Increased Short-Term Fluctuation in Optic Nerve Head Blood Flow in a Case of Normal-Tension Glaucoma by the Use of Laser Speckle Flowgraphy
Abstract
:1. Introduction
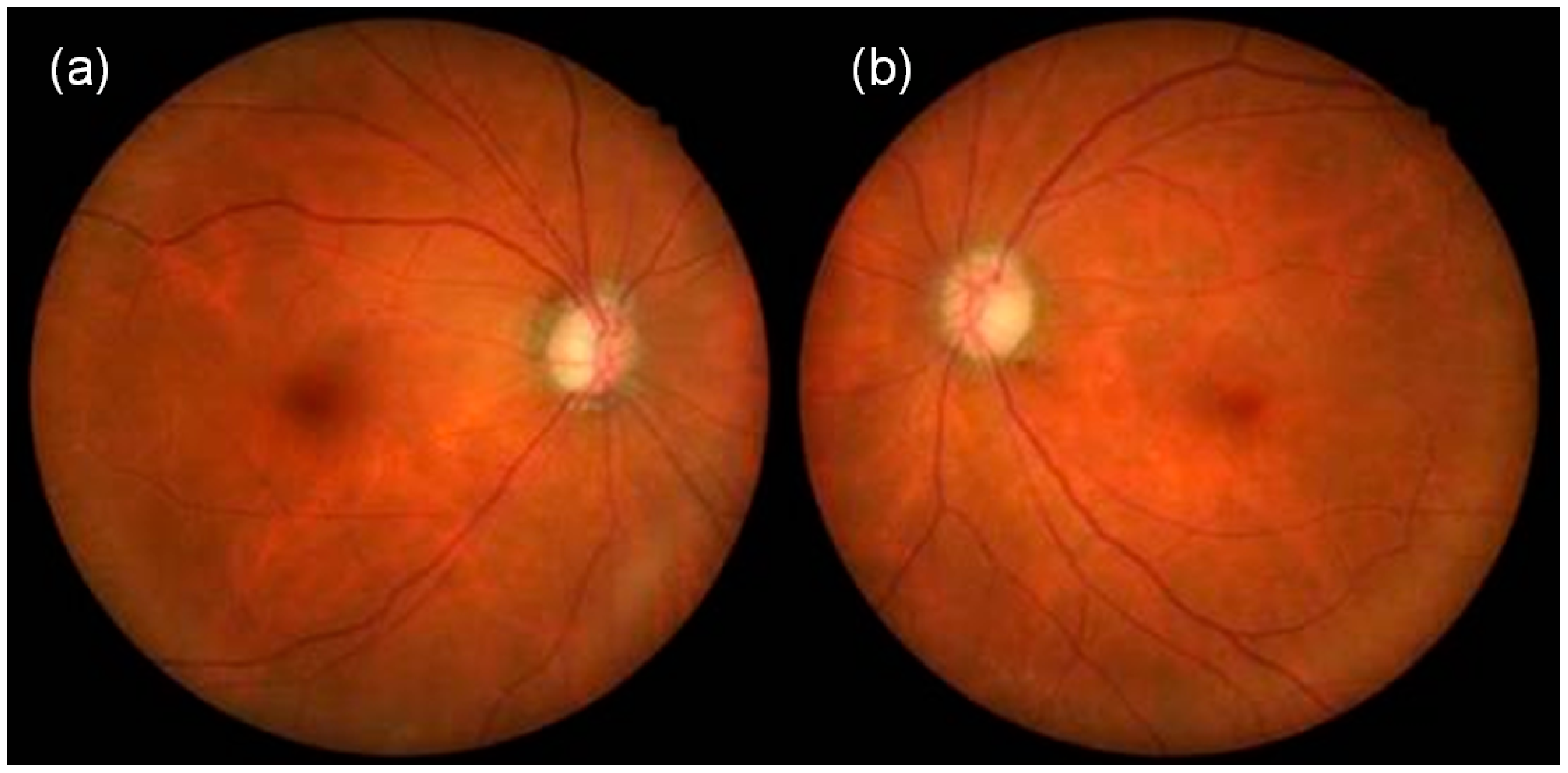
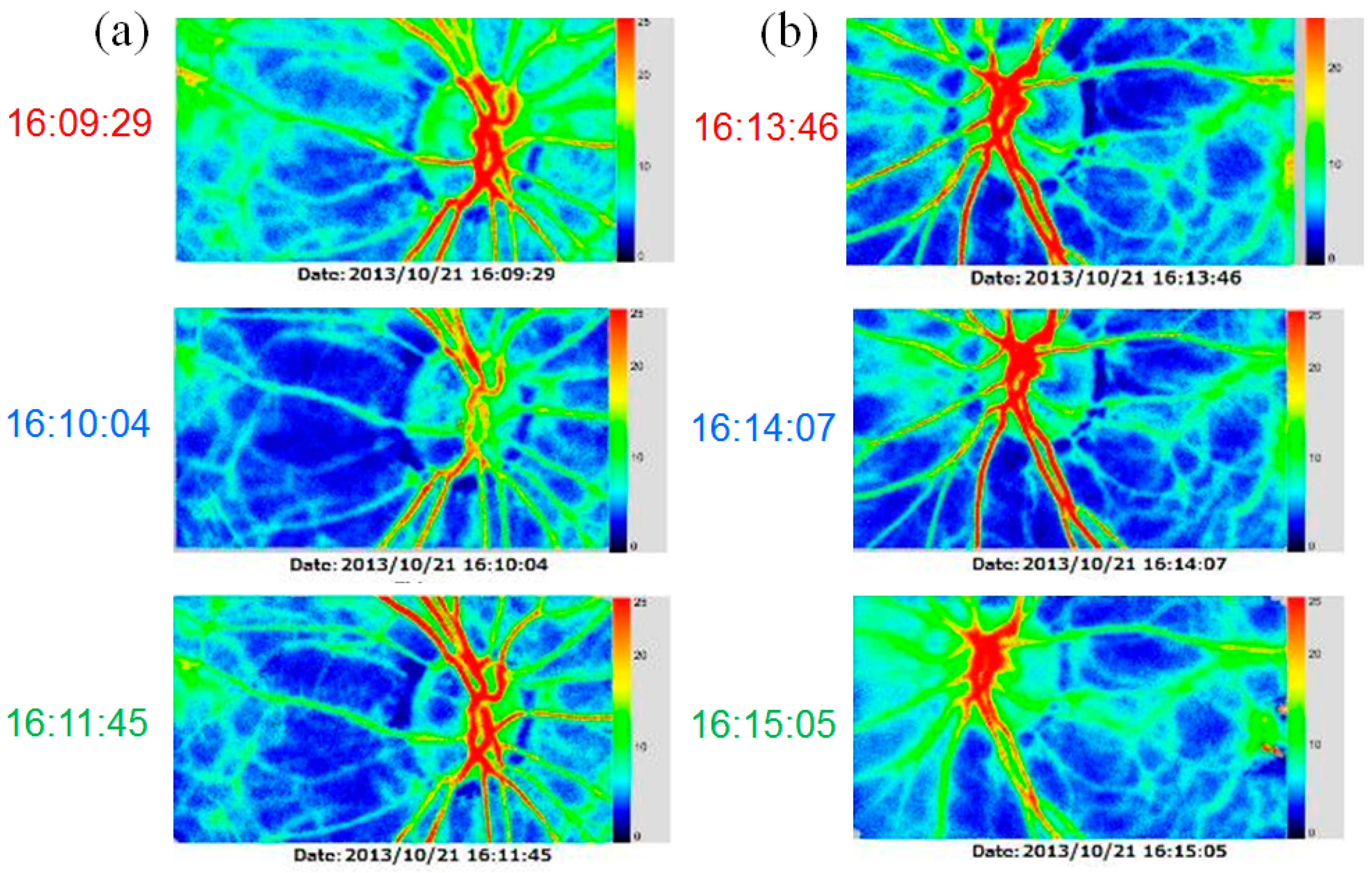
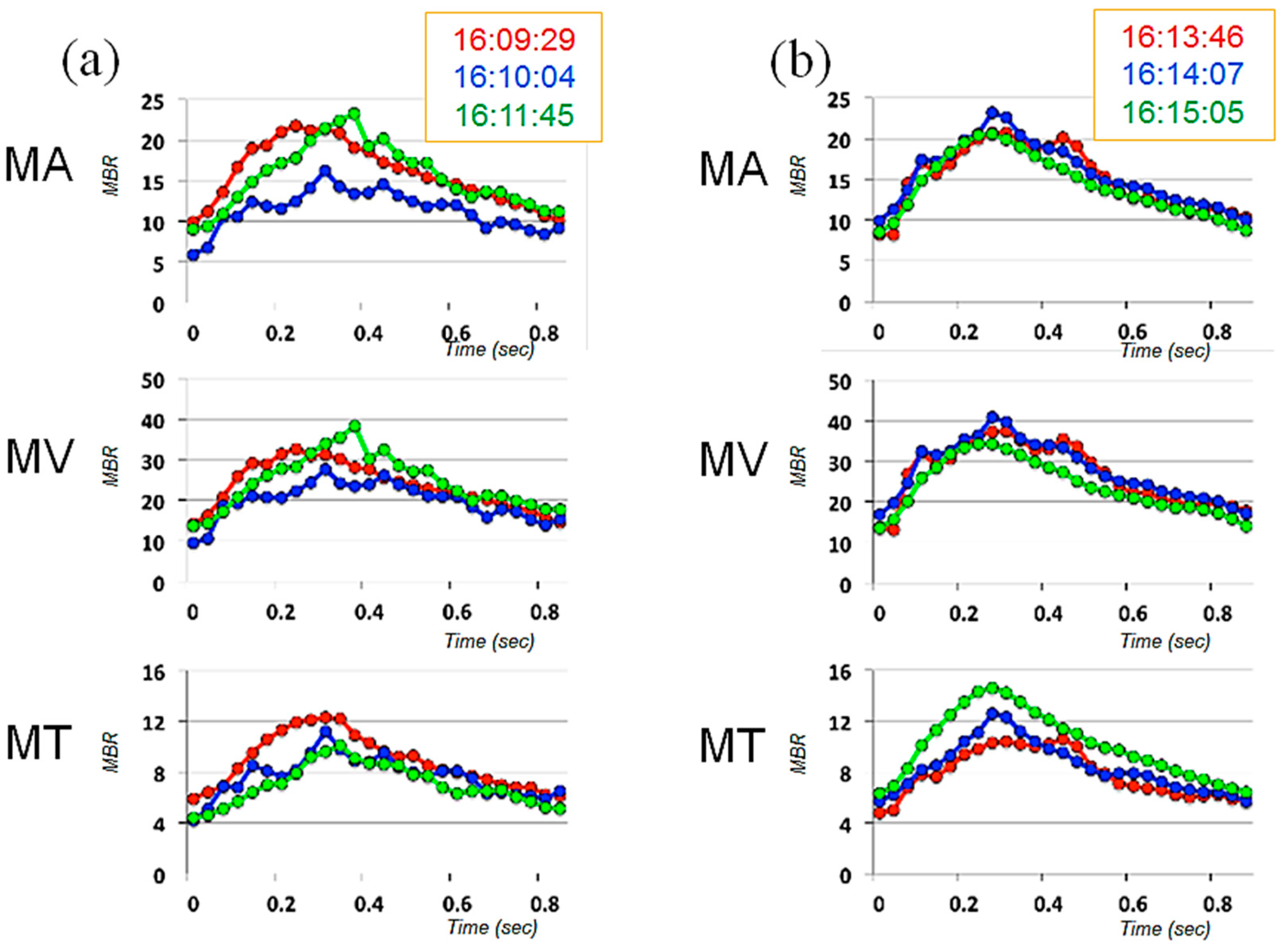
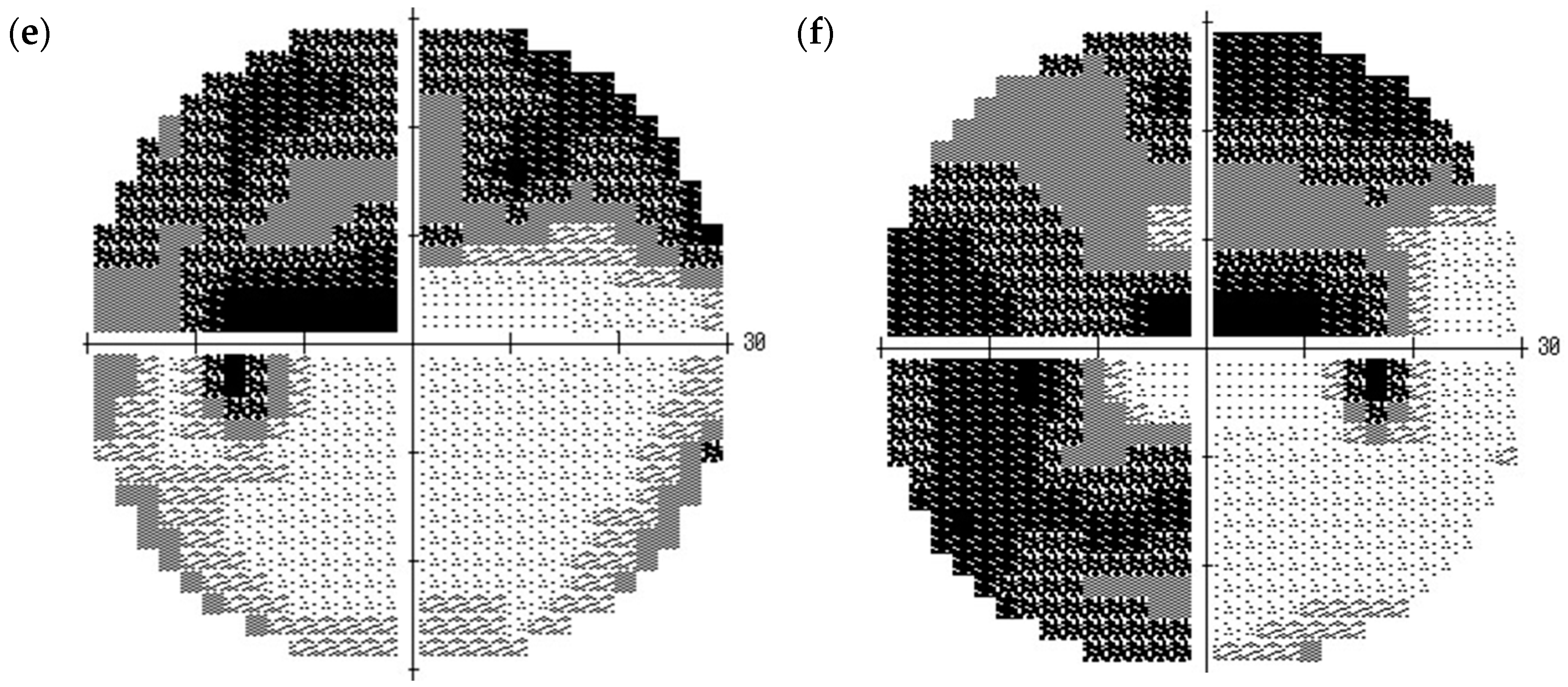
2. Case Presentation
3. Discussion
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Resnikoff, S.; Pascolini, D.; Etya’ale, D.; Kocur, I.; Pararajasegaram, R.; Pokharel, G.P.; Mariotti, S.P. Global data on visual impairment in the year 2002. Bull. World Health Organ. 2004, 82, 844–851. [Google Scholar] [PubMed]
- Collaborative Normal-Tension Glaucoma Study Group. The effectiveness of intraocular pressure reduction in the treatment of normal-tension glaucoma. Am. J. Ophthalmol. 1998, 126, 498–505. [Google Scholar]
- The AGIS Investigators. The Advanced Glaucoma Intervention Study (AGIS): 7. The relationship between control of intraocular pressure and visual field deterioration. Am. J. Ophthalmol. 2000, 130, 429–440. [Google Scholar]
- Heijl, A.; Leske, M.C.; Bengtsson, B.; Hyman, L.; Bengtsson, B.; Hussein, M.; Early Manifest Glaucoma Trial Group. Reduction of intraocular pressure and glaucoma progression: Results from the Early Manifest Glaucoma Trial. Arch. Ophthalmol. 2002, 120, 1268–1279. [Google Scholar] [CrossRef] [PubMed]
- Tielsch, J.M.; Katz, J.; Sommer, A.; Quigley, H.A.; Javitt, J.C. Hypertension, perfusion pressure, and primary open-angle glaucoma. A population-based assessment. Arch. Ophthalmol. 1995, 113, 216–221. [Google Scholar] [CrossRef] [PubMed]
- Leske, M.C.; Heijl, A.; Hyman, L.; Bengtsson, B.; Dong, L.; Yang, Z.; EMGT Group. Predictors of long-term progression in the Early Manifest Glaucoma Trial. Ophthalmology 2007, 114, 1965–1972. [Google Scholar] [CrossRef] [PubMed]
- Wentz, S.M.; Kim, N.J.; Wang, J.; Amireskandari, A.; Siesky, B.; Harris, A. Novel therapies for open-angle glaucoma. F1000Prime Rep. 2014, 6. [Google Scholar] [CrossRef] [PubMed]
- Chung, H.S.; Harris, A.; Evans, D.W.; Kagemann, L.; Garzozi, H.J.; Martin, B. Vascular aspects in the pathophysiology of glaucomatous optic neuropathy. Surv. Ophthalmol. 1999, 43, S43–S50. [Google Scholar] [CrossRef]
- Okuno, T.; Sugiyama, T.; Kojima, S.; Nakajima, M.; Ikeda, T. Diurnal variation in microcirculation of ocular fundus and visual field change in normal-tension glaucoma. Eye 2004, 18, 697–702. [Google Scholar] [CrossRef] [PubMed]
- Pemp, B.; Georgopoulos, M.; Vass, C.; Fuchsjäger-Mayrl, G.; Luksch, A.; Rainer, G.; Schmetterer, L. Diurnal fluctuation of ocular blood flow parameters in patients with primary open-angle glaucoma and healthy subjects. Br. J. Ophthalmol. 2009, 93, 486–491. [Google Scholar] [CrossRef] [PubMed]
- Flammer, J.; Orgül, S.; Costa, V.P.; Orzalesi, N.; Krieglstein, G.K.; Serra, L.M.; Renard, J.P.; Stefánsson, E. The impact of ocular blood flow in glaucoma. Prog. Retin. Eye Res. 2002, 21, 359–393. [Google Scholar] [CrossRef]
- Polak, K.; Luksch, A.; Berisha, F.; Fuchsjaeger-Mayrl, G.; Dallinger, S.; Schmetterer, L. Altered nitric oxide system in patients with open-angle glaucoma. Arch. Ophthalmol. 2007, 125, 494–498. [Google Scholar] [CrossRef] [PubMed]
- Flammer, J.; Mozaffarieh, M. What is the present pathogenetic concept of glaucomatous optic neuropathy? Surv. Ophthalmol. 2007, 52, S162–S173. [Google Scholar] [CrossRef] [PubMed]
- Nicolela, M.T. Clinical clues of vascular dysregulation and its association with glaucoma. Can. J. Ophthalmol. 2008, 43, 337–341. [Google Scholar] [CrossRef] [PubMed]
- Venkataraman, S.T.; Flanagan, J.G.; Hudson, C. Vascular reactivity of optic nerve head and retinal blood vessels in glaucoma—A review. Microcirculation 2010, 17, 568–581. [Google Scholar] [CrossRef] [PubMed]
- Aizawa, N.; Yokoyama, Y.; Chiba, N.; Omodaka, K.; Yasuda, M.; Otomo, T.; Nakamura, M.; Fuse, N.; Nakazawa, T. Reproducibility of retinal circulation measurements obtained using laser speckle flowgraphy-NAVI in patients with glaucoma. Clin. Ophthalmol. 2011, 5, 1171–1176. [Google Scholar] [PubMed]
- Sugiyama, T.; Araie, M.; Riva, C.E.; Schmetterer, L.; Orgul, S. Use of laser speckle flowgraphy in ocular blood flow research. Acta Ophthalmol. 2010, 88, 723–729. [Google Scholar] [CrossRef] [PubMed]
- Sugiyama, T. Basic technology and clinical applications of the updated model of laser speckle flowgraphy to ocular diseases. Photonics 2014, 1, 220–234. [Google Scholar] [CrossRef]


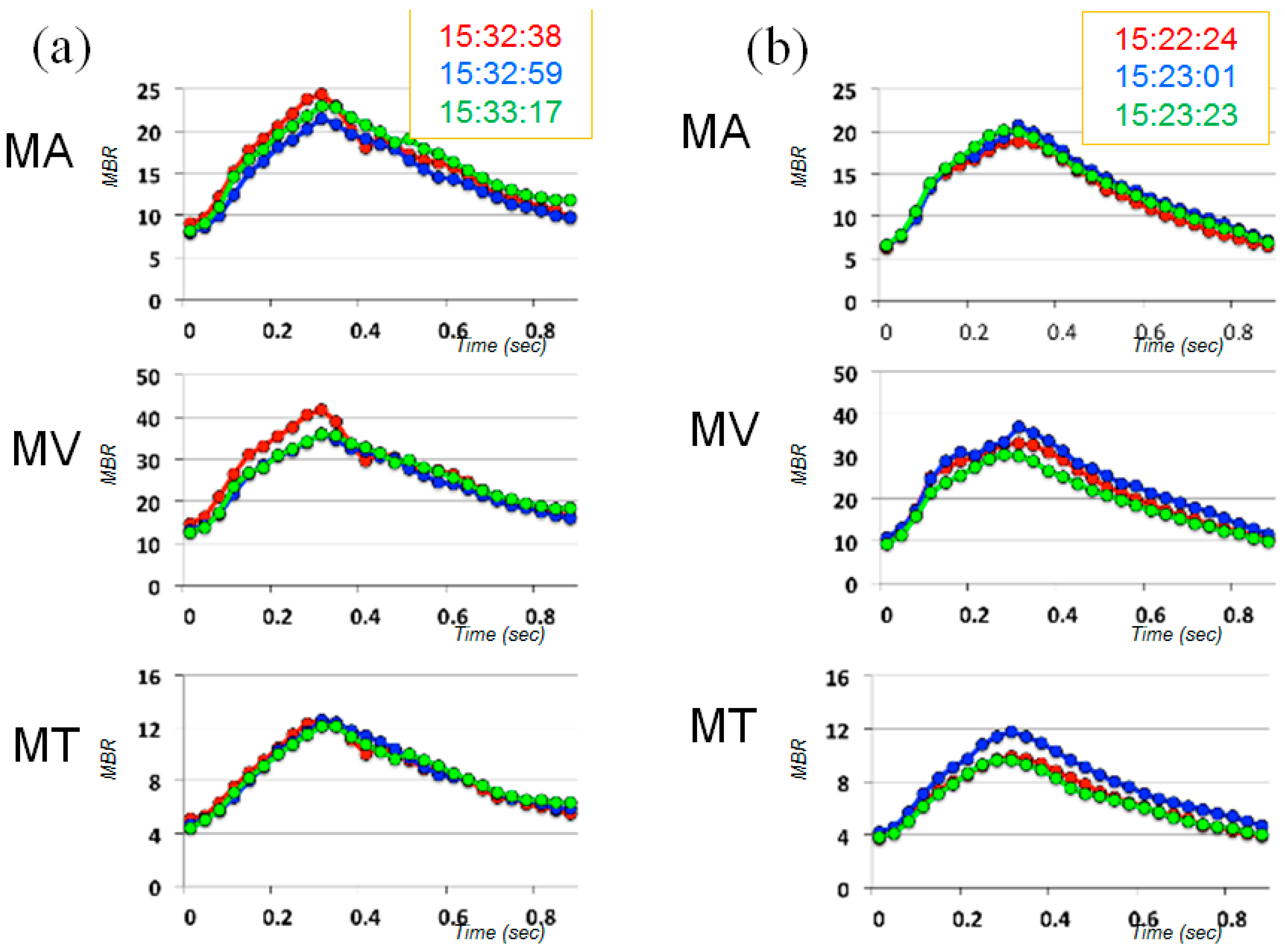
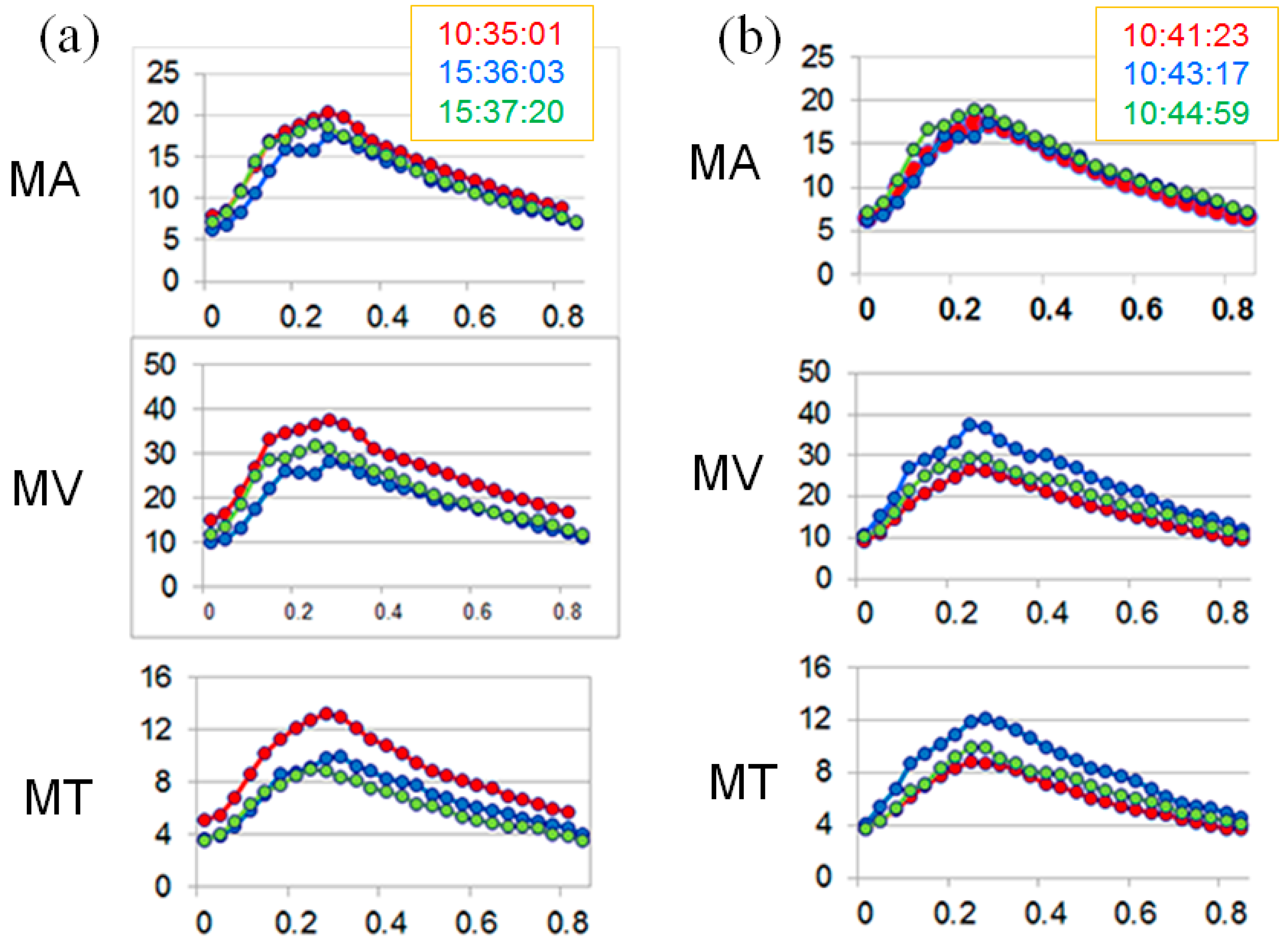
| Time | MA | MV | MT | |||
|---|---|---|---|---|---|---|
| Right | Left | Right | Left | Right | Left | |
| Day 1 | 15.3 | 5.4 | 10.9 | 9.6 | 10.5 | 11.2 |
| Day 2 | 8.4 | 10.1 | 8.9 | 10.0 | 19.3 | 16.5 |
| Day 3 | 5.8 | 3.8 | 4.9 | 7.8 | 2.9 | 8.2 |
| Day 4 | 8.7 | 6.7 | 10.7 | 14.6 | 21.0 | 14.0 |
| Normal [16] (Mean ± SD) | 2.9 ± 2.1 | 1.9 ± 1.2 | 2.1 ± 1.1 | |||
© 2016 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sugiyama, T.; Nakamura, H. Increased Short-Term Fluctuation in Optic Nerve Head Blood Flow in a Case of Normal-Tension Glaucoma by the Use of Laser Speckle Flowgraphy. Vision 2017, 1, 5. https://doi.org/10.3390/vision1010005
Sugiyama T, Nakamura H. Increased Short-Term Fluctuation in Optic Nerve Head Blood Flow in a Case of Normal-Tension Glaucoma by the Use of Laser Speckle Flowgraphy. Vision. 2017; 1(1):5. https://doi.org/10.3390/vision1010005
Chicago/Turabian StyleSugiyama, Tetsuya, and Hajime Nakamura. 2017. "Increased Short-Term Fluctuation in Optic Nerve Head Blood Flow in a Case of Normal-Tension Glaucoma by the Use of Laser Speckle Flowgraphy" Vision 1, no. 1: 5. https://doi.org/10.3390/vision1010005
APA StyleSugiyama, T., & Nakamura, H. (2017). Increased Short-Term Fluctuation in Optic Nerve Head Blood Flow in a Case of Normal-Tension Glaucoma by the Use of Laser Speckle Flowgraphy. Vision, 1(1), 5. https://doi.org/10.3390/vision1010005






