If not When, then Where? Ignoring Temporal Information Eliminates Reflexive but not Volitional Spatial Orienting
Abstract
:1. Introduction
1.1. Spatial Orienting to Central Cues can be Reflexive as well as Volitional
1.2. Attention in the Temporal Domain
1.3. The Influence of Attending to Time on Spatial Attention Effects
1.4. The Present Study
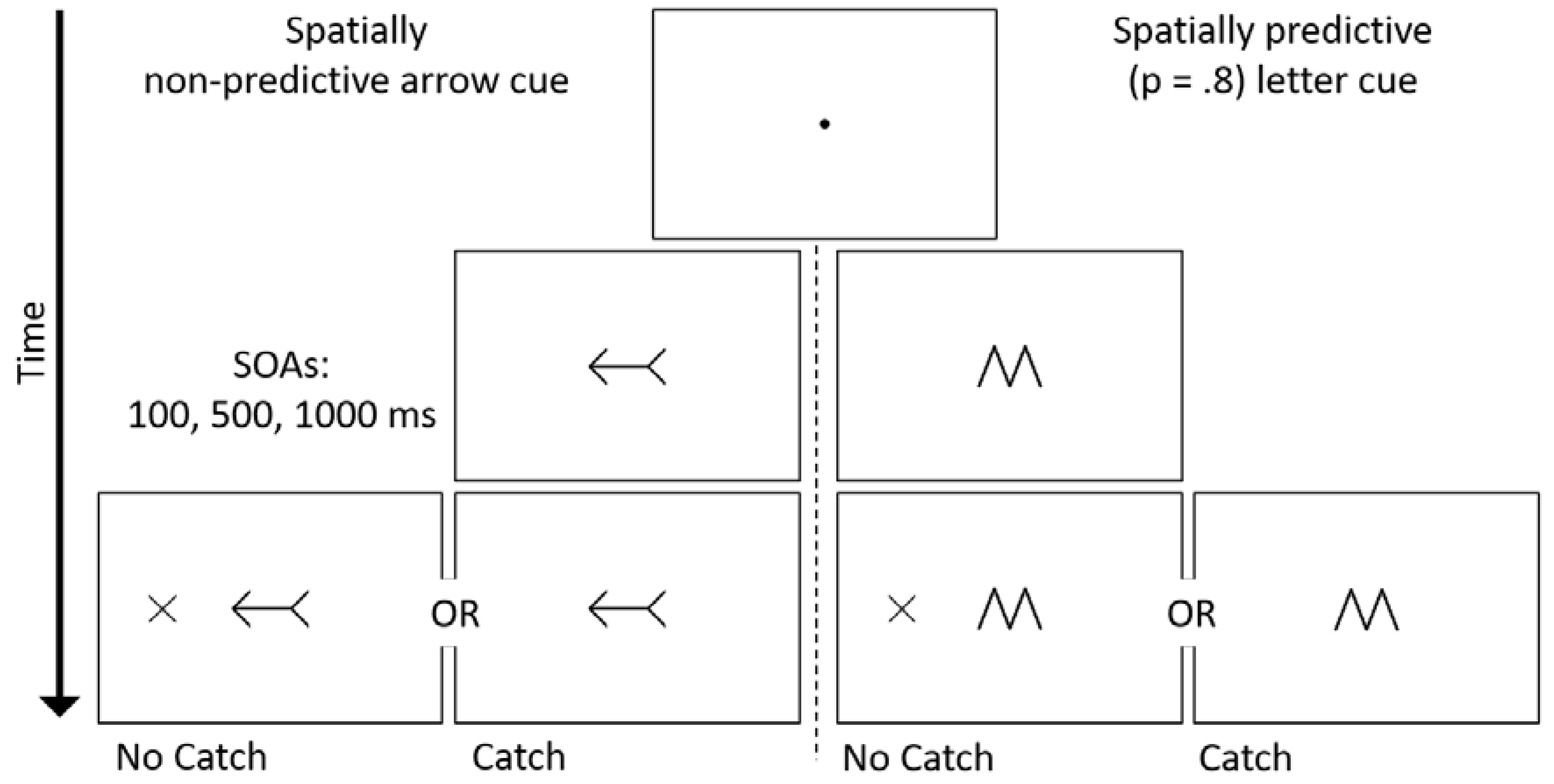
2. Methods
2.1. Participants
2.2. Stimuli and Procedure
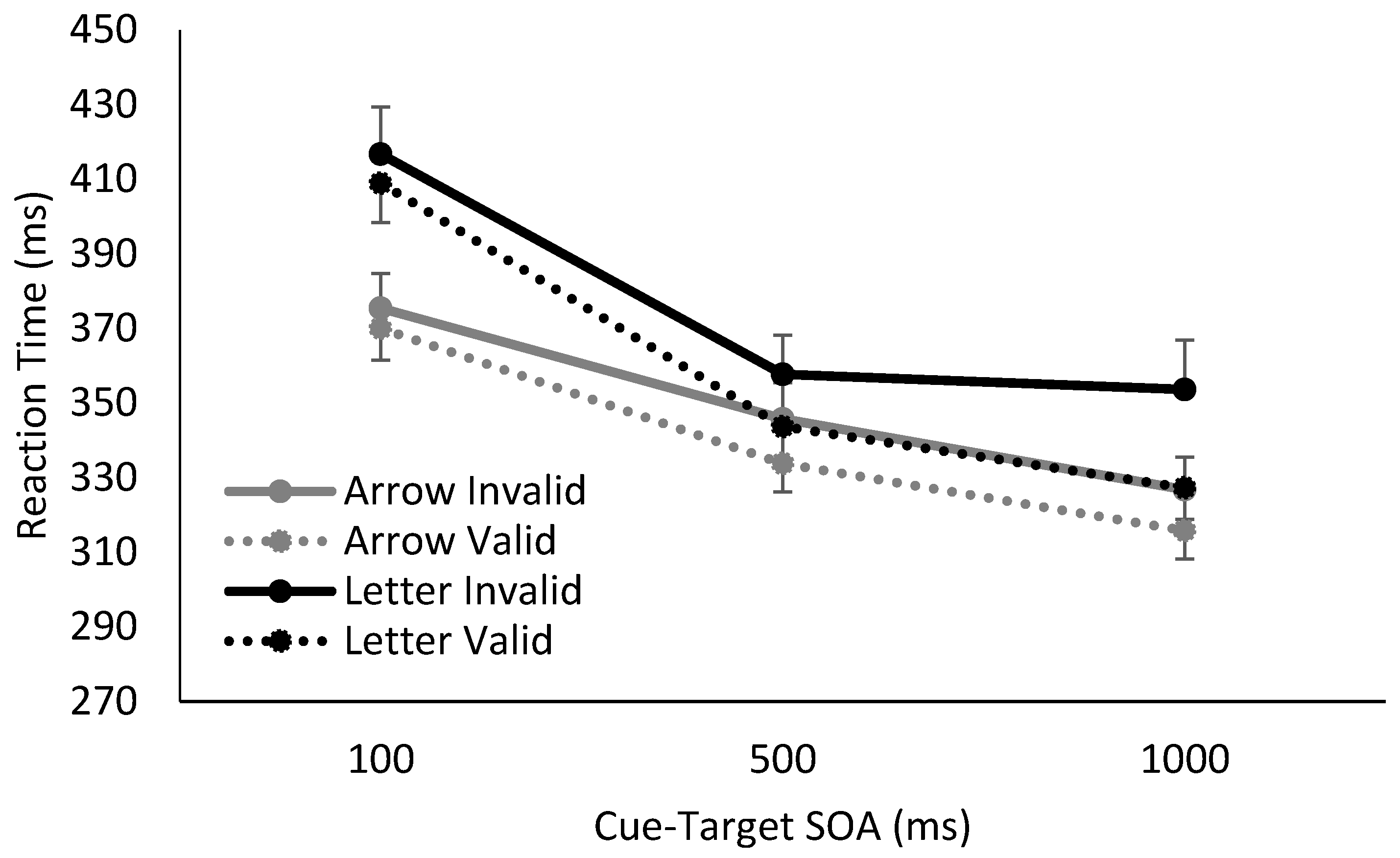
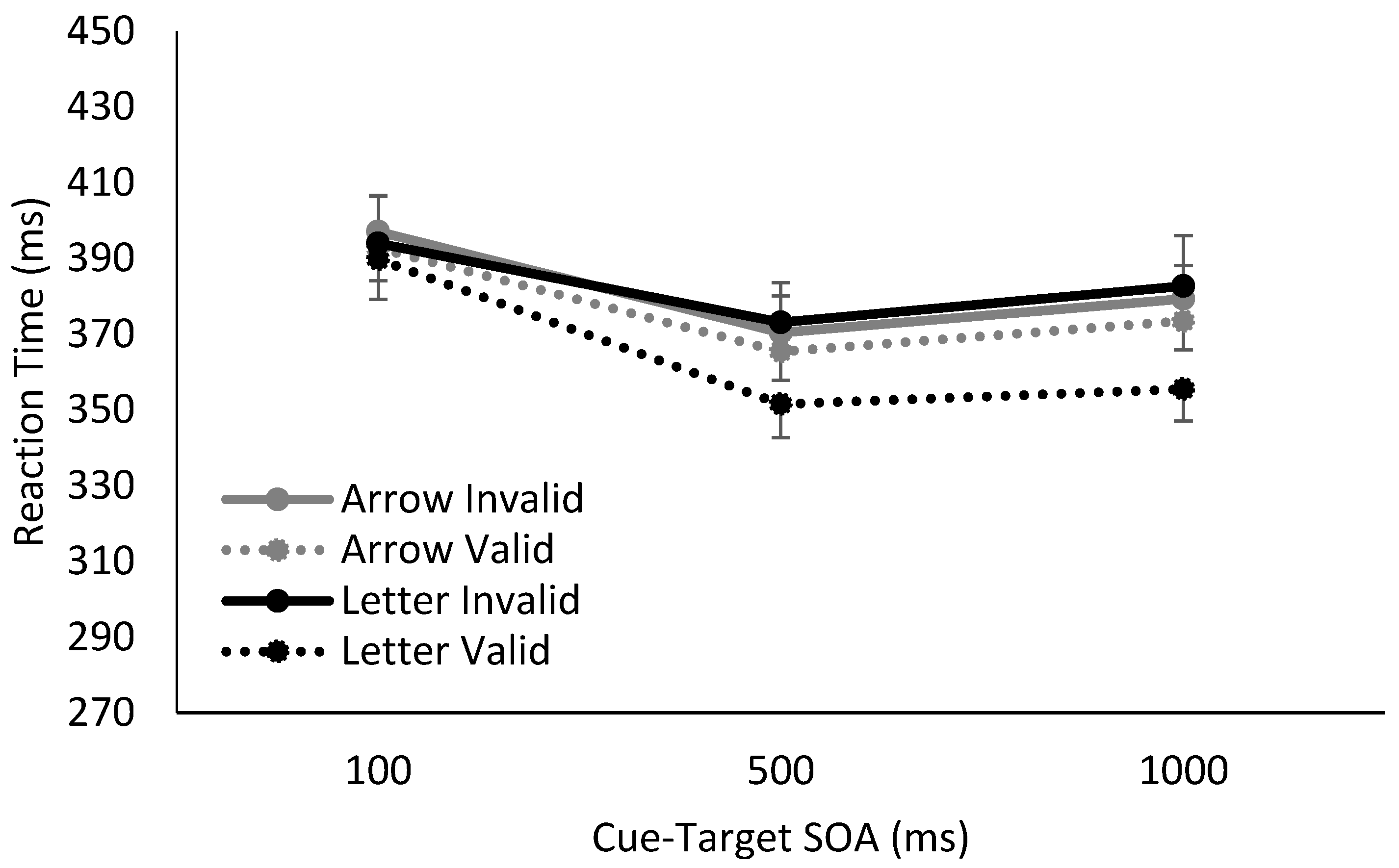
3. Results
4. Discussion
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Coull, J.T.; Nobre, A.C. Where and when to pay attention: The neural systems for directing attention to spatial locations and to time intervals as revealed by both PET and fMRI. J. Neurosci. 1998, 18, 7426–7435. [Google Scholar] [PubMed]
- Kubovy, M. Concurrent-Pitch Segregation and the Theory of Indispensable Attributes. In Perceptual Organization; Kubovy, M., Pomerantz, J.R., Eds.; Lawrence Erlbaum Associates: Hillsdale, NJ, USA, 1981; pp. 55–98. [Google Scholar]
- Desimone, R.; Duncan, J. Neural mechanisms of selective visual attention. Annu. Rev. Neurosci. 1995, 18, 193–222. [Google Scholar] [CrossRef] [PubMed]
- Correa, Á.; Lupiáñez, J.; Tudela, P. Attentional preparation based on temporal expectancy modulates processing at the perceptual level. Psychon. Bull. Rev. 2005, 12, 328–334. [Google Scholar] [CrossRef] [PubMed]
- Kingstone, A. Combining expectancies. Q. J. Exp. Psychol. 1992, 44, 69–104. [Google Scholar] [CrossRef]
- Moran, J.; Desimone, R. Selective attention gates visual processing in the extrastriate cortex. Science 1985, 229, 782–784. [Google Scholar] [CrossRef] [PubMed]
- Motter, B.C. Focal attention produces spatially selective processing in visual cortical areas V V2, and V4 in the presence of competing stimuli. J. Neurophysiol. 1993, 70, 909–919. [Google Scholar] [PubMed]
- Nobre, A.C. Orienting attention to instants in time. Neuropsychologia 2001, 39, 1317–1328. [Google Scholar] [CrossRef]
- Posner, M.I. Orienting of attention. Q. J. Exp. Psychol. 1980, 32, 3–25. [Google Scholar] [CrossRef] [PubMed]
- Posner, M.I.; Cohen, Y. Components of Visual Orienting. In Attention and Percormance X: Control of Language Processes; Bouma, H., Bouwhuis, D.G., Eds.; Erlbaum: Hillsdale, NJ, USA, 1984; pp. 531–556. [Google Scholar]
- Abrams, R.A.; Christ, S.E. Motion onset captures attention. Psychol. Sci. 2003, 14, 427–432. [Google Scholar] [CrossRef] [PubMed]
- Franconeri, S.L.; Simons, D.J. Moving and looming stimuli capture attention. Percept. Psychophys. 2003, 65, 999–1010. [Google Scholar] [CrossRef] [PubMed]
- Yantis, S.; Jonides, J.J. Attentional capture by abrupt onsets: New perceptual objects or visual masking? J. Exp. Psychol. Hum. Percept. Perform. 1996, 22, 1505–1513. [Google Scholar] [CrossRef] [PubMed]
- Jonides, J.J. Towards a model of the mind’s eye’s movement. Can. J. Psychol. Can. Psychol. 1980, 34, 103–112. [Google Scholar] [CrossRef]
- Brignani, D.; Guzzon, D.; Marzi, C.A.; Miniussi, C. Attentional orienting induced by arrows and eye-gaze compared with an endogenous cue. Neuropsychologia 2008, 47, 370–381. [Google Scholar] [CrossRef] [PubMed]
- Friesen, C.K.; Kingstone, A. The eyes have it! Reflexive orienting is triggered by nonpredictive gaze. Psychon. Bull. Rev. 1998, 5, 490–495. [Google Scholar] [CrossRef]
- Kuhn, G.; Kingstone, A. Look away! Eyes and arrows engage oculomotor responses automatically. Atten. Percept. Psychophys. 2009, 71, 314–327. [Google Scholar] [CrossRef] [PubMed]
- Langton, S.R.H.; Bruce, V. Reflexive visual orienting in response to the social attention of others. Vis. Cogn. 1999, 6, 541–567. [Google Scholar] [CrossRef]
- Ristic, J.; Friesen, C.K.; Kingstone, A. Are eyes special? It depends on how you look at it. Psychon. Bull. Rev. 2002, 9, 507–513. [Google Scholar] [CrossRef] [PubMed]
- Senju, A.; Tojo, Y.; Dairoku, H.; Hasegawa, T. Reflexive orienting in response to eye gaze and an arrow in children with and without autism. J. Child. Psychol. Psychiatry 2004, 45, 445–458. [Google Scholar] [CrossRef] [PubMed]
- Tipples, J. Eye gaze is not unique: Automatic orienting in response to uninformative arrows. Psychon. Bull. Rev. 2002, 9, 314–318. [Google Scholar] [CrossRef] [PubMed]
- O’Malley, S.; Besner, D. Is eye gaze direction always determined without intent? Psychon. Bull. Rev. 2014, 21, 1495–1500. [Google Scholar] [CrossRef] [PubMed]
- Johnson, M.H.; Posner, M.I.; Rothbart, M.K. Components of visual orienting in early infancy: Contingency learning, anticipatory looking, and disengaging. J. Cogn. Neurosci. 1991, 3, 335–344. [Google Scholar] [CrossRef] [PubMed]
- Guzzon, D.; Brignani, D.; Miniussi, C.; Marzi, C.A. Orienting of attention with eye and arrow cues and the effect of overtraining. Acta Psychol. (Amst) 2010, 134, 353–362. [Google Scholar] [CrossRef] [PubMed]
- Hommel, B.; Pratt, J.; Colzato, L.; Godijn, R. Symbolic control of visual attention. Psychol. Sci. 2001, 12, 360–365. [Google Scholar] [CrossRef] [PubMed]
- Ristic, J.; Kingstone, A. A new form of human spatial attention: Automated symbolic orienting. Vis. Cogn. 2012, 20, 244–264. [Google Scholar] [CrossRef]
- Ristic, J.; Landry, M.; Kingstone, A. Automated symbolic orienting: The missing link. Front. Psychol. 2012, 3, 560. [Google Scholar] [CrossRef] [PubMed]
- Eimer, M. Uninformative symbolic cues may bias visual-spatial attention: Behavioral and electrophysiological evidence. Biol. Psychol. 1997, 46, 67–71. [Google Scholar] [CrossRef]
- Gibson, B.S.; Bryant, T.A. Variation in cue duration reveals top-down modulation of involuntary orienting to uninformative symbolic cues. Percept. Psychophys. 2005, 65, 749–758. [Google Scholar] [CrossRef]
- Dodd, M.D.; Van der Stigchel, S.; Adil Leghari, M.; Fung, G.; Kingstone, A. Attentional SNARC: There’s something special about numbers (let us count the ways). Cognition 2008, 108, 810–818. [Google Scholar] [CrossRef] [PubMed]
- Duncan, J. Boundary conditions on parallel processing in human vision. Perception 1989, 18, 457–469. [Google Scholar] [CrossRef] [PubMed]
- Folk, C.L.; Remington, R.W.; Johnston, J.C. Involuntary covert orienting is contingent on attentional control settings. J. Exp. Psychol. Hum. Percept. Perform. 1992, 18, 1030–1044. [Google Scholar] [CrossRef] [PubMed]
- Niemi, P.; Näätänen, R. Foreperiod and simple reaction time. Psychol. Bull. 1981, 89, 133–162. [Google Scholar] [CrossRef]
- Woodrow, H. The measurement of attention. Psychol. Monogr. 1914, 5, 1–158. [Google Scholar] [CrossRef]
- Bertelson, P. The time course of preparation. Q. J. Exp. Psychol. 1967, 19, 272–279. [Google Scholar] [CrossRef] [PubMed]
- Coull, J.T.; Frith, C.D.; Büchel, C.; Nobre, A.C. Orienting attention in time: Behavioural and neuroanatomical distinction between exogenous and endogenous shifts. Neuropsychologia 2000, 38, 808–819. [Google Scholar] [CrossRef]
- Los, S.A.; Agter, F. Reweighting sequential effects across different distributions of foreperiods: Segregating elementary contributions to nonspecific preparation. Percept. Psychophys. 2005, 67, 1161–1170. [Google Scholar] [CrossRef] [PubMed]
- Gabay, S.; Henik, A. Temporal expectancy modulates inhibition of return in a discrimination task. Psychon. Bull. Rev. 2010, 17, 47–51. [Google Scholar] [CrossRef] [PubMed]
- Gabay, S.; Henik, A. The effects of expectancy on inhibition of return. Cognition 2007, 106, 1478–1486. [Google Scholar] [CrossRef] [PubMed]
- Callejas, A.; Lupiáñez, J.; Funes, M.J.; Tudela, P. Modulations among the alerting, orienting and executive control networks. Exp. Brain Res. 2005, 167, 27–37. [Google Scholar] [CrossRef] [PubMed]
- Näätänen, R. Time uncertainty and occurrence uncertainty of the stimulus in a simple reaction time task. Acta Psychol. 1972, 36, 492–503. [Google Scholar] [CrossRef]
- Tipper, C.; Kingstone, A. Is inhibition of return a reflexive effect? Cognition 2005, B55–B62. [Google Scholar] [CrossRef] [PubMed]
- Hayward, D.A.; Ristic, J. Exposing the cuing task: The case of gaze and arrow cues. Atten. Percept. Psychophys. 2015, 77, 1088–1104. [Google Scholar] [CrossRef] [PubMed]
- Alegria, J. Sequential effects of foreperiod duration: Some strategical factors in tasks involving time uncertainty. In Attention and Performance, V; Rabbit, P.M.A., Dornic, S., Eds.; Academic Press: London, UK, 1975; pp. 1–10. [Google Scholar]
- Nobre, A.C.; Coull, J.T. Attention and Time; Oxford University Press: New York, NY, USA, 2010. [Google Scholar]
- Hayward, D.A.; Ristic, J. Automated symbolic orienting is not modulated by explicit temporal attention. Acta Psychol. 2016, 171, 93–98. [Google Scholar] [CrossRef] [PubMed]
- Karlin, L. Reaction time as a function of foreperiod duration and variability. J. Exp. Psychol. 1959, 58, 185–191. [Google Scholar] [CrossRef] [PubMed]
- Weinbach, N.; Shofty, I.; Gabay, S.; Henik, A. Endogenous temporal and spatial orienting: Evidence for two distinct attentional mechanisms. Psychon. Bull. Rev. Rev. 2015, 22, 967–973. [Google Scholar] [CrossRef] [PubMed]
- Fischer, M.H.; Castel, A.D.; Dodd, M.D.; Pratt, J. Perceiving numbers causes spatial shifts of attention. Nat. Neurosci. 2003, 6, 555–556. [Google Scholar] [CrossRef] [PubMed]
- Müller, H.J.; Rabbitt, P.M. Reflexive and voluntary orienting of visual attention: Time course of activation and resistance to interruption. J. Exp. Psychol. Hum. Percept. Perform. 1989, 15, 315–330. [Google Scholar] [CrossRef] [PubMed]
- Carrasco, M. Visual attention: The past 25 years. Vis. Res. 2011, 51, 1484–1525. [Google Scholar] [CrossRef] [PubMed]
- Ristic, J.; Kingstone, A. Attention to arrows: pointing to a new direction. Q. J. Exp. Psychol. 2006, 59, 1921–1930. [Google Scholar] [CrossRef] [PubMed]
- Ristic, J.; Wright, A.; Kingstone, A. Attentional control and reflexive orienting to gaze and arrow cues. Psychon. Bull. Rev. 2007, 14, 964–969. [Google Scholar] [CrossRef] [PubMed]
- Gabay, S.; Chica, A.B.; Charras, P.; Funes, M.J.; Henik, A. Cue and target processing modulate the onset of inhibition of return. J. Exp. Psychol. Hum. Percept. Perform. 2012, 38, 42–52. [Google Scholar] [CrossRef] [PubMed]
- Olk, B.; Tsankova, E.; Raisa Petca, A.; Wilhelm, A.F.X. Measuring effects of voluntary attention: A comparison among predictive arrow, colour, and number cues. Q. J. Exp. Psychol. 2014, 67, 2025–2041. [Google Scholar] [CrossRef] [PubMed]
- Olk, B.; Cameron, B.; Kingstone, A. Enhanced orienting effects: Evidence for an interaction principle. Vis. Cogn. 2008, 16, 979–1000. [Google Scholar] [CrossRef]
- Corbetta, M.; Patel, G.; Shulman, G.L. The Reorienting System of the Human Brain: From Environment to Theory of Mind. Neuron 2008, 58, 306–324. [Google Scholar] [CrossRef] [PubMed]
| 1 | The manipulation of the cue’s temporal reliability also appears to have impacted RT at the shortest SOA, with the longest RTs observed when the amount of cue information to be processed was greatest, i.e., when spatially predictive letter cues were also temporally informative. |
| SOA (ms) | Probability of Target Appearance | |
|---|---|---|
| No Catch: No-Target Trials Absent | Catch: No-Target Trials Present | |
| 100 | 0.33 | 0.25 |
| 500 | 0.50 | 0.33 |
| 1000 | 1.00 | 0.50 |
| Target absent | n/a | 0.00 |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Laidlaw, K.E.W.; Kingstone, A. If not When, then Where? Ignoring Temporal Information Eliminates Reflexive but not Volitional Spatial Orienting. Vision 2017, 1, 12. https://doi.org/10.3390/vision1020012
Laidlaw KEW, Kingstone A. If not When, then Where? Ignoring Temporal Information Eliminates Reflexive but not Volitional Spatial Orienting. Vision. 2017; 1(2):12. https://doi.org/10.3390/vision1020012
Chicago/Turabian StyleLaidlaw, Kaitlin E. W., and Alan Kingstone. 2017. "If not When, then Where? Ignoring Temporal Information Eliminates Reflexive but not Volitional Spatial Orienting" Vision 1, no. 2: 12. https://doi.org/10.3390/vision1020012
APA StyleLaidlaw, K. E. W., & Kingstone, A. (2017). If not When, then Where? Ignoring Temporal Information Eliminates Reflexive but not Volitional Spatial Orienting. Vision, 1(2), 12. https://doi.org/10.3390/vision1020012






