Evaluating Human Balance Following an Exercise Intervention in Previously Sedentary, Overweight Adults
Abstract
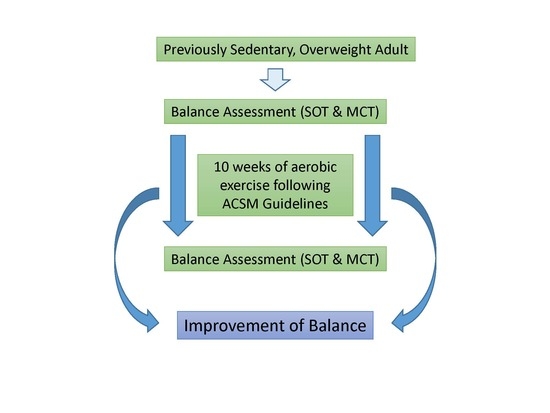
:1. Introduction
2. Materials and Methods
2.1. Participants
2.2. Measures and Data Analysis
2.3. Procedures
2.4. Statistical Analysis
3. Results
3.1. Physical Characteristics of Participants
3.2. Balance Evaluations
4. Discussion
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Corbeil, P.; Simoneau, M.; Rancourt, D.; Tremblay, A.; Teasdale, N. Increased risk for falling associated with obesity: Mathematical modeling of postural control. IEEE Trans. Neural Syst. Rehabil. Eng. 2001, 9, 126–136. [Google Scholar] [CrossRef] [PubMed]
- Goulding, A.; Jones, I.E.; Taylor, R.W.; Piggot, J.M.; Taylor, D. Dynamic and static tests of balance and postural sway in boys: Effects of previous wrist bone fractures and high adiposity. Gait Posture 2003, 17, 136–141. [Google Scholar] [CrossRef]
- Hue, O.; Simoneau, M.; Marcotte, J.; Berrigan, F.; Doré, J.; Marceau, P.; Marceau, S.; Tremblay, A.; Teasdale, N. Body weight is a strong predictor of postural stability. Gait Posture 2007, 26, 32–38. [Google Scholar] [CrossRef] [PubMed]
- Hita-Contreras, F.; Martínez-Amat, A.; Lomas-Vega, R.; Álvarez, P.; Mendoza, N.; Romero-Franco, N.; Aránega, A. Relationship of body mass index and body fat distribution with postural balance and risk of falls in Spanish postmenopausal women. Menopause 2012, 20, 202–208. [Google Scholar] [CrossRef] [PubMed]
- McGraw, B.; McClenaghan, B.A.; Williams, H.G.; Dickerson, J.; Ward, D.S. Gait and postural stability in obese and nonobese prepubertal boys. Arch. Phys. Med. Rehabil. 2000, 81, 484–489. [Google Scholar] [CrossRef] [PubMed]
- Del Porto, H.C.; Pechak, C.M.; Smith, D.R.; Reed-Jones, R.J. Biomechanical effects of obesity on balance. Int. J. Exerc. Sci. 2012, 5, 301–320. [Google Scholar]
- Bernard, P.L.; Geraci, M.; Hue, O.; Amato, M.; Seynnes, O.; Lantieri, D. Influence of obesity on postural capacities of teenagers. Preliminary study. Ann. Phys. Rehabil. Med. 2003, 46, 184–190. [Google Scholar]
- Deforche, B.I.; Hills, A.P.; Worringham, C.J.; Davies, P.S.; Murphy, A.J.; Bouckaert, J.J.; De Bourdeaudjui, I.M. Balance and postural skills in normal-weight and overweight prepubertal boys. Int. J. Pediatr. Obes. 2009, 4, 175–182. [Google Scholar] [CrossRef] [PubMed]
- Singh, D.; Park, W.; Levy, M.S.; Jung, E.S. The effects of obesity and standing time on postural sway during prolonged quiet standing. Ergonomics 2009, 52, 977–986. [Google Scholar] [CrossRef] [PubMed]
- Menegoni, F.; Galli, M.; Tacchini, E.; Vismara, L.; Cavigioli, M.; Capodaglio, P. Gender-specific effect of obesity on balance. Obesity 2009, 17, 1951–1956. [Google Scholar] [CrossRef] [PubMed]
- Fabris de Souza, S.A.; Faintuch, J.; Valezi, A.C.; Sant’Anna, A.F.; Gama-Rodrigues, J.J.; De Batista Fonseca, I.C.; De Melo, R.D. Postural changes in morbidly obese patients. Obes. Surg. 2005, 15, 1013–1016. [Google Scholar] [CrossRef] [PubMed]
- Matrangola, S.L.; Madigan, M.L. The effects of obesity on balance recovery using an ankle strategy. Hum. Movement Sci. 2011, 30, 584–595. [Google Scholar] [CrossRef] [PubMed]
- Błaszczyk, J.W.; Cieślinska-Świder, J.; Plewa, M.; Zahorska-Markiewicz, B.; Markiewicz, A. Effects of excessive body weight on postural control. J. Biomech. 2009, 42, 1295–1300. [Google Scholar] [CrossRef] [PubMed]
- Handrigan, G.; Hue, O.; Simoneau, M.; Corbeil, P.; Marceau, P.; Marceau, S.; Tremblay, A.; Teasdale, N. Weight loss and muscular strength affect static balance control. Int. J. Obes. 2010, 34, 936–942. [Google Scholar] [CrossRef] [PubMed]
- Maffiuletti, N.A.; Agosti, F.; Proietti, M.; Riva, D.; Resnik, M.; Lafortuna, C.L.; Sartorio, A. Postural instability of extremely obese individuals improves after a body weight reduction program entailing specific balance training. J. Endocrinol. Invest. 2005, 28, 2–7. [Google Scholar] [CrossRef] [PubMed]
- Teasdale, N.; Hue, O.; Marcotte, J.; Berrigan, F.; Simoneau, M.; Doré, J.; Marceau, P.; Marceau, S.; Tremblay, A. Reducing weight increases postural stability in obese and morbid obese men. Int. J. Obes. 2007, 31, 153–160. [Google Scholar] [CrossRef] [PubMed]
- Morris, C.E.; Garner, J.C.; Owens, S.G.; Valliant, M.W.; Loftin, M. Evaluation of the accuracy of a previously published equation to predict energy expenditure per unit distance following an exercise intervention in previously sedentary overweight adults. Gazz. Med. Ital. 2017, 176, 1–9, in press. [Google Scholar]
- Thomas, S.; Reading, J.; Shephard, R.J. Revision of the physical activity readiness questionnaire (par-q). Can. J. Sport Sci. 1992, 17, 338–345. [Google Scholar] [PubMed]
- Sallis, J.F.; Haskell, W.L.; Wood, P.D. Physical activity assessment methodology in the five-city project. Am. J. Epidemiol. 1985, 121, 91–106. [Google Scholar] [CrossRef] [PubMed]
- American College of Sports Medicine. ACSM’s Guidelines for Exercise Testing and Prescription, 9th ed.; Wolters Kluwer/Lippincott Williams, Wilkins: Philadelphia, PA, USA, 2014. [Google Scholar]
- Nashner, L. Computerized dynamic posturography. In The Handbook of Balance Function and Testing; Jacobson, G., Newman, C., Kartush, J., Eds.; Mosby Year Book: St. Louis, MO, USA, 1993; pp. 261–279. [Google Scholar]
- Guskiewicz, K.M.; Perrin, D.H. Research and clinical applications of assessing balance. J. Sport Rehabil. 1996, 5, 45–63. [Google Scholar] [CrossRef]
- Chander, H.; Garner, J.C.; Wade, C. Impact on balance while walking in occupational footwear. Footwear Sci. 2014, 6, 59–66. [Google Scholar] [CrossRef]
- Chander, H.; MacDonald, C.J.; Dabbs, N.C.; Allen, C.R.; Lamont, H.S.; Garner, J.C. Balance Performance in Female Collegiate Athletes. J. Sports Sci. Med. 2014, 2, 13–20. [Google Scholar]
- Chander, H.; Morris, C.E.; Wilson, S.J.; Garner, J.C.; Wade, C. Impact of alternative footwear on human balance. Footwear Sci. 2016, 8, 165–174. [Google Scholar] [CrossRef]
- Bravata, D.M.; Smith-Spangler, C.; Sundaram, V.; Gienger, A.L.; Lin, N.; Lewis, R.; Stave, C.D.; Olkin, I.; Sirard, J.R. Using pedometers to increase physical activity and improve health: A systematic review. J. Am. Med. Assoc. 2007, 298, 2296–2304. [Google Scholar] [CrossRef] [PubMed]
- Billot, M.; Simoneau, E.M.; Van Hoecke, J.; Martin, A. Age-related relative increases in electromyography activity and torque according to the maximal capacity during upright standing. Eur. J. Appl. Physiol. 2010, 109, 669–680. [Google Scholar] [CrossRef] [PubMed]
- Cattagni, T.; Scaglioni, G.; Laroche, D.; Van Hoecke, J.; Gremeaux, V.; Martin, A. Ankle muscle strength discriminates fallers from non-fallers. Front. Aging Neurosci. 2014, 6, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Kouzaki, M.; Shinohar, M. Steadiness in plantar flexor muscles and its relation to postural sway in young and elderly adults. Muscle Nerve 2010, 42, 78–87. [Google Scholar] [CrossRef] [PubMed]
- Kandel, E.R.; Schwartz, J.H.; Jessell, T.M. Principles of Neural Science, 4th ed.; McGraw-Hill, Health Professions Division: New York, NY, USA, 2000. [Google Scholar]
| Physical Characteristics | Mean | SD | Min | Max | |
|---|---|---|---|---|---|
| Body Mass (kg) | Pre | 94.1 | 20.1 | 61.2 | 126.9 |
| Post | 93.1 | 20.7 | 59.82 | 122.9 | |
| FFM (kg) | Pre | 59.6 | 13.3 | 38.7 | 82.1 |
| Post | 59.8 | 14.3 | 38.9 | 81.8 | |
| FM (kg) | Pre | 33.6 | 9.0 | 21.8 | 51.0 |
| Post | 33.3 | 9.1 | 20.9 | 49.0 | |
| AFM (kg) | Pre | 11.9 | 5.0 | 6.3 | 22.7 |
| Post | 11.6 | 4.1 | 5.7 | 18.4 | |
| VO2 max (mL/kg/min) | Pre | 34.5 | 5.8 | 25.0 | 43.0 |
| Post | 37.4 | 7.8 | 26.3 | 57.1 | |
| EQ Scores | Pre-Intervention | Post-Intervention | Latencies | Pre-Intervention | Post-Intervention |
|---|---|---|---|---|---|
| SOT Conditions | Mean ± SD | Mean ± SD | MCT Conditions | Mean ± SD | Mean ± SD |
| EO | 93.6 ± 1.6 | 92.9 ± 2.6 | BWS (ms) | 136.7 ± 10.3 | 134.3 ± 8.6 |
| EC | 92.0 ± 2.2 | 92.0 ± 2.0 | BWM (ms) | 126.7 ± 12.2 | 126.3 ± 10.1 |
| EOSRV | 92.0 ± 2.4 | 92.7 ± 2.2 | BWL (ms) | 120.7 ± 11.0 | 120.3 ± 12.5 |
| EOSRP | 83.0 ± 9.4 | 81.6 ± 13.7 | FWS (ms) | 150.7 ± 18.0 | 149.3 ± 17.3 |
| ECSRP | 68.1 ± 10.2 | 69.9 ± 7.1 | FWM (ms) | 131.0 ± 14.4 | 131.3 ± 13.4 |
| EOSRVP | 69.8 ± 11.1 | 72.8 ± 10.0 * | FWL (ms) | 125.7 ± 13.7 | 126.0 ± 11.8 |
| Composite | 80.2 ± 6.3 | 81.1 ± 6.7 | Composite (ms) | 128.5 ± 9.6 | 127.7 ± 11.5 |
| SOT Conditions | Pre-Intervention | Post-Intervention | SOT Conditions | Pre-Intervention | Post-Intervention |
|---|---|---|---|---|---|
| EO | Mean ± SD | Mean ± SD | EOSRP | Mean ± SD | Mean ± SD |
| MLVEL (cm/s) | 0.74 ± 0.1 | 0.75 ± 0.1 | MLVEL (cm/s) | 0.93 ± 0.2 | 0.91 ± 0.2 |
| APVEL (cm/s) | 1.03 ± 0.2 | 1.06 ± 0.3 | APVEL (cm/s) | 1.86 ± 0.7 | 1.64 ± 0.5 |
| MLRMS (cm) | 0.15 ± 0.1 | 0.18 ± 0.1 | MLRMS (cm) | 0.23 ± 0.1 | 0.26 ± 0.2 |
| APRMS (cm) | 0.32 ± 0.1 | 0.34 ± 0.1 | APRMS (cm) | 0.98 ± 0.7 | 1.11 ± 1.1 |
| EC | ECSRP | ||||
| MLVEL (cm/s) | 0.82 ± 0.2 | 0.81 ± 0.2 | MLVEL (cm/s) | 1.28 ± 0.3 | 1.24 ± 0.3 |
| APVEL (cm/s) | 1.36 ± 0.4 | 1.23 ± 0.40 * | APVEL (cm/s) | 3.40 ± 0.8 | 3.16 ± 0.7 |
| MLRMS (cm) | 0.19 ± 0.1 | 0.18 ± 0.09 | MLRMS (cm) | 0.38 ± 0.2 | 0.38 ± 0.2 |
| APRMS (cm) | 0.42 ± 0.1 | 0.40 ± 0.1 | APRMS (cm) | 1.69 ± 0.6 | 1.64 ± 0.5 |
| EOSRV | EOSRVP | ||||
| ML EL (cm/s) | 0.79 ± 0.1 | 0.75 ± 0.1 | MLVEL (cm/s) | 1.07 ± 0.2 | 1.06 ± 0.2 |
| APVEL (cm/s) | 1.20 ± 0.3 | 1.11 ± 0.2 * | APVEL (cm/s) | 2.77 ± 0.6 | 2.54 ± 0.5 |
| MLRMS (cm) | 0.17 ± 0.1 | 0.15 ± 0.1 | MLRMS (cm) | 0.29 ± 0.1 | 0.29 ± 0.2 |
| APRMS (cm) | 0.42 ± 0.1 | 0.37 ± 0.1 | APRMS (cm) | 1.76 ± 0.8 | 1.62 ± 0.8 |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Morris, C.E.; Chander, H.; Garner, J.C.; DeBusk, H.; Owens, S.G.; Valliant, M.W.; Loftin, M. Evaluating Human Balance Following an Exercise Intervention in Previously Sedentary, Overweight Adults. J. Funct. Morphol. Kinesiol. 2017, 2, 19. https://doi.org/10.3390/jfmk2020019
Morris CE, Chander H, Garner JC, DeBusk H, Owens SG, Valliant MW, Loftin M. Evaluating Human Balance Following an Exercise Intervention in Previously Sedentary, Overweight Adults. Journal of Functional Morphology and Kinesiology. 2017; 2(2):19. https://doi.org/10.3390/jfmk2020019
Chicago/Turabian StyleMorris, Cody E., Harish Chander, John C. Garner, Hunter DeBusk, Scott G. Owens, Melinda W. Valliant, and Mark Loftin. 2017. "Evaluating Human Balance Following an Exercise Intervention in Previously Sedentary, Overweight Adults" Journal of Functional Morphology and Kinesiology 2, no. 2: 19. https://doi.org/10.3390/jfmk2020019
APA StyleMorris, C. E., Chander, H., Garner, J. C., DeBusk, H., Owens, S. G., Valliant, M. W., & Loftin, M. (2017). Evaluating Human Balance Following an Exercise Intervention in Previously Sedentary, Overweight Adults. Journal of Functional Morphology and Kinesiology, 2(2), 19. https://doi.org/10.3390/jfmk2020019