Immune-Endocrine Interactions in the Fish Gonad during Infection: An Open Door to Vertical Transmission
Abstract
1. Introduction
2. Immune Privilege and the Immune Response in the Gonad
2.1. The Blood-Testis Barrier
2.2. Immune Effectors Out of the Blood-Testis Barrier
2.3. The Testicular Environment that Allows Immune Privilege
2.4. The Ovary Environment that Allows Immune Privilege
3. The Gonad Immune Responses in European Sea Bass—Preliminary Data
3.1. Cellular Innate Immune Functions
3.2. Description of Antimicrobial Peptides
4. Infection of the Gonad as a Perfect Way to Evade the Immune Response in Fish
5. Nodavirus as a Viral Model to Unravel Fish Immuno-Endocrine Interactions
6. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Food and Agriculture Organization (FAO). The State of World Fisheries and Aquaculture; FAO: Rome, Italy, 2016; 200p. [Google Scholar]
- Fukuda, Y.; Nguyen, H.D.; Furuhashi, M.; Nakai, T. Mass mortality of cultured sevenband grouper, Epinephelus septemfasciatus, associated with viral nervous necrosis. Fish Pathol. 1996, 31, 165–170. [Google Scholar] [CrossRef]
- Grau, A.; Crespo, S.; Pastor, E.; Gonzalez, P.; Carbonell, E. High infection by Zeuxapta seriolae (Monogenea: Heteraxinidae) associated with mass mortalities of amberjack Seriola dumerili Risso reared in sea cages in the Balearic Islands (western Mediterranean). Bull. Eur. Assoc. Fish Pathol. 2003, 23, 139–142. [Google Scholar]
- Villamil, L.; Figueras, A.; Toranzo, A.E.; Planas, M.; Novoa, B. Isolation of a highly pathogenic Vibrio pelagius strain associated with mass mortalities of turbot, Scophthalmus maximus (L.), larvae. J. Fish Dis. 2003, 26, 293–303. [Google Scholar] [CrossRef] [PubMed]
- Kibenge, F.S.B.; Godoy, M.G. Aquaculture Virology; Academic Press: London, UK, 2016; 568p, ISBN 9780128015735. [Google Scholar]
- Klein, S.L. Hormonal and immunological mechanisms mediating sex differences in parasite infection. Parasite Immunol. 2004, 26, 247–264. [Google Scholar] [CrossRef] [PubMed]
- Hecker, M.; Karbe, L. Parasitism in fish: An endocrine modulator of ecological relevance? Aquat. Toxicol. 2005, 72, 195–207. [Google Scholar] [CrossRef] [PubMed]
- Deane, E.E.; Li, J.; Woo, N.Y.S. Hormonal status and phagocytic activity in sea bream infected with vibriosis. Comp. Biochem. Physiol. B Biochem. Mol. Biol. 2001, 129, 687–693. [Google Scholar] [CrossRef]
- Wenger, M.; Shved, N.; Akgül, G.; Caelers, A.; Casanova, A.; Segner, H.; Eppler, E. Developmental oestrogen exposure differentially modulates IGF-I and TNF-α expression levels in immune organs of Yersinia ruckeri-challenged young adult rainbow trout (Oncorhynchus mykiss). Gen. Comp. Endocrinol. 2014, 205, 168–175. [Google Scholar] [CrossRef] [PubMed]
- Engelsma, M.Y.; Huising, M.O.; van Muiswinkel, W.B.; Flik, G.; Kwang, J.; Savelkoul, H.F.; Verburg-van Kemenade, B.M. Neuroendocrine-immune interactions in fish: A role for interleukin-1. Vet. Immunol. Immunopathol. 2002, 87, 467–479. [Google Scholar] [CrossRef]
- Harris, J.; Bird, D.J. Modulation of the fish immune system by hormones. Vet. Immunol. Immunopathol. 2000, 77, 163–176. [Google Scholar] [CrossRef]
- Lutton, B.; Callard, I. Evolution of reproductive-immune interactions. Integr. Comp. Biol. 2006, 46, 1060–1071. [Google Scholar] [CrossRef] [PubMed]
- Segner, H.; Verburg-van Kemenade, B.M.L.; Chadzinska, M. The immunomodulatory role of the hypothalamus-pituitary-gonad axis: Proximate mechanism for reproduction-immune trade offs? Dev. Comp. Immunol. 2017, 66, 43–60. [Google Scholar] [CrossRef] [PubMed]
- Chaves-Pozo, E.; García-Ayala, A.; Cabas, I. Effects of sex steroids on fish leukocytes. Biology 2018, 7, 9. [Google Scholar] [CrossRef] [PubMed]
- Iwanowicz, L.R.; Ottinger, C.A. Estrogens, estrogen receptors and their role as immunoregulators in fish. In Fish Defenses; Zaccone, G., Meseguer, J., Garcia Ayala, A., Kapoor, B.G., Eds.; Science Publisher: Enfield, UK, 2009; Volume 1, pp. 277–322. ISBN 978-1-57-808-327-5. [Google Scholar]
- Verburg-van Kemenade, B.M.L.; Cohen, N.; Chadzinska, M. Neuroendocrine-immune interaction: Evolutionarily conserved mechanisms that maintain allostasis in an ever-changing environment. Dev. Comp. Immunol. 2017, 66. [Google Scholar] [CrossRef] [PubMed]
- Szwejser, E.; Verburg-van Kemenade, B.M.L.; Maciuszek, M.; Chadzinska, M. Estrogen-dependent seasonal adaptations in the immune response of fish. Horm. Behav. 2017, 88, 15–24. [Google Scholar] [CrossRef] [PubMed]
- Hedger, M.P. Macrophages and the immune responsiveness of the testis. J. Reprod. Immunol. 2002, 57, 19–34. [Google Scholar] [CrossRef]
- Dunbar, B.S.; Prasad, S.; Carino, C.; Skinner, S.M. The ovary as an immune target. J. Soc. Gynecol. Investig. 2001, 8, S43–S48. [Google Scholar] [PubMed]
- Bukulmez, O.; Arici, A. Leukocytes in ovarian function. Hum. Reprod. Update 2000, 6, 1–15. [Google Scholar] [CrossRef] [PubMed]
- Fijak, M.; Meinhardt, A. The testis in immune privilege. Immunol. Rev. 2006, 213, 66–81. [Google Scholar] [CrossRef] [PubMed]
- Hedger, M.P. Immune Privilege of the testis: Meaning, mechanisms, and manifestations. In Infection, Immune Homeostasis and Immune Privilege, Birkhäuser Advances in Infectious Diseases; Stein-Streilein, J., Ed.; Springer: Basel, Switzerland, 2012. [Google Scholar]
- Wen, Q.; Tang, E.I.; Gao, Y.; Jesus, T.T.; Chu, D.S.; Lee, W.M.; Wong, C.K.C.; Liu, Y.-X.; Xiao, X.; Silvestrini, B.; et al. Signaling pathways regulating blood-tissue barriers—Lesson from the testis. Biochim. Biophys. Acta Biomembr. 2018, 1860, 141–153. [Google Scholar] [CrossRef] [PubMed]
- Russell, L. Movement of spermatocytes from the basal to the adluminal compartment of the rat testis. Am. J. Anat. 1977, 148, 313–328. [Google Scholar] [CrossRef] [PubMed]
- Su, L.; Mruk, D.D.; Cheng, C.Y. Drug transporters, the blood-testis barrier, and spermatogenesis. J. Endocrinol. 2011, 208, 207–223. [Google Scholar] [CrossRef] [PubMed]
- Smith, B.E.; Braun, R.E. Germ cell migration across Sertoli cell tight junctions. Science 2012, 338, 798–802. [Google Scholar] [CrossRef] [PubMed]
- França, L.R.; Hess, R.A.; Dufour, J.M.; Hofmann, M.C.; Griswold, M.D. The Sertoli cell: One hundred fifty years of beauty and plasticity. Andrology 2016, 4, 189–212. [Google Scholar] [CrossRef] [PubMed]
- Govero, J.; Esakky, P.; Scheaffer, S.M.; Fernandez, E.; Drury, A.; Platt, D.J.; Gorman, M.J.; Richner, J.M.; Caine, E.A.; Salazar, V.; et al. Zika virus infection damages the testes in mice. Nature 2016, 540, 438–442. [Google Scholar] [CrossRef] [PubMed]
- Houzet, L.; Matusali, G.; Dejucq-Rainsford, N. Origins of HIV-infected leukocytes and virions in semen. J. Infect. Dis. 2014, 210, S622–S630. [Google Scholar] [CrossRef] [PubMed]
- Jenabian, M.; Costiniuk, C.T.; Mehraj, V.; Ghazawi, F.M.; Fromenti, R.; Brousseau, J.; Brassard, P.; B’elanger, M.; Ancuta, P.; Bendayan, R.; et al. Immune tolerance properties of the testicular tissue as a viral sanctuary site in Art-treated Hiv-infected adults. Aids 2016, 30, 2777–2786. [Google Scholar] [CrossRef] [PubMed]
- Risley, M.S.; Tan, I.P.; Roy, C.; Saez, J.C. Cell-, age- and stage-dependent distribution of connexin 43 gap junctions in testes. J. Cell Sci. 1992, 103, 81–96. [Google Scholar] [PubMed]
- Batlouni, S.R.; Nóbrega, R.H.; França, L.R. Cell junctions in fish seminiferous epithelium. Fish Physiol. Biochem. 2009, 35, 207–217. [Google Scholar] [CrossRef] [PubMed]
- Schulz, R.W.; de França, L.R.; Lareyre, J.J.; LeGac, F.; Chiarini-García, H.; Nobrega, R.H.; Miura, T. Spermatogenesis in fish. Gen. Comp. Endocrinol. 2010, 165, 390–411. [Google Scholar] [CrossRef] [PubMed]
- Vogl, A.W.; Pfeiffer, D.C.; Mulholland, D.; Kimel, G.; Guttman, J. Unique and multifunctional adhesion junctions in the testis. Ectoplasmic specializations. Arch. Histol. Cytol. 2000, 63, 1–15. [Google Scholar] [CrossRef] [PubMed]
- Abraham, M.; Rahamim, E.; Tibika, H.; Golenser, E.; Kieselskin, M. The blood-testis barrier in Aphanius dispar (Teleostei). Cell Tissue Res. 1980, 211, 207–214. [Google Scholar] [CrossRef] [PubMed]
- Silva, M.; Godinho, H.P. Timing of some events of the gametogenesis in the male nile tilapia, Sarotherodon niloticus. Arch. dÁnatomie Microsc. 1983, 72, 231–237. [Google Scholar]
- Callard, G.V.; Callard, I.P. Spermatogenesis, in nonmammals. In Encyclopedia of Reproduction; Knobil, E., Neil, J.D., Eds.; Academic Press: New York, NY, USA, 1999; Volume 4, pp. 563–570. [Google Scholar]
- Batlouni, S.R.; Carreño, F.R.; Romagosa, E.; Borella, M.I. Cell junctions in the germinal epithelium may play an important role in spermatogenesis of the catfish P. fasciatum (Pisces, Siluriformes). J. Mol. Histol. 2005, 36, 97–110. [Google Scholar] [CrossRef] [PubMed]
- Head, J.R.; Neaves, W.B.; Billingham, R.E. Reconsideration of the lymphatic drainage of the rat testis. Transplantation 1983, 35, 91–95. [Google Scholar] [CrossRef] [PubMed]
- Möller, R. Arrangement and fine structure of lymphatic vessels in the human spermatic cord. Andrologia 2009, 12, 564–576. [Google Scholar] [CrossRef]
- Itoh, M.; Li, X.; Yano, A.; Xie, Q.; Takeuchi, Y. Patterns of efferent lymphatics of the mouse testis. J. Androl. 1998, 19, 466–472. [Google Scholar] [CrossRef] [PubMed]
- Ferguson, J.; Scothornet, R.J. Extended survival of pancreatic islet allografts in the testis of guinea-pigs. J. Anat. 1977, 124, 1. [Google Scholar] [PubMed]
- Bobzien, B.; Yasunami, Y.; Majercik, M.; Lacy, P.E.; Davie, J.M. Intratesticular transplants of islet xenografts (rat to mouse). Diabetes 1983, 32, 213–216. [Google Scholar] [CrossRef] [PubMed]
- Head, J.R.; Neaves, W.B.; Billingham, R.E. Immune privilege in the testis. I. Basic parameters of allograft survival. Transplantation 1983, 36, 423–431. [Google Scholar] [CrossRef] [PubMed]
- Yule, T.D.; Montoya, G.D.; Russell, L.D.; Williams, T.M.; Tung, K.S. Autoantigenic germ cells exist outside the blood testis barrier. J. Immunol. 1988, 141, 1161–1167. [Google Scholar] [PubMed]
- Hedger, M.P.; Hettiarachchi, S. Measurement of immunoglobulin g levels in adult rat testicular interstitial fluid and serum. J. Androl. 1994, 15, 583–590. [Google Scholar] [CrossRef] [PubMed]
- Pöllänen, P.; Saari, T.; Yeung, C.-H.; Cooper, T.G. Regulation of the transport of immunoglobulin G into the male rat reproductive tract. J. Reprod. Immunol. 1995, 28, 111–135. [Google Scholar] [CrossRef]
- Mclay, R.N.; Banks, W.A.; Kastin, A.J. Granulocyte macrophage-colony stimulating factor crosses the blood-testis barrier in mice. Biol. Reprod. 1997, 57, 822–826. [Google Scholar] [CrossRef] [PubMed]
- Chaves-Pozo, E.; Liarte, S.; García-Ayala, A. Immune and reproductive interaction: An essential clue for understanding gonad functions in gilthead seabream. In Recent Advances in Fish Reproductive Biology; García-Ayala, A., Meseguer, J., Chaves-Pozo, E., Eds.; Research Signpost: Kerala, India, 2010; pp. 127–139. ISBN 978-81-308-0397-5. [Google Scholar]
- García-García, M.; Liarte, S.; Gómez-González, N.E.; García-Alcázar, A.; Pérez-Sánchez, J.; Meseguer, J.; Mulero, V.; García-Ayala, A.; Chaves-Pozo, E. Cimetidine disrupts the renewal of testicular cells and the steroidogenesis in a hermaphrodite fish. Comp. Biochem. Physiol. C Toxicol. Pharmacol. 2016, 189, 44–53. [Google Scholar] [CrossRef] [PubMed]
- Valero, Y.; Sánchez-Hernández, M.; García-Alcázar, A.; García-Ayala, A.; Cuesta, A.; Chaves-Pozo, E. Characterization of the annual regulation of reproductive and immune parameters on the testis of European sea bass. Cell Tissue Res. 2015, 362, 215–229. [Google Scholar] [CrossRef] [PubMed]
- Wang, D.-S.; Jiao, B.; Hu, C.; Huang, X.; Liu, Z.; Cheng, C.H.K. Discovery of a gonad-specific IGF subtype in teleost. Biochem. Biophys. Res. Commun. 2008, 367, 336–341. [Google Scholar] [CrossRef] [PubMed]
- Zou, J.; Tafalla, C.; Truckle, J.; Secombes, C.J. Identification of a second group of type I IFNs in fish sheds light on IFN evolution in vertebrates. J. Immunol. 2007, 179, 3859–3871. [Google Scholar] [CrossRef] [PubMed]
- Purcell, M.K.; Laing, K.J.; Woodson, J.C.; Thorgaard, G.H.; Hansen, J.D. Characterization of the interferon genes in homozygous rainbow trout reveals two novel genes, alternate splicing and differential regulation of duplicated genes. Fish Shellfish Immunol. 2009, 26, 293–304. [Google Scholar] [CrossRef] [PubMed]
- Chaves-Pozo, E.; Zou, J.; Secombes, C.J.; Cuesta, A.; Tafalla, C. The rainbow trout (Oncorhynchus mykiss) interferon response in the ovary. Mol. Immunol. 2010, 47, 1757–1764. [Google Scholar] [CrossRef] [PubMed]
- Li, Z.; Hong, W.S.; Qiu, H.T.; Zhang, Y.T.; Yang, M.S.; You, X.X.; Chen, S.X. Cloning and expression of two hepcidin genes in the mudskipper (Boleophthalmus pectinirostris) provides insights into their roles in male reproductive immunity. Fish Shellfish Immunol. 2016, 56, 239–247. [Google Scholar] [CrossRef] [PubMed]
- Jin, J.-Y.; Zhou, L.; Wang, Y.; Li, Z.; Zhao, J.-G.; Zhang, Q.-Y.; Gui, J.-F. Antibacterial and antiviral roles of a fish β-defensin expressed both in pituitary and testis. PLoS ONE 2010, 5, e12883. [Google Scholar] [CrossRef] [PubMed]
- Mital, P.; Kaur, G.; Dufour, J.M. Immunoprotective sertoli cells: Making allogeneic and xenogeneic transplantation feasible. Reproduction 2010, 139, 495–504. [Google Scholar] [CrossRef] [PubMed]
- Nasr, I.W.; Wang, Y.; Gao, G.; Deng, S.; Diggs, L.; Rothstein, D.M.; Tellides, G.; Lakkis, F.G.; Dai, Z. Testicular immune privilege promotes transplantation tolerance by altering the balance between memory and regulatory T cells. J. Immunol. 2005, 174, 6161–6168. [Google Scholar] [CrossRef] [PubMed]
- Fijak, M.; Schneider, E.; Klug, J.; Bhushan, S.; Hackstein, H.; Schuler, G.; Wygrecka, M.; Gromoll, J.; Meinhardt, A. Testosterone replacement effectively inhibits the development of experimental autoimmune orchitis in rats: Evidence for a direct role of testosterone on regulatory T cell expansion. J. Immunol. 2011, 186, 5162–5172. [Google Scholar] [CrossRef] [PubMed]
- Chaves-Pozo, E.; Mulero, V.; Meseguer, J.; Garcia Ayala, A.; García-Ayala, A. Professional phagocytic granulocytes of the bony fish gilthead seabream display functional adaptation to testicular microenvironment. J. Leukoc. Biol. 2005, 78, 345–351. [Google Scholar] [CrossRef] [PubMed]
- Chaves-Pozo, E.; Liarte, S.; Fernández-Alacid, L.; Abellán, E.; Meseguer, J.; Mulero, V.; García-Ayala, A. Pattern of expression of immune-relevant genes in the gonad of a teleost, the gilthead seabream (Sparus aurata L.). Mol. Immunol. 2008, 45, 2998–3011. [Google Scholar] [CrossRef] [PubMed]
- Jin, H.J.; Shao, J.Z.; Xiang, L.X. Identification and characterization of suppressor of cytokine signaling 3 (SOCS-3) homologues in teleost fish. Mol. Immunol. 2007, 44, 1042–1051. [Google Scholar] [CrossRef] [PubMed]
- Jin, H.J.; Xiang, L.X.; Shao, J.Z. Identification and characterization of suppressor of cytokine signaling 1 (SOCS-1) homologues in teleost fish. Immunogenetics 2007, 59, 673–686. [Google Scholar] [CrossRef] [PubMed]
- Cabas, I.; Chaves-Pozo, E.; García-Alcázar, A.; Meseguer, J.; Mulero, V.; García-Ayala, A. Dietary intake of 17alpha-ethinylestradiol promotes leukocytes infiltration in the gonad of the hermaphrodite gilthead seabream. Mol. Immunol. 2011, 48, 2079–2086. [Google Scholar] [CrossRef] [PubMed]
- Chaves-Pozo, E.; Liarte, S.; Vargas-Chacoff, L.; García-López, A.; Mulero, V.; Meseguer, J.; Mancera, J.M.; García-Ayala, A. 17Beta-estradiol triggers postspawning in spermatogenically active gilthead seabream (Sparus aurata L.) males. Biol Reprod. 2007, 76, 142–148. [Google Scholar] [CrossRef] [PubMed]
- Sánchez-Hernández, M.; Chaves-Pozo, E.; Cabas, I.; Mulero, V.; García-Ayala, A.; García-Alcázar, A. Testosterone implants modify the steroid hormone balance and the gonadal physiology of gilthead seabream (Sparus aurata L.) males. J. Steroid. Biochem. Mol. Biol. 2013, 138C, 183–194. [Google Scholar] [CrossRef] [PubMed]
- Cabas, I.; Chaves-Pozo, E.; García-Alcázar, A.; Meseguer, J.; Mulero, V.; García-Ayala, A. The effect of 17alpha-ethynylestradiol on steroidogenesis and gonadal cytokine gene expression is related to the reproductive stage in marine hermaphrodite fish. Mar. Drugs 2013, 11, 4973–4992. [Google Scholar] [CrossRef] [PubMed]
- Li, X.; Strauss, L.; Kaatrasalo, A.; Mayerhofer, A.; Huhtaniemi, I.; Santti, R.; Mäkelä, S.; Poutanen, M. Transgenic Mice expressing P450 aromatase as a model for male infertility associated with chronic inflammation in the testis. Endocrinology 2006, 147, 1271–1277. [Google Scholar] [CrossRef] [PubMed]
- Yoshimura, Y.; Barua, A. Female reproductive system and immunology. In Avian Reproduction, Advances in Experimental Medicine and Biology; Springer: Singapore, 2017; pp. 33–57. [Google Scholar]
- Lutton, B.V.; Callard, I.P. Morphological relationships and leukocyte influence on steroid production in the epigonal organ-ovary complex of the skate, Leucoraja erinacea. J. Morphol. 2008, 269, 620–629. [Google Scholar] [CrossRef] [PubMed]
- Chaves-Pozo, E.; Liarte, S.; Mulero, I.; Abellán, E.; Meseguer, J.; García-Ayala, A.; Abellan, E.; Meseguer, J.; Garcia-Ayala, A. Early presence of immune cells in the developing gonad of the gilthead seabream (Sparus aurata Linnaeus, 1758). J. Reprod. Dev. 2009, 55, 440–445. [Google Scholar] [CrossRef] [PubMed]
- Liarte, S.; Chaves-Pozo, E.; García-Alcázar, A.; Mulero, V.; Meseguer, J.; García-Ayala, A. Testicular involution prior to sex change in gilthead seabream is characterized by a decrease in DMRT1 gene expression and by massive leukocyte infiltration. Reprod. Biol. Endocrinol. 2007, 5, 20. [Google Scholar] [CrossRef] [PubMed]
- Zhang, L.; Gallo, R.L. Antimicrobial peptides. Curr. Biol. 2016, 26, R14–R19. [Google Scholar] [CrossRef] [PubMed]
- Valero, Y.; García-Alcázar, A.; Esteban, M.A.; Cuesta, A.; Chaves-Pozo, E.; García-Alcazar, A.; Esteban, M.A.; Cuesta, A.; Chaves-Pozo, E. Antimicrobial response is increased in the testis of European sea bass, but not in gilthead seabream, upon nodavirus infection. Fish Shellfish Immunol. 2015, 44, 203–213. [Google Scholar] [CrossRef] [PubMed]
- Grandjean, V.; Vincent, S.; Martin, L.; Rassoulzadegan, M.; Cuzin, F. Antimicrobial protection of the mouse testis: Synthesis of defensins of the cryptdin family. Biol. Reprod. 1997, 57, 1115–1122. [Google Scholar] [CrossRef] [PubMed]
- Silphaduang, U.; Hincke, M.T.; Nys, Y.; Mine, Y. Antimicrobial proteins in chicken reproductive system. Biochem. Biophys. Res. Commun. 2006, 340, 648–655. [Google Scholar] [CrossRef] [PubMed]
- King, A.E.; Critchley, H.O.; Kelly, R.W. Innate immune defences in the human endometrium. Reprod. Biol. Endocrinol. 2003, 1, 116. [Google Scholar] [CrossRef] [PubMed]
- King, A.E.; Fleming, D.C.; Critchley, H.O.; Kelly, R.W. Differential expression of the natural antimicrobials, beta-defensins 3 and 4, in human endometrium. J. Reprod. Immunol. 2003, 59, 1–16. [Google Scholar] [CrossRef]
- Rodríguez, A.; Esteban, M.A.; Meseguer, J. Phagocytosis and peroxidase release by seabream (Sparus aurata L.) leucocytes in response to yeast cells. Anat. Rec. A Discov. Mol. Cell. Evol. Biol. 2003, 272, 415–423. [Google Scholar] [CrossRef] [PubMed]
- Ortuño, J.; Esteban, M.A.; Meseguer, J. Kinetics of hydrogen peroxide production during in vitro respiratory burst of seabream (Sparus aurata L.) head-kidney leucocytes, as measured by a flow cytometric method. Fish Shellfish Immunol. 2000, 10, 725–729. [Google Scholar] [CrossRef] [PubMed]
- Cuesta, A.; Esteban, M.A.; Meseguer, J. Natural cytotoxic activity of gilthead seabream (Sparus aurata L.) leucocytes. Assessment by flow cytometry and microscopy. Vet. Immunol. Immunopathol. 1999, 71, 161–171. [Google Scholar] [CrossRef]
- Secombes, C.J. Immunological activation of rainbow trout macrophages induced in vitro by sperm autoantibodies and factors derived from testis sensitised leucocytes. Vet. Immunol. Immunopathol. 1986, 12, 193–201. [Google Scholar] [CrossRef]
- Saito, E.; Nakamura, O.; Yamada, H.; Tsutsui, S.; Watanabe, T. Suppression of lymphocyte proliferation by ovarian cavity fluid from the viviparous fish Neoditrema ransonnetii (Perciformes; Embiotocidae). Fish Shellfish Immunol. 2009, 27, 549–555. [Google Scholar] [CrossRef] [PubMed]
- Cammarata, M.; Benenati, G.; Odom, E.W.; Salerno, G.; Vizzini, A.; Vasta, G.R.; Parrinello, N. Isolation and characterization of a fish F-type lectin from gilt head bream (Sparus aurata) serum. Biochim. Biophys. Acta 2007, 1770, 150–155. [Google Scholar] [CrossRef] [PubMed]
- Dong, M.; Liang, Y.; Ramalingam, R.; Tang, S.W.; Shen, W.; Ye, R.; Gopalakrishnan, S.; Au, D.W.T.; Lam, Y.W. Proteomic characterization of the interactions between fish serum proteins and waterborne bacteria reveals the suppression of anti-oxidative defense as a serum-mediated antimicrobial mechanism. Fish Shellfish Immunol. 2017, 62, 96–106. [Google Scholar] [CrossRef] [PubMed]
- An, J.; Liu, T.; She, R.; Wu, Q.; Tian, J.; Shi, R.; Hao, W.; Ren, X.; Yang, Y.; Lu, Y.; et al. Replication of hepatitis E virus in the ovary and promotion of oocyte apoptosis in rabbits infected with HEV-4. Oncotarget 2018, 9, 4475–4484. [Google Scholar] [CrossRef] [PubMed]
- Song, B.-H.; Yun, S.-I.; Woolley, M.; Lee, Y.-M. Zika virus: History, epidemiology, transmission, and clinical presentation. J. Neuroimmunol. 2017, 308, 50–64. [Google Scholar] [CrossRef] [PubMed]
- Blaszkowska, J.; Goralska, K. Parasites and fungi as a threat for prenatal and postnatal human development. Ann. Parasitol. 2014, 60, 225–234. [Google Scholar] [PubMed]
- Sheldon, I.M.; Owens, S.-E.; Turner, M.L. Innate immunity and the sensing of infection, damage and danger in the female genital tract. J. Reprod. Immunol. 2017, 119, 67–73. [Google Scholar] [CrossRef] [PubMed]
- Armour, N.K.; Ferguson-Noel, N. Evaluation of the egg transmission and pathogenicity of Mycoplasma gallisepticum isolates genotyped as ts-11. Avian Pathol. 2015, 44, 296–304. [Google Scholar] [CrossRef] [PubMed]
- Breuil, G.; Pepin, J.F.P.; Boscher, S.; Thiery, R. Experimental vertical transmission of nodavirus from broodfish to eggs and larvae of the sea bass, Dicentrarchus labrax (L.). J. Fish Dis. 2002, 25, 697–702. [Google Scholar] [CrossRef]
- Kocan, R.M.; Hershberger, P.K.; Elder, N.E. Survival of the North American strain of viral hemorrhagic septicemia virus (VHSV) in filtered seawater and seawater containing ovarian fluid, crude oil and serum-enriched culture medium. Dis. Aquat. Organ. 2001, 44, 75–78. [Google Scholar] [CrossRef] [PubMed]
- Sinyakov, M.S.; Belotsky, S.; Shlapobersky, M.; Avtalion, R.R. Vertical and horizontal transmission of tilapia larvae encephalitis virus: The bad and the ugly. Virology 2011, 410, 228–233. [Google Scholar] [CrossRef] [PubMed]
- Smail, D.A.; Munro, E.S. Isolation and quantification of infectious pancreatic necrosis virus from ovarian and seminal fluids of Atlantic salmon, Salmo salar L. J. Fish Dis. 2008, 31, 49–58. [Google Scholar] [CrossRef] [PubMed]
- Fioravanti, M.L.; Caffara, M.; Florio, D.; Gustinelli, A.; Marcer, F. Sphaerospora dicentrarchi and S. testicularis (Myxozoa: Sphaerosporidae) in farmed European seabass (Dicentrarchus labrax) from Italy. Folia Parasitol. 2004, 51, 208–210. [Google Scholar] [CrossRef] [PubMed]
- Sitja-Bobadilla, A.; Alvarez-Pellitero, P. Population dynamics of Sphaerospora dicentrarchi Sitja-Bobadilla et Alvarez-Pellitero, 1992 and S. testicularis Sitja-Bobadilla et Alvarez-Pellitero, 1990 (Myxosporea: Bivalvulida) infections in wild and cultured Mediterranean sea bass (Dicentrarchus labrax L.). Parasitology 1993, 106, 39–45. [Google Scholar] [PubMed]
- Sitja-Bobadilla, A.; Alvarez-Pellitero, P. Pathologic effects of Sphaerospora dicentrarchi Sitjà-Bobadilla and Alvarez-Pellitero, 1992 and S. testicularis Sitjà-Bobadilla and Alvarez-Pellitero, 1990 (Myxosporea: Bivalvulida) parasitic in the Mediterranean sea bass Dicentrarchus labrax L. (Teleostei: Serranidae) and the cell-mediated immune reaction: A light and electron microscopy study. Parasitol. Res. 1993, 79, 119–129. [Google Scholar] [PubMed]
- Alvarez-Pellitero, P.; Sitja-Bobadilla, A. Pathology of Myxosporea in marine fish culture. Dis. Aquat. Organ. 1993, 17, 229–238. [Google Scholar] [CrossRef]
- Pappas, G. The changing Brucella ecology: Novel reservoirs, new threats. Int. J. Antimicrob. Agents 2010, 36 (Suppl. 1), S8–S11. [Google Scholar] [CrossRef] [PubMed]
- Nymo, I.H.; Seppola, M.; Al Dahouk, S.; Bakkemo, K.R.; Jiménez de Bagüés, M.P.; Godfroid, J.; Larsen, A.K. Experimental challenge of Atlantic cod (Gadus morhua) with a Brucella pinnipedialis strain from Hooded seal (Cystophora cristata). PLoS ONE 2016, 11, e0159272. [Google Scholar] [CrossRef] [PubMed]
- Taylor, P.W. Detection of Flavobacterium psychrophilum in eggs and sexual fluids of pacific salmonids by a polymerase chain reaction assay: Implications for vertical transmission of bacterial coldwater disease. J. Aquat. Anim. Health 2004, 16, 104–108. [Google Scholar] [CrossRef]
- Brown, L.L.; Cox, W.T.; Levine, R.P. Evidence that the causal agent of bacterial cold-water disease Flavobacterium psychrophilum is transmitted within salmonid eggs. Dis. Aquat. Organ. 1997, 29, 213–218. [Google Scholar] [CrossRef]
- Ekman, E.; Börjeson, H.; Johansson, N. Flavobacterium psychrophilum in Baltic salmon Salmo salar brood fish and their offspring. Dis. Aquat. Organ. 1999, 37, 159–163. [Google Scholar] [CrossRef] [PubMed]
- Cipriano, R.C. Intraovum infection caused by Flavobacterium psychrophilum among eggs from captive Atlantic salmon broodfish. J. Aquat. Anim. Health 2005, 17, 275–283. [Google Scholar] [CrossRef]
- Madsen, L.; Moller, J.D.; Dalsgaard, I. Flavobacterium psychrophilum in rainbow trout, Oncorhynchus mykiss (Walbaum), hatcheries: Studies on broodstock, eggs, fry and environment. J. Fish Dis. 2005, 28, 39–47. [Google Scholar] [CrossRef] [PubMed]
- Chaves-Pozo, E.; Montero, J.; Cuesta, A.; Tafalla, C. Viral hemorrhagic septicemia and infectious pancreatic necrosis viruses replicate differently in rainbow trout gonad and induce different chemokine transcription profiles. Dev. Comp. Immunol. 2010, 34, 648–658. [Google Scholar] [CrossRef] [PubMed]
- Al-Hussinee, L.; Lumsden, J.S. Detection of VHSV IVb withing the gonads of Great Lakes fish using in situ hybridization. Dis. Aquat. Organ. 2011, 95, 81–86. [Google Scholar] [CrossRef] [PubMed]
- Ahne, W.; Negele, R.D. IPNV transmission via eyed eggs. In Fish and Shellfish Pathology; Ellis, A., Ed.; Academic Press: London, Orlando, USA, 1985; pp. 261–269. [Google Scholar]
- World Organisation for Animal Health. OIE, Aquatic Animal Health Code, 13rd ed.; OIE, Ed.; World Organisation for Animal Health: Paris, France, 2010; ISBN 978-92-9044-777-1. [Google Scholar]
- Sitjà-Bobadilla, A.; Alvarez-Pellitero, P. Ultrastructural and cytochemical observations on the sporogenesis of Sphaerospora testicularis (Protozoa: Myxosporea) from Mediterranean sea bass, Dicentrarchus labrax (L.). Eur. J. Protistol. 1993, 29, 219–229. [Google Scholar] [CrossRef]
- Mao, M.-G.; Wen, S.-H.; Perálvarez-Marín, A.; Li, H.; Jiang, J.-L.; Jiang, Z.-Q.; Li, X.; Sun, H.; Lü, H.-Q. Evidence for and characterization of nervous necrosis virus infection in Pacific cod (Gadus macrocephalus). Arch. Virol. 2015, 160, 2237–2248. [Google Scholar] [CrossRef] [PubMed]
- Valero, Y.; Arizcun, M.; Esteban, M.A.; Bandín, I.; Olveira, J.G.; Patel, S.; Cuesta, A.; Chaves-Pozo, E. Nodavirus colonizes and replicates in the testis of gilthead seabream and European sea bass modulating its immune and reproductive functions. PLoS ONE 2015, 10, e0145131. [Google Scholar] [CrossRef] [PubMed]
- Dalla Valle, L.; Zanella, L.; Patarnello, P.; Paolucci, L.; Belvedere, P.; Colombo, L. Development of a sensitive diagnostic assay for fish nervous necrosis virus based on RT-PCR plus nested PCR. J. Fish Dis. 2000, 23, 321–327. [Google Scholar] [CrossRef]
- Mushiake, K.; Nishizawa, T.; Nakai, T.; Furusawa, I.; Muroga, K. Control of VNN in striped jack: Selection of spawners based on the detection of SJNNV gene by polymerase chain reaction (PCR). Fish Pathol. 1994, 29, 177–182. [Google Scholar] [CrossRef]
- Shao, L.; Zhao, J.; Zhang, H. Spring viraemia of carp virus enters grass carp ovary cells via clathrin-mediated endocytosis and macropinocytosis. J. Gen. Virol. 2016, 97, 2824–2836. [Google Scholar] [CrossRef] [PubMed]
- Suebsing, R.; Pradeep, P.J.; Jitrakorn, S.; Sirithammajak, S.; Kampeera, J.; Turner, W.A.; Saksmerprome, V.; Withyachumnarnkul, B.; Kiatpathomchai, W. Detection of natural infection of infectious spleen and kidney necrosis virus in farmed tilapia by hydroxynapthol blue-loop-mediated isothermal amplification assay. J. Appl. Microbiol. 2016, 121, 55–67. [Google Scholar] [CrossRef] [PubMed]
- Dawar, F.U.; Hu, X.; Zhao, L.; Dong, X.; Xiong, Y.; Zhou, M.; Liang, R.; Sarath Babu, V.; Li, J.; Mei, J.; et al. Transcriptomic analysis reveals differentially expressed genes and a unique apoptosis pathway in channel catfish ovary cells after infection with the channel catfish virus. Fish Shellfish Immunol. 2017, 71, 58–68. [Google Scholar] [CrossRef] [PubMed]
- Ahne, W.; Bjorklund, H.V.; Essbauer, S.; Fijan, N.; Kurath, G.; Winton, J.R. Spring viremia of carp (SVC). Dis. Aquat. Org. Dis. Aquat. Org. 2002, 52, 261–272. [Google Scholar] [CrossRef] [PubMed]
- Nabi, I.R.; Le, P.U. Caveolae/raft-dependent endocytosis. J. Cell Biol. 2003, 161, 673–677. [Google Scholar] [CrossRef] [PubMed]
- Guo, C.-J.; Wu, Y.-Y.; Yang, L.-S.; Yang, X.-B.; He, J.; Mi, S.; Jia, K.-T.; Weng, S.-P.; Yu, X.-Q.; He, J.-G. Infectious spleen and kidney necrosis virus (a fish iridovirus) enters Mandarin fish fry cells via caveola-dependent endocytosis. J. Virol. 2012, 86, 2621–2631. [Google Scholar] [CrossRef] [PubMed]
- Hedrick, R.P.; McDowell, T.S.; Groff, J.M.; Yun, S.; Wingfield, W.H. Isolation and some properties of an iridovirus like agent from white sturgeon Acipenser transmontanus. Dis. Aquat. Organ. 1992, 12, 75–81. [Google Scholar] [CrossRef]
- LaPatra, S.E.; Groff, J.M.; Jones, G.R.; Munn, B.; Patterson, T.L.; Holt, R.A.; Hauck, A.K.; Hedrick, R.P. Occurrence of white sturgeon iridovirus infections among cultured white sturgeon in the Pacific Northwest. Aquaculture 1994, 126, 201–210. [Google Scholar] [CrossRef]
- Nylund, A.; Hovland, T.; Watanabe, K.; Endresen, C. Presence of infectious salmon anaemia virus (ISAV) in tissues of Atlantic salmon, Salmo salar L., collected during three separate outbreaks of the disease. J. Fish Dis. 1995, 18, 135–145. [Google Scholar] [CrossRef]
- Marshall, S.H.; Ramírez, R.; Labra, A.; Carmona, M.; Muñoz, C. Bona fide evidence for natural vertical transmission of infectious salmon anemia virus in freshwater brood stocks of farmed Atlantic salmon (Salmo salar) in Southern Chile. J. Virol. 2014, 88, 6012–6018. [Google Scholar] [CrossRef] [PubMed]
- Gomez-Casado, E.; Estepa, A.; Coll, J.M. A comparative review on European-farmed finfish RNA viruses and their vaccines. Vaccine 2011, 29, 2657–2671. [Google Scholar] [CrossRef] [PubMed]
- Breuil, G.; Bonami, J.R.; Pepin, J.F.; Pichot, Y. Viral infection (picorna-like virus) associated with mass mortalities in hatchery-reared sea-bass (Dicentrarchus labrax) larvae and juveniles. Aquaculture 1991, 97, 109–116. [Google Scholar] [CrossRef]
- Grotmol, S.; Bergh, O.; Totland, G.K. Transmission of viral encephalopathy and retinopathy (VER) to yolk-sac larvae of the Atlantic halibut Hippoglossus hippoglossus: Occurrence of nodavirus in various organs and a possible route of infection. Dis. Aquat. Organ. 1999, 36, 95–106. [Google Scholar] [CrossRef] [PubMed]
- Korsnes, K.; Karlsbakk, E.; Nylund, A.; Nerland, A.H. Horizontal transmission of nervous necrosis virus between turbot Scophthalmus maximus and Atlantic cod Gadus morhua using cohabitation challenge. Dis. Aquat. Organ. 2012, 99, 13–21. [Google Scholar] [CrossRef] [PubMed]
- Kuo, H.C.; Wang, T.Y.; Hsu, H.H.; Chen, P.P.; Lee, S.H.; Chen, Y.M.; Tsai, T.J.; Wang, C.K.; Ku, H.T.; Lee, G.B.; et al. Nervous necrosis virus replicates following the embryo development and dual infection with iridovirus at juvenile stage in grouper. PLoS ONE 2012, 7, e36183. [Google Scholar] [CrossRef] [PubMed]
- Watanabe, K.I.; Nishizawa, T.; Yoshimizu, M. Selection of brood stock candidates of barfin flounder using an ELISA system with recombinant protein of barfin flounder nervous necrosis virus. Dis. Aquat. Organ. 2000, 41, 219–223. [Google Scholar] [CrossRef] [PubMed]
- Chaves-Pozo, E.; Guardiola, F.A.; Meseguer, J.; Esteban, M.A.; Cuesta, A. Nodavirus infection induces a great innate cell-mediated cytotoxic activity in resistant, gilthead seabream, and susceptible, European sea bass, teleost fish. Fish Shellfish Immunol. 2012, 33, 1159–1166. [Google Scholar] [CrossRef] [PubMed]
- Valero, Y.; Boughlala, B.; Arizcun, M.; Patel, S.; Fiksdal, I.U.; Esteban, M.Á.; De Juan, J.; Meseguer, J.; Chaves-Pozo, E.; Cuesta, A. Genes related to cell-mediated cytotoxicity and interferon response are induced in the retina of European sea bass upon intravitreal infection with nodavirus. Fish Shellfish Immunol. 2018, 74, 627–636. [Google Scholar] [CrossRef] [PubMed]
- Valero, Y.; Arizcun, M.; Esteban, M.Á.; Cuesta, A.; Chaves-Pozo, E. Transcription of histones H1 and H2B is regulated by several immune stimuli in gilthead seabream and European sea bass. Fish Shellfish Immunol. 2016, 57, 107–115. [Google Scholar] [CrossRef] [PubMed]
- Valero, Y.; Morcillo, P.; Meseguer, J.; Buonocore, F.; Esteban, M.A.; Chaves-Pozo, E.; Cuesta, A. Characterization of the interferon pathway in the teleost fish gonad against the vertically transmitted viral nervous necrosis virus. J. Gen. Virol. 2015, 96, 2176–2187. [Google Scholar] [CrossRef] [PubMed]
- Cordero, H.; Li, C.H.; Chaves-Pozo, E.; Esteban, M.Á.; Cuesta, A. Molecular identification and characterization of haptoglobin in teleosts revealed an important role on fish viral infections. Dev. Comp. Immunol. 2017, 76, 189–199. [Google Scholar] [CrossRef] [PubMed]
- Tokarz, J.; Möller, G.; Hrabě de Angelis, M.; Adamski, J. Steroids in teleost fishes: A functional point of view. Steroids 2015, 103, 123–144. [Google Scholar] [CrossRef] [PubMed]
- Cabas, I.; Rodenas, M.C.; Abellan, E.; Meseguer, J.; Mulero, V.; Garcia-Ayala, A.; Abellán, E.; Meseguer, J.; Mulero, V.; García-Ayala, A. Estrogen signaling through the G protein-coupled estrogen receptor regulates granulocyte activation in fish. J. Immunol. 2013, 191, 4628–4639. [Google Scholar] [CrossRef] [PubMed]
- Kovats, S. Estrogen receptors regulate innate immune cells and signaling pathways. Cell. Immunol. 2015, 294, 63–69. [Google Scholar] [CrossRef] [PubMed]
- Liarte, S.; Chaves-Pozo, E.; Abellán, E.; Meseguer, J.; Mulero, V.; Canario, A.V.; García-Ayala, A. Estrogen-responsive genes in macrophages of the bony fish gilthead seabream: A transcriptomic approach. Dev. Comp. Immunol. 2011, 35, 840–849. [Google Scholar] [CrossRef] [PubMed]
- Liarte, S.; Cabas, I.; Chaves-Pozo, E.; Arizcun, M.; Meseguer, J.; Mulero, V.; García-Ayala, A. Natural and synthetic estrogens modulate the inflammatory response in the gilthead seabream (Sparus aurata L.) through the activation of endothelial cells. Mol. Immunol. 2011, 48, 1917–1925. [Google Scholar] [CrossRef] [PubMed]
- López-Muñoz, A.; Liarte, S.; Gómez-González, N.E.; Cabas, I.; Meseguer, J.; García-Ayala, A.; Mulero, V. Estrogen receptor 2b deficiency impairs the antiviral response of zebrafish. Dev. Comp. Immunol. 2015, 53, 55–62. [Google Scholar] [CrossRef] [PubMed]
- Roa, J.; Navarro, V.M.; Tena-Sempere, M. Kisspeptins in reproductive biology: Consensus knowledge and recent developments. Biol. Reprod. 2011, 85, 650–660. [Google Scholar] [CrossRef] [PubMed]
- Migaud, H.; Ismail, R.; Cowan, M.; Davie, A. Kisspeptin and seasonal control of reproduction in male European sea bass (Dicentrarchus labrax ). Gen. Comp. Endocrinol. 2012, 179, 384–399. [Google Scholar] [CrossRef] [PubMed]
- Escobar, S.; Felip, A.; Gueguen, M.M.; Zanuy, S.; Carrillo, M.; Kah, O.; Servili, A. Expression of kisspeptins in the brain and pituitary of the european sea bass (Dicentrarchus labrax). J. Comp. Neurol. 2013, 521, 933–948. [Google Scholar] [CrossRef] [PubMed]
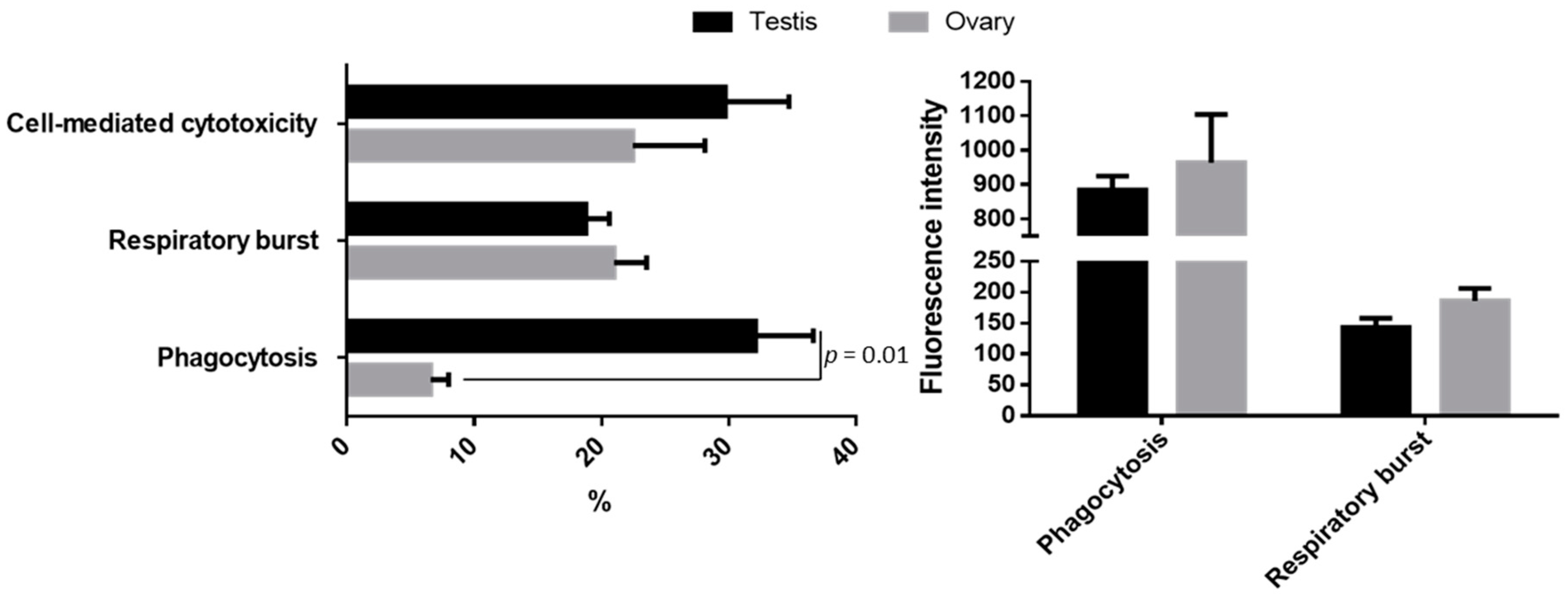
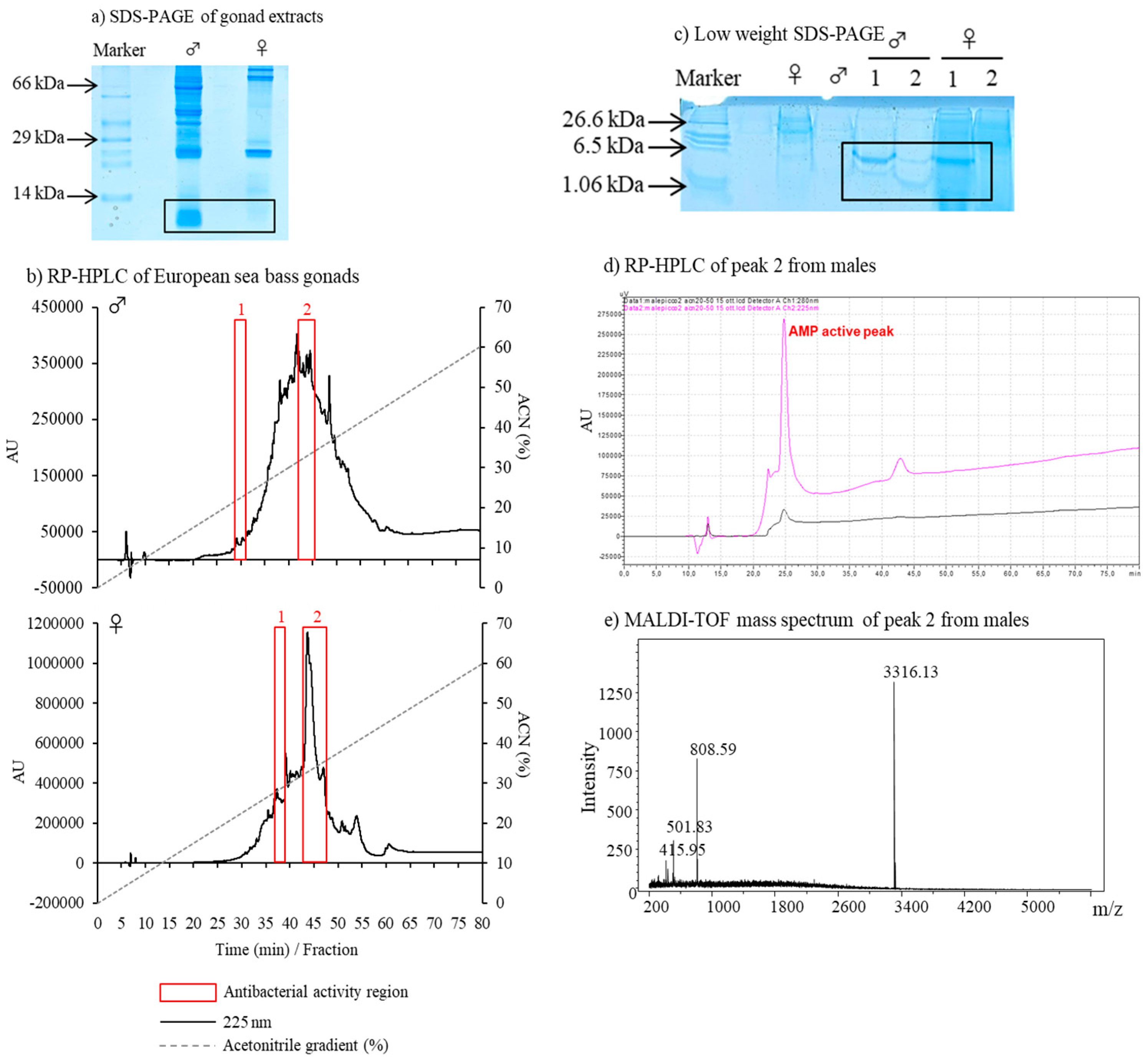
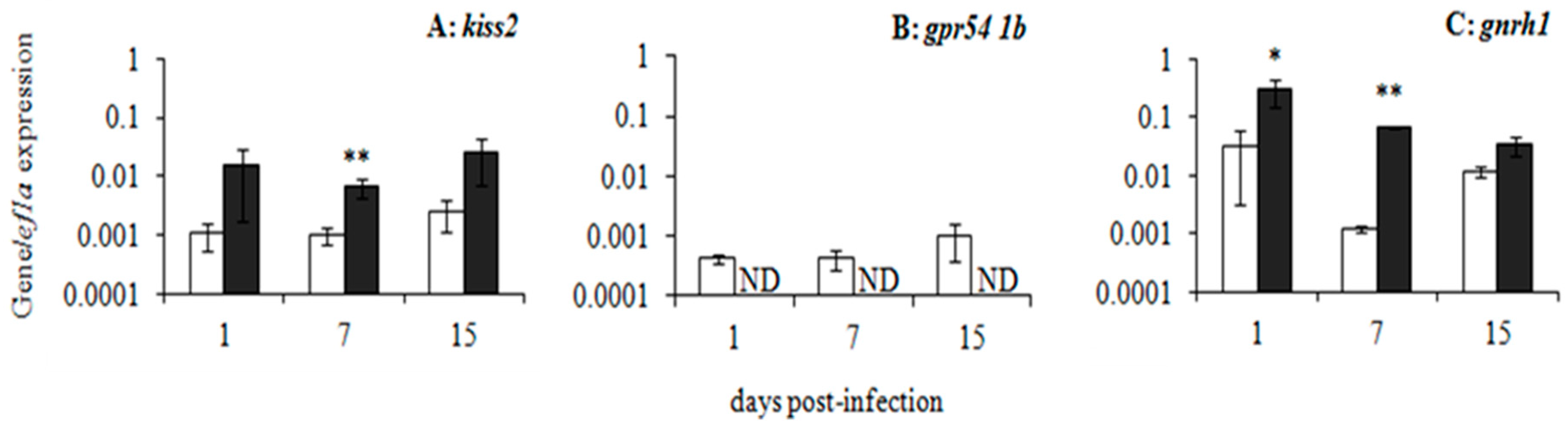
| Time Tested | Sex | Region | Micrococcus luteus | Escherichia coli | Candida albicans |
|---|---|---|---|---|---|
| Naïve | Testis | Complete | 1:2 | - | - |
| Ovary | Complete | 1:2 | - | - | |
| After Sep-Pak purification | Testis | 10% ACN | - | - | - |
| 40% ACN | 1:64 | 1:8 | 1:4 | ||
| 80% ACN | - | - | - | ||
| Ovary | 10% ACN | - | - | - | |
| 40% ACN | 1:32 | 1:2 | - | ||
| 80% ACN | - | - | - | ||
| After 1st RP-HPLC purification | Testis | Region 1 | 1:8 | ND | ND |
| Region 2 | 1:16 | ND | ND | ||
| Ovary | Region 1 | 1:4 | ND | ND | |
| Region 2 | 1:16 | ND | ND | ||
| After 2nd RP-HPLC purification | Testis | Region 1 | - | ND | ND |
| Region 2 | 1:1 | ND | ND | ||
| Ovary | Region 1 | - | ND | ND | |
| Region 2 | - | ND | ND |
| Etiological Agent | Fish Species | Tissue | Location | Detection Method | Reference |
|---|---|---|---|---|---|
| Parasites | |||||
| Sphaerospora testicularis | European sea bass (Dicentrarchus labrax) | Testis | Extracellular: Interstitial and seminiferous tubules | Transmission electron and light microscopy | [96,111] |
| Bacteria | |||||
| Brucella pinnipedialis | Atlantic cod (Gadus morhua) | Ovary | ND | Bacterial growth culture | [101] |
| Flavobacterium psychrophilum | Coho salmon (Oncorhynchus kisutch) | Ovary | Gametes | Conventional PCR | [102] |
| Chinook salmon (Oncorhynchus tshawytscha) | Ovary | Gametes | Conventional PCR | ||
| Shum salmon (Oncorhynchus keta) | Ovary | Gametes | Conventional PCR | ||
| Rainbow trout (Oncorhynchus mykiss) | Ovary | Gametes | Conventional PCR | ||
| Baltic salmon (Salmo salar) | Ovary | Gametes | Bacterial growth culture | [104] | |
| Virus | |||||
| NNV | Pacific cod (Gadus macrocephalus) | Gonad | ND | qPCR | [112] |
| European sea bass | Testis | Sertoli | in situ PCR and re-isolation in cell culture | [113] | |
| Sperm | Conventional PCR | [114] | |||
| Ovary | ND | Conventional PCR | |||
| Shi drum (Umbrina cirrosa) | Testis | Sperm | Conventional PCR | [114] | |
| Ovary | ND | Conventional PCR | |||
| Gilthead seabream (Sparus aurata) | Testis | Tunica albuginea, Sertoli and germ cells Tunica albuginea Germ cells | in situ PCR, immunohistochemistry and re-isolation in cell culture | [113] | |
| Striped jack (Pseudocaranx dentex) | Ovary | ND | Conventional PCR | [115] | |
| Testis | ND | Conventional PCR | |||
| VHSV | Rainbow trout | Ovary | Efferent duct’s epithelial and interstitial cells | qPCR and immunohistochemistry | [108] |
| IPNV | Rainbow trout | Ovary | RTG-2 cell line | Ovarian cell line | [107] |
| SVCV | Grass carp (Ctenopharyngodon idella) | Ovary | CO cell line | Grass carp ovary cell line | [116] |
| ISKNV | Nile tilapia (Oreochromis niloticus) | Ovary | Follicle cells | in situ hydroxynapthol blue-loop-mediated isothermal amplification | [117] |
| CCV | Channel catfish (Ictalurus punctatus) | Ovary | CCO cell line | Ovarian cell line | [118] |
| mda5 | lgp2 | mavs | traf3 | tank | tbk1 | irf3 | irf7 | ifn | mx | pkr | |
|---|---|---|---|---|---|---|---|---|---|---|---|
| Sea bass erb2 | - | 0.73 | 0.58 | 0.60 | 0.46 | 0.69 | 0.69 | 0.59 | 0.67 | 0.75 | 0.67 |
| - | 0.00 | 0.02 | 0.01 | 0.07 | 0.00 | 0.00 | 0.01 | 0.00 | 0.01 | 0.00 | |
| Gilthead seabream era | 0.89 | - | - | - | - | 0.77 | 0.80 | - | 0.88 | 0.82 | 0.82 |
| 0.00 | - | - | - | - | 0.00 | 0.00 | - | 0.00 | 0.00 | 0.00 |
| c3 | lyz | hamp | bdef | pis | h1 | h2b | |
|---|---|---|---|---|---|---|---|
| Gilthead seabream era | 0.79 | 0.67 | 0.42 | 0.85 | 0.88 | 0.77 | 0.79 |
| 0.00 | 0.00 | 0.04 | 0.00 | 0.00 | 0.00 | 0.00 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Valero, Y.; Cuesta, A.; Cammarata, M.; Esteban, M.A.; Chaves-Pozo, E. Immune-Endocrine Interactions in the Fish Gonad during Infection: An Open Door to Vertical Transmission. Fishes 2018, 3, 24. https://doi.org/10.3390/fishes3020024
Valero Y, Cuesta A, Cammarata M, Esteban MA, Chaves-Pozo E. Immune-Endocrine Interactions in the Fish Gonad during Infection: An Open Door to Vertical Transmission. Fishes. 2018; 3(2):24. https://doi.org/10.3390/fishes3020024
Chicago/Turabian StyleValero, Yulema, Alberto Cuesta, Matteo Cammarata, María Angeles Esteban, and Elena Chaves-Pozo. 2018. "Immune-Endocrine Interactions in the Fish Gonad during Infection: An Open Door to Vertical Transmission" Fishes 3, no. 2: 24. https://doi.org/10.3390/fishes3020024
APA StyleValero, Y., Cuesta, A., Cammarata, M., Esteban, M. A., & Chaves-Pozo, E. (2018). Immune-Endocrine Interactions in the Fish Gonad during Infection: An Open Door to Vertical Transmission. Fishes, 3(2), 24. https://doi.org/10.3390/fishes3020024






