Changes in Tonic Alertness but Not Voluntary Temporal Preparation Modulate the Attention Elicited by Task-Relevant Gaze and Arrow Cues
Abstract
:1. Introduction
1.1. Measuring Attentional Effects
1.1.1. Tonic Alertness
1.1.2. Voluntary Temporal Preparation
1.2. Effects of Tonic Alertness and Voluntary Temporal Preparation on Attentional Orienting
1.3. The Present Study
2. Materials and Methods
2.1. Participants
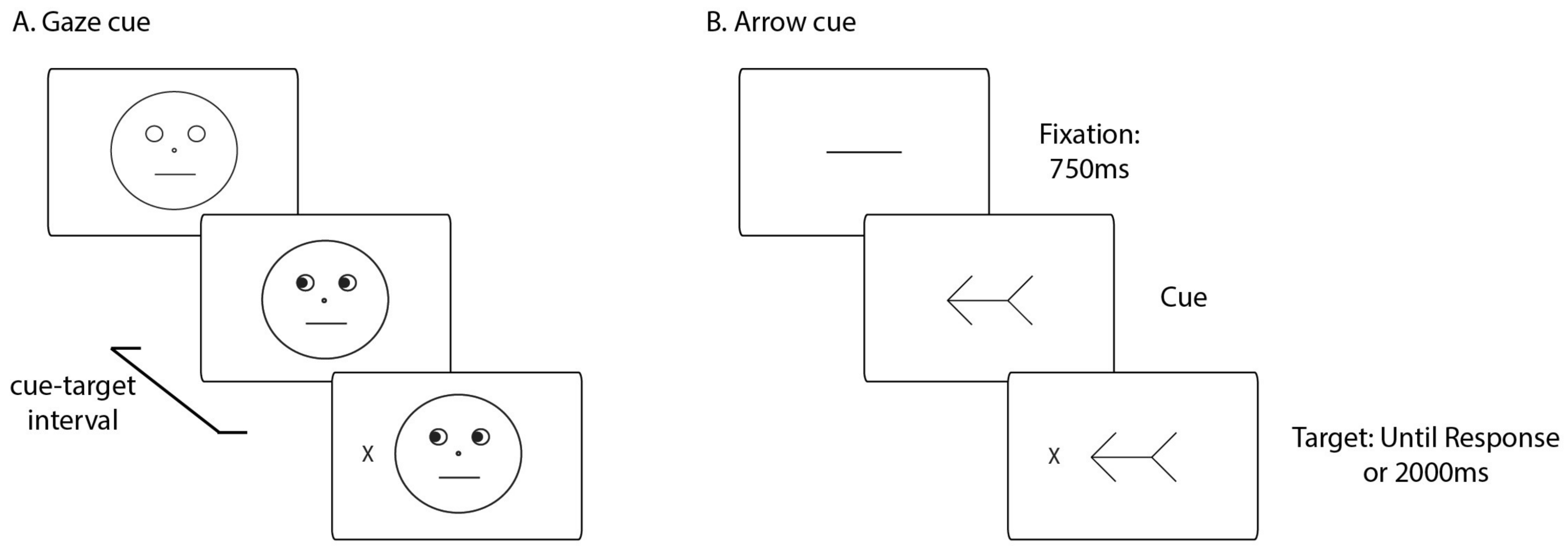
2.2. Apparatus and Stimuli
2.3. Design
2.4. Procedure
3. Results
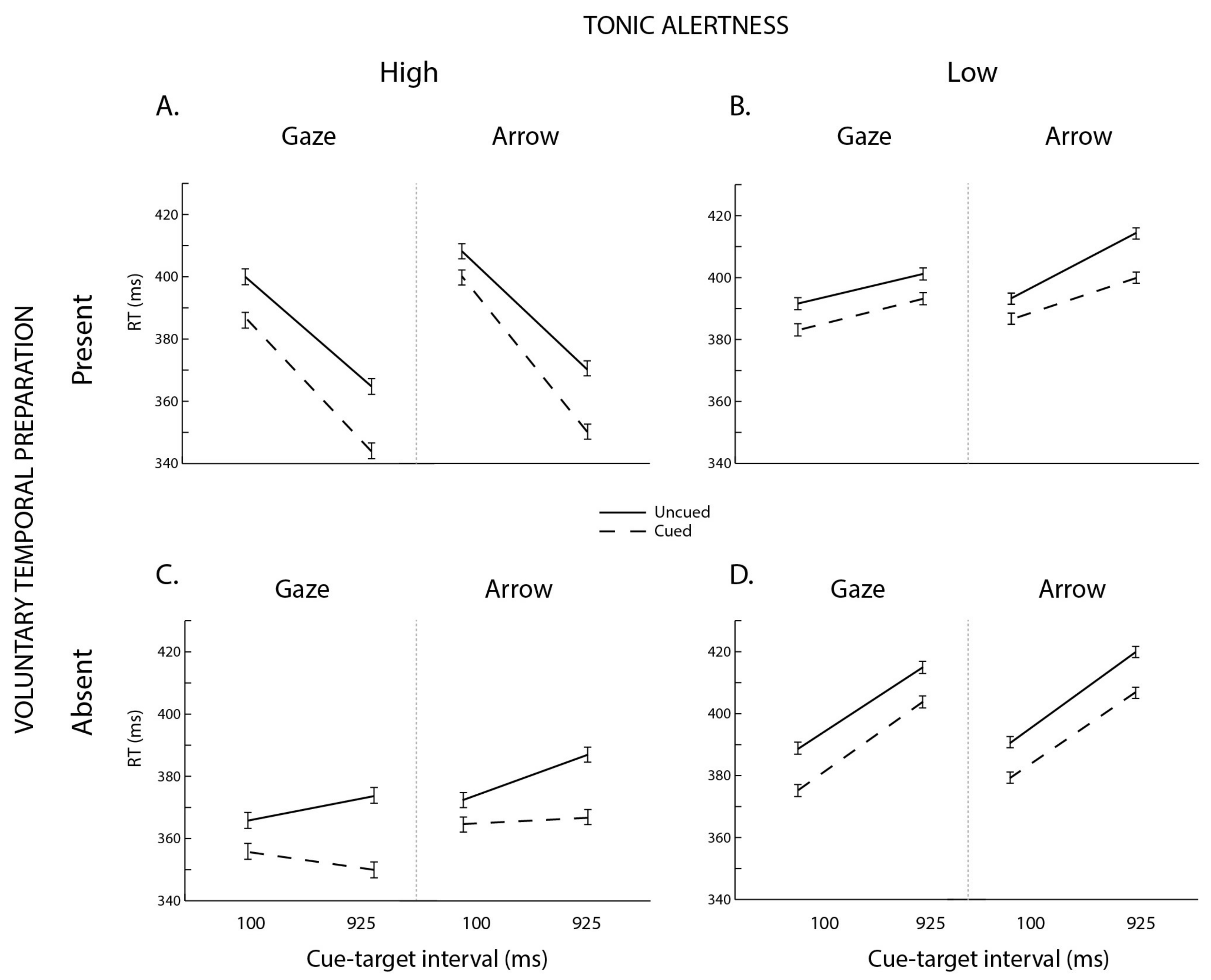
3.1. Omnibus ANOVA
3.2. Follow-Up Analyses
4. Discussion
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Dodd, M.D.; Van der Stigchel, S.; Adil Leghari, M.; Fung, G.; Kingstone, A. Attentional SNARC: There’s something special about numbers (let us count the ways). Cognition 2008, 108, 810–818. [Google Scholar] [CrossRef] [PubMed]
- Hayward, D.A.; Ristic, J. Exposing the cuing task: The case of gaze and arrow cues. Atten. Percept. Psychophys. 2015, 77, 1088–1104. [Google Scholar] [CrossRef] [PubMed]
- Posner, M.I. Orienting of attention. Q. J. Exp. Psychol. 1980, 32, 3–25. [Google Scholar] [CrossRef] [PubMed]
- Ristic, J.; Landry, M. Combining attention: A novel way of conceptualizing the links between attention, sensory processing, and behavior. Attent. Percept. Psychophys. 2015, 77, 36–49. [Google Scholar] [CrossRef] [PubMed]
- Ristic, J.; Wright, A.; Kingstone, A. The number line effect reflects top-down control. Psychon. Bull. Rev. 2006, 13, 862–868. [Google Scholar] [CrossRef] [PubMed]
- Posner, M.I.; Snyder, C.R.R.; Davidson, B.J. Attention and the detection of signals. J. Exp. Psychol. Gen. 1980, 109, 160–174. [Google Scholar] [CrossRef]
- Müller, H.J.; Rabbitt, P.M.A. Reflexive and voluntary orienting of visual attention: Time course of activation and resistance to interruption. J. Exp. Psychol. Hum. Percept. Perform. 1989, 15, 315–330. [Google Scholar] [CrossRef] [PubMed]
- Klein, R.M. Inhibition of return. Trends Cogn. Sci. 2000, 4, 138–147. [Google Scholar] [CrossRef]
- Posner, M.I.; Cohen, Y. Components of visual orienting. In Attention and Performance X: Control of Language Processes; Bouma, H., Bouwhuis, D., Eds.; Erlbaum: Hillsdale, NJ, USA, 1984; pp. 531–556. [Google Scholar]
- Driver, J.; Davis, G.; Ricciardelli, P.; Kidd, P.; Maxwell, E.; Baron-Cohen, S. Gaze perception triggers reflexive visuospatial orienting. Vis. Cogn. 1999, 6, 509–540. [Google Scholar] [CrossRef]
- Friesen, C.K.; Kingstone, A. The eyes have it! Reflexive orienting is triggered by nonpredictive gaze. Psychon. Bull. Rev. 1998, 5, 490–495. [Google Scholar] [CrossRef]
- Langton, S.R.H.; Bruce, V. Reflexive visual orienting in response to the social attention of others. Vis. Cogn. 1999, 6, 541–567. [Google Scholar] [CrossRef]
- Lambert, A.; Roser, M.; Wells, L.; Heffer, C. The spatial correspondence hypothesis and orienting in response to central and peripheral cues. Vis. Cogn. 2006, 13, 65–88. [Google Scholar] [CrossRef]
- Fischer, M.H.; Castel, A.D.; Dodd, M.D.; Pratt, J. Perceiving numbers causes spatial shifts of attention. Nat. Neurosci. 2003, 6, 555–556. [Google Scholar] [CrossRef] [PubMed]
- Gabay, S.; Henik, A. The effects of expectancy on inhibition of return. Cognition 2008, 106, 1478–1486. [Google Scholar] [CrossRef] [PubMed]
- Gabay, S.; Henik, A. Temporal expectancy modulates inhibition of return in a discrimination task. Psychon. Bull. Rev. 2010, 17, 47–51. [Google Scholar] [CrossRef] [PubMed]
- Hayward, D.A.; Ristic, J. Measuring attention using the Posner cuing paradigm: The role of across and within trial target probabilities. Front. Hum. Neurosci. 2013, 7, 205. [Google Scholar] [CrossRef] [PubMed]
- Laidlaw, K.E.W.; Kingstone, A. If not when, then where? Ignoring temporal information eliminates reflexive but not volitional spatial orienting. Vision 2017, 1, 12. [Google Scholar] [CrossRef]
- Tipper, C.M.; Kingstone, A. Is inhibition of return a reflexive effect? Cognition 2005, 97, B55–B62. [Google Scholar] [CrossRef] [PubMed]
- Weinbach, N.; Henik, A. Temporal orienting and alerting—The same or different? Front. Psychol. 2012, 3, 236. [Google Scholar] [CrossRef] [PubMed]
- Callejas, A.; Lupiáñez, J.; Funes, M.J.; Tudela, P. Modulations among the alerting, orienting and executive control networks. Exp. Brain Res. 2005, 167, 27–37. [Google Scholar] [CrossRef] [PubMed]
- Näätänen, R. Time uncertainty and occurrence uncertainty of the stimulus in a simple reaction time task. Acta Psychol. 1972, 36, 492–503. [Google Scholar] [CrossRef]
- Los, S.A. Foreperiod and sequential effects: Theory and data. In Attention and Time; Nobre, A.C., Coull, J.T., Eds.; Oxford University Press: Oxford, UK, 2010. [Google Scholar]
- Niemi, P.; Näätänen, R. Foreperiod and simple reaction time. Psychol. Bull. 1981, 89, 133–162. [Google Scholar] [CrossRef]
- Bertelson, P. Time course of preparation. Q. J. Exp. Psychol. 1967, 19, 272–279. [Google Scholar] [CrossRef] [PubMed]
- Ristic, J.; Kingstone, A. A new form of human spatial attention: Automated symbolic orienting. Vis. Cogn. 2012, 20, 244–264. [Google Scholar] [CrossRef]
- Ristic, J.; Landry, M.; Kingstone, A. Automated symbolic orienting: The missing link. Front. Psychol. 2012, 3, 560. [Google Scholar] [CrossRef] [PubMed]
- Hayward, D.A.; Ristic, J. The uniqueness of social attention revisited: Working memory load interferes with endogenous but not social orienting. Exp. Brain Res. 2013, 231, 405–414. [Google Scholar] [CrossRef] [PubMed]
- Ristic, J.; Kingstone, A. Attention to arrows: Pointing to a new direction. Q. J. Exp. Psychol. 2006, 59, 1921–1930. [Google Scholar] [CrossRef] [PubMed]
- Ristic, J.; Kingstone, A. Rethinking attentional development: Reflexive and volitional orienting in children and adults. Dev. Sci. 2009, 12, 289–296. [Google Scholar] [CrossRef] [PubMed]
- Näätänen, R. The diminishing time-uncertainty with the lapse of time after the warning signal in reaction-time experiments with varying fore-periods. Acta Psychol. 1970, 34, 399–419. [Google Scholar] [CrossRef]
- Hayward, D.A.; Ristic, J. Automated symbolic orienting is not modulated by explicit temporal attention. Acta Psychol. 2016, 171, 93–98. [Google Scholar] [CrossRef] [PubMed]
- Correa, A.; Lupiàñez, J.; Tudela, P. Attentional preparation based on temporal expectancy modulates processing at the perceptual level. Psychon. Bull. Rev. 2005, 12, 328–334. [Google Scholar] [CrossRef] [PubMed]
- Milliken, B.; Lupiáñez, J.; Roberts, M.; Stevanovski, B. Orienting in space and time: Joint contributions to exogenous spatial cuing effects. Psychon. Bull. Rev. 2003, 10, 877–883. [Google Scholar] [CrossRef] [PubMed]
- Jonides, J. Voluntary versus automatic control over the mind’s eye’s movement. In Attention and Performance; Long, J.B., Baddeley, A.D., Eds.; Erlbaum: Hillsdale, NJ, USA, 1981; pp. 187–203. [Google Scholar]
- Ristic, J.; Friesen, C.K.; Kingstone, A. Are eyes special? It depends on how you look at it. Psychon. Bull. Rev. 2002, 9, 507–513. [Google Scholar] [CrossRef] [PubMed]
- Tipples, J. Eye gaze is not unique: Automatic orienting in response to uninformative arrows. Psychon. Bull. Rev. 2002, 9, 314–318. [Google Scholar] [CrossRef] [PubMed]
- Blair, C.D.; Ristic, J. Combined attention controls complex behavior by suppressing unlikely events. Brain Cogn. 2018, 120, 17–25. [Google Scholar] [CrossRef] [PubMed]
| Target Present | No Target | ||||||||
|---|---|---|---|---|---|---|---|---|---|
| Cue–target time | 100 ms | 375 ms | 650 ms | 925 ms | |||||
| cued | uncued | cued | uncued | cued | uncued | cued | uncued | ||
| High Alertness | |||||||||
| Aging | 192 | 64 | 192 | 64 | 192 | 64 | 192 | 64 | 64 |
| Nonaging | 384 | 128 | 192 | 64 | 96 | 32 | 48 | 16 | 64 |
| Low Alertness | |||||||||
| Aging | 240 | 80 | -- | -- | -- | -- | 240 | 80 | 216 |
| Nonaging | 336 | 112 | -- | -- | -- | -- | 168 | 56 | 224 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hayward, D.A.; Ristic, J. Changes in Tonic Alertness but Not Voluntary Temporal Preparation Modulate the Attention Elicited by Task-Relevant Gaze and Arrow Cues. Vision 2018, 2, 18. https://doi.org/10.3390/vision2020018
Hayward DA, Ristic J. Changes in Tonic Alertness but Not Voluntary Temporal Preparation Modulate the Attention Elicited by Task-Relevant Gaze and Arrow Cues. Vision. 2018; 2(2):18. https://doi.org/10.3390/vision2020018
Chicago/Turabian StyleHayward, Dana A., and Jelena Ristic. 2018. "Changes in Tonic Alertness but Not Voluntary Temporal Preparation Modulate the Attention Elicited by Task-Relevant Gaze and Arrow Cues" Vision 2, no. 2: 18. https://doi.org/10.3390/vision2020018




