Stress for Vertebral Bodies and Intervertebral Discs with Respect to Squatting Depth
Abstract
:1. Introduction
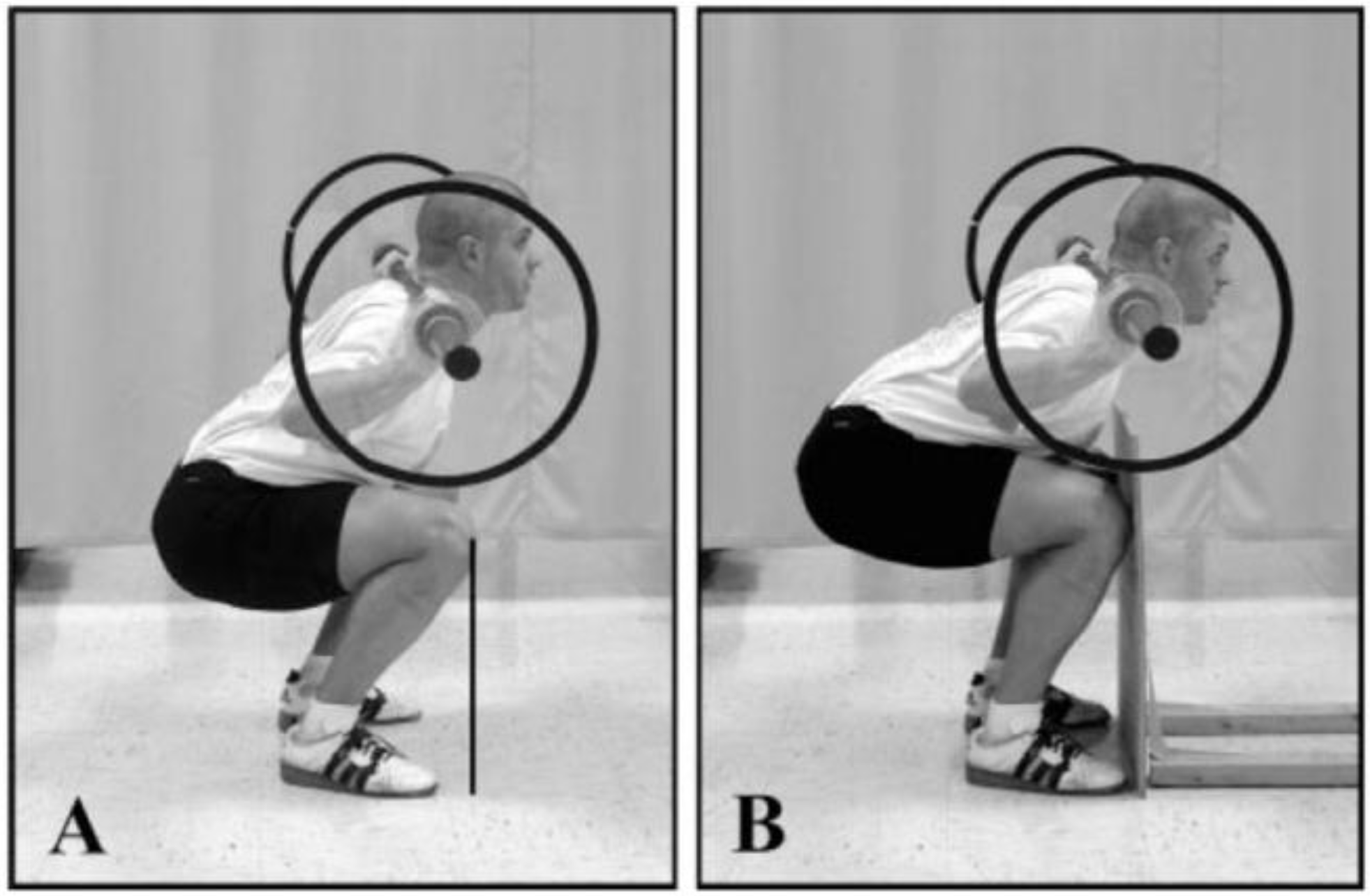
2. Materials and Methods
3. Results
4. Discussion
4.1. Comparison of the Calculated Compressive Forces in the Half and Quarter Squat with Experimentally-Determined Strength Values of Vertebral Bodies and Segments
4.2. Comparison of the Calculated Compressive Forces in the Parallel and Deep Squat with Experimentally-Determined Strength Values of Vertebral Bodies and Segments
4.3. Incidence and Risk of Injuries of the Vertebral Column in Deep Squats
4.4. Risk of Injuries of the Knee Joint and Vertebral Column Due to the Restriction of the Anterior Knee Displacement in the Squat
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
Abbreviations
| BMC | Bone mineral content |
| BMD | Bone mineral density |
| MPa | Megapascal (N/mm2) |
| N | Newton |
| CSA | Muscle cross-sectional area |
| 1-RM | One repetition maximum |
| RM | Repetition maximum |
References
- Wretenberg, P.; Feng, Y.; Arborelius, U.P. High- and low-bar squatting techniques during weight-training. Med. Sci. Sports Exerc. 1996, 28, 218–224. [Google Scholar] [CrossRef] [PubMed]
- Caterisano, A.; Moss, R.F.; Pellinger, T.K.; Woodruff, K.; Lewis, V.C.; Booth, W.; Khadra, T. The effect of back squat depth on the EMG activity of 4 superficial hip and thigh muscles. J. Strength Cond. Res. 2002, 16, 428–432. [Google Scholar] [CrossRef] [PubMed]
- Wilson, G.J. Strength and power in sport. In Applied Anatomy and Biomechanics in Sport, 3rd ed.; Bloomfield, J., Ackland, T.R., Elliott, B.C., Eds.; Blackwell: Berlin, Germany, 1998; pp. 110–208. [Google Scholar]
- Cotter, J.A.; Chaudhari, A.M.; Jamison, S.T.; Devor, S.T. Knee joint kinetics in relation to commonly prescribed squat loads and depths. J. Strength Cond. Res. 2013, 27, 1765–1774. [Google Scholar] [CrossRef] [PubMed]
- Fry, A.C.; Aro, T.A.; Bauer, J.A.; Kraemer, W.J. A comparison of methods for determining kinematic properties of three barbell squat exercises. J. Hum. Mov. Stud. 1993, 24, 83–95. [Google Scholar]
- Wretenberg, P.; Feng, Y.; Lindberg, F.; Arborelius, U.P. Joint moments of force and quadriceps muscle activity during squatting exercise. Scand. J. Med. Sci. Sports 1993, 3, 244–250. [Google Scholar] [CrossRef]
- Ratamess, N.A.; Alvar, B.A.; Evetoch, T.K.; Housh, T.J.; Kibler, W.B.; Kraemer, W.J.; Triplett, N.T. American College of Sports Medicine position stand. Progression models in resistance training for healthy adults. Med. Sci. Sports Exerc. 2009, 41, 687–708. [Google Scholar]
- Kraemer, W.J.; Ratamess, N.A. Fundamentals of resistance training: Progression and exercise prescription. Med. Sci. Sports Exerc. 2004, 36, 674–688. [Google Scholar] [CrossRef] [PubMed]
- Schmidtbleicher, D. Training for power events. In Strength and Power in Sport, 2nd ed.; Komi, P., Ed.; Blackwell Science: Oxford, UK, 1992; pp. 381–395. [Google Scholar]
- Schoenfeld, B. The mechanisms of muscle hypertrophy and their application to resistance training. J. Strength Cond. Res. 2010, 24, 2857–2872. [Google Scholar] [CrossRef] [PubMed]
- Stone, M.H.; Stone, M.; Sands, W. Principles and Practice of Resistance Training; Human Kinetics: Champaign, IL, USA, 2007. [Google Scholar]
- Wernbom, M.; Augustsson, J.; Thomeé, R. The influence of frequency, intensity, volume and mode of strength training on whole muscle cross-sectional area in humans. Sports Med. 2007, 37, 225–264. [Google Scholar] [CrossRef] [PubMed]
- Fry, A.C. The role of resistance exercise intensity on muscle fibre adaptations. Sports Med. 2004, 34, 663–679. [Google Scholar] [CrossRef] [PubMed]
- Wirth, K.; Zawieja, M. Experiences from weightlifting for competitive strength training. Part 2: Differences in periodization and designing of strength training in weightlifting and other sports with high demands on speed-strength. Leistungssport 2008, 38, 50–54. (In German) [Google Scholar]
- Zatsiorsky, V.M.; Kraemer, W. Science and Practice of Strength Training, 2nd ed.; Human Kinetics: Champaign, IL, USA, 2006. [Google Scholar]
- Hoff, J.; Helgerud, J. Maximal strength training enhances running economy and aerobic endurance performance. In Football (Soccer). New Developments in Physical Training Research; Hoff, J., Helgerud, J., Eds.; NTNU: Trondheim, Norway, 2003; pp. 39–55. [Google Scholar]
- Hartmann, H.; Wirth, K.; Klusemann, M.; Dalic, J.; Matuschek, C.; Schmidtbleicher, D. Influence of squatting depth on jumping performance. J. Strength Cond. Res. 2012, 26, 3243–3261. [Google Scholar] [CrossRef] [PubMed]
- Wirth, K.; Zawieja, M. Experiences from weightlifting for competitive strength training. Part 1: Importance of competition and training exercises of weightlifting for the development of speed-strength. Leistungssport 2008, 38, 10–13. (In German) [Google Scholar]
- Cappozzo, A.; Felici, F.; Figura, F.; Gazzani, F. Lumbar spine loading during half-squat exercises. Med. Sci. Sports Exerc. 1985, 17, 613–620. [Google Scholar] [CrossRef] [PubMed]
- Kuo, C.-S.; Hu, H.-T.; Huang, K.-Y.; Zhong, Z.-C.; Hseih, M.-L. Biomechanical analysis of the lumbar spine on facet joint and intradiscal pressure—A finite element study. BMC Musculoskelet. Disord. 2010, 11, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Bressel, E.; Willardson, J.M.; Thompson, B.; Fontana, F.E. Effect of instruction, surface stability, and load intensity on trunk muscle activity. J. Electromyogr. Kinesiol. 2009, 19, e500–e504. [Google Scholar] [CrossRef] [PubMed]
- Nuzzo, J.L.; McCaulley, G.O.; Cormie, P.; Cavill, M.J.; McBride, J.M. Trunk muscle activity during stability ball and free weight exercises. J Strength Cond. Res. 2008, 22, 95–102. [Google Scholar] [CrossRef] [PubMed]
- Potvin, J.R.; Norman, R.W.; McGill, S.M. Reduction in anterior shear forces on the L4/L5 disc by the lumbar musculature. Clin. Biomech. 1991, 6, 88–96. [Google Scholar] [CrossRef]
- Dintiman, G.; Ward, B. Sports Speed, 3rd ed.; Human Kinetics: Champaign, IL, USA, 2003. [Google Scholar]
- Helgerud, J.; Kemi, O.J.; Hoff, J. Pre-season concurrent strength and endurance development in elite soccer players. In Football (Soccer). New Developments in Physical Training Research; Hoff, J., Helgerud, J., Eds.; NTNU: Trondheim, Norway, 2003; pp. 55–66. [Google Scholar]
- Rønnestad, B.R. Comparing the performance-enhancing effects of squats on a vibration platform with conventional squats in recreationally resistance-trained men. J. Strength Cond. Res. 2004, 18, 839–845. [Google Scholar] [CrossRef] [PubMed]
- Schoenfeld, B.J. Squatting kinematics and kinetics and their application to exercise performance. J. Strength Cond. Res. 2010, 24, 3497–3506. [Google Scholar] [CrossRef] [PubMed]
- Lander, J.E.; Simonton, R.L.; Giacobbe, J.K.F. The effectiveness of weight-belts during the squat exercise. Med. Sci. Sports Exerc. 1990, 22, 117–126. [Google Scholar] [CrossRef] [PubMed]
- Aggrawal, N.D.; Kaur, R.; Kumar, S.; Mathur, D.N. A study of changes in the spine in weight lifters and other athletes. Br. J. Sports Med. 1979, 13, 58–61. [Google Scholar] [CrossRef] [PubMed]
- Baranto, A.; Hellström, M.; Cederlund, C.-G.; Nyman, R.; Swärd, L. Back pain and MRI changes in the thoraco-lumbar spine of top athletes in four different sports: A 15-year follow-up study. Knee Surg. Sports Traumatol. Arthrosc. 2009, 17, 1125–1134. [Google Scholar] [CrossRef] [PubMed]
- Dalichau, S.; Scheele, K. Are there any influences of sports mechanic demands in weightlifting on the thoracolumbar curvature? Schweiz. Z. Sportmed. Sporttraumatol. 2001, 49, 119–123. (In German) [Google Scholar]
- Räty, H.P.; Battié, M.C.; Videman, T.; Sarna, S. Lumbar mobility in former elite male weightlifters, soccer players, long-distance runners and shooters. Clin. Biomech. 1997, 12, 325–330. [Google Scholar] [CrossRef]
- List, R.; Gulay, T.; Stoop, M.; Lorenzetti, S. Kinematics of the trunk and the lower extremities during restricted and unrestricted squats. J. Strength Cond. Res. 2013, 27, 1529–1538. [Google Scholar] [CrossRef] [PubMed]
- Lorenzetti, S.; Gülay, T.; Stoop, M.; List, R.; Gerber, H.; Schellenberg, F.; Stüssi, E. Comparison of the angles and corresponding moments in the knee and hip during restricted and unrestricted squats. J. Strength Cond. Res. 2012, 28, 2829–2836. [Google Scholar] [CrossRef] [PubMed]
- McKean, M.R.; Dunn, P.K.; Burkett, B.J. The lumbar and sacrum movement pattern during the back squat exercise. J. Strength Cond. Res. 2010, 24, 2731–2741. [Google Scholar] [CrossRef] [PubMed]
- Walsh, J.C.; Quinlan, J.F.; Stapleton, R.; FitzPatrick, D.P.; McCormack, D. Three-dimensional motion analysis of the lumbar spine during “free squat” weight lift training. Am. J. Sports Med. 2007, 35, 927–932. [Google Scholar] [CrossRef] [PubMed]
- Brinckmann, P.; Biggemann, M.; Hilweg, D. Prediction of the compressive strength of human lumbar vertebrae. Clin. Biomech. Suppl. 1989, 2, S1–S27. [Google Scholar]
- Hutton, W.C.; Adams, M.A. Can the lumbar spine be crushed in heavy lifting? Spine 1982, 7, 586–590. [Google Scholar] [CrossRef] [PubMed]
- Hutton, W.C.; Cyron, B.M.; Stott, J.R.R. The compressive strength of lumbar vertebrae. J. Anat. 1979, 129, 753–758. [Google Scholar] [PubMed]
- Jäger, M.; Luttmann, A. Compressive strength of lumbar elements related to age, gender, and other influencing factors. In Electromyographical Kinesiology; Anderson, P.A., Hobart, D.J., Danoff, J.V., Eds.; Excerpta Medica: Amsterdam, The Netherlands; New York, NY, USA; Oxford, UK, 1991; pp. 291–294. [Google Scholar]
- Porter, R.W.; Adams, M.A.; Hutton, W.C. Physical activity and the strength of the lumbar spine. Spine 1989, 14, 201–203. [Google Scholar] [CrossRef] [PubMed]
- Adams, M.A.; Hutton, W.C. Gradual disc prolapse. Spine 1985, 10, 524–531. [Google Scholar] [CrossRef] [PubMed]
- Krismer, M.; Haid, Ch.; Rabl, W. The contribution of anulus fibers to torque resistance. Spine 1996, 21, 2551–2557. [Google Scholar] [CrossRef] [PubMed]
- Hickey, D.S.; Hukins, D.L. Relation between the structure of the annulus fibrosus and the function and failure of the intervertebral disc. Spine 1980, 5, 106–116. [Google Scholar] [CrossRef] [PubMed]
- Schmidt, H.; Kettler, A.; Heuer, F.; Simon, U.; Claes, L.; Wilke, H.-J. Intradiscal pressure, shear strain, and fiber strain in the intervertebral disc under combined loading. Spine 2007, 32, 748–755. [Google Scholar] [CrossRef] [PubMed]
- Bader, D.L.; Bouten, C. Biomechanics of soft tissues. In Clinical Biomechanics; Dvir, Z., Ed.; Churchill Livingstone: New York, NY, USA; Edinburgh, Scotland; London, UK; Philadelphia, PA, USA, 2000; pp. 35–64. [Google Scholar]
- Cormie, P.; McBride, J.M.; McCaulley, G.O. Power-time, force-time, and velocity-time curve analysis of the countermovement jump: Impact of training. J. Strength Cond. Res. 2009, 23, 177–186. [Google Scholar] [CrossRef] [PubMed]
- Granhed, H.; Jonson, R.; Hansson, T. The loads on the lumbar spine during extreme weight lifting. Spine 1987, 12, 146–149. [Google Scholar] [CrossRef] [PubMed]
- Jäger, M.; Luttmann, A. Biomechanical analysis and assessment of lumbar stress during load lifting using a dynamic 19-segment human model. Ergonomics 1989, 32, 93–112. [Google Scholar] [CrossRef] [PubMed]
- Gilsanz, V.; Boechat, M.I.; Gilsanz, R.; Loro, M.L.; Roe, T.F.; Goodman, W.G. Gender differences in vertebral sizes in adults: Biomechanical implications. Radiology 1994, 190, 678–682. [Google Scholar] [CrossRef] [PubMed]
- Ebbesen, E.N.; Thomsen, J.S.; Beck-Nielsen, H.; Nepper-Rasmussen, H.J.; Mosekilde, L. Age- and gender-related differences in vertebral bone mass, density, and strength. J. Bone Miner. Res. 1999, 14, 1394–1403. [Google Scholar] [CrossRef] [PubMed]
- Ebbesen, E.N.; Thomsen, J.S.; Beck-Nielsen, H.; Nepper-Rasmussen, H.J.; Mosekilde, L. Lumbar vertebral body compressive strength evaluated by dual-energy X-ray absorptiometry, quantitative computed tomography, and ashing. Bone 1999, 25, 713–724. [Google Scholar] [CrossRef]
- Conroy, B.P.; Kraemer, W.J.; Maresh, C.M.; Fleck, S.J.; Stone, M.H.; Fry, A.C.; Miller, P.D.; Dalsky, G.P. Bone mineral density in elite junior Olympic weightlifters. Med. Sci. Sports Exerc. 1993, 25, 1103–1109. [Google Scholar] [CrossRef] [PubMed]
- Fleck, S.J.; Kraemer, W.J. Designing Resistance Training Programs, 3rd ed.; Human Kinetics: Champaign, IL, USA, 2004. [Google Scholar]
- Lang, T.F.; Saeed, I.H.; Streeper, T.; Carballido-Gamio, J.; Harnish, R.J.; Frassetto, L.A.; Lee, S.M.; Sibonga, J.D.; Keyak, J.H.; Spiering, B.A.; et al. Spatial heterogeneity in the response of the proximal femur to two lower-body resistance exercise regimens. J. Bone Miner. Res. 2014, 29, 1337–1345. [Google Scholar] [CrossRef] [PubMed]
- Loehr, J.A.; Lee, S.M.; English, K.L.; Sibonga, J.; Smith, S.M.; Spiering, B.A.; Hagan, R.D. Musculoskeletal adaptations to training with the advanced resistive exercise device. Med. Sci. Sports Exerc. 2011, 43, 146–156. [Google Scholar] [CrossRef] [PubMed]
- Almstedt, H.C.; Canepa, J.A.; Ramirez, D.A.; Shoepe, T.C. Changes in bone mineral density in response to 24 weeks of resistance training in college-age men and women. J. Strength Cond. Res. 2011, 25, 1098–1103. [Google Scholar] [CrossRef] [PubMed]
- Dalsky, G.P. Exercise: Its effect on bone mineral content. Clin. Obstet. Gynecol. 1987, 30, 820–832. [Google Scholar] [CrossRef] [PubMed]
- Chilibeck, P.D.; Calder, A.; Sale, D.G.; Webber, C.E. Twenty weeks of weight training increases lean tissue mass but not bone mineral mass or density in healthy, active young women. Can. J. Physiol. Pharmacol. 1996, 74, 1180–1185. [Google Scholar] [CrossRef] [PubMed]
- Dinç, H.; Savci, G.; Demirci, A.; Sadikoğlu, M.Y.; Tuncel, E.; Yavuz, H. Quantitative computed tomography for measuring bone mineral density in athletes. Calcif. Tissue Int. 1996, 58, 398–401. [Google Scholar] [CrossRef] [PubMed]
- Karlsson, M.K.; Johnell, O.; Obrant, K.J. Bone mineral density in weight lifters. Calcif. Tissue Int. 1993, 52, 212–215. [Google Scholar] [CrossRef] [PubMed]
- Sabo, D.; Benrd, L.; Pfeil, J.; Reiter, A. Bone quality in the lumbar spine in high-performance athletes. Eur. Spine J. 1996, 5, 258–263. [Google Scholar] [CrossRef] [PubMed]
- Heinonen, A.; Oja, P.; Kannus, P.; Sievänen, H.; Mänttäri, A.; Vuori, I. Bone mineral density of female athletes in different sports. Bone Miner. 1993, 23, 1–14. [Google Scholar] [CrossRef]
- Tszuzuku, S.; Ikegami, Y.; Yabe, K. Effects of high-intensity resistance training on bone mineral density in young male powerlifters. Calcif. Tissue Int. 1998, 63, 283–286. [Google Scholar] [CrossRef]
- Holm, S.; Nachemson, A. Variations in the nutrition of the canine intervertebral disc induced by motion. Spine 1983, 8, 866–874. [Google Scholar] [CrossRef] [PubMed]
- Säämänen, A.-M.; Puustjärvi, K.; Ilves, K.; Lammi, M.; Kiviranta, I.; Jurvelin, J.; Helminen, H.J.; Tammi, M. Effect of running exercise on proteoglycans and collagen content in the intervertebral disc of young dogs. Intern. J. Sports Med. 1993, 14, 48–51. [Google Scholar] [CrossRef] [PubMed]
- Brisby, H.; Wei, A.Q.; Molloy, T.; Chung, S.A.; Murrell, G.A.; Diwan, A.D. The effect of running exercise on intervertebral disc extracellular matrix production in a rat model. Spine 2010, 35, 1429–1436. [Google Scholar] [CrossRef] [PubMed]
- Sasaki, N.; Henriksson, H.B.; Runesson, E.; Larsson, K.; Sekiguchi, M.; Kikuchi, S.-I.; Konno, S.-I.; Rydevik, B.; Brisby, H. Physical exercise affects cell proliferation in lumbar intervertebral disc regions in rats. Spine 2012, 37, 1440–1447. [Google Scholar] [CrossRef] [PubMed]
- Wuertz, K.; Godburn, K.; MacLean, J.J.; Barbir, A.; Donnelly, J.S.; Roughley, P.J.; Alini, M.; Iatridis, J.C. In vivo remodeling of intervertebral discs in response to short- and long-term dynamic compression. J. Orthop. Res. 2009, 27, 1235–1242. [Google Scholar] [CrossRef] [PubMed]
- Alini, M.; Eisenstein, S.M.; Ito, K.; Little, Ch.; Kettler, A.A.; Masuda, K.; Melrose, J.; Ralphs, J.; Stokes, I.; Wilke, H.J. Are animal models useful for studying human disc disorders/degeneration? Eur. Spine J. 2008, 17, 2–19. [Google Scholar] [CrossRef] [PubMed]
- Ishihara, H.; McNally, D.S.; Urban, J.P.G.; Hall, A.C. Effects of hydrostatic pressure on matrix synthesis in different regions of the intervertebral disk. J. Appl. Physiol. 1996, 80, 839–846. [Google Scholar] [PubMed]
- Wilke, H.J.; Neef, P.; Caimi, M.; Hoogland, T.; Claes, E. New in vivo measurements of pressures in the intervertebral disc in daily life. Spine 2009, 24, 755–762. [Google Scholar] [CrossRef]
- Neidlinger-Wilke, C.; Würtz, K.; Urban, J.P.G.; Börm, W.; Arand, M.; Ignatius, A.; Wilke, H.-J.; Claes, L.E. Regulation of gene expression in intervertebral disc cells by low and high hydrostatic pressure. Eur. Spine J. 2006, 15 (Suppl. S3), S372–S378. [Google Scholar] [CrossRef] [PubMed]
- Parkkinen, J.J.; Lammi, M.J.; Helminen, H.J.; Tammi, M. Local stimulation of proteoglycan synthesis in articular cartilage explants by dynamic compression in vitro. J. Orthop. Res. 1992, 10, 610–620. [Google Scholar] [CrossRef] [PubMed]
- Tittel, K. Descriptive and functional anatomy of men; Urban and Fischer: Jena, Germany, 1994. (In German) [Google Scholar]
- Neumann, P.; Keller, T.; Ekström, L.; Hult, E.; Hansson, T. Structural properties of the anterior longitudinal ligament. Correlation with lumbar bone mineral content. Spine 1993, 18, 637–645. [Google Scholar] [CrossRef] [PubMed]
- Belavy, D.L.; Albracht, K.; Bruggemann, G.-P.; Vergroesen, P.-P.A.; van Dieen, J.H. Can exercise positively influence the intervertebral disc? Sports Med. 2016, 46, 473–485. [Google Scholar] [CrossRef] [PubMed]
- Granhed, H.; Morelli, B. Low back pain among retired wrestlers and heavyweight lifters. Am. J. Sports Med. 1988, 16, 530–533. [Google Scholar] [CrossRef] [PubMed]
- Videman, T.; Battié, M.C.; Gibbons, L.E.; Manninen, H.; Gill, K.; Fisher, L.D.; Koskenvuo, M. Lifetime exercise and disk degeneration: An MRI study of monozygotic twins. Med. Sci. Sports Exerc. 1997, 29, 1350–1356. [Google Scholar] [CrossRef] [PubMed]
- Fair, J.D. The tragic history of the military press in Olympic and world championship competition, 1928–1972. J. Sport Hist. 2001, 28, 345–374. [Google Scholar]
- Stone, M.H.; Fry, A.C.; Ritchie, M.; Stoessel-Ross, L.; Marsit, J.L. Injury potential and safety aspects of weightlifting movements. Strength Cond. J. 1994, 16, 15–21. [Google Scholar] [CrossRef]
- Byrd, R.; Pierce, K.; Rielly, L.; Brady, J. Young weightlifters’ performance across time. Sports Biomech. 2003, 2, 133–140. [Google Scholar] [CrossRef] [PubMed]
- Calhoon, G.; Fry, A.C. Injury rates and profiles of elite competitive weightlifters. J. Athl. Train. 1999, 34, 232–238. [Google Scholar] [PubMed]
- Hamill, B.P. Relative safety of weightlifting and weight training. J. Strength Cond. Res. 1994, 8, 53–57. [Google Scholar] [CrossRef]
- Lavallee, M.E.; Balam, T. An overview of strength training injuries: Acute and chronic. Curr. Sports Med. Rep. 2010, 9, 307–313. [Google Scholar] [CrossRef] [PubMed]
- Adams, M.A.; May, S.; Freeman, B.J.C.; Morrison, H.P.; Dolan, P. Effects of backward bending on lumbar intervertebral discs. Relevance to physical therapy treatments for low back pain. Spine 2000, 25, 431–437. [Google Scholar] [CrossRef] [PubMed]
- Cipriani, D.J.; Terry, M.E.; Haines, M.A.; Tibibnia, A.P.; Lyssanova, O. Effect of stretch frequency and sex on the rate of gain and rate of loss in muscle flexibility during a hamstring-stretching program: A randomized single-blind longitudinal study. J. Strength Cond. Res. 2012, 26, 2119–2129. [Google Scholar] [CrossRef] [PubMed]
- Lander, J.E.; Hundley, J.R.; Simonton, R.L. The effectiveness of weight-belts during multiple repetitions of the squat exercise. Med. Sci. Sports Exerc. 1992, 24, 603–609. [Google Scholar] [CrossRef] [PubMed]
- Trafimow, J.H.; Schipplein, O.D.; Novak, G.J.; Andersson, G.P. The effects of quadriceps fatigue on the technique in lifting. Spine 1993, 18, 364–367. [Google Scholar] [CrossRef] [PubMed]
- Hartmann, H.; Wirth, K.; Klusemann, M. Analysis of the load on the knee joint and vertebral column with changes in squatting depth and weight load. Sports Med. 2013, 43, 993–1008. [Google Scholar] [CrossRef] [PubMed]
- Fry, A.C.; Smith, Ch.; Schilling, B.K. Effect of knee position on hip and knee torques during the barbell squat. J. Strength Cond. Res. 2003, 17, 629–633. [Google Scholar] [CrossRef] [PubMed]
- Bandi, W. The retropatellar knee joint-diseases. Pathomechanics and pathologic anatomy, clinic and therapy; Verlag Hans Huber, Bern, Stuttgart: Wien, Austria, 1977. (In German) [Google Scholar]
- Huberti, H.H.; Hayes, W.C. Patellofemoral contact pressures. The influence of Q-angle and tendofemoral contact. J. Bone Jt. Surg. 1984, 66, 715–724. [Google Scholar]
- Nisell, R.; Ekholm, J. Patellar forces during knee extension. Scand. J. Rehabil. Med. 1985, 17, 63–74. [Google Scholar] [PubMed]
- Hehne, H.J. Biomechanics of the patellofemoral joint and its clinical relevance. Clin. Orthop. Relat. Res. 1990, 258, 73–85. [Google Scholar] [CrossRef] [PubMed]
- Eckstein, F.; Lemberger, B.; Gratzke, C.; Hudelmaier, M.; Glaser, C.; Englmeier, K.-H.; Reiser, M. In vivo cartilage deformation after different types of activity and its dependence on physical training status. Ann. Rheum. Dis. 2005, 64, 291–295. [Google Scholar] [CrossRef] [PubMed]
- Hille, E.; Schulitz, K.-P. Determination of contact surfaces of the femoropatellar joint with reference to the localization of chondromalacia. Z. Orthop. Ihre Grenzgeb. 1984, 122, 40–47. (In German) [Google Scholar] [CrossRef] [PubMed]
- Ahmed, A.M.; Burke, D.L.; Yu, A. In-vitro measurement of static pressure distribution in synovial joints-Part II: Retropatellar surface. J. Biomech. Eng. 1983, 105, 226–236. [Google Scholar] [PubMed]
- Glitsch, U.; Lundershausen, N.; Knieps, D.; Johannknecht, A.; Ellegast, R. Biomechanical analysis of knee joint stress for activities in squatting and kneeling. In 49th Annual Congress of the German Society for Occupational and Environmental Medicine e.V., 11–14 March 2009; DGAUM Verlag: Aachen, Germany, 2009; pp. 391–394. (In German)[Google Scholar]
- Zelle, J.; Barink, M.; de Waal Malefijt, M.; Verdonschot, N. Thigh-calf contact: Does it affect the loading of the knee in the high-flexion range? J. Biomech. 2009, 42, 587–593. [Google Scholar] [CrossRef] [PubMed]
- Collins, J.J. Antagonistic-synergistic muscle action at the knee during competitive weightlifting. Med. Biol. Eng. Comput. 1994, 32, 168–174. [Google Scholar] [CrossRef] [PubMed]
- McGill, S.M. The biomechanics of low back pain injury: Implications on current practice in industry and the clinic. J. Biomech. 1997, 30, 465–475. [Google Scholar] [CrossRef]
- Nisell, R.; Ekholm, J. Joint load during the parallel squat in powerlifting and force analysis of in vivo bilateral quadriceps tendon rupture. Scand. J. Sports Sci. 1986, 8, 63–70. [Google Scholar]
- Keiner, M.; Sander, A.; Wirth, K.; Caruso, O.; Immesberger, P.; Zawieja, M. Strength performance in youth: Trainability of adolescents and children in the back and front squats. J. Strength Cond. Res. 2013, 27, 357–362. [Google Scholar] [CrossRef] [PubMed]
- Faigenbaum, A.D.; Lloyd, R.S.; MacDonald, J.; Myer, G.D. Citius, Altius, Fortius: Beneficial effects of resistance training for young athletes. Br. J. Sports Med. 2016, 50, 3–7. [Google Scholar] [CrossRef] [PubMed]
- Comfort, P.; Stewart, A.; Bloom, L.; Clarkson, B. Relationships between strength, sprint, and jump performance in well-trained youth soccer players. J. Strength Cond. Res. 2014, 28, 173–177. [Google Scholar] [CrossRef] [PubMed]
- Haff, G.G.; Nimphius, S. Training principles for power. Strength Cond. J. 2012, 34, 2–12. [Google Scholar] [CrossRef]
- Kerr, A. Strength and Conditioning in children and adolescent sport. In Sports Injuries in Children and Adolescents: An Essential Guide; Solomon, A., Ed.; Xlibris Corporation: Bloomington, IN, USA, 2013; pp. 355–366. [Google Scholar]
- Lloyd, R.S.; Faigenbaum, A.D.; Myer, G.D.; Stone, M.H.; Oliver, J.L.; Jeffreys, I.; Moody, J.; Brewer, C.; Pierce, K. UKSCA position statement: Youth resistance training. UK Strength Cond. Assoc. 2012, 26, 26–39. [Google Scholar]
- Sander, A.; Keiner, M.; Wirth, K.; Schmidtbleicher, D. Influence of a 2-year strength training programme on power performance in elite youth soccer players. Eur. J. Sport Sci. 2012, 13, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Lloyd, R.S.; Faigenbaum, A.D.; Stone, M.H.; Oliver, J.L.; Jeffreys, I.; Moody, J.A.; Brewer, C.; Pierce, K.C.; McCambridge, T.M.; Howard, R.; et al. Position statement on youth resistance training: The 2014 International Consensus. Br. J. Sports Med. 2014, 48, 498–505. [Google Scholar] [CrossRef] [PubMed]
- Bass, S.I.; Saxon, L.; Daly, R.M.; Turner, C.H.; Robling, A.G.; Seeman, E.; Stuckey, S. The effect of mechanical loading on the size and shape of bone in pre-, peri-, and postpubertal girls: A study in tennis players. J. Bone Miner. Res. 2002, 17, 2274–2280. [Google Scholar] [CrossRef] [PubMed]
- Kontulainen, S.; Sievänen, H.; Kannus, P.; Pasanen, M.; Vuori, I. Effect of long-term impact-loading on mass, size, and estimated strength of humerus and radius of female racquet-sports players: A peripheral quantitative computed tomography study between young and old starters and controls. J. Bone Miner. Res. 2003, 18, 352–359. [Google Scholar] [CrossRef] [PubMed]
- Warden, S.J.; Fuchs, R.K. Exercise and bone health: Optimising bone structure during growth is key, but all is not vain during ageing. Br. J. Sports Med. 2009, 43, 885–887. [Google Scholar] [CrossRef] [PubMed]
- Gunter, K.B.; Almstedt, H.C.; Janz, K.F. Physical activity in childhood may be the key to optimizing lifespan skeletal health. Exerc. Sport Sci. Rev. 2012, 40, 13–21. [Google Scholar] [CrossRef] [PubMed]
- Keiner, M.; Sander, A.; Wirth, K.; Schmidtbleicher, D. The impact of 2 years of additional athletic training on the jump performance of young athletes. Sci. Sports 2013, 29, e39–e46. [Google Scholar] [CrossRef]
- Bloomquist, K.; Langberg, H.; Karlsen, S.; Madsgaard, S.; Boesen, M.; Raastad, T. Effect of range of motion in heavy load squatting on muscle and tendon adaptations. Eur. J. Appl. Physiol. 2013, 113, 2133–2142. [Google Scholar] [CrossRef] [PubMed]
| Author | n | Structure | Compressive Strength (N) |
|---|---|---|---|
| Jäger and Luttmann [40] | 17 | L5 | 5000 |
| 8 | L5/S1-disc | 8000 |
| Author | Structure | Compressive Strength (N) | Age (Years) | Sex |
|---|---|---|---|---|
| Hutton et al. [39] | L3 | 12,931 | 46 | male |
| L5 | 15,559 | 21 | male | |
| Porter et al. [41] | L2/L3 | 13,954 | 25 | male |
| Brinckmann et al. [37] | L3/L4 | 6200 | 22 | female |
| L3/L4 | 8200 | 39 | male | |
| L4/L5 | 8800 | 22 | male | |
| Hutton and Adams [38] | L5/S1 | 12,740 | 22 | male |
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hartmann, H.; Wirth, K.; Mickel, C.; Keiner, M.; Sander, A.; Yaghobi, D. Stress for Vertebral Bodies and Intervertebral Discs with Respect to Squatting Depth. J. Funct. Morphol. Kinesiol. 2016, 1, 254-268. https://doi.org/10.3390/jfmk1020254
Hartmann H, Wirth K, Mickel C, Keiner M, Sander A, Yaghobi D. Stress for Vertebral Bodies and Intervertebral Discs with Respect to Squatting Depth. Journal of Functional Morphology and Kinesiology. 2016; 1(2):254-268. https://doi.org/10.3390/jfmk1020254
Chicago/Turabian StyleHartmann, Hagen, Klaus Wirth, Christoph Mickel, Michael Keiner, Andre Sander, and Dennis Yaghobi. 2016. "Stress for Vertebral Bodies and Intervertebral Discs with Respect to Squatting Depth" Journal of Functional Morphology and Kinesiology 1, no. 2: 254-268. https://doi.org/10.3390/jfmk1020254
APA StyleHartmann, H., Wirth, K., Mickel, C., Keiner, M., Sander, A., & Yaghobi, D. (2016). Stress for Vertebral Bodies and Intervertebral Discs with Respect to Squatting Depth. Journal of Functional Morphology and Kinesiology, 1(2), 254-268. https://doi.org/10.3390/jfmk1020254








