Molecular Analysis of Carbon Ion-Induced Mutations in DNA Repair-Deficient Strains of Saccharomyces cerevisiae
Abstract
1. Introduction
2. Materials and Methods
2.1. Strains and Growth Conditions
2.2. Irradiation
2.3. Survival Rate and Mutation Frequency
2.4. Sequence Analysis
3. Results
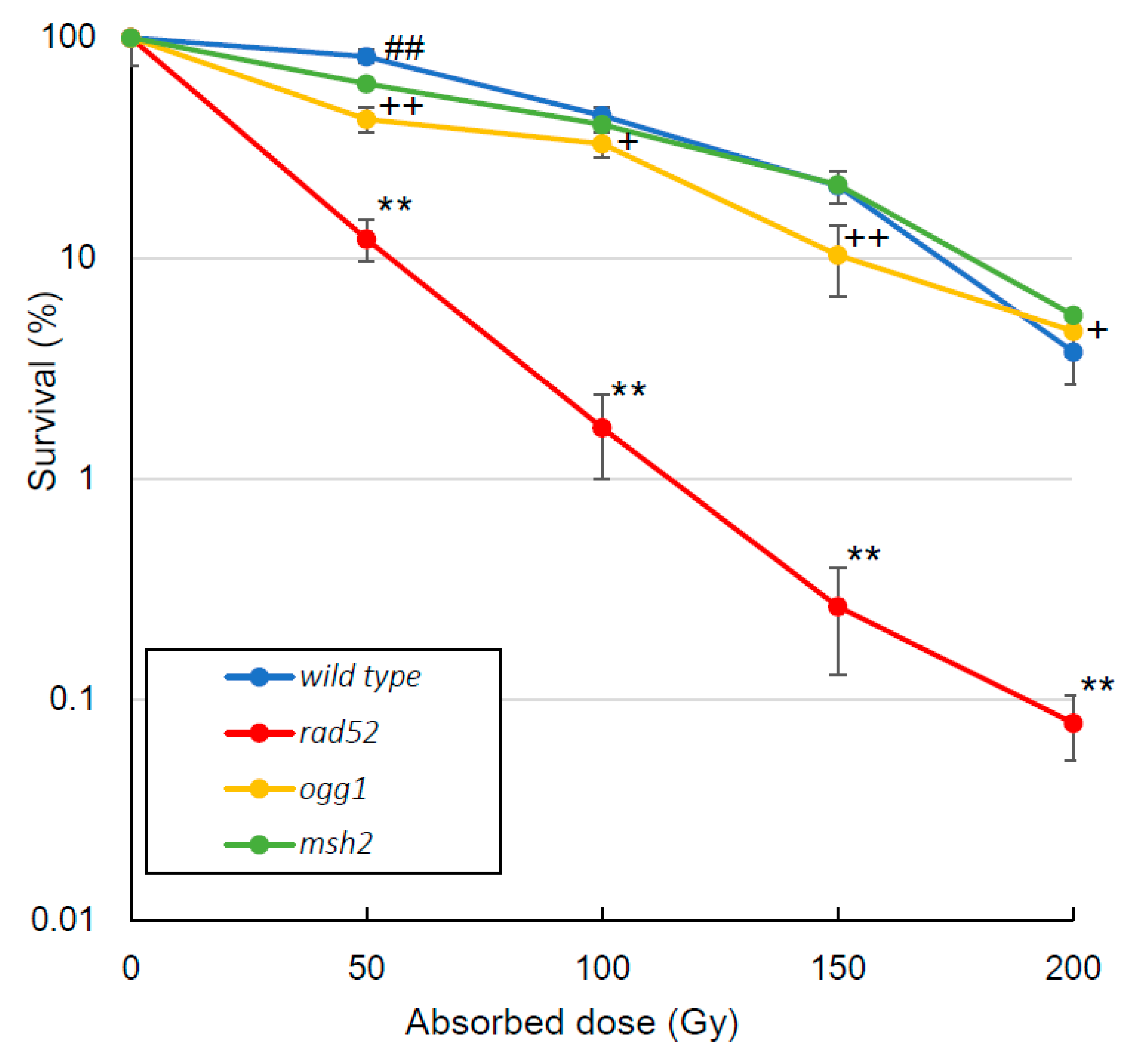
3.1. Survival Rate
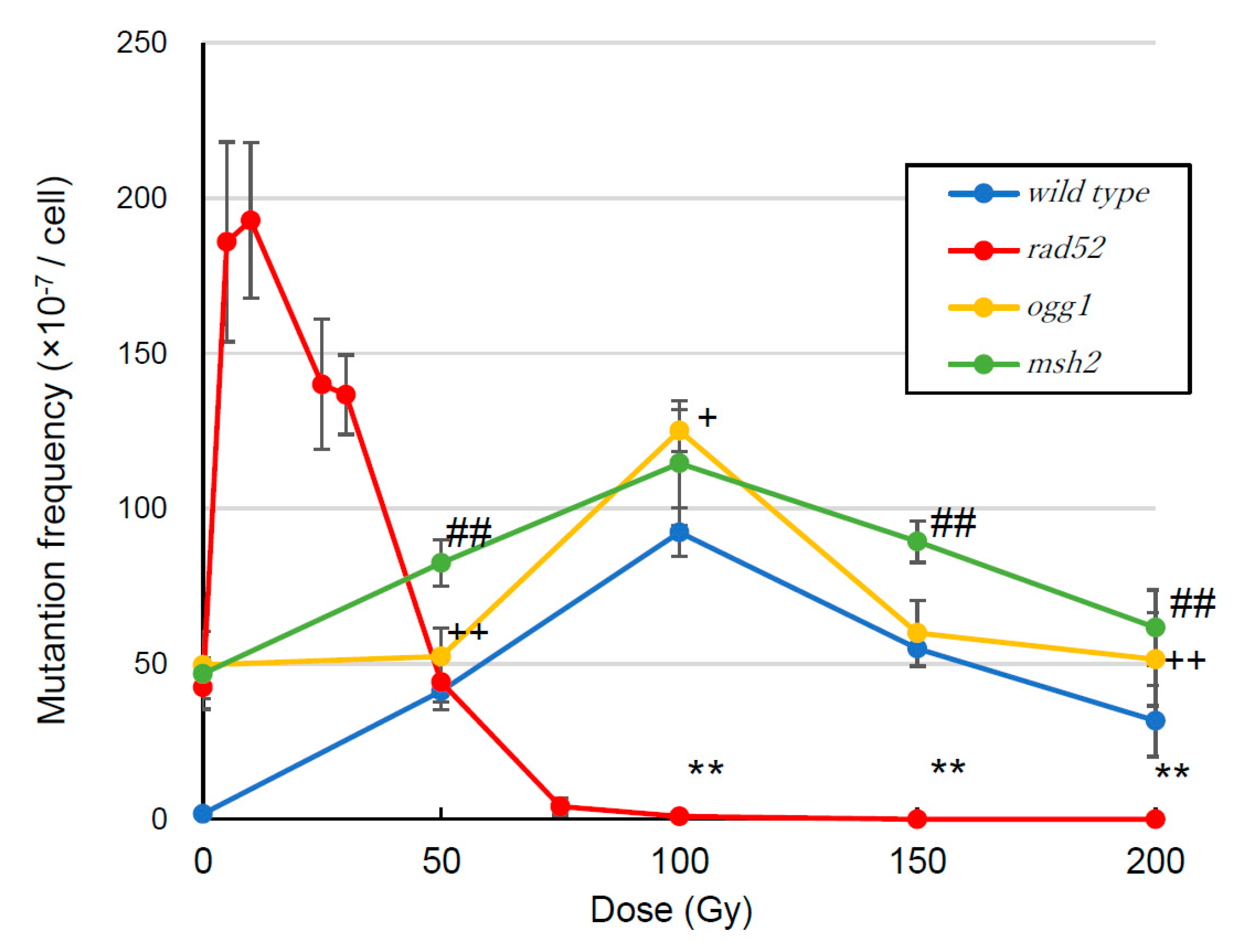
3.2. Mutation Frequency
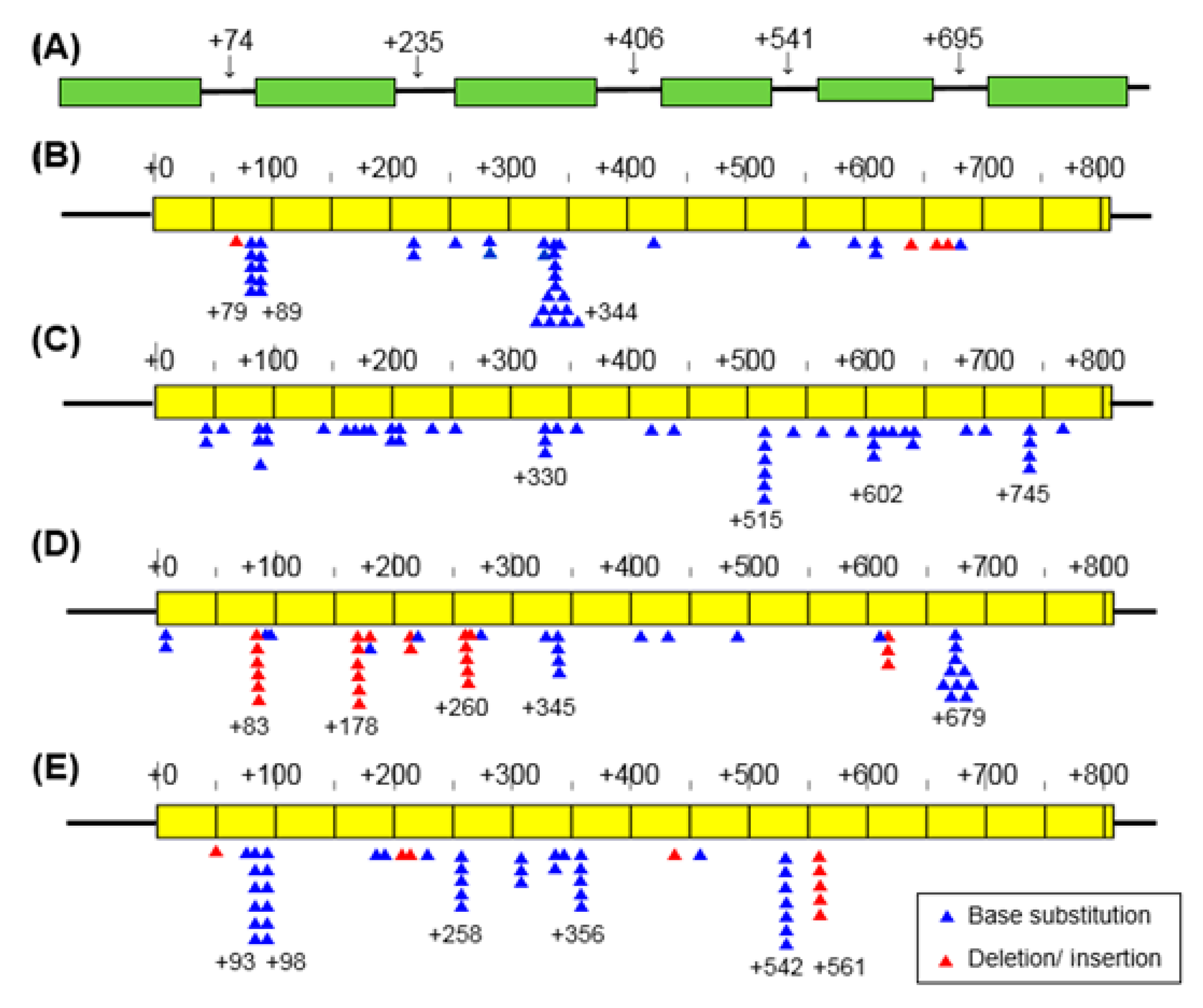
3.3. Mutation Spectrum
4. Discussion
4.1. Survival Rate
4.2. Mutation Frequency
4.3. Mutation Spectrum
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Goodhead, D.T. Mechanisms for the biological effectiveness of high-LET radiations. J. Radiat. Res. 1999, 40, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Hamada, N.; Funayama, T.; Wada, S.; Sakashita, T.; Kakizaki, T.; Ni, M.; Kobayashi, Y. LET-dependent survival of irradiated normal human fibroblasts and their descendants. Radiat. Res. 2006, 166, 24–30. [Google Scholar]
- Hase, Y.; Yamaguchi, M.; Inoue, M.; Tanaka, A. Reduction of survival and induction of chromosome aberrations in tobacco irradiated by carbon ions with different linear energy transfers. Int. J. Radiat. Biol. 2002, 78, 799–806. [Google Scholar] [CrossRef]
- Imamura, M.; Murata, T.; Akagi, K.; Tanaka, Y.; Imamura, M.; Inoue, K.; Mizuma, N.; Kobayashi, Y.; Watanabe, H.; Hachiya, M.; et al. Relationship between LET and RBE values for Escherichia coli determined using carbon ion beams from the TIARA cyclotron and HIMAC synchrotron. J. Gen. Appl. Microbiol. 1997, 43, 175–177. [Google Scholar] [CrossRef][Green Version]
- Shikazono, N.; Yokota, Y.; Kitamura, S.; Suzuki, C.; Watanabe, H.; Tano, S.; Tanaka, A. Mutation rate and novel tt mutants of Arabidopsis thaliana induced by carbon ions. Genetics 2008, 163, 1449–1455. [Google Scholar]
- Kitamura, S.; Shikazono, N.; Tanaka, A. TRANSPARENT TESTA 19 is involved in the accumulation of both anthocyanins and proanthocyanidins in Arabidopsis. Plant J. 2004, 37, 104–114. [Google Scholar] [CrossRef] [PubMed]
- Tanaka, A.; Tano, S.; Chantes, T.; Yokota, Y.; Shikazono, N.; Watanabe, H. New Arabidopsis mutant induced by ion beams affects flavonoid synthesis with spotted pigmentation in testa. Genes Genet. Syst. 1997, 72, 141–148. [Google Scholar] [CrossRef][Green Version]
- Hase, Y.; Tanaka, A.; Baba, T.; Watanabe, H. FRL1 is required for petal sepal development in Arabidopsis. Plant J. 2000, 24, 21–32. [Google Scholar]
- Okamura, M. Wide variety of flower-color and -shape mutants regenerated from leaf cultures irradiated with ion beams. Nucl. Instr. Meth. Phys. Res. B 2003, 206, 574–578. [Google Scholar] [CrossRef]
- Tanaka, A.; Nozawa, S.; Hase, Y.; Narumi, I.; Ishikawa, H.; Koike, A. Ion Beam Breeding of Rice for the Mutation Breeding Project of the Forum for Nuclear Cooperation in Asia (FNCA). QST Takasaki Ann. Rep. 2015, 2017, 103. [Google Scholar]
- Araie, H.; Hase, Y.; Oono, Y.; Suzuki, I.; Shiraiwa. Mutagenesis of the Oil-producing Algae by Ion Beam Irradiation. QST Takasaki Ann. Rep. 2015, 2017, 129. [Google Scholar]
- Matuo, Y.; Nishijima, S.; Hase, Y.; Sakamoto, A.; Tanaka, A.; Shimizu, K. Specificity of mutations induced by carbon ions in budding yeast Saccharomyces cerevisiae. Mutat. Res. 2006, 602, 7–13. [Google Scholar]
- Inoue, M.; Kamiya, H.; Fujikawa, K.; Ootsuyama, Y.; Murata-Kamiya, N.; Osaki, T.; Yasumoto, K.; Kasai, H. Induction of Chromosomal Gene Mutations in Escherichia coli by Direct Incorporation of Oxidatively Damaged Nucleotides. J. Biol. Chem. 1998, 273, 11069–11074. [Google Scholar] [CrossRef] [PubMed]
- Jackson, S.P. Sensing and repairing DNA double-strand breaks. Carcinogenesis 2002, 23, 687–696. [Google Scholar] [CrossRef] [PubMed]
- Krogh, B.O.; Symington, L.S. Recombination proteins in yeast. Annu. Rev. Genet. 2004, 38, 233–271. [Google Scholar] [CrossRef]
- Boiteux, S. Repair of 8-oxoguanine in Saccharomyces cerevisiae: interplay of DNA repair and replication mechanisms. Free Radic. Biol. Med. 2002, 32, 1244–1253. [Google Scholar]
- Güldener, U.; Heck, S.; Fielder, T.; Beinhauer, J.; Hegemann, J.H. A new efficient gene disruption cassette for repeated use in budding yeast. Nucleic Acids Res. 1996, 24, 2519–2524. [Google Scholar]
- Guthrie, C. Guide to Yeast Genetics and Molecular Biology; Academic Press: San Diego, CA, USA, 1991; pp. 302–318. [Google Scholar]
- Kurashima, S.; Satoh, T.; Saitoh, Y.; Yokota, W. Irradiation Facilities of the Takasaki Advanced Radiation Research Institute. Quantum Beam Sci. 2017, 1, 2. [Google Scholar] [CrossRef]
- Matuo, Y.; Izumi, Y.; Furusawa, Y.; Shimizu, K. Biological effects of carbon ion beams with various LETs on budding yeast Saccharomyces cerevisiae. Mutat. Res. 2018, 810, 45–51. [Google Scholar]
- Tanaka, S.; Livingstone-Zatchej, M.; Thoma, F. Chromatin structure of the yeast URA3 gene at high resolution provides insight into structure and positioning of nucleosomes in the chromosomal context. J. Mol. Biol. 1996, 257, 919–934. [Google Scholar] [CrossRef]
- Delacote, F.; Lopez, S. Importance of the cell cycle phase for the choice of the appropriate DSB repair pathway, for genome stability maintenance: The trans-S double-strand break repair model. Cell Cycle 2008, 7, 33–38. [Google Scholar] [CrossRef] [PubMed]
- Carlson, K.D.; Washington, A.T. Mechanism of efficient and accurate nucleotide incorporation opposite 7,8-dihydro-8-oxoguanine by Saccharomyces cerevisiae DNA polymerase eta. Mol. Cell. Biol. 2005, 25, 2169–2176. [Google Scholar] [CrossRef] [PubMed]
- Azzam, E.I.; Jay-Gerin, J.P.; Pain, D. Ionizing radiation-induced metabolic oxidative stress and prolonged cell injury. Cancer Lett. 2012, 327, 48–60. [Google Scholar]
- Lee, H.; Popodi, E.; Tang, H.; Foster, P.L. Rate and molecular spectrum of spontaneous mutations in the bacterium Escherichia coli as determined by whole-genome sequencing. PNAS 2012, 109, 2774–2783. [Google Scholar] [CrossRef] [PubMed]
- Kasai, H. Analysis of a form of oxidative DNA damage, 8-hydroxy-2′-deoxyguanosine, as a marker of cellular oxidative stress during carcinogenesis. Mutat. Res. 1997, 387, 147–163. [Google Scholar] [CrossRef]
- Terri, T.N. MSH2 and MSH6 Are Required for Removal of Adenine Misincorporated Opposite 8-Oxo-Guanine in S. cerevisiae. Mol. Cell 1999, 4, 439–444. [Google Scholar]
- Marsischky, G.T.; Filosi, N.; Kane, M.F.; Kolodner, R. Redundancy of Saccharomyces cerevisiae MSH3 and MSH6 in MSH2-dependent mismatch repair. Genes Dev. 1996, 10, 407–420. [Google Scholar] [CrossRef]
- Yuan, O.Z.; Mark, L.S.; David, W.H.; Dmitri, A.P. Precise estimates of mutation rate and spectrum in yeast. PNAS 2014, 111, 2310–2318. [Google Scholar]
- Sollazzo, A.; Shakeri-Manesh, S.; Fotouhi, A.; Czub, J.; Haghdoost, S.; Wojcik, A.A. Interaction of low and high LET radiation in TK6 cells—mechanistic aspects and significance for radiation protection. J. Radiol. Prot. 2016, 36, 721–735. [Google Scholar] [CrossRef]
- Wellinger, R.E.; Thoma, F. Nucleosome structure and positioning modulate nucleotide excision repair in the non-transcribed strand of an active gene. EMBO J. 1997, 16, 5046–5056. [Google Scholar] [CrossRef]
| Type of Mutation | rad52 | ogg1 | msh2 | Wild-type | ||||
|---|---|---|---|---|---|---|---|---|
| Number | Percentage | Number | Percentage | Number | Percentage | Number | Percentage | |
| Base substitution | ||||||||
| Transversions | 31 | 73.8 | 47 | 94.0 | 16 | 32.0 | 35 | 68.6 |
| G:C to T:A | 26 | 61.9 | 36 | 72.0 | 13 | 26.0 | 21 | 41.2 |
| G:C to C:G | 3 | 7.1 | 0 | 0.0 | 2 | 4.0 | 12 | 23.5 |
| A:T to C:G | 1 | 2.4 | 7 | 14.0 | 0 | 0.0 | 1 | 2.0 |
| A:T to T:A | 1 | 2.4 | 4 | 8.0 | 1 | 2.0 | 1 | 2.0 |
| Transitions | 7 | 16.6 | 3 | 6.0 | 10 | 20.0 | 7 | 13.7 |
| G:C to A:T | 7 | 16.6 | 3 | 6.0 | 9 | 18.0 | 7 | 13.7 |
| A:T to G:C | 0 | 0.0 | 0 | 0.0 | 1 | 2.0 | 0 | 0.0 |
| Deletions | 2 | 4.8 | 0 | 0.0 | 23 | 46.0 | 8 | 15.7 |
| Insertions | 2 | 4.8 | 0 | 0.0 | 1 | 2.1 | 1 | 2.0 |
| Total | 42 | 100 | 50 | 100.0 | 50 | 100.0 | 51 | 100.0 |
| Genotype | Mutation | Occurrence |
|---|---|---|
| Wild-type | A2 → A1 | 1/51 |
| C2 → C1 | 1/51 | |
| C1 → C0 | 5/51 | |
| T4 → T3 | 1/51 | |
| T5 → T6 | 1/51 | |
| rad52 | T1 → T0 | 1/42 |
| T1 → T2 | 2/42 | |
| A2 → A1 | 1/42 | |
| msh2 | A5 → A4 | 6/50 |
| A4 → A3 | 6/50 | |
| C3 → C2 | 3/50 | |
| C1 → C0 | 1/50 | |
| T6 → T5 | 6/50 | |
| T5 → T4 | 1/50 | |
| T6 → T7 | 1/50 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Matuo, Y.; Izumi, Y.; N. Sakamoto, A.; Hase, Y.; Satoh, K.; Shimizu, K. Molecular Analysis of Carbon Ion-Induced Mutations in DNA Repair-Deficient Strains of Saccharomyces cerevisiae. Quantum Beam Sci. 2019, 3, 14. https://doi.org/10.3390/qubs3030014
Matuo Y, Izumi Y, N. Sakamoto A, Hase Y, Satoh K, Shimizu K. Molecular Analysis of Carbon Ion-Induced Mutations in DNA Repair-Deficient Strains of Saccharomyces cerevisiae. Quantum Beam Science. 2019; 3(3):14. https://doi.org/10.3390/qubs3030014
Chicago/Turabian StyleMatuo, Youichirou, Yoshinobu Izumi, Ayako N. Sakamoto, Yoshihiro Hase, Katsuya Satoh, and Kikuo Shimizu. 2019. "Molecular Analysis of Carbon Ion-Induced Mutations in DNA Repair-Deficient Strains of Saccharomyces cerevisiae" Quantum Beam Science 3, no. 3: 14. https://doi.org/10.3390/qubs3030014
APA StyleMatuo, Y., Izumi, Y., N. Sakamoto, A., Hase, Y., Satoh, K., & Shimizu, K. (2019). Molecular Analysis of Carbon Ion-Induced Mutations in DNA Repair-Deficient Strains of Saccharomyces cerevisiae. Quantum Beam Science, 3(3), 14. https://doi.org/10.3390/qubs3030014






