Abstract
Mechanical vibration, applied to single or few muscles, can be a selective stimulus for muscle spindles, able to modify neuromuscular management, inducing short and long-term effects, are now mainly employed in clinic studies. Several studies reported as treatments with focal vibratory (FVT) can influence neuromuscular parameters also in healthy people. However, the application modalities and the consequent effects are remarkably fragmented. This paper aims to review these studies and to characterize the FVT effectiveness on long-term conditional capacities in relation to FVT characteristics. A systematic search of studies published from 1985 to 2020 in English on healthcare databases was performed. Articles had to meet the following criteria: (1) treatment based on a locally applied vibration on muscle belly or tendon; (2) healthy adults involved; (3) outcomes time analysis enduring for more than 24 h. Twelve studies were found, all of them presented an excellent quality score of ≥75%. All selected papers reported positive changes, comparable with traditional long-lasting training effects. Muscle force and power were the most investigated parameters. The after-effects persisted for up to several months. Among the different FV administration modalities, the most effective seems to show a stimulus frequency of ≈100 Hz, repeated more times within three-five days on a voluntary contracted muscle.
1. Introduction
The production and control of a human voluntary movement is a complex process involving both nervous and muscular systems. Muscular performance changes are obtained by acting a tune of activation between these two systems. In this regard, a well-defined and structured intervention able to change one or more performance parameters for a long time is often called motor conditioning. Traditionally, this is achieved by means of training programs composed by an adequate exercise stint and workload [1]. These programs require remarkable compliance from the subject and long-lasting training application for enhancing force, motor endurance or ability [1,2]. Moreover, intervention based on exercise execution could not fit with a subject’s eventual status, because of a muscular frailty or exercise intolerance (e.g., elderly, post trauma subjects). To overcome these limits, in the main attempt both to reduce the training duration and to enhance the subjects’ compliance and performance, in recent years, different interventions based on electrical [3] or mechanical vibratory stimulation have been proposed.
Regarding the mechanical stimulus, the most adopted technique, particularly in sport sciences, consists of an elicitation, typically starting from the feet to the whole body (WBV) [4]. The rational of the WBV approach is based on the ability of the mechanical vibratory stimulus to excite muscular proprioceptors, particularly muscle spindles [5,6,7,8,9]. These latter exert a decisive influence on the motor control and could induce a subsequent neural conditioning [10,11]. Even if a large consensus exists on the effectiveness of WBV to induce positive motor effects [12,13,14], some authors have reported no effects [15], or potential harmful consequences [16]. Moreover, WBV does not allow action on single muscles, or on a muscular group, a condition that is often desirable in athletes’ training or in muscle reconditioning post trauma.
The focal vibration (FV) is another modality to apply mechanical vibration, and it allows us to locally act on well-defined body part. Many studies have been carried out to assess the FV effects on motor parameters both in healthy and pathologic individuals. In this respect, a large variety of FV treatments (FVT), different from each other in the administration modalities and the tested functional parameters, are present in the literature. As an example, FVT durations differ from a single day to several weeks, and the application number varies between one and more times per week. The muscle state may be contracted or relaxed during stimulation. The tested performance parameters are different, belonging to several physiological functionalities, from the simple force amplitude to the force time course or movement coordination, with outcome follow-ups ranging from hours to months [17,18,19,20,21,22,23,24,25]. Such protocol differences are likely the origin of different reported results. Even if positive effects seem to stand out, they vary according to the after-effect type, magnitude, persistence. The differences among the tested protocols demand definition of the possible beneficial applicative effects and the most effective stimulus characteristics. For this reason, by analyzing the literature on FVTs, some authors have tried to clarify the possible role of FVT in motor disease therapy [26]. Whereas others have been provided to illustrate the acute and chronic neuromuscular changes as the effect of FV [27]; however, these authors concluded that it is still difficult to define practical recommendations.
Therefore, the present paper aims to review the literature in order to grow knowledge on the efficacy of FVT for inducing persistent (at least 24 h from treatment ending) enhancements of conditional (those regarding metabolic processes are strictly dependent on the anatomical and physiological characteristics of some apparatuses, and are distinguished in: strength, endurance, speed and flexibility, understood as joint mobility and muscle stiffness) abilities in healthy subjects. The choice to limit the analysis of healthy subjects is to avoid the presence of any pathology as a possible confounding factor in evaluating the physiological effects of FV. Attention is also focused on looking for a dose-effect relationship between the motor parameters’ changes and the intervention characteristics (i.e., stimulus frequency, administration modalities and muscle state during treatment). Finally, the interpretative suggestions forwarded by the different studies are compared to hypothesize a possible common mainframe for considering the FVT as motor conditioning, with applications in motor and sport sciences.
2. Materials and Methods
The Preferred Reporting Items for Systematic reviews and Meta-Analyses (PRISMA) guidelines were followed in this review [28].
2.1. Data Sources
A systematic literature search was conducted, from January 1985 to June 2020, in the online databases PubMed, Web of Science, and The Cochrane Library. The following Medical Subject Headings (MeSH) of the United States National Library of Medicine (NLM) and search terms were included in our Boolean search syntax: (focal OR local OR segmental) AND vibration AND (healthy OR player OR athlete). The search was limited to English language, human species, full text availability and only original studies. Chain searching by backtracking relevant publications, scanning reference lists of relevant articles, on the basis of the authors’ knowledge and consultations with experts, in the field were performed systematically.
2.2. Selection Criteria
Two reviewers (LF, GMF) extracted relevant data according to a structured script from each study. The structured script included abstract, study design, sample characteristics (sample size, sex), experimental and control group characteristics, outcome measure and timing of results. Inclusion criteria were decided by the consensus statements of these two reviewers. In cases where LF and GMF did not reach agreement on inclusion of an article, AR and VEP were contacted. Inclusion criteria were selected by (a) population: healthy subjects, that is, subjects recruited did not have any illnesses or impairment that prevented the possibility of carrying out a physical activity aimed at improving conditional capacity; (b) intervention: treatment adopting localized mechanical vibration with a clear a description of apparatus; (c) outcomes: neuromuscular parameters regarding conditional abilities; (d) effects duration: at least 24 h from treatment ending.
2.3. Study Eligibility
Studies were excluded if they (a) analyzed outcomes in a short follow up period (<24h); (b) the outcomes were not regarding conditional motor abilities; (c) applied FVT by summing up the FV application with some type of physical exercise, in the same applicative session or in the period from treatment ending to the subsequent 24 h; (d) the administration of the stimulus involves, at the same time, many different muscles; (e) did not present an original investigation (reviews or proceedings); (f) did not publish in the English language.
2.4. Assessment of Methodological Quality
The study quality of each publication was evaluated by LF, GMF, AR, VEP, according to the 16-item checklist Downs and Black modified scale [29]. The quality scores were classified as “low” methodological quality for scores ≤50%; “good” for scores between 51% and 75%; and “excellent” if the score was >75%.
3. Results
Following the Boolean search syntax (see above) a total of 748 study records were identified. From these, 654 were excluded through title-abstract assessment and 34 were discarded as duplicates. From the remaining 60 studies without duplications, 50 ones were discarded verifying the study eligibility. Two studies were added, resulting from the chain searching. Consequently, a total of 12 eligible studies were included in the present review [30,31,32,33,34,35,36,37,38,39,40,41]. The study selection flow chart is shown in Figure 1.
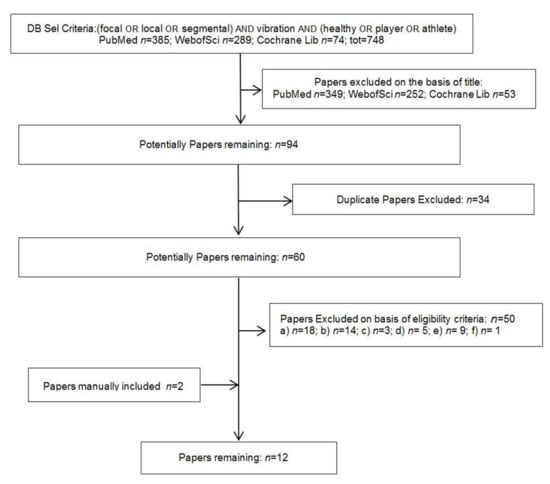
Figure 1.
Flow diagram of studies selection process.
3.1. Assessment of Methodological Quality
According to the assessment of methodological quality, three studies showed a score of 75%, seven ranged between 81% and 87%, two studies resulted in scores of over 90%. The items showing the lowest score concerned with the evaluation of the statistical power size (two out 12 studies) and the number of dropouts (stated in three out of 12 papers). The inter-rater reliability analysis showed a good coherence among the observers, 0.91 being the kappa value.
3.2. Outcome Measures and After-Effects
The main outcomes are the maximal voluntary contraction (MVC) and the muscle power. However, some studies analyzed the force output decline (fatigue) and the body balance. All the studies reported statistically significant improvements of, at least, one tested parameter. On the other hand, no side effects were evidenced during and after FVT in the different studies. Follow-up ranged between 24 h and 360 days, without treatment repetitions. The twelve selected studies and their main points, analyzed in the present review, are summarized.
3.3. Bias
In relation to the review’s aims, some biases were recognized in the selected papers. The different studies, commonly, paid poor attention to describe all the modalities of FV administration. In this respect, the features relative the contact between the stimulatory apparatus and the tissues are often absent. Since such detail might deeply influence the transmission of the mechanical stimulus to the muscle receptors, it constitutes a variable which might alter the definition of the dose-effect relation. Moreover, in some papers, the follow-up was not large enough to define the after-effect persistence. In this regard, some studies do not clearly describe the level of physical activity before and after FVT and, consequently, it cannot be excluded that the effect duration was due to a possible increase of physical activities exerted after the treatment.
4. Discussion
The aim of this review is to verify whether an intervention adopting FV could be considered a motor conditioning, that is, inducing long-lasting motor performance changes. Moreover, the review attempts to identify the relationship between protocol and parameter changes, by analyzing the intervention modalities. Twelve papers were listed, after a literature selection including only trials enrolling healthy adults (see above, selection criteria), with outcomes measured at least 24 h after intervention ending, the mechanical focal stimuli being the only adopted treatment. In all of these, stimulus characteristics and administration protocols were well defined. It seems primarily and immediately relevant that all trials reported positive and persistent neuromuscular changes and none of the tested individuals reported negative collateral effects. Moreover, only in two cases were improvements obtained by stimulating three or five muscles of the same limb [35,38], whereas in the other studies, improvements were evoked by treating only one muscle.
4.1. Outcomes
As expected, a quick analysis revealed that the maximal muscular strength and power were the more investigated motor parameters and the quadriceps muscle was the more extensively tested muscle. In this regard, MVC increased up to ≈+30% in young subjects [35], whereas, in the elderly, MVC improvements of ≈+40% and ≈+50% were reported, respectively, in males and females [33]. It is noticeable that another study, not included since the evaluation was performed only immediately after the end of FVT [42], supported the MVC increase reported in a previous study by the same authors [23]. Only in a single study did MVC not increase, even if the rate of the force development (RFD) improved (≈+27%) [30]. Muscle power also evidenced significant and persistent after-effects. In volleyball players, after FVT on both quadriceps muscles, muscle power was evaluated by the squat jump displacement (SJ), a typical Sport Sciences functional test. In this study, at the end of the follow-up period (240 days), muscle power improved by ≈+26%. On the other hand, in counter movement jump (CMJ), power was enhanced by ≈+13%. These values in treated subjects were significantly different from those of the untreated participants, even if both followed the same team-training protocol; the control group, at the end of the follow-up, respectively improved by ≈+9% (SJ) and by ≈+7.5% (CMJ) [36]. FVT effects were also tested in a high intensity effort as in the Wingate test (WnT), repeated more times with short rest periods, able to simulate a Sprint Interval Training [41]. These authors reported a 10% increase in the power peak and of 8% in total exercise work performed. The Jump task also showed in the elderly (≥65 years old), at the end of the follow-up (90 days), an improvement of about +40% [32,36].
4.2. Focal Vibration and Fatigue
Aside from maximal muscular strength and power, fatigue was also investigated after FVT. The decline of the motor output, induced by prolonged muscle activity, an important parameter in the sportive performance, as well as a relevant limiting factor in rehabilitative practice, was investigated in two studies, before and after FVT, by analyzing both the mechanical performance [30,38] and the myoelectric manifestations of fatigue [31]. The mechanical performance, evaluated by the number of the motor task repetitions, improved by about +40% [30] and +30% [38], whereas the fatigue index, explored by an analysis of the myoelectrical signals, improved by about +20% [31]. It is remarkable how, the protocols being different, the first two studies respectively evidenced such improvements 15 and 10 days after the end of FVT, whereas, in the third study, the follow-up was after 48 h.
4.3. After-Effect Duration
Some authors tested at different intervals the FVT results, allowing a gross time course of the after-effects [30,32,35,37,38,40,41]. The FVT after-effects, with respect to their temporal development, commonly show an early significant manifestation, as stated by the authors, after a few minutes [35,38,41], or after 24 h [32,36,37], even if two studies, with FVT different protocols with respect to the others, evidenced by the earliest statistically significant results after only two or four weeks of FVT [33,39]. Two studies extended the follow-up and a surprisingly long persistence of the FVT after-effects was evidenced by Brunetti’s group, up to 240 [36] and 360 days [37]. These data seem to be confirmed by other authors too, showing the absence of any significant decay of the after-effect throughout the follow-up period, ranging in these reports between 15 days and 16 weeks [32,33,35,39,40,41]. These long-lasting after-effects might be reasonably sustained by some other factors related to an improved motor efficiency, due to FVT, eliciting execution of a spontaneous higher workload daily. This hypothesis could be particularly reasonable in athletes [36] and elderly people [37], even if participants were asked not to change their lifestyle. In particular, in elderly people it is reasonable to suppose the onset of a virtuous circle induced by better self-confidence related to balance improvements after FVT [43].
4.4. FVT Versus Traditional Training Protocols
Evidence, concerning almost all papers, shows that the magnitudes of the FVT after-effects are roughly comparable with the improvement levels commonly obtained by the traditional long-lasting training protocols. As an example, eight weeks of training of leg extensions are reported as necessary to induce +27% in MVC [1]. Also, it is worth noting the performance improvements obtained in elderly people on force parameters [32,33,37], since, in this population, power and strength ameliorations can only be improved after traditional training protocols, requiring high compliance and long duration [44,45]. In the same elderly population, several papers showed an effectiveness of FVT on balance parameters [32,37], by treating quadriceps muscles and this result appears to be potentially important, the body equilibrium being in decline in elderly people related to fall risk [46]. In this regard, other studies, here excluded because of the inclusion criteria, supported, with large samples, FVT as a valid strategy to counteract falls in the elderly population [43,47].
4.5. Protocol Parameters
Since the different studies varied the FV application modalities, to better focus on FVT applicability, some considerations must be forwarded to better define the FV parameters which can influence the intervention efficacy. Attention was focused on the vibration frequency, on FV dose administration and on the muscle state during FVT.
4.5.1. Vibration Frequency and Amplitude
Firstly, by analyzing Table 1, an evident datum is that the most adopted vibration frequency is around 100 Hz, even though several trials adopted stimulus frequencies at 300 Hz [30,33,38,39]. Moreover, only a single study shows after-effects by applying a frequency lower than 100 Hz, however its follow-up is the shortest one (24 h) [34]. Stimulus frequency may be a critical factor, because the relative spindles neural inflow is reported as efficacious to induce changes in spinal alfa-gamma loop [48] and supra-spinal activation [34]. Moreover, spindle afferents are highly responsive to a vibration frequency range of 80–120 Hz, showing a responsiveness of about 1:1 [7,8]. On the other hand, the amplitude is a parameter that largely varyies in the different studies, but less relevant since the stretch-amplitude range, able to elicit a spindle afferent discharge, is extended between a few microns and 2 mm [9]. Moreover, this parameter may be largely altered and decreased by the soft tissue, interposed between the vibrating device and the muscle spindles.

Table 1.
Main characteristics of the studies included in the review. The outcome values at the end of the follow-up are reported (* p < 0.01; ** p < 0.001). 1 Fatigue index: fatigue evaluation computed as muscle fiber velocity conduction. 2 Postural Sway Area: the area which encloses the data points relative to oscillations in the horizontal plane of the center of gravity even when a person is standing. 3 Velocity of Sway: velocity of the center of gravity oscillations.
4.5.2. Dose Administration
The single session of FVT lasted from a minimum of 10 consecutive minutes, to 1 h. Moreover, the single sessions were differently distributed in the time. Two main schemes can de individuated. The first one, homogeneous in different researches [30,32,34,36,37,41], consisted of short single sessions, lasting only 10 min, repeated three times/day, separated by only 1–2 resting minutes. In the second scheme, sessions were carried on once a day [31,33,34,35,38,39,40], in this scheme each session lasted 15 [33], 30 [31,35,38,40], or 60 min [34,39]. Finally, the two schemes showed a different number of stimulation days. The first one is concentrated in three sessions/day, during only three consecutive days [30,32,36,37,41]. The second scheme show a more dispersed FVT, being the sessions variously distributed throughout the time, that is, one session/day, during three or five days/week and during 2–26 consecutive weeks [31,33,34,35,38,39,40]. Results seem comparable about the magnitude, whereas the after-effect persistence is, perhaps, favored by the shorter but more concentrated protocol.
4.5.3. Treated Muscles
Despite the variety of the treated muscles in the different studies, they share some common aspects. Firstly, all the treated muscles are anti-gravity and, moreover, inferior limb muscles are clearly privileged (10 studies, out of 12). Quadriceps largely constitute the preferential target, both for the number of studies (8) and tested subjects (239) [30,32,33,35,36,37,40,41]. Triceps surae [34,35] and Tibialis anterior were tested in only two studies, respectively involving 65 and 80 individuals [33,34]. Whereas, in the superior limb, Biceps brachii, Deltoid and Pectoralis were examined in two studies, including 58 participants [31,38]. Two studies applied FVT respectively on two [38] and five [31] muscles in the same session, whereas, all the others reported the treatment on one muscle.
4.5.4. State of the Muscle during the Treatment
A further difference in the various protocols is the muscle condition during FV application: relaxed or contracted. Some studies requested a slight voluntary muscle contraction [30,31,32,33,36,41], in others the muscle is completely relaxed [31,34,35,38,39,40], whereas, in other experimental protocols, either the two conditions were tested and compared [30,32,41]. In this respect, the more persistent effects seem to be elicited in studies where FVT was executed during a voluntary, isometric muscle contraction. The research that explored FV after-effects both in contracted and relaxed condition, with the same protocol [32,36,41], obtained effective significant after-effects only in the contracted state. It might be observed that in relaxed conditions a good mechanical coupling is not performed between the stimulatory device and the stressed anatomical part. Consequently, the mechanical energy applied by vibrator can be altered and dissipated because of a dumping effect. Therefore, the physical characteristics of the applied stimulus may be unknown, not quantifiable and, on some occasions, the magnitude might be not adequate to properly elicit the spindles [49]. On the contrary, a state of muscle contraction, even if at a low level, will increase the segmental rigidity of the vibrated section and will improve the mechanical coupling, ensuring a more faithful propagation of the vibratory energy transmission and in frequency characteristics [50]. It must be noted that FVT after-effects were studied by Transcranial Magnetic Stimulation (TMS) in the motor cortical areas, in healthy and diseased individuals. These studies showed significant rearrangements of the motor cortex, only when FV is performed on a contracted muscle, being absent in a relaxed muscle condition [51,52]. These last studies supported the necessity of the voluntary muscle contraction, during vibration, with references specifically related to the induction of the cortical plasticity. Despite this, now, it is not possible to clarify if, in which conditions and how much the muscle status (relaxed/contracted) influenced the FVT results.
4.6. Suggested Physiological Mechanisms
As is well known, mechanical vibration, if directly applied to muscle or tendon, is an adequate way to achieve an intense and highly specific spindle activation and the selected literature is in agreement in considering the muscle spindle activation as an essential step in the development of muscular functionality after-effects [5]. Moreover, neurophysiological studies show that an appropriate FVT can induce a central neural rearrangement able to explain long lasting motor performance changes [51,52,53,54,55,56,57,58].
Now the question concerns how an intense proprioceptive activation could induce such remarkable effects in muscle strength or power, fatigue and balance, thus implying a main central nervous system role in the observed after-effects and whether other mechanisms can be involved.
It should be considered that several papers, adopting FVT, showed statistically significant motor changes a few minutes after treatment ending [35,38,39,40,41]. In these instances, it is unlikely to consider possible morphological changes as being responsible for the observed immediate improvements, whereas it is strongly suggested that a primarily and direct influence of the proprioceptive activation on the central or local motor command, as much as motor units recruitment, firing frequency, synchronization and coordination between agonist and antagonist muscles [59,60]. The persistence (days or weeks) of the after-effects supports the hypothesis that a primary role of the central nervous system influenced, in some way, by the FVT. At the same time, a neural rearrangement, especially at a cortical level, might explain the cross effects also detected on the non-treated limb [30,39]. The longer persistence (months) of motor changes may be due to a spontaneous enhancement of the daily spontaneous activities [32,37], or of a more efficacious training consequent to motor changes induced by FVT [41].
With regard to the motor performance improvements, it is well known that the interplay between agonist and antagonist muscle activities, in a system composed of several muscles, bone segments and joints, take on a determinant role from both the biomechanical and motor task efficiency point of view [61]. Such improved centrally managed joint efficiency might also be a direct reason for the decreased fatigue. Such observations are supported by several studies with TMS which showed a rearrangement of the cortical excitability, selectively involving the motor areas pertinent to the treated muscles after FVT [52,53,54,55,56,57,58,59]. In particular, Marconi’s group, after FVT, applied according to a clustered modality, evidenced a long-lasting advantageous cortical modulation of the agonist-antagonist muscle balance, correlated to the improvement of motor coordination of the joint belonging to the vibrated muscle and its antagonist [51,52]. It is worth noting that motor improvements, induced by traditional physical trainings, are correlated with similar rearrangements of cortical excitability [62,63,64].
From a biomechanical point of view, during movement, the main role of an antagonist is to provide a braking force finalized to control both time and accuracy [65,66,67,68] and, in turn, has a protective role for the joint by modulating its stiffness [69]. Therefore, during the movement the antagonist force output may exceed the level necessary to brake, in particular when the acceleratory phase is ending, and this occurs in especially in non-skilled subjects [70]. Consequently, the agonist-antagonist coordination, by modulating the joint functional stiffness, allows us to manage the movement smoothness [71]. In turn, the smoothness amelioration in a motor task is considered as a direct expression of an improved motor coordination and, consequently, of a more efficacious motor execution [72]. In this regard, Aprile et al., analyzing the movement smoothness, evidenced significant improvements after FVT [38]. This is coherent with Marconi’s neurophysiological data [51,52] and might additionally explain the outcomes of the jumping test where performance is a direct consequence of agonist–antagonist interplay [32,36].
Finally, the ameliorated balance of agonist/antagonist activation might allow a more efficient movement, according to an energetic point of view by inducing a reduction of the internal work in favor of the external one [73]. Such considerations may explain the capacity to perform the exercise with an increase of workout after FVT [37], as well as a lesser fatigability in isotonic movements [30,38], a reduction of the RFD [30] and the improved body stability evidenced by testing the balance in static and dynamic conditions [32,37]. The datum showing a significant decrease of the joint laxity, after FVT, also appears to be coherent and relevant [36].
Nevertheless, it must be underlined that, if the cortical agonist/antagonist is supported by experimental evidence, we cannot exclude other, still unidentified but possible mechanisms, both in subcortical and spinal motor networks and in the sensory function. FVT likely involves spindle afferents and it is known that their influence on motoneuron behavior, that is, both by favoring the recruitment [9] and by modulating the strength output decline [74]. Muscle strength enhancements, in sportive training, are improved by changes in alpha-motoneuron discharge [60,61]. Finally, the space perception results should be modified after FVT, this being potentially relevant for the balance improvements [75,76].
5. Conclusions
This review shows that FV seems to be a brief and efficient stimulus able to enhance the conditional capacities in healthyindividuals. These changes could be attributed to a better agonist/antagonist interplay because of a rearrangement in central and segmental nervous pathways. Consequently, power and absolute force is increased, kinematic is smoother and articular efficiency is higher, whereas force output declines as time is delayed. All listed papers showed long-lasting positive motor changes, without negative collateral effects, the FVT acting as a motor conditioning. Table 1 shows that FV can be administered with several treatment protocols and properties varying mechanical stimulus characteristics, stimulus administration modalities and intervention duration as evident from the paper selected. Between them, based on effects duration, administration modalities and motor improvements, the more efficient FVT seems to be characterized by a stimulus frequency of about 100 Hz, a three–five days administration, on a contracted muscle.
Author Contributions
L.F., A.R., V.E.P., and G.M.F. conceived the outline of the review, and wrote the protocol; L.F. and G.M.F. reviewed the literature, contributed to figure preparation, literature selection, and provided suggestions for the draft planning; L.F., A.R., V.E.P., and G.M.F. wrote the original draft and provided suggestions to the revisions. All authors have read and agreed to the published version of the manuscript.
Funding
This research received no external funding.
Acknowledgments
We would like to express our gratitude to the original study authors as well as to the respective study participants included in this review.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Lum, D.; Barbosa, T.M. Brief Review: Effects of isometric strength training on strength and dynamic performance. Int. J. Sports Med. 2019, 40, 363–375. [Google Scholar] [CrossRef] [PubMed]
- Mikkola, J.S.; Rusko, H.K.; Nummela, A.T.; Paavolainen, L.M.; Häkkinen, K. Concurrent endurance and explosive type strength training increases activation and fast force production of leg extensor muscles in endurance athletes. J. Strength Cond. Res. 2007, 21, 613–620. [Google Scholar]
- Filipovic, A.; Kleinöder, H.; Dörmann, U.; Mester, J. Electromyostimulation—A systematic review of the effects of different electromyostimulation methods on selected strength parameters in trained and elite athletes. J. Strength Cond. Res. 2012, 26, 2600–2614. [Google Scholar] [CrossRef]
- Costantino, C.; Gimigliano, R.; Olvirri, S.; Gimigliano, F. Whole body vibration in sport: A critical review. J. Sports Med. Phys. Fit. 2014, 54, 757–764. [Google Scholar]
- Bianconi, R.; van der Meulen, J.J. The response to vibration of the end organs of mammalian muscle spindles. J. Neurophysiol. 1963, 26, 177–190. [Google Scholar] [CrossRef]
- Burke, D.; Hagbarth, K.E.; Löfstedt, L.; Wallin, B.G. The responses of human muscle spindle endings to vibration of non-contracting muscles. J. Physiol. 1976, 261, 673–693. [Google Scholar] [CrossRef]
- Burke, D.; Hagbarth, K.-E.; Löfstedt, L.; Wallin, B.G. The responses of human muscle spindle endings to vibration during isometric contraction. J. Physiol. 1976, 261, 695–711. [Google Scholar] [CrossRef] [PubMed]
- Roll, J.P.; Vedel, J.P.; Roll, R. Eye, head and skeletal muscle spindle feedback in the elaboration of body references. Prog. Brain Res. 1989, 80, 113–123. [Google Scholar]
- Matthwes, P.B.C. Mammalian Muscle Receptors and Their Central Actions; Edward Arnold: London, UK, 1972. [Google Scholar]
- Thompson, A.K.; Wolpaw, J.R. Restoring walking after spinal cord injury: Operant conditioning of spinal reflexes can help. Neuroscientist 2015, 21, 203–215. [Google Scholar] [CrossRef]
- Bongiovanni, L.G.; Hagbarth, K.E. Tonic vibration reflexes elicited during fatigue from maximal voluntary contractions in man. J. Physiol. 1990, 423, 1–14. [Google Scholar] [CrossRef] [PubMed]
- Rittweger, J. Vibration as an exercise modality: How it may work, and what its potential might be. Eur. J. Appl. Physiol. 2010, 108, 877–904. [Google Scholar] [CrossRef]
- Cochrane, D.J. Vibration exercise: The potential benefits. Int. J. Sports Med. 2011, 32, 75–99. [Google Scholar] [CrossRef]
- Stania, M.; Juras, G.; Słomka, K.; Chmielewska, D.; Kŕol, P. The application of whole-body vibration in physiotherapy—A narrative review. Physiol. Int. 2016, 103, 133–145. [Google Scholar] [CrossRef]
- Hortobágyi, T.; Lesinski, M.; Fernandez-del-Olmo, M. Small and inconsistent effects of whole body vibration on athletic performance: A systematic review and meta-analysis. Eur. J. Appl. Physiol. 2015, 115, 1605–1625. [Google Scholar] [CrossRef] [PubMed]
- Abercromby, A.F.J.; Amonette, W.E.; Layne, C.S.; McFarlin, B.K.; Hinman, M.R.; Paloski, W.H. Vibration Exposure and Biodynamic Responses during Whole-Body Vibration Training. Med. Sci. Sports Exerc. 2007, 39, 1794–1800. [Google Scholar] [CrossRef]
- Shinohara, M. Effects of Prolonged Vibration on Motor Unit Activity and Motor Performance. Med. Sci. Sports Exerc. 2005, 37, 2120–2125. [Google Scholar] [CrossRef] [PubMed]
- Ushiyama, J.; Masani, K.; Kouzaki, M.; Hiroaki, K.; Fukunaga, T. Difference in after effects following prolonged Achilles tendon vibration on muscle activity during maximal voluntary contraction among plantar flexor synergists. J. Appl. Physiol. 2005, 98, 1427–1433. [Google Scholar] [CrossRef] [PubMed]
- Luo, J.; Clarke, M.; McNamara, B.; Moran, K. Influence of resistance load on neuromuscular response to vibration training. J. Strength Cond. Res. 2009, 23, 420. [Google Scholar] [CrossRef]
- Couto, B.P.; Silva, H.R.; Barbosa, M.P.; Szmuchrowski, L.A. Chronic Effects of Different Frequencies of Local Vibrations. Int. J. Sports Med. 2012, 32, 123–129. [Google Scholar] [CrossRef] [PubMed]
- Fry, A.; Folland, J.P. Prolonged infrapatellar tendon vibration does not influence quadriceps maximal or explosive isometric force production in man. Eur. J. Appl. Physiol. 2014, 114, 1757–1766. [Google Scholar] [CrossRef]
- Goebel, R.T.; Kleinöder, H.; Yue, Z.; Gosh, R.; Mester, J. Effect of Segment-Body Vibration on Strength Parameters. Sports Med. Open 2015, 1, 14. [Google Scholar] [CrossRef]
- Celletti, C.; Fara, M.A.; Filippi, G.M.; La Torre, G.; Tozzi, R.; Vanacore, N.; Camerota, F. Focal Muscle Vibration and Physical Exercise in Postmastectomy Recovery: An Explorative Study. Biomed. Res. Int. 2017, 2017, 7302892. [Google Scholar] [CrossRef] [PubMed]
- Cochrane, D.J. Effectiveness of using wearable vibration therapy to alleviate muscle soreness. Eur. J. Appl. Physiol. 2017, 117, 501–509. [Google Scholar] [CrossRef]
- Contemori, S.; Dieni, C.V.; Sullivan, J.A.; Ferraresi, A.; Occhigrossi, C.; Calabrese, F.; Pettorossi, V.E.; Biscarini, A.; Panichi, R. Sensory infow manipulation induces learning-like phenomena in motor behavior. Eur. J. Appl. Physiol. 2020, 120, 811–828. [Google Scholar] [CrossRef] [PubMed]
- Germann, D.; El Bouse, A.; Shnier, J.; Abdelkader, N.; Kazemi, M. Effects of local vibration therapy on various performance parameters: A narrative literature review. J. Can. Chiropr. Assoc. 2018, 62, 170–181. [Google Scholar] [PubMed]
- Souron, R.; Besson, T.; Millet, G.Y.; Lapole, T. Acute and chronic neuromuscular adaptations to local vibration training. Eur. J. Appl. Physiol. 2017, 117, 1939–1964. [Google Scholar] [CrossRef]
- Moher, D.; Liberati, A.; Tetzlaff, J.; Altman, D.G. Group P. Preferred reporting items for systematic reviews and meta-analyses: The PRISMA statement. PLoS Med. 2009, 6, e1000097. [Google Scholar] [CrossRef]
- Downs, S.H.; Black, N. The feasibility of creating a checklist for the assessment of the methodological quality both of randomised and non-randomised studies of health care interventions. J. Epidemiol. Community Health 1998, 52, 377–384. [Google Scholar] [CrossRef] [PubMed]
- Fattorini, L.; Ferraresi, A.; Rodio, A.; Azzena, G.B.; Filippi, G.M. Motor performance changes induced by muscle vibration. Eur. J. Appl. Physiol. 2006, 98, 79–87. [Google Scholar] [CrossRef] [PubMed]
- Casale, R.; Ring, H.; Rainoldi, A. High frequency vibration conditioning stimulation centrally reduces myoelectrical manifestation of fatigue in healthy subjects. J. Electromyogr. Kinesiol. 2009, 19, 998–1004. [Google Scholar] [CrossRef]
- Filippi, G.M.; Brunetti, O.; Botti, F.M.; Panichi, R.; Roscini, M.; Camerota, F.; Cesari, M.; Pettorossi, V.E. Improvement of stance control and muscle performance induced by focal muscle vibration in young-elderly women: A randomized controlled trial. Arch. Phys. Med. Rehabil. 2009, 90, 2019–2025. [Google Scholar] [CrossRef]
- Pietrangelo, T.; Mancinelli, R.; Toniolo, L.; Cancellara, L.; Paoli, A.; Puglielli, C.; Iodice, P.; Doria, C.; Bosco, G.; D’Amelio, L.; et al. Effects of local vibrations on skeletal muscle trophism in elderly people: Mechanical, cellular, and molecular events. Int. J. Mol. Med. 2009, 24, 503–512. [Google Scholar] [CrossRef]
- Lapole, T.; Pérot, C. Effects of repeated Achilles tendon vibration on triceps surae force production. J. Electromyogr. Kinesiol. 2010, 20, 648–654. [Google Scholar] [CrossRef]
- Iodice, P.; Bellomo, R.G.; Gialluca, G.; Fano, G.; Saggini, R. Acute and cumulative effects of focused high-frequency vibrations on the endocrine system and muscle strength. Eur. J. Appl. Physiol. 2011, 111, 897–904. [Google Scholar] [CrossRef]
- Brunetti, O.; Botti, F.M.; Roscini, M.; Brunetti, A.; Panichi, R.; Filippi, G.M.; Biscarini, A.; Pettorossi, V.E. Focal vibration of quadriceps muscle enhances leg power and decreases knee joint laxity in female volleyball players. J. Sports Med. Phys. Fit. 2012, 52, 595–596. [Google Scholar]
- Brunetti, O.; Botti, F.M.; Brunetti, A.; Biscarini, A.; Scarponi, A.M.; Filippi, G.M.; Pettorossi, V.E. Effects of focal vibration on bone mineral density and motor performance of postmenopausal osteoporotic women. J. Sports Med. Phys. Fit. 2015, 55, 118–127. [Google Scholar]
- Aprile, I.; di Sipio, E.; Germanotta, M.; Simbolotti, C.; Padua, L. Muscle focal vibration in healthy subjects: Evaluation of the effects on upper limb motor performance measured using a robotic device. Eur. J. Appl. Physiol. 2016, 116, 729–737. [Google Scholar] [CrossRef] [PubMed]
- Souron, R.; Farabet, A.; Féasson, L.; Belli, A.; Millet, G.Y.; Lapole, T. Eight weeks of local vibration training increases dorsiflexor muscle cortical voluntary activation. J. Appl. Physiol. 2017, 122, 1504–1515. [Google Scholar] [CrossRef]
- Feltroni, L.; Monteleone, S.; Petrucci, L.; Carlisi, E.; Mazzacane, B.; Schieppati, M.; Dalla Toffola, E. Potentiation of muscle strength by focal vibratory stimulation on quadriceps femoris. G Ital. Med. Lav. Ergon. 2018, 40, 90–96. [Google Scholar]
- Filippi, G.M.; Fattorini, L.; Summa, A.; Zagaglia, A.; Rodio, A. Effects of focal vibration on power and work in multiple wingate tests. Biol. Sport 2020, 37, 25–35. [Google Scholar] [CrossRef] [PubMed]
- Souron, R.; Besson, T.; Lapole, T.; Millet, G.Y. Neural adaptations in quadriceps muscle after 4 weeks of local vibration training in young versus older subjects. Appl. Physiol. Nutr. Metab. 2018, 43, 427–436. [Google Scholar] [CrossRef] [PubMed]
- Celletti, C.; Fattorini, L.; Camerota, F.; Ricciardi, D.; La Torre, G.; Landi, F.; Filippi, G.M. Focal muscle vibration as a possible intervention to prevent falls in elderly women: A pragmatic randomized controlled trial. Aging Clin. Exp. Res. 2015, 27, 857–863. [Google Scholar] [CrossRef]
- Caserotti, P.; Aagaard, P.; Puggaard, L. Changes in power and force generation during coupled eccentric–concentric versus concentric muscle contraction with training and aging. Eur. J. Appl. Physiol. 2008, 103, 151–161. [Google Scholar] [CrossRef]
- Andersson, E.A.; Frank, P.; Pontén, M.; Ekblom, B.; Ekblom, M.; Moberg, M.; Sahlin, K. Improving strength, power, muscle aerobic capacity, and glucose tolerance through short-term progressive strength training among elderly people. J. Vis. Exp. 2017, 125, 55518. [Google Scholar] [CrossRef]
- Cuevas-Trisan, R. Balance problems and fall risks in the elderly. Phys. Med. Rehabil. Clin. N. Am. 2017, 28, 727–737. [Google Scholar] [CrossRef]
- Attanasio, G.; Camerota, F.; Ralli, M.; Galeoto, G.; La Torre, G.; Galli, M.; de Vincentiis, M.; Greco, A.; Celletti, C. Does focal mechanical stimulation of the lower limb muscles improve postural control and sit to stand movement in elderly? Aging Clin. Exp. Res. 2018, 30, 1161–1166. [Google Scholar] [CrossRef]
- Roll, J.P.; Vedel, J.P. Alteration of proprioceptive messages induced by tendon vibration in man: A microneurographic study. Exp. Brain Res. 1989, 76, 213–222. [Google Scholar] [CrossRef]
- Fallon, J.B.; Macefield, V.G. Vibration sensitivity of human muscle spindles and golgi tendon organs. Muscle Nerve 2007, 36, 21–29. [Google Scholar] [CrossRef]
- Fattorini, L.; Tirabasso, A.; Lunghi, A.; Sacco, F.; Marchetti, E. Muscular forearm activation in hand-grip tasks with superimposition of mechanical vibrations. J. Electromyogr. Kinesiol. 2016, 26, 143–145. [Google Scholar] [CrossRef]
- Marconi, B.; Filippi, G.M.; Koch, G.; Pecchioli, C.; Salerno, S.; Don, R.; Camerota, F.; Saraceni, V.M.; Caltagirone, C. Long-term effects on motor cortical excitability induced by repeated muscle vibration during contraction in healthy subjects. J. Neurol. Sci. 2008, 275, 51–59. [Google Scholar] [CrossRef]
- Marconi, B.; Filippi, G.M.; Koch, G.; Giacobbe, V.; Pecchioli, C.; Versace, V.; Camerota, F.; Saraceni, V.M.; Caltagirone, C. Long-term effects on cortical excitability and motor recovery induced by repeated muscle vibration in chronic stroke patients. Neurorehabil. Neural Repair 2011, 25, 48–60. [Google Scholar] [CrossRef]
- Steyvers, M.; Levin, O.; van Baelen, M.; Swinnen, S.P. Corticospinal excitability changes following prolonged muscle tendon vibration. Neuroreport 2003, 14, 2001–2004. [Google Scholar] [CrossRef]
- Rosenkranz, K.; Rothwell, J.C. Differential effect of muscle vibration on intracortical inhibitory circuits in humans. J. Physiol. 2003, 551, 649–660. [Google Scholar] [CrossRef]
- Rosenkranz, K.; Rothwell, J.C. The effect of sensory input and attention on the sensorimotor organization of the hand area of the human motor cortex. J. Physiol. 2004, 561, 307–320. [Google Scholar] [CrossRef] [PubMed]
- Rosenkranz, K.; Williamon, A.; Butler, K.; Cordivari, C.; Lees, A.J.; Rothwell, J.C. Pathophysiological differences between musician’s dystonia and writer’s cramp. Brain 2005, 128, 918–931. [Google Scholar] [CrossRef] [PubMed]
- Rosenkranz, R.; Rothwell, J.C. Differences between the effects of three plasticity inducing protocols on the organization of the human motor cortex. Eur. J. Neurosci. 2006, 23, 822–829. [Google Scholar] [CrossRef]
- Rosenkranz, K.; Rothwell, J.C. Spatial attention affects sensorimotor reorganisation in human motor cortex. Exp. Brain Res. 2006, 170, 97–108. [Google Scholar] [CrossRef] [PubMed]
- Gabriel, D.A.; Kamen, G.; Frost, G. Neural adaptations to resistive exercise mechanisms and recommendations for training practices. Sports Med. 2006, 36, 133–149. [Google Scholar] [CrossRef] [PubMed]
- Mrówczynski, W.; Celichowski, J.; Raikova, R.; Krutki, P. Physiological consequences of doublet discharges on motoneuronal firing and motor unit force. Front. Cell Neurosci. 2015, 10, 81. [Google Scholar] [CrossRef]
- Mason, J.; Howatson, G.; Frazer, A.K.; Pearce, A.J.; Jaberzadeh, S.; Avela, J.; Kidgell, D.J. Modulation of intracortical inhibition and excitation in agonist and antagonist muscles following acute strength training. Eur. J. Appl. Physiol. 2019, 119, 2185–2214. [Google Scholar] [CrossRef]
- Dai, W.; Pi, Y.L.; Ni, Z.; Tan, X.Y.; Zhang, J.; Wu, Y. Maintenance of balance between motor cortical excitation and inhibition after long-term training. Neuroscience 2016, 336, 114–122. [Google Scholar] [CrossRef]
- Sato, D.; Yamazaki, Y.; Yamashiro, K.; Onishi, H.; Baba, Y.; Ikarashi, K.; Maruyama, A. Elite competitive swimmers exhibit higher motor cortical inhibition and superior sensorimotor skills in a water environment. Behav. Brain Res. 2020, 395, 112835. [Google Scholar] [CrossRef]
- Siddique, U.; Rahman, S.; Frazer, A.; Leung, M.; Pearce, A.J.; Kidgell, D.J. Task-dependent modulation of corticospinal excitability and inhibition following strength training. J. Electromyogr. Kinesiol. 2020, 52, 102411. [Google Scholar] [CrossRef]
- Lestienne, F. Effects of inertial load and velocity on the braking process of voluntary limb movements. Exp. Brain Res. 1979, 35, 407–418. [Google Scholar] [CrossRef] [PubMed]
- Marsden, C.D.; Obeso, J.A.; Rothwell, J.C. The function of the antagonist muscle during fast limb movements in man. J. Physiol. 1983, 335, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Wierzbicka, M.M.; Wiegner, A.W.; Shahani, B.T. Role of agonist and antagonist muscles in fast arm movements in man. Exp. Brain Res. 1986, 63, 331–340. [Google Scholar] [CrossRef]
- Wierzbicka, M.M.; Wiegner, A.W. Accuracy of motor responses in subjects with and without control of antagonist muscle. J. Neurophysiol. 1996, 75, 2533–2541. [Google Scholar] [CrossRef]
- Karst, G.M.; Hasan, Z. Antagonist muscle activity during human forearm movements under varying kinematic and loading conditions. Exp. Brain Res. 1987, 67, 391–401. [Google Scholar] [CrossRef]
- Teulings, H.L.; Contreras-Vidal, J.L.; Stelmach, G.E.; Adler, C.H. Parkinsonism reduces coordination of fingers, wrist, and arm in fine motor control. Exp. Neurol. 1997, 146, 159–170. [Google Scholar] [CrossRef] [PubMed]
- Cavagna, G.A.; Kaneko, M. Mechanical work and efficiency in level walking and running. J. Appl. Physiol. 1977, 18, 467–471. [Google Scholar] [CrossRef]
- Fattorini, L.; Tirabasso, A.; Lunghi, A.; Sacco, F.; Marchetti, E. Muscular synchronization and hand-arm fatigue. Int. J. Ind. Ergon. 2017, 62, 13–16. [Google Scholar] [CrossRef]
- Di Prampero, P.E.; Capelli, C.; Pagliaro, P.; Antonutto, G.; Girardis, M.; Zamparo, P.; Soule, R.G. Energetics of best performances in middle-distance running. J. Appl. Physiol. 1993, 74, 2318–2324. [Google Scholar] [CrossRef] [PubMed]
- Gandevia, S.C. Spinal and supraspinal factors in human muscle fatigue. Physiol. Rev. 2001, 81, 1725–1789. [Google Scholar] [CrossRef]
- Kerkhoff, G. Modulation and rehabilitation of spatial neglect by sensory stimulation. Prog. Brain Res. 2003, 142, 257–314. [Google Scholar]
- Pettorossi, V.E.; Panichi, R.; Botti, F.M.; Biscarini, A.; Filippi, G.M.; Schieppati, M. Long-lasting effects of neck muscle vibration and contraction on self-motion perception of vestibular origin. Clin. Neurophysiol. 2015, 126, 1886–1894. [Google Scholar] [CrossRef] [PubMed]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).