The Effects of Haemophilia on the Postural Control of Adolescents
Abstract
:1. Introduction
2. Materials and Methods
3. Results
3.1. Anthropometric Characteristics
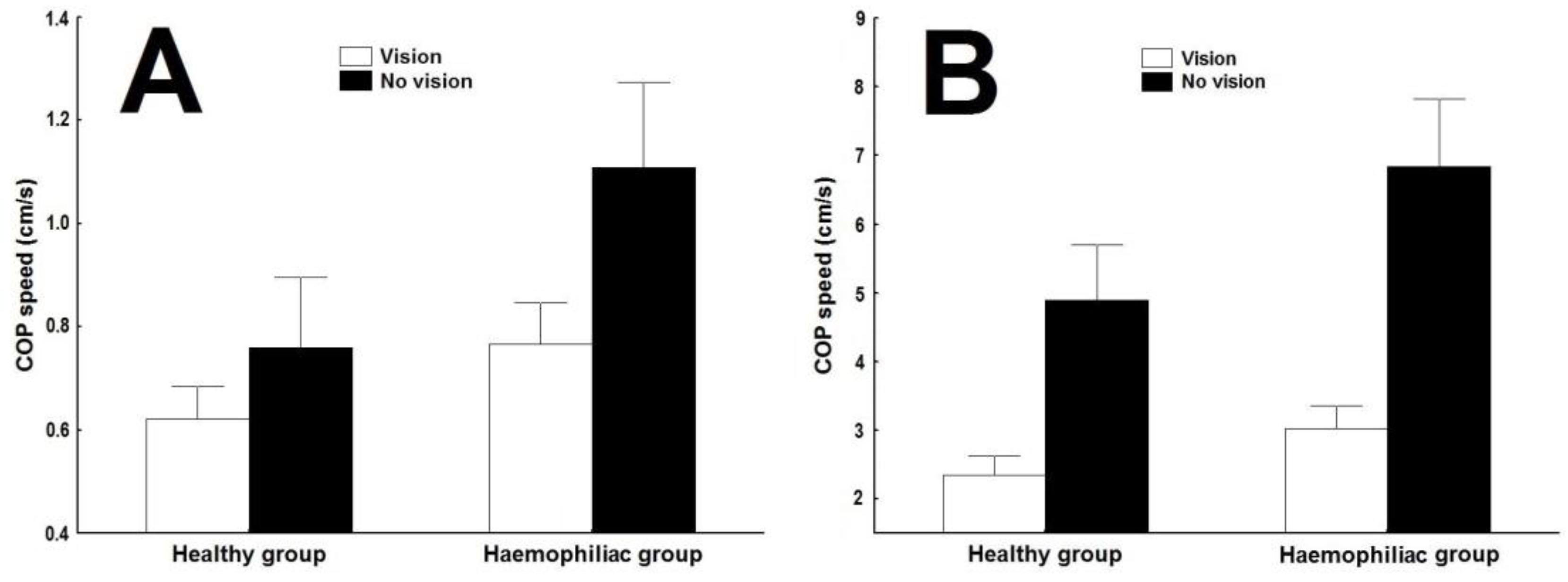
3.2. COP Speed
4. Discussion
4.1. Haemophilia, Postural Control, and Altered Vision Condition
4.2. Haemophilia, Postural Control, and Altered Proprioception Condition
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Loram, I.D.; Kelly, S.M.; Lakie, M. Human balancing of an inverted pendulum: Is sway size controlled by ankle impedance? J. Physiol. 2001, 532, 879–891. [Google Scholar] [CrossRef] [PubMed]
- Johansson, R.; Magnusson, M.; Akesson, M. Identification of human postural dynamics. IEEE Trans. Biomed. Eng. 1988, 35, 858–869. [Google Scholar] [CrossRef] [PubMed]
- Loram, I.D.; Lakie, M. Direct measurement of human ankle stiffness during quiet standing: The intrinsic mechanical stiffness is insufficient for stability. J. Physiol. 2002, 545, 1041–1053. [Google Scholar] [CrossRef] [PubMed]
- Massion, J. Postural control system. Curr. Opin. Neurobiol. 1994, 4, 877–887. [Google Scholar] [CrossRef]
- Horak, F.B.; Shupert, C.L.; Mirka, A. Components of postural dyscontrol in the elderly: A review. Neurobiol. Aging 1989, 10, 727–738. [Google Scholar] [CrossRef]
- Maki, B.E.; Holliday, P.J.; Topper, A.K. A prospective study of postural balance and risk of falling in an ambulatory and independent elderly population. J. Gerontol. 1994, 49, M72–M84. [Google Scholar] [CrossRef] [PubMed]
- Muir, S.W.; Berg, K.; Chesworth, B.; Klar, N.; Speechley, M. Quantifying the magnitude of risk for balance impairment on falls in community-dwelling older adults: A systematic review and meta-analysis. J. Clin. Epidemiol. 2010, 63, 389–406. [Google Scholar] [CrossRef] [PubMed]
- Cahill, M.R.; Colvin, B.T. Haemophilia. Postgrad. Med. J. 1997, 73, 201–206. [Google Scholar] [CrossRef] [PubMed]
- Hilberg, T.; Herbsleb, M.; Gabriel, H.H.; Jeschke, D.; Schramm, W. Proprioception and isometric muscular strength in haemophiliac subjects. Haemophilia 2001, 7, 582–588. [Google Scholar] [CrossRef] [PubMed]
- Fearn, M.; Hill, K.; Williams, S.; Mudge, L.; Walsh, C.; McCarthy, P.; Walsh, M.; Street, A. Balance dysfunction in adults with haemophilia. Haemophilia 2010, 16, 606–614. [Google Scholar] [CrossRef] [PubMed]
- Kurz, E.; Anders, C.; Herbsleb, M.; Puta, C.; Czepa, D.; Hilberg, T. Ankle muscle activation in people with haemophilia. Haemophilia 2012, 18, 948–954. [Google Scholar] [CrossRef] [PubMed]
- Buzzard, B.M. Proprioceptive training in haemophilia. Haemophilia 1998, 4, 528–531. [Google Scholar] [PubMed]
- Gallach, J.E.; Querol, F.; González, L.M.; Pardo, A.; Aznar, J.A. Posturographic analysis of balance control in patients with haemophiliac arthropathy. Haemophilia 2008, 14, 329–335. [Google Scholar] [CrossRef] [PubMed]
- Souza, F.M.; McLaughlin, P.; Pereira, R.P.; Minuque, N.P.; Mello, M.H.; Siqueira, C.; Villaça, P.; Tanaka, C. The effects of repetitive haemarthrosis on postural balance in children with haemophilia. Haemophilia 2013, 19, e212–e217. [Google Scholar] [CrossRef] [PubMed]
- Teasdale, N.; Stelmach, G.E.; Breunig, A. Postural sway characteristics of the elderly under normal and altered visual and support surface conditions. J. Gerontol. 1991, 46, B238–B244. [Google Scholar] [CrossRef] [PubMed]
- Wu, G.; Chiang, J.H. The effects of surface compliance on foot pressure in stance. Gait Posture 1996, 4, 122–129. [Google Scholar] [CrossRef]
- Chiang, J.H.; Wu, G. The influence of foam surfaces on biomechanical variables contributing to postural control. Gait Posture 1997, 5, 239–245. [Google Scholar] [CrossRef]
- Maki, B.E.; Holliday, P.J.; Fernie, G.R. A comparison of spontaneous-and induced sway balance tests. J. Am. Geriatr. Soc. 1990, 38, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Geurts, A.C.; Nienhuis, B.; Mulder, T.W. Intrasubject variability of selected force-platform parameters in the quantification of postural control. Arch. Phys. Med. Rehabil. 1993, 74, 1144–1150. [Google Scholar] [PubMed]
- Baratto, L.; Morasso, P.G.; Re, C.; Spada, G. A new look at posturographic analysis in the clinical context: Sway-density versus other parameterization techniques. Mot. Control 2002, 6, 246–270. [Google Scholar] [CrossRef]
- Raymakers, J.A.; Samson, M.M.; Verhaar, H.J.J. The assessment of body sway and the choice of the stability parameter(s). Gait Posture 2005, 21, 48–58. [Google Scholar] [CrossRef] [PubMed]
- Peterka, R.J. Sensorimotor integration in human postural control. J. Neurophysiol. 2002, 88, 1097–1118. [Google Scholar] [PubMed]
- Winter, D.A. Human balance and posture control during standing and walking. Gait Posture 1995, 3, 193–214. [Google Scholar] [CrossRef]
- Teasdale, N.; Simoneau, M. Attentional demands for postural control: The effects of aging and sensory reintegration. Gait Posture 2001, 14, 203–210. [Google Scholar] [CrossRef]
- Rodriguez-Merchan, E.C. Articular Bleeding in Hemophilia. Cardiovasc. Hematol. Disord. Drug Targets 2016, 16, 21–24. [Google Scholar] [CrossRef] [PubMed]
- Runge, C.F.; Shupert, C.L.; Horak, F.B.; Zajac, F.E. Ankle and hip postural strategies defined by joint torques. Gait Posture 1999, 10, 161–170. [Google Scholar] [CrossRef]
- Patel, M.; Fransson, P.A.; Lush, D.; Petersen, H.; Magnusson, M.; Johansson, R.; Gomez, S. The effects of foam surface properties on standing body movement. Acta Otolaryngol. 2008, 128, 952–960. [Google Scholar] [CrossRef] [PubMed]
- Gurfinkel, V.S.; Lipshits, M.I.; Mori, S.; Popov, K.E. Stabilization of body position as the main task of postural regulation. Hum. Physiol. 1981, 7, 155–165. [Google Scholar] [PubMed]
- Soames, R.W.; Atha, J. The role of the antigravity musculature during quiet standing in man. Eur. J. Appl. Physiol. Occup. Physiol. 1981, 47, 159–167. [Google Scholar] [CrossRef] [PubMed]
- Corbeil, P.; Blouin, J.S.; Teasdale, N. Effects of intensity and locus of painful stimulation on postural stability. Pain 2004, 108, 43–50. [Google Scholar] [CrossRef] [PubMed]
- Forestier, N.; Teasdale, N.; Nougier, V. Alteration of the position sense at the ankle induced by muscular fatigue in humans. Med. Sci. Sports Exerc. 2002, 34, 117–122. [Google Scholar] [CrossRef] [PubMed]
- Hill, K.; Fearn, M.; Williams, S.; Mudge, L.; Walsh, C.; McCarthy, P.; Walsh, M.; Street, A. Effectiveness of a balance training home exercise programme for adults with haemophilia: A pilot study. Haemophilia 2010, 16, 162–169. [Google Scholar] [CrossRef] [PubMed]
| Variables | Healthy Group n = 12 | Haemophiliac Group n = 8 |
|---|---|---|
| Age (years) | 14.8 ± 1.4 (13–17) | 15.0 ± 1.4 (14–17) |
| Height (m) | 1.69 ± 0.1 (1.54–1.82) | 1.66 ± 0.1 (1.52–1.79) |
| Weight (kg) | 56.7 ± 9.8 (40–69) | 56.1 ± 8.0 (40.8–65.7) |
| BMI (kg/m2) | 19.8 ± 1.8 (16.8–22.3) | 20.4 ± 3.2 (17.4–27.3) |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Thabti, H.A.; Beausoleil, S.; Hue, O. The Effects of Haemophilia on the Postural Control of Adolescents. J. Funct. Morphol. Kinesiol. 2017, 2, 24. https://doi.org/10.3390/jfmk2030024
Thabti HA, Beausoleil S, Hue O. The Effects of Haemophilia on the Postural Control of Adolescents. Journal of Functional Morphology and Kinesiology. 2017; 2(3):24. https://doi.org/10.3390/jfmk2030024
Chicago/Turabian StyleThabti, Hafedh Alaeddine, Sarah Beausoleil, and Olivier Hue. 2017. "The Effects of Haemophilia on the Postural Control of Adolescents" Journal of Functional Morphology and Kinesiology 2, no. 3: 24. https://doi.org/10.3390/jfmk2030024
APA StyleThabti, H. A., Beausoleil, S., & Hue, O. (2017). The Effects of Haemophilia on the Postural Control of Adolescents. Journal of Functional Morphology and Kinesiology, 2(3), 24. https://doi.org/10.3390/jfmk2030024






