entities, processes and locations necessary to keep track of the aneurysms after introducing the simulator were significantly multiplied. The simulator being an expensive new piece of equipment also attracted interest from management expecting it to result in saved money and/or success stories of improved patient treatment. As we shall discuss later, these stakeholders are all involved in a process of bringing the aneurysms and the work with the patients to locations outside the patients themselves. Importantly, the extreme crowdedness described here was not the rule; it happened in the very first procedure following simulation, and it was not reproduced on that scale.
Before the introduction of the patient-specific rehearsal the operators usually prepared themselves doing what can be called a mental rehearsal. Several of the interventional radiologists we talked to said that when doing measurements of the aneurysm and when ordering the stent-grafts they went through the patient’s CT images visualizing how to tackle challenging anatomy. It can be particularly challenging to insert instruments through tortious vessels, and the operators on beforehand reflect upon which instruments to use and the angle of the C-arm to create optimal visualization of the anatomy while limiting use of X-ray. The difference between the mental rehearsal, which they already did, and the simulated patient-specific rehearsal would in theory be that the simulated rehearsal contained practical (physical) aspects like stent-graft deployment.
Unsurprisingly to anyone familiar with the introduction of sophisticated, digital tools, the first phase of the project was more characterized by practical problems than by the actual work expected to be done on and by the simulator. There were issues with the file formats required by the simulator—where this format was different to the format usually used by the surgeons and radiologists. The file format that the simulator requires is STL (STereoLithography), which is a format widely used for rapid prototyping and 3D printing. STL files describes the surface geometry of a three-dimensional object dividing it into triangles. In addition, a considerable amount of work to prepare this file and align it with the simulator software was necessary. Hence the question of how to organize this data work in the future was accentuated—indicating the organizational scaffolding that is always necessary for technology to function according to the intention. Eventually one specific radiographer received the responsibility and dedicated time to create the files and to import them into the simulator.
4.2. Simulation and the Acquring of Competences
As access to the aorta aneurysms is given through the femoral arteries, there are in principle two ways of entering the body with the necessary instruments: surgical access or percutaneous access. Surgical access involves intersecting with a scalpel, a task that is traditionally reserved for surgeons and that necessitates the presence of a scrub nurse and a circulating nurse. When the access is given percutaneously, however, the use of a needle instead of a scalpel makes the fundamental difference that the flesh is not exposed, and hence this task is unproblematic to the radiologists together with radiographers. Percutaneous access is also reported by our radiologist informants to reduce the risk of complications for the patients, in addition to recovery time, although the number of complications pertaining to surgical access in EVAR procedures is small [
59,
60]. Percutaneous access therefore potentially necessitates less personnel in the OR and thus would have economic advantages. On the other hand, surgeons have increasingly trained and practiced to develop skills to produce, interpret and act upon images such as X-ray and CT. As aneurysms are treated with increasingly more sophisticated methods involving more imaging and interpretation work and less blood and craftsmanship—first through the turn from open to minimally invasive surgery, and then from surgical to percutaneous access—the professions are catching up both by developing and making their professional competence relevant, and by negotiating the division of labor with adjacent professions. An argument that the vascular surgeons would use was that they needed the surgical access as cases for surgical training, if all patients were to be operated with percutaneous access, they would not be able to train their surgical trainees.
With the simulator comes new opportunities for training through interaction. In this study, many of the informants conveyed to us that the simulator provided an arena for competence-building interactions between those with much experience and those with less experience. Several of the informants held that the simulator in particular produces a larger surface of contact between novices and experts; for example, the inexperienced surgeons are given a much better opportunity to ask questions in the simulator sessions than during the real procedures, and also for the more experienced to observe the less experienced. We were told about a certain procedure where the experienced radiologist (G) did not pay so close attention to the less experienced surgeon (R): “If we had only simulated this situation before, G would have known that R wasn’t that experienced, and he would have paid closer attention to him”.
4.2.1. A sociotechnical Test Arena
The simulator worked as a sociotechnical test- and practice arena where representations and interaction are central resources. One of our informants told us that “in the old days, they got a lot of experience from the diagnostic work on patients. Today, this diagnostic rest heavily on the CT images, so the surgical trainees do not get that experience. But the simulator can be a substitution for that”. From the moment when a doctor suspects that patients may be suffering from an aneurysm of a certain size, the patients’ bodily structures are transformed into three-dimensional digital images that are easily submitted to all kinds of treatment—such as rotation, zooming and teleporting—in contrast to the more recalcitrant nature of humans of flesh and bones. During all phases of aneurysm treatment—from diagnosis, preparations, through the operation and in the post-operative controls—different kinds of images (particularly CT images) are held together with other types of sensory aids such as contrast liquids (injected into the patients’ vein system during operation to produce clearer imager particularly for obtaining an accurate positioning of the stent grafts) and measuring scales (measuring scales are used at different occasions such as when making measurements on the CT images before ordering stent-graft components or during procedures to measure actual distances within the body), and machineries such as the EVAR simulator, hence multiply the patients and their condition in a fashion that allows different types of professionals and materials to contribute with their particular expertise.
4.2.2. New Forms of Articulation Work
The coordination of these contributions is ensured through multiple strategies. While formalized processes guide some phases of the work, such as the introductory phase in the OR when the main plans and uncertainties are accounted for through e.g., checklists, other processes are drawing on non-formalized experience-based and pragmatic lines of work. One example is when the curving of the patients’ arteries makes the guidance of the stent challenging, and idiosyncratic dexterity makes up an important part of the repertoire. The type of wriggling and bending and wire manipulation we observed was reported as only anecdotally referred to in the syllabus or practical training, but acquired through years of experience as professional competence. The same is the case in those instances when—not so seldom—the planned type of instruments is switched with a different one during the operation, due to what is learned about the patients’ arteries when images are supplemented with real patients.
The increased number of persons and roles following the introduction of the simulator also requires new forms of articulation work: in one of the simulator sessions we participated in, a problem with the stent deployment mechanism occurred. This occasioned a discussion of how such simulator functionality problems should be addressed, and how responsibilities should be shared between the different parties of radiographer, radiologists, surgeons and simulator company; each of them is an expert only on different parts of the process. The simulator company preferred that one person from the EVAR team should come to them. However, one of the radiologists stressed that the competence is distributed in the team—the operators master some aspects, and the radiographer master other (e.g., preparing the image files and importing them into the simulator) and that it would not be right to send one of them. It was decided to postpone the discussion of how to manage such issues with the simulator company a few days. Another type of ”secondary work” coming with the simulator is the importing and pre-processing of the CT images, tasks that eventually was given to a dedicated radiographer. As one informant said: “What will be most difficult and most important to spend enough time on in the beginning, is the data-technical aspects of the simulator. The simulation itself will be easier to handle”.
4.2.3. Low-Stake Environments and the Creation of Shared Understanding
The simulator granted a position between the traditional phases of planning and operation. By rehearsing on the operation using patient-specific images, one may see the simulator as bridging two phases—the planning and the operation—that are otherwise conceptually, spatially and temporally divided. In addition, it brings together the numerous human and non-human actors in an environment that allows for “configurative exploration” (we use this term to encompass cognitive modi, interpretation paths and organisational arrangements in one term) at a different scale than is recommendable in the OR. Prentice [
2] discusses the difference between high-stakes and low-stakes environments and emphasizes what is lost in a low-stakes environment. It seems although in our case that the simulator sessions, by being low-stakes add levels of reflection that remain tacit during the high-stakes environments such as the OR. On several occasions the surgeon and the interventional radiologist used the simulator session to discuss division of tasks during the actual procedure the next day, and during the procedure on patients they talked about experiences from the simulator session creating a shared understanding. The simulator sessions also gave more room for inexperienced surgeons or interventional radiologists to spend more time, ask more questions to the other more experienced operator and thus obtain more training on steps of the EVAR procedure. An example of this occurred during a simulator session when the interventional radiologist requests one more rehearsal. They start again; the vascular surgeon inserts the main body into the simulator whereafter they do a digital subtraction angiography. “This time I did it all by myself” the vascular surgeon says, with an apparently strengthened self-confidence. Visions to improve the naturalistic power of such integration further were expressed by one of the doctors attending the training session: “In the future, the simulator should be integrated in the angio-lab, so that the user interface will be the same as that used for operations”, indicating that the simulator facilitated valuable training and preparation, which potentially could be removed if not streamlined with clinical practice in the angio-lab. We did not discuss this issue of interface with the manufacturer, which of course also pertains to whether the simulator is manufactured to support training or patient planning—this could be something to elaborate on in further studies.
While the patient-specific EVAR simulator is designed and marketed as a tool for training operating skills and preparing for particular patients—above described as bridging both phases and resources—our informants portray it as a tool for bridging the professions and preparing the ground for collaboration. To this end, there also exists a formal collaboration agreement between the vascular surgeons and the interventional radiologists at the hospital. While the experienced surgeons and interventional radiologists indicate that they do not really need the simulator for training skills as the procedures are so embodied that the simulator does not represent an essential addition, they speak of the simulator in the same context as they speak of the formal collaboration agreement, explicitly explaining that the simulator and the collaboration agreement serve the same purpose. The gathering of both professions around the simulator “does something with the maturity that they bring with them into the OR to do the procedure” (interventional radiologist). In the following we will elaborate on relational aspects of the simulator.
4.3. Relational Aspects of the Simulator
The organization of work between vascular surgeons and interventional radiologists differs much between different hospitals, and the different possible organizational configurations seem to suggest that vascular surgeons and interventional radiologists are to some extent interchangeable. However, there are no indications in our study that the vascular surgeons and interventional radiologists have a wish to merge these professions. To the contrary, they express a strong professional identity, and this enters into the continuous, though not very visible work of defining EVAR procedures and defining the role of the vascular surgeons and interventional radiologists, respectively.
4.3.1. Access and the Division of Labor
In our study hospital, the “ownership” of the patients is with the vascular surgeons and their clinic. That means that although the interventional radiologists take part in both the planning of the procedures and the actual operations, the economic–administrative model—such as “unit price financing”—of the activities makes the surgical department the main economic beneficiary of the patients. While the division of labor is otherwise in play, the surgeons are the only ones offering a ward for the patients, giving administrative and clinical weight to the ownership.
There are several rationales for engaging both surgeons and radiologists in EVAR procedures. Some are essentialist, that is, owing to the nature of the procedures and the different professional competences. Others are pragmatic, that is, pertaining to a continuously negotiated division of labor based on organizational aspects. Yet others are somewhere in between, enjoying the controversial status of not belonging unambiguously to either the realm of professional competence or organizational aspects.
In the development of aneurysm repair from open to minimal invasive surgery, interventional radiologists were central for visualizing the inner body and the surgical work through imaging technologies such as X-ray and CT. With time, the division of labor, at this hospital, has evolved such that much of the work of the surgeons and radiologist can be performed by both. With the development of professional competence and the blurring of the differences between what surgeons and radiologists can do, radiologists—supported by the momentum of the developments in digital imaging technologies and their role in changing the nature of medical work—are aspiring to strengthen their position relative to the surgeons. In this situation, the practice of access serves to stabilize the division of labor, slowing down or preventing a development feared by surgeons: that their contributions to and influence over EVAR surgery will be diminished. The laconic and “humorously” meant comment from an interventional radiologist performing a simple task during a procedure, illustrates, that what is included and what is not included in the professional practice, is socially regulated, and at the same time at stake: “This is all we interventional radiologists are allowed to do!” In contradiction, at a simulator session a few days later, the vascular surgeon asked what role the interventional radiologist wanted, to which the answer was: “the main role” (smiling). In a moment of high-contextual communication, the surgeon handed the levers to the interventional radiologist.
4.3.2. A Collaborative Artefact
It is important to underscore that what is at stake with the changing nature of aneurysm repair procedures is not one-sidedly the relation and power between the two positions, but also the robustness implied in having and nurturing these two professions with their differences. This is explicitly stated in the written collaboration agreement between the vascular surgeons and the interventional radiologists, articulating that the hospital today has two separate teams covering the requirements related to endovascular treatment, and that it is important to maintain the competence within both teams to ensure sufficient readiness. At the same time, the operators are occupied with building a collaborative environment. One informant told us that “the simulator works as a bridge between the operators, it supports the teamwork. When both operators go through the procedure together on the simulator in advance, it does something with the maturity they bring to the OR to do the actual procedure”.
The initiative for this project came from the interventional radiologists, and due to the personnel situation, the radiologists are also in a position to spend more time with the simulator than the surgeons, with the unspoken possibility of making it a tool more directed towards supporting the radiologists than the surgeons. It was also a radiographer that was given the responsibility to prepare the files that are imported into the simulator.
The picture painted by our informants, however, is that while the proprietary aspects of EVAR are something that the local teams seek to use as a resource for optimal collaboration, the more central and distant institutions like the labor unions tend to be stronger oriented towards protectionism and, as a consequence, not necessarily fuelling collaboration.
5. Discussion
While the simulator was acquired by our study hospital with primary expectations to improve technical success, fluoroscopy angle and operating time, and the realism of the simulator was considered an important parameter to achieve this (results from these investigations can be found in [
28,
29]), the results from the present study show that there are a number of other, interrelated themes indicating that the simulator also makes a difference in other, less expected—but not necessarily less important—ways. These belong not only to the technical sphere, but also to the social and organizational spheres.
The organizing and carrying out of EVAR procedures differ between hospitals in different cities. In some hospitals, the least invasive endovascular approach, the percutaneous approach, is the norm. In EVAR procedures using the percutaneous approach, interventional radiologists and their team of radiographers are allowed to perform the operation without assistance and presence of vascular surgeons and OR nurses. At other hospitals, such as our case hospital, vascular surgeons and interventional radiologists are present during the full length of the endovascular part of the procedure, together with both teams of radiographers and OR nurses. Yet at other hospitals vascular surgeons undertake the procedure without the presence of interventional radiologists.
In the previous section, we have elaborated on some central aspects of what the introduction of a simulator into a clinical environment and a social collegium can mean in practice. In the following, we shall continue to analyze these themes and explore what new insights can be developed when we approach them as themes of boundary work and sensework.
The medical field and hospital environment are not unique when it comes to development of specialized technology and professional specialization. As a refrain of modernity, new human and (particularly) non-human actors are increasingly populating our workplaces, particularly in or through the IT departments. As is well documented in the literature [
1,
2,
3], the nature of surgical work is changing with the introduction of new imaging and simulator tools. EVAR procedures are becoming increasingly technologically complex procedures, where simulators are aspiring to acquire an important role, particularly in the planning phase. New techniques are added and mixed into older ones, sometimes producing new opportunities, other times not. In an operation that we observed, the surgeon and the radiologist discussed how they could use some small pieces of metal from a former operation that the patient had undergone, as points of reference. These would not be visible in the simulation as the simulation is based on segmented images of the patient aorta and not images of the whole patient.
With this development of the increased role of biomedical informatics, the relation between medical professionals and the physical subjects that are the final target of their work, is changing [
61]. Whether this change is towards more or less intimacy is perhaps a question of perspective. Many specialists’ first encounter with patients is actually with numerous images and other kinds of sensor data from the patients. A considerable proportion of the work is actually done on representations that are separated from the patients in time and space. CT images of aneurysms may be produced in another hospital and by other individuals than those using them for diagnosis and further operation planning. The rationale for this separation process is the cost-effectiveness that modernistic arrangements of work ascribe to it. However, not before images and patients are re-united in the OR—or virtually in the simulator—is it proven how well they fit; will the doctors manage to guide the selected stent through the blood vessels from the femoral artery to the aneurysm and position it accurately and permanently? Now is the time to bring together the lessons from the work on the computer screen, the discussions between colleagues, the experiences from similar operations and the rehearsal on the simulator.
How does this development influence the relation between professions, and the nature of work? In the following we will discuss these themes in terms of boundary work and sensework, respectively, before we indicate the connection between these as a central part of the conclusion.
5.1. Simulation as Boundary Work
The discourse on the relationship between surgeons and radiologists tends to be reduced by somewhat essentialist descriptions of the qualifications and modus operandi of representatives of the two professions. The following report from that discourse illustrates this:
“The question of which groups were to control MRI images was unresolved in the early days of MRI. In recent years, practitioners from a broader range of specialities have used the technology, and so the power to interpret the scans is once more at stake. For example, in The New York Times a radiologist claimed that ‘a radiologist who has spent four to five years learning how to interpret those images’ would best be able to guarantee the quality and cost-effectiveness of image interpretation [62] (Levin 2004). Michael J. Wolk, then President of the American College of Cardiology, responded immediately that ‘[in] fact, cardiologists are the physicians best prepared to interpret cardiovascular images’ [63] (Wolk 2004).”
What we see in our study, however, are two things that are not so much discussed; first, the possibility of one of the professions taking over the other is real, but it is real because developments in technology—not the least, better images and tools to support interpretation—make it real; second, the boundary work taking place does not necessarily—although it is most often the way it is framed—reflect distinction practices [
5], moving divisional lines in a zero-sum game. In our study, the surgeons and radiologists are actively exploiting the leeway at this boundary to strengthen collaboration and eventually the quality of work. The way they talk of and administer both the simulator and the collaboration agreement illustrates this. In addition, as one of our informants underscores: “The most important is to think of what is best for the patient, and not which department that gets paid” (interventional radiologist, our emphasis).
In this context, the simulator can be understood as an actor taking a role in the boundary work amongst the two professions, a boundary that they by no means wish to efface, but rather use as a warrant for robustness—remember the collaboration agreement—for the best for the patient. While the simulator provided opportunities for new alliances in the negotiation of professional identity and ensuring that both professions’ expertise was still unique and not cannibalized, we saw that the two professions exploited the occasion for developing the alliance between them.
Framed this way, the way from boundary work to sensework does not seem so long. In addition, actually, Bailey et al. [
64] in a highly condensed fashion express this relationship when they state that
“manipulating physical objects through digital interfaces prompts changes in the organization of work, alters the way people make sense of—and come to trust—the objects with which they work, and transforms workers’ roles.”
In our study, one very concrete way that the simulator brought about changes in the organizing of work was the new practices for the meetings between the two operators where the upcoming procedure is discussed in plenum one day before the actual operation. These previously loosely organized meetings were moved to the simulator room and organized as a simulator session. The radiographer, that was given the responsibility of preparing the CT images and eventually the simulator sessions, acted as a mediator between the operators. One of the vascular surgeons said that “there is one good thing about the simulator, and that is [the name of the radiographer]”, indicating that the simulator session had a pronounced positive effect on commitment and engagement from the participants. This does not only refrain the analyses of Barley [
1] and Burri [
5] on how tools may enter into the negotiation of social order through distinction, but also hints to something we shall see more of; namely how materiality may
reverse distinctions and bring professions closer together.
As an example of material agency, this influence of artefacts bears resemblance to the influence of the X-ray apparatuses in the OR in the early days of EVAR-procedures, as they acted as attractors on the radiologists, and as arguments for bringing their expertise and intervention into the actual procedures.
We find many examples of how the simulator works to reduce the importance of the distinction between surgeons and radiologist, while at the same time offering opportunities for identity formation and territorial achievements. Burri [
5] highlights the significance of
where machines are installed and
who has the skills for operating them, and despite the collaboration agreement and the explicit good relation between the surgeons and the radiologists, it is not without significance that the initiative to acquire the simulator came from the radiologists. The radiologists are in a position to spend more time with the simulator thus—consciously or unconsciously—shaping it to support them in particular, and also a radiographer was given the responsibility to prepare the image files for simulation. In this way, although the ownership to the simulator formally is divided between them, one can see the contours of a symbolic ownership gradually developing. Is it therefore that radiologists are more numerous than the surgeons in the practice sessions; is it about commitment? Are we here witness to subtle distinction practices that will turn out more visible in the future? We are at least witnessing the politics of the simulator.
5.2. Simulation as Sensework
One pronounced development trend in surgery and radiology, as this study also demonstrates, is the increasingly heterogeneously populated operating environments; not only by different medical professionals, simulators and visual representations, but also by technicians, salespersons, project coordinators, and new image file converters and procedures. Taken together with expectations from the hospital management that the effects of the simulator also will be traceable in the hospital financial statements, the simulator tends to recruit a range of different cognitive, organizational and strategic modi operandi into the field of EVAR. Not surprisingly, given the early stage of technological and process innovation, the work involves a significant amount of pragmatic practices and workarounds—as with the file format conversion in order to load images into the simulator software. Although not explicitly communicated, the responsibility for this work was given to one of the radiographers, with all that this could imply regarding mental ownership—illustrating the close interrelation between sensework and boundary work.
Understanding EVAR through the lenses of sensework directs attention towards organizational work from the inside, with the nitty-gritty details of work on numerous and often separated information sources, such as digital sensor data and visual representations, as the point of departure. In the case of EVAR procedures, the pieces of digital sensor data and different sorts of representations are nothing less than extensions of the patients’ identities [
3], carefully shaped to fit into the modern medical machinery of information treatment.
An effect of the simulator on the EVAR procedures is the extension of the focus from the bodily patient alone to an extended patient situated in what we might think of as a more synthetic situation [
65]. Here, the haptic feedback is artificial, resulting not from friction between medical instruments and bodily structures, but between computer simulated “digital friction”. The patient is thus multiplied into different representations [
3], some of which are far more mobile and also combinable than the bodily patient [
42,
54]. The translation of the patient into numerous representational states that are produced, circulated and recombined—easily and swiftly so due to their digital constitution—seems to offer creative interpretation work of higher importance in the diagnostic part of medicine.
With the technification and digitalization of the patient, and a parallel development of specialization and splitting into more and more specific work processes, the work with the patients increasingly takes place in locations outside the patients themselves, such as digital 3D representations and simulators. In this context, patient specific EVAR simulators contribute as phenomenological links between the patients and their digital representations.
Individually embodied skills may also change with the introduction of ever more sophisticated tools like robots. The shift from craftsmanship to sensework also implies a relatively stronger weight on the immaterial aspects of surgery, although not necessarily at cost of the material.
This allows for a more active and creative exploration in simulated conditions, what Prentice [
2] calls low-stakes environments, that are more forgiving than the bodily patient alone, accentuating the explorative and retrospective nature of decisions that Weick, Sutcliffe, and Obstfeld [
53] has theorized in general and Haavik [
4] has documented in the OR. This bringing of the patient into a new and extended, technoscientific reality [
3], a new landscape for sensework that is less explored and with fewer established boundaries demarcating the professions, facilitates for significant unpredictability with respect to outcomes of boundary work. This may explain why there are so great differences in the division of labor and the formal organization of EVAR in different hospitals, which is an empirical finding in this study as well as in the work of Barley [
1] in the case of the introduction of identical CT scanners in two radiological departments; technology may have a shaping effect on organization, but this effect cannot be understood in isolation from organizational and social context.
6. Conclusions
The surgical procedure of aneurysm repair has undergone substantial developments in the last decades, and the patient-specific EVAR simulator is among the more recent contribution to this development. In parallel with this, the professions of vascular surgery and interventional radiology performing EVAR are developing with new technologies and organizing models in the hospitals. This is the context for our study, where we set out to explore what role the EVAR simulator may take in relation to both the EVAR procedure and the development and interrelation of the professions. Our findings relate both to the particularities of EVAR simulators and changing professional practices and relations, and contributes with generic findings on the relationship between sensework and boundary work.
While the EVAR simulator has the potential for elevating professional skills and optimizing the procedures in terms of such parameters as reduced operating time, less use of X-rays and better equipment selection, it demonstrates an even more prominent role in negotiating, ordering and stabilizing the professional relationship between vascular surgeons and interventional radiologists. Other studies have documented similar findings in simulation training for spine surgeons, where the relationship between neurosurgeons and orthopedic surgeons were stabilized [
66]. Thus, the article contributes to the discourse on socio-material understanding of professional work and interdisciplinary relations.
This study is the first to our knowledge that investigates the role of simulators for professional identity and relationship among surgeons and radiologists, although similar qualitative studies in the surgical field exist [
67,
68].
A task for future research on this topic could be to continue exploring this phenomenon in depth, and to map the more long-term effects of simulators in multidisciplinary medical environments. It would also be useful to direct more attention to radiographers, anesthetics personnel and OR nurses to produce a more complete picture of the sociotechnical environment. Comparative studies in similar medical environments where identical technologies are accompanied with different interdisciplinary relations than in the present case would be useful to shed more light on the significance of social context prior to introduction of new technologies. Further, similar studies in other empirical fields should follow up on the more generic themes and findings.