The Differential Contribution of Macular Pigments and Foveal Anatomy to the Perception of Maxwell’s Spot and Haidinger’s Brushes
Abstract
1. Introduction
2. Materials and Methods
3. Results
3.1. Experiment 1: Horizontal Radius of MS and HB, Macular Pigment Density and OCT Morphometry
3.2. Experiment 2: Light Box Generation of MS
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Walls, G.L.; Mathews, R.W. New means of studying color blindness and normal foveal color vision, with some results and their genetical implications. Univ. Calif. Publ. Psychol. 1952, 7, 1–172. [Google Scholar]
- Miles, W.R. Comparison of functional and structural areas in human fovea. I. Method of entoptic plotting. J. Neurophysiol. 1954, 17, 22–38. [Google Scholar] [CrossRef] [PubMed]
- Maxwell, J.C. On the unequal sensibility of the Foramen Centrale to light of different colours. Brit. Assoc. Rep. 1856, 2, 12. [Google Scholar]
- Haidinger, W. Ueber das directe Erkennen des polarisirten Lights und der Lage der Polarisationsebene. Ann. Phys. 1844, 139, 29–39. [Google Scholar] [CrossRef]
- O’Shea, R.P.; Temple, S.E.; Misson, G.P.; Wade, N.J.; Bach, M. Historical context, scientific context, and translation of Haidinger’s (1844) discovery of naked-eye visibility of the polarization of light. arXiv 2020, arXiv:2010.15252. [Google Scholar]
- Misson, G.P.; Temple, S.E.; Anderson, S.J. Polarization perception in humans: On the origin of and relationship between Maxwell’s spot and Haidinger’s brushes. Sci. Rep. 2020, 10, 108. [Google Scholar] [CrossRef]
- Helmholtz, H. Treatise on Physiological Optics, 3rd ed.; Germany Version; Optical Society of America: Washington, DC, USA, 1924; Volume 2, pp. 304–308. [Google Scholar]
- Spencer, J.A. An investigation of Maxwell’s Spot. Br. J. Physiol. Opt. 1967, 24, 103–147. [Google Scholar]
- Temple, S.E.; McGregor, J.E.; Miles, C.; Graham, L.; Miller, J.; Buck, J.; Scott-Samuel, N.E.; Roberts, N.W. Perceiving polarization with the naked eye: Characterization of human polarization sensitivity. Proc. R. Soc. B 2015, 282, 20150338. [Google Scholar] [CrossRef]
- McGregor, J.; Temple, S.E.; Horváth, G. Human Polarization Sensitivity. In Polarized Light and Polarization Vision in Animal Sciences, 2nd ed.; Horváth, G., Ed.; Springer: Berlin/Heidelberg, Germany, 2014; pp. 303–315. [Google Scholar]
- Hemenger, R.P. Dichroism of the macular pigment and Haidinger’s brushes. J. Opt. Soc. Am. 1982, 72, 734–737. [Google Scholar] [CrossRef]
- Magnussen, S.; Spillmann, L.; Stürzel, F.; Werner, J.S. Filling-in of the foveal blue scotoma. Vis. Res. 2001, 41, 2961–2967. [Google Scholar] [CrossRef]
- Chen, Y.; Lan, W.; Schaeffel, F. Size of the foveal blue scotoma related to the shape of the foveal pit but not to macular pigment. Vis. Res. 2015, 106, 81–89. [Google Scholar] [CrossRef]
- Stiles, W. 18th Thomas Young Oration. The basic data of colour-matching. Phys. Soc. Year Book 1955, 44, 44–65. [Google Scholar]
- Ruddock, K.H. Evidence for macular pigmentation from colour matching data. Vis. Res. 1963, 3, 417–429. [Google Scholar] [CrossRef]
- Nussbaum, J.J.; Pruett, R.C.; Delori, F.C. Historic perspectives. Macular yellow pigment. The first 200 years. Retina 1981, 1, 296–310. [Google Scholar] [CrossRef]
- Brindley, G.S. Physiology of the Retina and Visual Pathway; Edward Arnold: London, UK, 1970. [Google Scholar]
- Delori, F.C.; Goger, D.G.; Keilhauer, C.; Salvetti, P.; Staurenghi, G. Bimodal spatial distribution of macular pigment: Evidence of a gender relationship. J. Opt. Soc. Am. A 2006, 23, 521–538. [Google Scholar] [CrossRef]
- Trezona, P.W. Rod participation in the ‘blue’ mechanism and its effect on colour matching. Vis. Res. 1970, 10, 317–332. [Google Scholar] [CrossRef]
- Isobe, K.; Motokawa, K. Functional Structure of the Retinal Fovea and Maxwell’s Spot. Nature 1955, 175, 306–307. [Google Scholar] [CrossRef]
- Howells, O.; Eperjesi, F.; Bartlett, H. Measuring macular pigment optical density in vivo: A review of techniques. Graefe’s Arch. Clin. Exp. Ophthalmol. 2011, 249, 315–347. [Google Scholar] [CrossRef]
- Delori, F.C. Autofluorescence method to measure macular pigment optical densities fluorometry and autofluorescence imaging. Arch. Biochem. Biophys. 2004, 430, 156–162. [Google Scholar] [CrossRef]
- Hammond, B.J.; Fuld, K. Interocular differences in macular pigment density. Investig. Ophthalmol. Vis. Sci. 1992, 33, 350–355. [Google Scholar]
- Hammond, J.B.R.; Wooten, B.R.; Snodderly, D.M. Individual variations in the spatial profile of human macular pigment. J. Opt. Soc. Am. A 1997, 14, 1187–1196. [Google Scholar] [CrossRef] [PubMed]
- Trieschmann, M.; Spital, G.; Lommatzsch, A.; van Kuijk, E.; Fitzke, F.; Bird, A.C.; Pauleikhoff, D. Macular pigment: Quantitative analysis on autofluorescence images. Graefe’s Arch. Clin. Exp. Ophthalmol. 2003, 241, 1006–1012. [Google Scholar] [CrossRef] [PubMed]
- Berendschot, T.T.; van Norren, D. Macular pigment shows ringlike structures. Investig. Ophthalmol. Vis. Sci. 2006, 47, 709–714. [Google Scholar] [CrossRef] [PubMed]
- Sharifzadeh, M.; Bernstein, P.S.; Gellermann, W. Nonmydriatic fluorescence-based quantitative imaging of human macular pigment distributions. J. Opt. Soc. Am. A Opt. Image Sci. Vis. 2006, 23, 2373–2387. [Google Scholar] [CrossRef] [PubMed]
- Trieschmann, M.; van Kuijk, F.J.G.M.; Alexander, R.; Hermans, P.; Luthert, P.; Bird, A.C.; Pauleikhoff, D. Macular pigment in the human retina: Histological evaluation of localization and distribution. Eye 2007, 22, 132–137. [Google Scholar] [CrossRef]
- Liew, S.H.M.; Gilbert, C.E.; Spector, T.D.; Mellerio, J.; Kuijk, F.J.V.; Beatty, S.; Fitzke, F.; Marshall, J.; Hammond, C.J. Central retinal thickness is positively correlated with macular pigment optical density. Exp. Eye Res. 2006, 82, 915–920. [Google Scholar] [CrossRef]
- van der Veen, R.L.; Ostendorf, S.; Hendrikse, F.; Berendschot, T.T. Macular pigment optical density relates to foveal thickness. Eur. J. Ophthalmol. 2009, 19, 836–841. [Google Scholar] [CrossRef]
- Nolan, J.M.; Stringham, J.M.; Beatty, S.; Snodderly, D.M. Spatial profile of macular pigment and its relationship to foveal architecture. Investig. Ophthalmol. Vis. Sci. 2008, 49, 2134–2142. [Google Scholar] [CrossRef]
- Meyer zu Westrup, V.; Dietzel, M.; Pauleikhoff, D.; Hense, H.-W. The Association of Retinal Structure and Macular Pigment Distribution. Investig. Ophthalmol. Vis. Sci. 2014, 55, 1169–1175. [Google Scholar] [CrossRef]
- Kirby, M.L.; Galea, M.; Loane, E.; Stack, J.; Beatty, S.; Nolan, J.M. Foveal Anatomic Associations with the Secondary Peak and the Slope of the Macular Pigment Spatial Profile. Investig. Ophthalmol. Vis. Sci. 2009, 50, 1383–1391. [Google Scholar] [CrossRef]
- Balaratnasingam, C.; Chae, B.; Remmer, M.H.; Gomez, E.; Suzuki, M.; Engelbert, M.; Spaide, R.F. The Spatial Profile of Macular Pigments Is Related to the Topological Characteristics of the Foveal Avascular Zone. Investig. Ophthalmol. Vis. Sci. 2015, 56, 7859–7865. [Google Scholar] [CrossRef]
- Green-Gomez, M.; Bernstein, P.S.; Curcio, C.A.; Moran, R.; Roche, W.; Nolan, J.M. Standardizing the Assessment of Macular Pigment Using a Dual-Wavelength Autofluorescence Technique. Transl. Vis. Sci. Technol. 2019, 8, 41. [Google Scholar] [CrossRef]
- Conrady, C.D.; Bell, J.P.; Besch, B.M.; Gorusupudi, A.; Farnsworth, K.; Ermakov, I.; Sharifzadeh, M.; Ermakova, M.; Gellermann, W.; Bernstein, P.S. Correlations Between Macular, Skin, and Serum Carotenoids. Investig. Ophthalmol. Vis. Sci. 2017, 58, 3616–3627. [Google Scholar] [CrossRef]
- Rasband, W.S. ImageJ. U.S. National Institutes of Health: Bethesda, MD, USA, 1997–2018. Available online: https://imagej.nih.gov/ij/ (accessed on 2 November 2022).
- Dubis, A.M.; McAllister, J.T.; Carroll, J. Reconstructing foveal pit morphology from optical coherence tomography imaging. Br. J. Ophthalmol. 2009, 93, 1223–1227. [Google Scholar] [CrossRef]
- Ctori, I.; Gruppetta, S.; Huntjens, B. The effects of ocular magnification on Spectralis spectral domain optical coherence tomography scan length. Graefes Arch. Clin. Exp. Ophthalmol. 2015, 253, 733–738. [Google Scholar] [CrossRef]
- Misson, G.P.; Temple, S.E.; Anderson, S.J. Computational simulation of Haidinger’s brushes. J. Opt. Soc. Am. A 2018, 35, 946–952. [Google Scholar] [CrossRef]
- Altman, D.G. Practical Statistics for Medical Research; Taylor & Francis: Abingdon, UK, 1990. [Google Scholar]
- Stanworth, A.; Naylor, E.J. The measurement and clinical significance of the Haidinger effect. Trans. Ophthalmol. Soc. UK 1955, 75, 67–79. [Google Scholar]
- Temple, S.E.; Roberts, N.W.; Misson, G.P. Haidinger’s brushes elicited at varying degrees of polarization rapidly and easily assesses total macular pigmentation. J. Opt. Soc. Am. A 2019, 36, B123–B131. [Google Scholar] [CrossRef]
- Misson, G.P. A Mueller matrix model of Haidinger’s brushes. Ophthal. Physiol. Opt. 2003, 23, 441–447. [Google Scholar] [CrossRef]
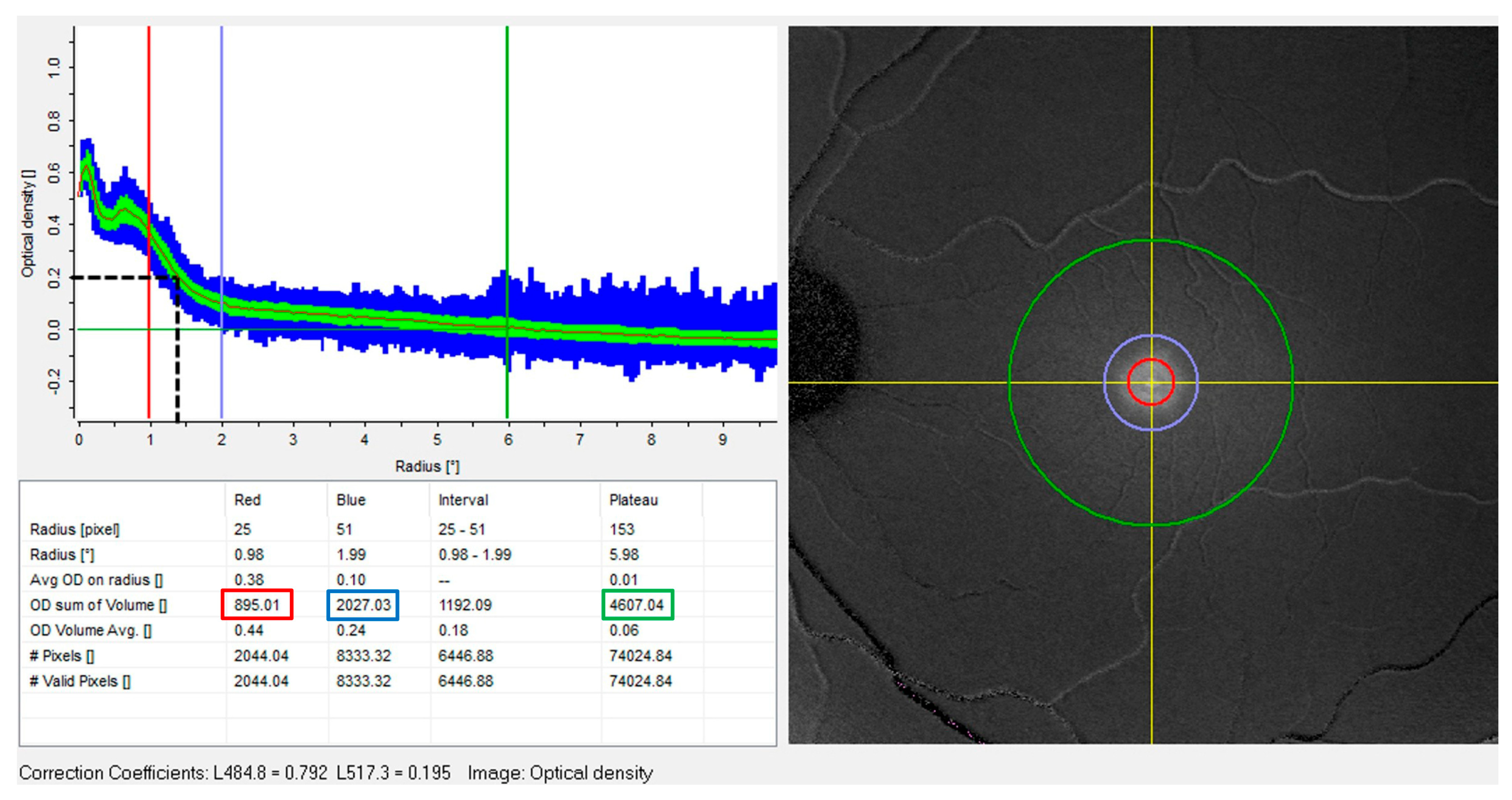


| Parameter | Description | n | Min | Max | Mean | sd |
|---|---|---|---|---|---|---|
| MSr | Maxwell spot radius (°) | 49 | 0.2 | 2.3 | 1.4 | 0.5 |
| HBr | Haidinger brush radius (°) | 48 | 0.0 | 3.0 | 1.6 | 0.7 |
| MPVc | central macular pigment volume (arbitrary units) | 49 | 25 | 101 | 56.9 | 19.0 |
| MPV1 | sum of macular pigment volume within 1° radius (arbitrary units) | 49 | 408 | 1629 | 918.9 | 331.0 |
| MPV2 | sum of macular pigment volume within 2° radius (arbitrary units) | 49 | 846 | 3843 | 2172.1 | 823.7 |
| MPV6 | sum of macular pigment volume within 6° radius (arbitrary units) | 49 | 1581 | 9500 | 5086.4 | 1983.9 |
| MPr0.2 | radius at which MPOD = 0.2 (°) | 49 | 0.60 | 2.23 | 1.50 | 0.41 |
| Ft | foveolar thickness (μm) | 49 | 199 | 272 | 229.9 | 15.1 |
| Fr | foveolar radius (°) | 49 | 0.51 | 1.13 | 0.81 | 0.16 |
| Ph | foveal pit height (μm) | 49 | 68 | 166 | 123.2 | 21.4 |
| Pr | foveal pit radius (°) | 49 | 2.80 | 5.09 | 3.96 | 0.44 |
| Spot Density Profile | Ring Density Profile | |||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Parameter | n | Min | Max | Mean | sd | n | Min | Max | Mean | sd | p | |
| Maxwell spot radius | MSr | 24 | 0.2 | 2.1 | 1.2 | 0.5 | 23 | 0.5 | 2.3 | 1.5 | 0.5 | 0.020 * |
| Haidinger brush radius | HBr | 23 | 0.0 | 2.3 | 1.5 | 0.8 | 23 | 1.1 | 3.0 | 1.9 | 0.6 | 0.004 * |
| Macular pigment optical density | ||||||||||||
| MPVc | 26 | 25 | 101 | 64.4 | 22.2 | 23 | 29 | 64 | 48.4 | 9.2 | 0.002 * | |
| MPV1 | 26 | 408 | 1629 | 971.2 | 421.4 | 23 | 452 | 1142 | 859.7 | 174.3 | 0.225 | |
| MPV2 | 26 | 846 | 3843 | 2165.4 | 1005.2 | 23 | 1116 | 3278 | 2179.8 | 576.4 | 0.951 | |
| MPV6 | 26 | 1581 | 9500 | 5188.7 | 2359.3 | 23 | 2630 | 7143 | 4970.7 | 1495.2 | 0.698 | |
| MPr0.2 | 26 | 0.60 | 2.23 | 1.43 | 0.48 | 23 | 0.77 | 2.10 | 1.57 | 0.32 | 0.229 | |
| OCT morphometry | ||||||||||||
| Foveolar thickness | Ft | 26 | 199 | 272 | 239.2 | 14.2 | 23 | 207 | 232 | 219.3 | 7.2 | <0.001 * |
| Foveolar radius | Fr | 26 | 0.51 | 1.06 | 0.76 | 0.17 | 23 | 0.67 | 1.13 | 0.87 | 0.13 | 0.010 * |
| Foveal pit height | Ph | 26 | 68 | 155 | 110.9 | 17.4 | 23 | 113 | 166 | 137.2 | 16.5 | <0.001 * |
| Foveal pit radius | Pr | 26 | 3.38 | 4.72 | 3.87 | 0.35 | 23 | 2.80 | 5.09 | 4.06 | 0.51 | 0.126 |
| Spot Perceptual Profile | Ring Perceptual Profile | |||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Parameter | n | Min | Max | Mean | sd | n | Min | Max | Mean | sd | p | |
| Maxwell spot radius | MSr | 14 | 0.2 | 2.1 | 1.3 | 0.6 | 24 | 0.5 | 2.3 | 1.6 | 0.5 | 0.136 |
| Haidinger brush radius | HBr | 14 | 0.0 | 2.3 | 1.4 | 0.9 | 25 | 0.6 | 3.0 | 1.8 | 0.6 | 0.039 * |
| Macular pigment optical density | ||||||||||||
| MPVc | 16 | 25 | 101 | 60.3 | 24.1 | 24 | 29 | 78 | 50.7 | 12.2 | 0.156 | |
| MPV1 | 16 | 408 | 1620 | 897.5 | 445.8 | 24 | 629 | 1173 | 898.6 | 168.9 | 0.992 | |
| MPV2 | 16 | 846 | 3843 | 2034.9 | 1082.1 | 24 | 1423 | 3278 | 2247.5 | 538.0 | 0.475 | |
| MPV6 | 16 | 1581 | 8800 | 5028.5 | 2400.4 | 24 | 2630 | 7143 | 5089. 7 | 1438.1 | 0.928 | |
| MPr0.2 | 16 | 0.60 | 2.15 | 1.36 | 0.52 | 24 | 1.25 | 2.10 | 1.61 | 0.27 | 0.091 | |
| OCT morphometry | ||||||||||||
| Foveolar thickness | Ft | 16 | 199 | 258 | 237.0 | 14.0 | 24 | 207 | 254 | 223.9 | 11.8 | 0.006 * |
| Foveolar radius | Fr | 16 | 0.51 | 1.06 | 0.77 | 0.18 | 24 | 0.61 | 1.13 | 0.87 | 0.15 | 0.092 |
| Foveal pit height | Ph | 16 | 87 | 134 | 109.6 | 12.8 | 24 | 94 | 166 | 133.3 | 18.3 | <0.001 * |
| Foveal pit radius | Pr | 16 | 3.48 | 4.35 | 3.79 | 0.26 | 24 | 2.80 | 5.09 | 4.09 | 0.51 | 0.017 * |
| MPOD Category | ||||||
|---|---|---|---|---|---|---|
| 0 | 1 | 2 | 3 | Sum | ||
| Chart category | 0 | 0 | 1 | 0 | 0 | 1 |
| 1 | 0 | 16 | 0 | 0 | 16 | |
| 2 | 0 | 0 | 11 | 0 | 11 | |
| 3 | 0 | 1 | 5 | 7 | 13 | |
| Sum | 0 | 18 | 16 | 7 | 41 | |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Misson, G.P.; Heitmar, R.; Armstrong, R.; Anderson, S.J. The Differential Contribution of Macular Pigments and Foveal Anatomy to the Perception of Maxwell’s Spot and Haidinger’s Brushes. Vision 2023, 7, 11. https://doi.org/10.3390/vision7010011
Misson GP, Heitmar R, Armstrong R, Anderson SJ. The Differential Contribution of Macular Pigments and Foveal Anatomy to the Perception of Maxwell’s Spot and Haidinger’s Brushes. Vision. 2023; 7(1):11. https://doi.org/10.3390/vision7010011
Chicago/Turabian StyleMisson, Gary P., Rebekka Heitmar, Richard Armstrong, and Stephen J. Anderson. 2023. "The Differential Contribution of Macular Pigments and Foveal Anatomy to the Perception of Maxwell’s Spot and Haidinger’s Brushes" Vision 7, no. 1: 11. https://doi.org/10.3390/vision7010011
APA StyleMisson, G. P., Heitmar, R., Armstrong, R., & Anderson, S. J. (2023). The Differential Contribution of Macular Pigments and Foveal Anatomy to the Perception of Maxwell’s Spot and Haidinger’s Brushes. Vision, 7(1), 11. https://doi.org/10.3390/vision7010011






