Shoulder Muscle Strength Assessment: A Comparative Study of Hand-Held Dynamometers and Load Cell Measurements
Abstract
1. Introduction
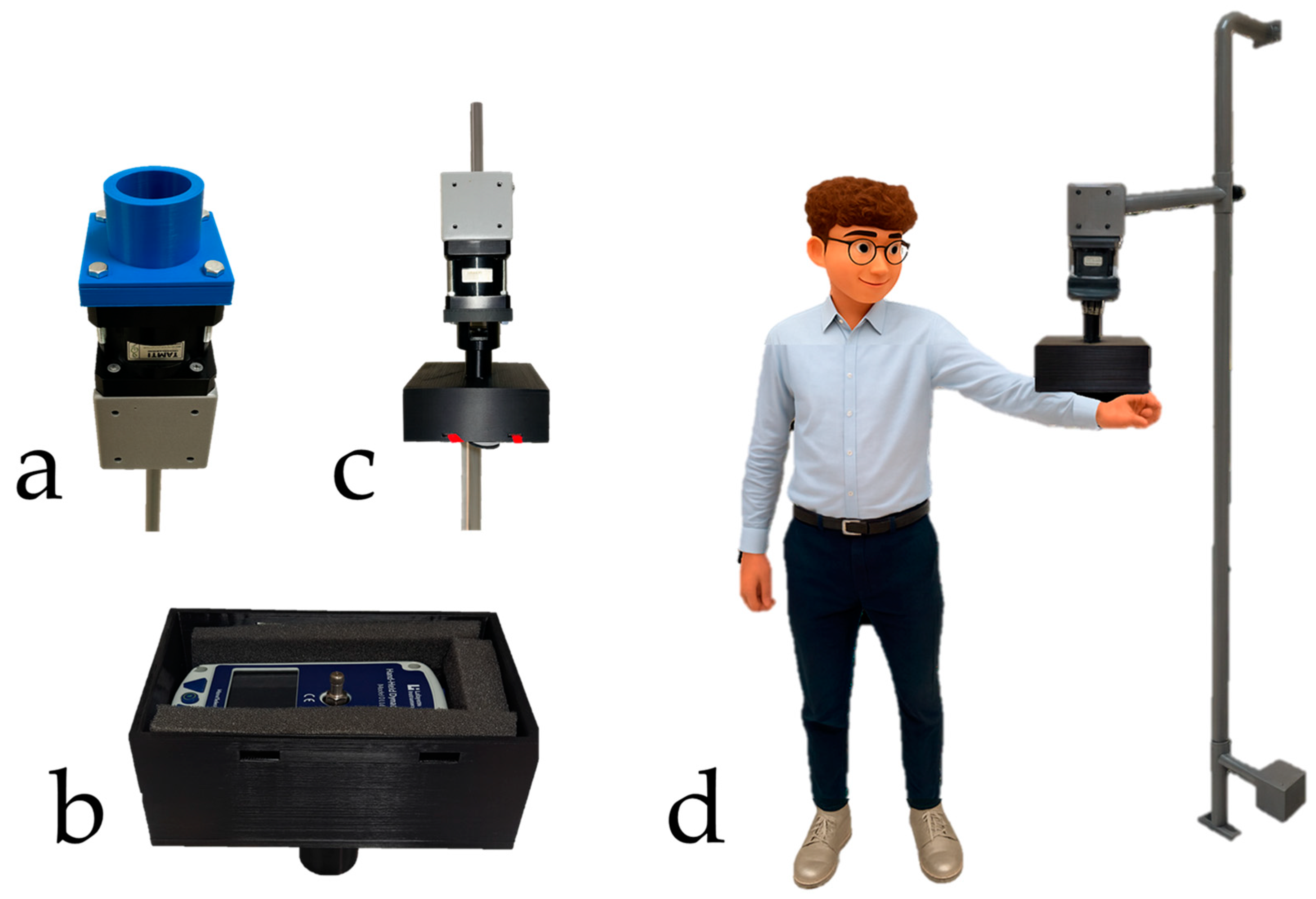
2. Materials and Methods
3. Results
4. Discussion and Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| HHD | Hand-held Dynamometer |
| ST | Standard Deviation |
| RMSE | Root Mean Square Error |
| MOD | Mean of Difference |
| LOA | Limits of Agreement |
References
- Matsuki, K.; Matsuki, K.O.; Mu, S.; Yamaguchi, S.; Ochiai, N.; Sasho, T.; Sugaya, H.; Toyone, T.; Wada, Y.; Takahashi, K.; et al. In vivo 3-dimensional analysis of scapular kinematics: Comparison of dominant and nondominant shoulders. J. Shoulder Elb. Surg. 2011, 20, 659–665. [Google Scholar] [CrossRef] [PubMed]
- Maffulli, N.; Longo, U.G.; Berton, A.; Loppini, M.; Denaro, V. Biological factors in the pathogenesis of rotator cuff tears. Sports Med. Arthrosc. Rev. 2011, 19, 194–201. [Google Scholar] [CrossRef]
- Rockwood, C.A. The Shoulder; Elsevier Health Sciences: Amsterdam, The Netherlands, 2009; Volume 1. [Google Scholar]
- Longo, U.G.; Risi Ambrogioni, L.; Berton, A.; Candela, V.; Massaroni, C.; Carnevale, A.; Stelitano, G.; Schena, E.; Nazarian, A.; DeAngelis, J.; et al. Scapular Dyskinesis: From Basic Science to Ultimate Treatment. Int. J. Environ. Res. Public Health 2020, 17, 2974. [Google Scholar] [CrossRef]
- Cools, A.M.; De Wilde, L.; Van Tongel, A.; Ceyssens, C.; Ryckewaert, R.; Cambier, D.C. Measuring shoulder external and internal rotation strength and range of motion: Comprehensive intra-rater and inter-rater reliability study of several testing protocols. J. Shoulder Elb. Surg. 2014, 23, 1454–1461. [Google Scholar] [CrossRef]
- Mera, B.J. Current Perspectives on Rotator Cuff Disease. Osteology 2022, 2, 62–69. [Google Scholar] [CrossRef]
- González-Rosalén, J.; Benítez-Martínez, J.C.; Medina-Mirapeix, F.; Cuerda-Del Pino, A.; Cervelló, A.; Martín-San Agustín, R. Intra- and Inter-Rater Reliability of Strength Measurements Using a Pull Hand-Held Dynamometer Fixed to the Examiner’s Body and Comparison with Push Dynamometry. Diagnostics 2021, 11, 1230. [Google Scholar] [CrossRef]
- Franceschi, F.; Longo, U.G.; Ruzzini, L.; Rizzello, G.; Denaro, V. Arthroscopic management of calcific tendinitis of the subscapularis tendon. Knee Surg. Sports Traumatol. Arthrosc. 2007, 15, 1482–1485. [Google Scholar] [CrossRef]
- Maffulli, N.; Longo, U.G. Conservative management for tendinopathy: Is there enough scientific evidence? Rheumatology 2008, 47, 390–391. [Google Scholar] [CrossRef]
- Constant, C.R.; Gerber, C.; Emery, R.J.; Søjbjerg, J.O.; Gohlke, F.; Boileau, P. A review of the Constant score: Modifications and guidelines for its use. J. Shoulder Elb. Surg. 2008, 17, 355–361. [Google Scholar] [CrossRef]
- Vrotsou, K.; Ávila, M.; Machón, M.; Mateo-Abad, M.; Pardo, Y.; Garin, O.; Zaror, C.; González, N.; Escobar, A.; Cuéllar, R. Constant-Murley Score: Systematic review and standardized evaluation in different shoulder pathologies. Qual. Life Res. 2018, 27, 2217–2226. [Google Scholar] [CrossRef]
- Booker, S.; Alfahad, N.; Scott, M.; Gooding, B.; Wallace, W.A. Use of scoring systems for assessing and reporting the outcome results from shoulder surgery and arthroplasty. World J. Orthop. 2015, 6, 244–251. [Google Scholar] [CrossRef] [PubMed]
- Kirkley, A.; Griffin, S.; Dainty, K. Scoring systems for the functional assessment of the shoulder. Arthroscopy 2003, 19, 1109–1120. [Google Scholar] [CrossRef] [PubMed]
- Ziegler, P.; Kühle, L.; Stöckle, U.; Wintermeyer, E.; Stollhof, L.E.; Ihle, C.; Bahrs, C. Evaluation of the Constant score: Which is the method to assess the objective strength? BMC Musculoskelet. Disord. 2019, 20, 403. [Google Scholar] [CrossRef]
- Burrus, C.; Deriaz, O.; Luthi, F.; Konzelmann, M. Role of pain in measuring shoulder strength abduction and flexion with the Constant-Murley score. Ann. Phys. Rehabil. Med. 2017, 60, 258–262. [Google Scholar] [CrossRef]
- Durfee, W.K.; Iaizzo, P. Rehabilitation and Muscle Testing; John Wiley and Sons: Hoboken, NJ, USA, 2006. [Google Scholar]
- Wikholm, J.B.; Bohannon, R.W. Hand-held Dynamometer Measurements: Tester Strength Makes a Difference. J. Orthop. Sports Phys. Ther. 1991, 13, 191–198. [Google Scholar] [CrossRef]
- Chamorro, C.; Arancibia, M.; Trigo, B.; Arias-Poblete, L.; Jerez-Mayorga, D. Absolute Reliability and Concurrent Validity of Hand-Held Dynamometry in Shoulder Rotator Strength Assessment: Systematic Review and Meta-Analysis. Int. J. Environ. Res. Public Health 2021, 18, 9293. [Google Scholar] [CrossRef]
- Decleve, P.; Van Cant, J.; De Buck, E.; Van Doren, J.; Verkouille, J.; Cools, A.M. The Self-Assessment Corner for Shoulder Strength: Reliability, Validity, and Correlations With Upper Extremity Physical Performance Tests. J. Athl. Train. 2020, 55, 350–358. [Google Scholar] [CrossRef]
- Hirano, M.; Katoh, M. Absolute reliability of shoulder joint horizontal adductor muscle strength measurements using a handheld dynamometer. J. Phys. Ther. Sci. 2015, 27, 2125–2127. [Google Scholar] [CrossRef][Green Version]
- Holt, K.L.; Raper, D.P.; Boettcher, C.E.; Waddington, G.S.; Drew, M.K. Hand-held dynamometry strength measures for internal and external rotation demonstrate superior reliability, lower minimal detectable change and higher correlation to isokinetic dynamometry than externally-fixed dynamometry of the shoulder. Phys. Ther. Sport 2016, 21, 75–81. [Google Scholar] [CrossRef]
- Croci, E.; Born, P.; Eckers, F.; Nüesch, C.; Baumgartner, D.; Müller, A.M.; Mündermann, A. Test-retest reliability of isometric shoulder muscle strength during abduction and rotation tasks measured using the Biodex dynamometer. J. Shoulder Elb. Surg. 2023, 32, 2008–2016. [Google Scholar] [CrossRef] [PubMed]
- Tanveer, F.; Arslan, S.A.; Darain, H.; Ahmad, A. Reliability of Hand-Held Dynamometer for assessing Isometric Lumbar Muscles Strength in Asymptomatic Healthy Population. Pak. J. Med. Sci. 2021, 37, 461–465. [Google Scholar] [CrossRef]
- Byl, N.N.; Richards, S.; Asturias, J. Intrarater and interrater reliability of strength measurements of the biceps and deltoid using a hand held dynamometer. J. Orthop. Sports Phys. Ther. 1988, 9, 395–398. [Google Scholar] [CrossRef]
- Cadogan, A.; Laslett, M.; Hing, W.; McNair, P.; Williams, M. Reliability of a new hand-held dynamometer in measuring shoulder range of motion and strength. Man. Ther. 2011, 16, 97–101. [Google Scholar] [CrossRef]
- Beshara, P.; Davidson, I.; Pelletier, M.; Walsh, W.R. The Intra- and Inter-Rater Reliability of a Variety of Testing Methods to Measure Shoulder Range of Motion, Hand-behind-Back and External Rotation Strength in Healthy Participants. Int. J. Environ. Res. Public Health 2022, 19, 14442. [Google Scholar] [CrossRef]
- Stone, C.A.; Nolan, B.; Lawlor, P.G.; Kenny, R.A. Hand-held dynamometry: Tester strength is paramount, even in frail populations. J. Rehabil. Med. 2011, 43, 808–811. [Google Scholar] [CrossRef] [PubMed]
- Wadsworth, C.T.; Krishnan, R.; Sear, M.; Harrold, J.; Nielsen, D.H. Intrarater reliability of manual muscle testing and hand-held dynametric muscle testing. Phys. Ther. 1987, 67, 1342–1347. [Google Scholar] [CrossRef] [PubMed]
- Reed, R.L.; Den Hartog, R.; Yochum, K.; Pearlmutter, L.; Ruttinger, A.C.; Mooradian, A.D. A comparison of hand-held isometric strength measurement with isokinetic muscle strength measurement in the elderly. J. Am. Geriatr. Soc. 1993, 41, 53–56. [Google Scholar] [CrossRef] [PubMed]
- Brinkmann, J.R. Comparison of a hand-held and fixed dynamometer in measuring strength of patients with neuromuscular disease. J. Orthop. Sports Phys. Ther. 1994, 19, 100–104. [Google Scholar] [CrossRef]
- Bohannon, R.W. Intertester reliability of hand-held dynamometry: A concise summary of published research. Percept. Mot. Ski. 1999, 88, 899–902. [Google Scholar] [CrossRef]
- McMahon, L.M.; Burdett, R.; Whitney, S.L. Effects of muscle group and placement site on reliability of hand-held dynamometry strength measurements. J. Orthop. Sports Phys. Ther. 1992, 15, 236–242. [Google Scholar] [CrossRef]
- Lu, T.W.; Hsu, H.C.; Chang, L.Y.; Chen, H.L. Enhancing the examiner’s resisting force improves the reliability of manual muscle strength measurements: Comparison of a new device with hand-held dynamometry. J. Rehabil. Med. 2007, 39, 679–684. [Google Scholar] [CrossRef]
- Beshay, N.; Lam, P.H.; Murrell, G.A. Assessing the reliability of shoulder strength measurement: Hand-held versus fixed dynamometry. Shoulder Elb. 2011, 3, 244–251. [Google Scholar] [CrossRef]
- Ruschel, C.; Haupenthal, A.; Jacomel, G.F.; Fontana, H.B.; Santos, D.P.; Scoz, R.D.; Roesler, H. Validity and reliability of an instrumented leg-extension machine for measuring isometric muscle strength of the knee extensors. J. Sport Rehabil. 2015, 24, 122. [Google Scholar] [CrossRef]
- Macedo, M.C.; Souza, M.A.; Ferreira, K.R.; Campos, L.O.; Souza, I.S.O.; Barbosa, M.A.; Brito, C.J.; Intelangelo, L.; Barbosa, A.C. Validity and Test-Retest Reliability of a Novel Push Low-Cost Hand-Held Dynamometer for Knee Strength Assessment during Different Force Ranges. Diagnostics 2022, 12, 186. [Google Scholar] [CrossRef]
- Machado Rodrigues, F.; Demeyer, H.; Hornikx, M.; Camillo, C.A.; Calik-Kutukcu, E.; Burtin, C.; Janssens, W.; Troosters, T.; Osadnik, C. Validity and reliability of strain gauge measurement of volitional quadriceps force in patients with COPD. Chron. Respir. Dis. 2017, 14, 289–297. [Google Scholar] [CrossRef]
- Hong, J.-S.; Ko, J.-B.; Ju, M.-M.; Lee, B.-K.; Park, D.-S.; Lee, S.-H. The Reliability and Validity of an Isometric Knee Strength Measurement Device in Older Adult Individuals. Sensors 2025, 25, 2981. [Google Scholar] [CrossRef]
- Choi, J.; Yeoh, W.L.; Loh, P.Y.; Muraki, S. Force and electromyography responses during isometric force release of different rates and step-down magnitudes. Hum. Mov. Sci. 2019, 67, 102516. [Google Scholar] [CrossRef]
- Liu, J.; Qu, X.; Stone, M.H. Evaluation of force-time curve analysis methods in the isometric mid-thigh pull test. Sports Biomech. 2023, 22, 1381–1397. [Google Scholar] [CrossRef] [PubMed]
- González-Rosalén, J.; Cuerda-Del Pino, A.; Sánchez-Barbadora, M.; Martín-San Agustín, R. Validity and reliability of the DiCI for the measurement of shoulder flexion and abduction strength in asymptomatic and symptomatic subjects. PeerJ 2021, 9, e11600. [Google Scholar] [CrossRef] [PubMed]
- Antonacci, C.; Ghelli, G.; Carnevale, A.; Silvestri, S.; Schena, E.; Longo, U.G. A novel system integrating load cell for assessing shoulder muscle strength. In Proceedings of the 2024 IEEE International Workshop on Metrology for Industry 4.0 & IoT (MetroInd4. 0 & IoT), Firenze, Italy, 29–31 May 2024; pp. 591–596. [Google Scholar]
- Bradley, H.; Pierpoint, L. Normative Values of Isometric Shoulder Strength Among Healthy Adults. Int. J. Sports Phys. Ther. 2023, 18, 977–988. [Google Scholar] [CrossRef] [PubMed]
- Chai, T. Root Mean Square. In Encyclopedia of Mathematical Geosciences; Springer: Berlin/Heidelberg, Germany, 2022; pp. 1–3. [Google Scholar]
- Chicco, D.; Warrens, M.J.; Jurman, G. The coefficient of determination R-squared is more informative than SMAPE, MAE, MAPE, MSE and RMSE in regression analysis evaluation. Peerj Comput. Sci. 2021, 7, e623. [Google Scholar] [CrossRef]
- Wittstein, J.R.; Queen, R.; Abbey, A.; Toth, A.; Moorman, C.T. Isokinetic strength, endurance, and subjective outcomes after biceps tenotomy versus tenodesis: A postoperative study. Am. J. Sports Med. 2011, 39, 857–865. [Google Scholar] [CrossRef] [PubMed]
- Constant, C.R.; Murley, A.H. A clinical method of functional assessment of the shoulder. Clin. Orthop. Relat. Res. 1987, 214, 160–164. [Google Scholar] [CrossRef]
- Cools, A.M.; Vanderstukken, F.; Vereecken, F.; Duprez, M.; Heyman, K.; Goethals, N.; Johansson, F. Eccentric and isometric shoulder rotator cuff strength testing using a hand-held dynamometer: Reference values for overhead athletes. Knee Surg. Sports Traumatol. Arthrosc. 2016, 24, 3838–3847. [Google Scholar] [CrossRef]
- Bland, J.M.; Altman, D.G. Statistical methods for assessing agreement between two methods of clinical measurement. Int. J. Nurs. Stud. 2010, 47, 931–936. [Google Scholar] [CrossRef]
- Awatani, T.; Morikita, I.; Shinohara, J.; Mori, S.; Nariai, M.; Tatsumi, Y.; Nagata, A.; Koshiba, H. Intra- and inter-rater reliability of isometric shoulder extensor and internal rotator strength measurements performed using a hand-held dynamometer. J. Phys. Ther. Sci. 2016, 28, 3054–3059. [Google Scholar] [CrossRef]
- Ludbrook, J. Statistical techniques for comparing measurers and methods of measurement: A critical review. Clin. Exp. Pharmacol. Physiol. 2002, 29, 527–536. [Google Scholar] [CrossRef]
- Andrews, A.W.; Thomas, M.W.; Bohannon, R.W. Normative values for isometric muscle force measurements obtained with hand-held dynamometers. Phys. Ther. 1996, 76, 248–259. [Google Scholar] [CrossRef] [PubMed]
- Merry, K.; Napier, C.; Chung, V.; Hannigan, B.C.; MacPherson, M.; Menon, C.; Scott, A. The Validity and Reliability of Two Commercially Available Load Sensors for Clinical Strength Assessment. Sensors 2021, 21, 8399. [Google Scholar] [CrossRef]
- Maffiuletti, N.A.; Aagaard, P.; Blazevich, A.J.; Folland, J.; Tillin, N.; Duchateau, J. Rate of force development: Physiological and methodological considerations. Eur. J. Appl. Physiol. 2016, 116, 1091–1116. [Google Scholar] [CrossRef]
- Aagaard, P.; Simonsen, E.B.; Andersen, J.L.; Magnusson, P.; Dyhre-Poulsen, P. Increased rate of force development and neural drive of human skeletal muscle following resistance training. J. Appl. Physiol. 2002, 93, 1318–1326. [Google Scholar] [CrossRef] [PubMed]
- Hoshikawa, K.; Yuri, T.; Giambini, H.; Kiyoshige, Y. Shoulder scaption is dependent on the behavior of the different partitions of the infraspinatus muscle. Surg. Radiol. Anat. 2021, 43, 653–659. [Google Scholar] [CrossRef]
- Wang, Y.; Sun, Y.; Gao, L.; Ma, Z.; Yang, X. Development and performance verification of an isometric dynamometer for lower extremity. Sci. Rep. 2025, 15, 26145. [Google Scholar] [CrossRef] [PubMed]
- Cavuoto, L.A.; Pajoutan, M.; Mehta, R.K. Reliability analyses and values of isometric shoulder flexion and trunk extension strengths stratified by body mass index. PLoS ONE 2019, 14, e0219090. [Google Scholar] [CrossRef] [PubMed]


| Known Weights [N] | HHD Mean Value (SD) [N] | Load Cell Mean Value (SD) [N] | RMSE [N] |
|---|---|---|---|
| 9.81 | 10.45 (0.09) | 10.40 (0.03) | 0.55 |
| 19.62 | 19.76 (0.04) | 19.79 (0.02) | 0.41 |
| 29.43 | 30.04 (0.46) | 30.29 (0.11) | 0.46 |
| 39.24 | 38.63 (0.43) | 39.37 (0.09) | 0.40 |
| 49.05 | 48.83 (0.31) | 50.08 (0.08) | 1.29 |
| 98.10 | 93.38 (0.60) | 98.81 (0.25) | 5.06 |
| Partecipant IDs | Arm Side | HHD Mean Value (SD) [N] | Rising Phase—Load Cell Mean Value (SD) [N] | Plateau Phase—Load Cell Mean Value (SD) [N] |
|---|---|---|---|---|
| 1 | Right | 39.80 (7.07) | 27.11 (3.95) | 28.27 (4.42) |
| Left | 44.94 (7.53) | 40.96 (8.59) | 45.58 (19.94) | |
| 2 | Right | 30.36 (3.61) | 20.55 (6.36) | 24.10 (7.99) |
| Left | 31.36 (4.26) | 22.60 (4.76) | 26.30 (5.92) | |
| 3 | Right | 32.98 (3.28) | 44.53 (9.82) | 47.56 (10.95) |
| Left | 33.78 (10.96) | 26.86 (3.74) | 28.34 (4.27) | |
| 4 | Right | 19.80 (1.44) | 11.60 (2.70) | 12.11 (2.93) |
| Left | 19.76 (2.34) | 12.25 (1.95) | 13.15 (2.26) | |
| 5 | Right | 25.18 (1.31) | 20.91 (3.51) | 24.12 (4.99) |
| Left | 23.82 (1.67) | 17.14 (1.71) | 19.21 (2.33) |
| Partecipant IDs | Arm Side | HHD Mean Value (SD) [N] | Rising Phase—Load Cell Mean Value (SD) [N] | Plateau Phase—Load Cell Mean Value (SD) [N] |
|---|---|---|---|---|
| 1 | Right | 81.66 (8.83) | 73.46 (9.47) | 80.26 (10.81) |
| Left | 82.34 (2.08) | 78.58 (11.32) | 89.78 (14.37) | |
| 2 | Right | 50.42 (6.48) | 57.01 (6.87) | 69.33 (9.27) |
| Left | 62.02 (5.53) | 55.00 (10.88) | 70.25 (11.71) | |
| 3 | Right | 43.24 (9.76) | 51.54 (12.15) | 54.42 (12.85) |
| Left | 46.32 (5.60) | 54.78 (12.65) | 57.55 (13.13) | |
| 4 | Right | 31.26 (1.92) | 17.12 (3.03) | 18.36 (3.39) |
| Left | 22.94 (4.40) | 21.60 (3.57) | 23.37 (4.21) | |
| 5 | Right | 43.12 (11.80) | 28.61 (2.41) | 32.27 (2.03) |
| Left | 37.06 (2.89) | 35.90 (11.92) | 41.42 (13.21) |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Antonacci, C.; Carnevale, A.; Mancini, L.; de Sire, A.; D’Hooghe, P.; Mercurio, M.; Papalia, R.; Schena, E.; Longo, U.G. Shoulder Muscle Strength Assessment: A Comparative Study of Hand-Held Dynamometers and Load Cell Measurements. Instruments 2026, 10, 2. https://doi.org/10.3390/instruments10010002
Antonacci C, Carnevale A, Mancini L, de Sire A, D’Hooghe P, Mercurio M, Papalia R, Schena E, Longo UG. Shoulder Muscle Strength Assessment: A Comparative Study of Hand-Held Dynamometers and Load Cell Measurements. Instruments. 2026; 10(1):2. https://doi.org/10.3390/instruments10010002
Chicago/Turabian StyleAntonacci, Carla, Arianna Carnevale, Letizia Mancini, Alessandro de Sire, Pieter D’Hooghe, Michele Mercurio, Rocco Papalia, Emiliano Schena, and Umile Giuseppe Longo. 2026. "Shoulder Muscle Strength Assessment: A Comparative Study of Hand-Held Dynamometers and Load Cell Measurements" Instruments 10, no. 1: 2. https://doi.org/10.3390/instruments10010002
APA StyleAntonacci, C., Carnevale, A., Mancini, L., de Sire, A., D’Hooghe, P., Mercurio, M., Papalia, R., Schena, E., & Longo, U. G. (2026). Shoulder Muscle Strength Assessment: A Comparative Study of Hand-Held Dynamometers and Load Cell Measurements. Instruments, 10(1), 2. https://doi.org/10.3390/instruments10010002









