How May the Increase in Pink Salmon (Oncorhynchus gorbucsha) Cause the Populations of the Atlantic Salmon (Salmo salar) to Decline?
Abstract
1. Background
1.1. Invasive Species Pink Salmon Spreads in the North Atlantic
1.2. The Decline of Atlantic Salmon and the Increase in Pink Salmon Coincide
1.3. The Prey Fauna of Salmon Is Much Wider in the North Atlantic than in the Baltic Sea
1.4. The Survival of Atlantic Salmon Depends on the Diet of Post-Smolts—A High-Lipid Fish Diet Reduces Survival
1.5. A Diet Rich in Fish-Based Marine Lipids Causes Thiamine Deficiency
1.6. The Lipid Peroxidation of n–3 PUFAs Produces Toxic Malondialdehyde
1.7. Invertebrates Are Lower in n–3 HUFAs than Marine Fish
1.8. Studies on the Effects of Pink Salmon Have Focused on Other Oncorhynchus Species
2. The Life Cycles of Atlantic Salmon and Pink Salmon Differ
2.1. Atlantic Salmon
| Life Phase | Atlantic Salmon | Pink Salmon | References |
|---|---|---|---|
| In the river | |||
| Ascending | June–Aug | July–Aug | [74] |
| Spawning | Sept–Oct | Aug–early Sept | [74,77] |
| Embryo | Sept/Oct–May | Aug/early Sept–Oct/Apr | [74,77] |
| Hatching | May | Oct–Apr | [74,77] |
| Yolk-sac fry | May–June | Oct–Apr | [74,77] |
| Alevin | May–June | Apr | |
| Parr | 3–8 years | – | [74] |
| Parr food | Chironomid and Ephemeroptera larvae (pink salmon eggs) | [8] | |
| Smolt migration | June–July | Apr–June | [74,78] |
| Smolt food | Trichoptera, Plecoptera and Ephemeroptera larvae (pink salmon alevins/smolts) | Chironomid, Plecoptera and Ephemeroptera larvae | [8,76,78] |
| In the sea | |||
| Post-smolts in the estuary | A couple of weeks | At least four weeks | [8,30] |
| Post-smolt food in the estuary | Fish fry, Euphausiids, (pink salmon smolts) | Copepods, Euphausiids, amphipods, fish fry | [79,80] |
| Post-smolt, lipid content, % | 1.3; 1.4 | 0.6–2.1 | [81,82,83] |
| Adult, open sea residence period | 1–4 years | 1+ year | [74,84] |
| Adult food | |||
| Adult, lipid content, % | 2.1–22 | 1.2–7.6 | [82,83,85,86,87,88] |
2.2. Pink Salmon
3. Possible Impacts of Pink Salmon on Atlantic Salmon
3.1. Pink Salmon Spawn Earlier than Atlantic Salmon
3.2. Pink Salmon Eggs Are Energy-Rich Food for Atlantic Salmon Parr
3.3. Pink Salmon Eggs and Alevins Provide Atlantic Salmon Parr and Smolts with an Excess of Marine Lipids

3.4. Due to the Short River Period, a Large Proportion of Pink Salmon Fry Survive Until the Sea
3.5. Pink Salmon Post-Smolts Are of a Suitable Size as Prey for Atlantic Salmon Post-Smolts
3.6. Post-Smolts Need Protein to Grow
3.7. Post-Smolts of Pink Salmon and Atlantic Salmon Do Not Compete for the Same Food
4. Conclusions and Suggestions
4.1. General
4.2. Research to Further Assess the Hypothesis Presented
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| 1-SW | one-sea-winter Atlantic salmon; i.e., those which return after their first sea-winter |
| 2-SW | two-sea-winter Atlantic salmon; i.e., those which return after their second sea-winter |
| 3-SW | three-sea-winter Atlantic salmon; i.e., those which return after their third sea-winter |
| 4-SW | four-sea-winter Atlantic salmon; i.e., those which return after their fourth sea-winter |
| ALA | alpha-linolenic acid, 18:3n–3 |
| ARA | arachidonic acid, 20:4n–6 |
| ATP | adenosine triphosphate |
| DHA | docosahexaenoic acid, 22:6n–3 |
| DPA | docosapentaenoic acid, 22:5n–3 |
| eDNA | environmental DNA, referring to genetic material shed by organisms |
| EPA | eicosapentaenoic acid, 20:5n–3 |
| HUFA | highly unsaturated long–chain fatty acid |
| LNA | linoleic acid, 18:2n–6 |
| M74 syndrome | thiamine deficiency in fish called in the Baltic Sea region |
| MDA | malondialdehyde |
| MUFA | monounsaturated fatty acid |
| n–3 HUFA | long-chain, highly unsaturated fatty acid of n–3 series |
| n–3 PUFA | polyunsaturated fatty acid of n–3 series |
| Na+/K+-ATPase | Na+/K+-adenosine triphosphatase |
| NAD/NADH | nicotinamide adenine dinucleotide/nicotinamide adenine dinucleotide hydrogen |
| NADP/NADPH | nicotinamide adenine dinucleotide phosphate/nicotinamide adenine dinucleotide phosphate hydrogen |
| PCA | Principal Component Analysis |
| PUFA | polyunsaturated fatty acid |
| SFA | saturated fatty acid |
| TDC | Thiamine Deficiency Complex, thiamine deficiency in fish called in North America |
| THIAM | unbound or free thiamine |
| TPP | thiamine pyrophosphate derivative of thiamine |
| TTP | thiamine triphosphate derivative of thiamine |
| UI | unsaturation index, the sum of double bonds in fatty acids |
References
- Ruggerone, G.T.; Springer, A.M.; van Vliet, G.B.; Connors, B.; Irvine, J.R.; Shaul, L.D.; Sloat, M.R.; Atlas, W.I. From diatoms to killer whales: Impacts of pink salmon on North Pacific ecosystems. Mar. Ecol. Prog. Ser. 2023, 719, 1–40. [Google Scholar] [CrossRef]
- Orell, P.; Erkinaro, J. Kyttyrälohi Jäämeren Lohijoissa, Kirjallisuuskatsaus Vieraslajin Biologiaan, Leviämiseen ja Mahdollisiin Vaikutuksiin; Luonnonvara-ja biotalouden tutkimus No. 75/2023; Luonnonvarakeskus: Helsinki, Finland, 2023; 32p. Available online: http://urn.fi/URN:ISBN:978-952-380-756-3 (accessed on 5 February 2025). (In Finnish)
- Rasputina, E.N.; Shustov, Y.A.; Tyrkin, I.A. Eggs of pink salmon Oncorhynchus gorbuscha as additional nontraditional food of juvenile atlantic salmon Salmo salar in rivers of the Kola Peninsula. Russ. J. Biol. Invasions 2016, 7, 294–296. [Google Scholar] [CrossRef]
- Diaz-Pauli, B.; Berntsen, H.H.; Thorstad, E.B.; Homrum, E.Ì.; Lusseau, S.M.; Wennevik, V.; Utne, K.R. Geographic distribution, abundance, diet, and body size of invasive pink salmon (Oncorhynchus gorbuscha) in the Norwegian and Barents Seas, and in Norwegian rivers. ICES J. Mar. Sci. 2023, 80, 76–90. [Google Scholar] [CrossRef]
- Northern Hemisphere Pink Salmon Expert Group. A Review of Pink Salmon in the Pacific, Arctic, and Atlantic oceans; North Pacific Anadromous Fish Commission Technical Report 21; North Pacific Anadromous Fish Commission: Vancouver, BC, Canada, 2023; 58p. [Google Scholar]
- Sandlund, O.T.; Berntsen, H.H.; Fiske, P.; Kuusela, J.; Muladal, R.; Niemelä, E.; Uglem, I.; Forseth, T.; Mo, T.A.; Thorstad, E.B.; et al. Pink salmon in Norway: The reluctant invader. Biol. Invasions 2019, 21, 1033–1054. [Google Scholar] [CrossRef]
- Staveley, T.A.B.; Hellström, M.; Birgersson, V.; Hernvall, P.; Schibli, H.; Axelsson, E.; Larliander, L.; Molander, L.; Thorstad, E.B.; Berntsen, H.H.; et al. Detection of Non-Native Pink Salmon (Oncorhynchus gorbuscha) in Swedish Rivers Using eDNA. Environ. DNA 2025, 7, e70117. [Google Scholar] [CrossRef]
- Erkinaro, J.; Orell, P.; Kytökorpi, M.; Pohjola, J.P.; Power, M. Active feeding of downstream migrating juvenile pink salmon (Oncorhynchus gorbuscha) revealed in a large Barents Sea river using diet and stable isotope analysis. J. Fish Biol. 2024, 104, 797–806. [Google Scholar] [CrossRef] [PubMed]
- NASCO. NASCO Implementation Plan for Salmon Management in Finland; NASCO Ad Hoc Review Group: Vancouver, BC, Canada, 2008; p. 12. [Google Scholar]
- Anonymous. Status of the Tana/Teno River Salmon Populations in 2020; Report from the Tana Monitoring and Research Group nr 1/2020; Tana Monitoring and Research Group: Tromsö, Norway; Trondheim, Norway; Oulu, Finland, 2020; 124p, Available online: https://jukuri.luke.fi/handle/11111/56652 (accessed on 2 June 2025).
- Anonymous. Status of the Tana/Teno River Salmon Populations in 2024; Report from the Tana/Teno Monitoring and Research Group nr 1/2025; Tana/Teno Monitoring and Research Group: Tromsö, Norway; Trondheim, Norway; Oulu, Finland, 2025; 99p, Available online: https://urn.fi/URN:NBN:fi-fe2025022013046 (accessed on 2 June 2025).
- Staveley, T.A.B.; Bergendahl, I.A.; Bárðarson, H.; Berntsen, H.H.; Eliasen, K.; Erkinaro, J.; Nydaard, R.; Sivebæk, F.; Thorstad, E.B. Status and future perspectives of pink salmon in the Nordic region. Boreal Environ. Res. 2025, 30, 149–162. [Google Scholar] [CrossRef]
- HELCOM. Atlas of the Baltic Sea; Vlasov, N., Ed.; HELCOM: Helsinki, Finland, 2010. [Google Scholar]
- Airaksinen, R.; Hallikainen, A.; Rantakokko, P.; Ruokojärvi, P.; Vuorinen, P.J.; Parmanne, R.; Verta, M.; Mannio, J.; Kiviranta, H. Time trends and congener profiles of PCDD/Fs, PCBs, and PBDEs in Baltic herring off the coast of Finland during 1978–2009. Chemosphere 2014, 114, 165–171. [Google Scholar] [CrossRef]
- Vuorinen, P.J.; Keinänen, M.; Kiviranta, H.; Koistinen, J.; Kiljunen, M.; Myllylä, T.; Pönni, J.; Peltonen, H.; Verta, M.; Karjalainen, J. Biomagnification of organohalogens in Atlantic salmon (Salmo salar) from its main prey species in three areas of the Baltic Sea. Sci. Total Environ. 2012, 421–422, 129–143. [Google Scholar] [CrossRef]
- Ruokojärvi, P.; Airaksinen, R.; Keinänen, M.; Kiviranta, H.; Koponen, J.; Mannio, J.; Myllylä, T.; Nieminen, J.; Raitaniemi, J.; Rantakokko, P.; et al. Dioxin and PCB concentrations in Baltic salmon have decreased remarkably during the 2000s’. Organohalogen Compd. 2017, 79, 134–137. [Google Scholar]
- Vuorinen, P.J.; Roots, O.; Keinänen, M. Review of organohalogen toxicants in fish from the Gulf of Finland. J. Mar. Syst. 2017, 171, 141–150. [Google Scholar] [CrossRef]
- Pitcher, C.R.; Hiddink, J.G.; Jennings, S.; Collie, J.; Parma, A.M.; Amoroso, R.; Mazor, T.; Sciberras, M.; McConnaughey, R.A.; Rijnsdorp, A.D.; et al. Trawl impacts on the relative status of biotic communities of seabed sedimentary habitats in 24 regions worldwide. Proc. Natl. Acad. Sci. USA 2022, 119, e2109449119. [Google Scholar] [CrossRef] [PubMed]
- Hiddink, J.G.; Kaiser, M.J.; Sciberras, M.; McConnaughey, R.A.; Mazor, T.; Hilborn, R.; Collie, J.S.; Pitcher, C.R.; Parma, A.M.; Suuronen, P.; et al. Selection of indicators for assessing and managing the impacts of bottom trawling on seabed habitats. J. Appl. Ecol. 2020, 57, 1199–1209. [Google Scholar] [CrossRef]
- Sciberras, M.; Hiddink, J.G.; Jennings, S.; Szostek, C.L.; Hughes, K.M.; Kneafsey, B.; Clarke, L.J.; Ellis, N.; Rijnsdorp, A.D.; McConnaughey, R.A.; et al. Response of benthic fauna to experimental bottom fishing: A global meta-analysis. Fish Fish. 2018, 19, 698–715. [Google Scholar] [CrossRef]
- Kouts, M.; Maljutenko, I.; Elken, J.; Liu, Y.; Hansson, M.; Viktorsson, L.; Raudsepp, U. Recent regime of persistent hypoxia in the Baltic Sea. Environm. Res. Commun. 2021, 3, 075004. [Google Scholar] [CrossRef]
- Vuorinen, P.J.; Käkelä, R.; Pakarinen, T.; Heinimaa, P.; Ritvanen, T.; Nikonen, S.; Rokka, M.; Keinänen, M. Thiamine deficiency M74 developed in salmon (Salmo salar) stocks in two Baltic Sea areas after the hatching of large year-classes of two clupeid species—Detected by fatty acid signature analysis. Fishes 2024, 9, 35. [Google Scholar] [CrossRef]
- SYKE. Marinefinland.fi. Available online: https://itameri.fi/en/ (accessed on 12 December 2025).
- Hansson, S.; Karlsson, L.; Ikonen, E.; Christensen, O.; Mitans, A.; Uzars, D.; Petersson, E.; Ragnarsson, B. Stomach analyses of Baltic salmon from 1959–1962 and 1994–1997: Possible relations between diet and yolk-sac-fry mortality (M74). J. Fish Biol. 2001, 58, 1730–1745. [Google Scholar] [CrossRef]
- Jacobsen, J.A.; Hansen, L.P. Feeding habits of wild and escaped farmed Atlantic salmon, Salmo salar L., in the Northeast Atlantic. ICES J. Mar. Sci. 2001, 58, 916–933. [Google Scholar] [CrossRef]
- Long, A.P.; Vaughan, L.; Tray, E.; Thomas, K.; Maoiléidigh, N.Ó.; Poole, R.; Cotter, D.; Doogan, A.; Brophy, D. Recent marine growth declines in wild and ranched Atlantic salmon Salmo salar from a western European catchment discovered using a 62-year time series. ICES J. Mar. Sci. 2023, 80, 1697–1709. [Google Scholar] [CrossRef]
- Pardo, S.A.; Bolstad, G.H.; Dempson, J.B.; April, J.; Jones, R.A.; Raab, D.; Hutchings, J.A. Trends in marine survival of Atlantic salmon populations in eastern Canada. ICES J. Mar. Sci. 2021, 78, 2460–2473. [Google Scholar] [CrossRef]
- Keinänen, M.; Raitaniemi, J.; Pönni, J.; Ritvanen, T.; Myllylä, T.; Vuorinen, P.J. Reduced numbers of returning Atlantic salmon (Salmo salar) and thiamine deficiency are both associated with the consumption of high-lipid prey fish. Fishes 2025, 10, 16. [Google Scholar] [CrossRef]
- Salminen, M. Relationships between smolt size, postsmolt growth and sea age at maturity in Atlantic salmon ranched in the Baltic Sea. J. Appl. Ichthyol. 1997, 13, 121–130. [Google Scholar] [CrossRef]
- Thorstad, E.B.; Whoriskey, F.; Uglem, I.; Moore, A.; Rikardsen, A.H.; Finstad, B. A critical life stage of the Atlantic salmon Salmo salar: Behaviour and survival during the smolt and initial post-smolt migration. J. Fish Biol. 2012, 81, 500–542. [Google Scholar] [CrossRef]
- Kallio-Nyberg, I.; Saloniemi, I.; Jutila, E.; Jokikokko, E. Effect of hatchery rearing and environmental factors on the survival, growth and migration of Atlantic salmon in the Baltic Sea. Fish. Res. 2011, 109, 285–294. [Google Scholar] [CrossRef]
- Keinänen, M.; Nikonen, S.; Käkelä, R.; Ritvanen, T.; Rokka, M.; Myllylä, T.; Pönni, J.; Vuorinen, P.J. High lipid content of prey fish and n-3 PUFA peroxidation impair the thiamine status of feeding-migrating Atlantic salmon (Salmo salar) and is reflected in hepatic biochemical indices. Biomolecules 2022, 12, 526. [Google Scholar] [CrossRef] [PubMed]
- Keinänen, M.; Käkelä, R.; Ritvanen, T.; Myllylä, T.; Pönni, J.; Vuorinen, P.J. Fatty acid composition of sprat (Sprattus sprattus) and herring (Clupea harengus) in the Baltic Sea as potential prey for salmon (Salmo salar). Helgol. Mar. Res. 2017, 71, 4. [Google Scholar] [CrossRef]
- Mikkonen, J.; Keinänen, M.; Casini, M.; Pönni, J.; Vuorinen, P.J. Relationships between fish stock changes in the Baltic Sea and the M74 syndrome, a reproductive disorder of Atlantic salmon (Salmo salar). ICES J. Mar. Sci. 2011, 68, 2134–2144. [Google Scholar] [CrossRef]
- Keinänen, M.; Uddström, A.; Mikkonen, J.; Casini, M.; Pönni, J.; Myllylä, T.; Aro, E.; Vuorinen, P.J. The thiamine deficiency syndrome M74, a reproductive disorder of Atlantic salmon (Salmo salar) feeding in the Baltic Sea, is related to the fat and thiamine content of prey fish. ICES J. Mar. Sci. 2012, 69, 516–528. [Google Scholar] [CrossRef]
- Grisdale-Helland, B.; Gatlin, D.M.; Helland, S.J. Optimization of dietary macronutrients for Atlantic salmon post-smolts using increasing ration levels. Aquaculture 2013, 408, 88–94. [Google Scholar] [CrossRef]
- Tocher, D.R. Metabolism and functions of lipids and fatty acids in teleost fish. Rev. Fish. Sci. 2003, 11, 107–184. [Google Scholar] [CrossRef]
- Zhang, Z.; Miar, Y.; Huyben, D.; Colombo, S.M. Omega-3 long-chain polyunsaturated fatty acids in Atlantic salmon: Functions, requirements, sources, de novo biosynthesis and selective breeding strategies. Rev. Aquacult. 2024, 16, 1030–1041. [Google Scholar] [CrossRef]
- Futia, M.H.; Connerton, M.J.; Weidel, B.C.; Rinchard, J. Diet predictions of Lake Ontario salmonines based on fatty acids and correlations between their fat content and thiamine concentrations. J. Great Lakes Res. 2019, 45, 934–948. [Google Scholar] [CrossRef]
- Kaga, T.; Sato, S.; Azumaya, T.; Davis, N.D.; Fukuwaka, M.-a. Lipid content of chum salmon Oncorhynchus keta affected by pink salmon O. gorbuscha abundance in the central Bering Sea. Mar. Ecol. Prog. Ser. 2013, 478, 211–221. [Google Scholar] [CrossRef]
- Jobling, M.; Bendiksen, E.Å. Dietary lipids and temperature interact to influence tissue fatty acid compositions of Atlantic salmon, Salmo salar L., parr. Aquac. Res. 2003, 34, 1423–1441. [Google Scholar] [CrossRef]
- Hasegawa, K.; Yano, Y.; Honda, K.; Ogura, Y. DHA and EPA levels in a piscivorous fish changed by preying upon stocked salmon fry. Sci. Rep. 2023, 13, 15278. [Google Scholar] [CrossRef]
- Mantua, N.J.; Bell, H.; Todgham, A.E.; Daniels, M.E.; Rinchard, J.; Ludwig, J.M.; Field, J.C.; Lindley, S.T.; Rowland, F.E.; Richter, C.A.; et al. Widespread thiamine deficiency in California salmon linked to an anchovy-dominated marine prey base. Proc. Natl. Acad. Sci. USA 2025, 122, e2426011122. [Google Scholar] [CrossRef]
- Vuorinen, P.J.; Juntunen, E.-P.; Iivari, J.; Koski, P.; Nikonen, S.; Rokka, M.; Ritvanen, T.; Pakkala, J.; Heinimaa, P.; Keinänen, M. Lipid-related thiamine deficiency cause mortality of river lampreys (Lampetra fluviatilis) during pre-spawning fasting. Reg. Stud. Mar. Sci. 2023, 62, 14. [Google Scholar] [CrossRef]
- Woodward, B. Dietary vitamin requirements of cultured young fish, with emphasis on quantitative estimates for salmonids. Aquaculture 1994, 124, 133–168. [Google Scholar] [CrossRef]
- Lonsdale, D.; Marrs, C. Thiamine Deficiency Disease, Dysautonomia, and High Calorie Malnutrition; Academic Press: London, UK; San Diego, CA, USA, 2019. [Google Scholar]
- Kriketos, A.D.; Peters, J.C.; Hill, J.O. Cellular and whole-animal energetics. In Biochemical and Physiological Aspects of Human Nutrition; Stipanuk, M.H., Ed.; Saunders/Elsevier: Philadelphia, PA, USA, 2000; pp. 411–424. [Google Scholar]
- Combs, G.F., Jr.; McClung, J.P. Thiamin. In The Vitamins, Fundamental Aspects in Nutrition and Health, 5th ed.; Academic Press: London, UK; San Diego, CA, USA; Cambridge, MA, USA; Oxford, UK, 2017; pp. 297–314. [Google Scholar]
- Lee, B.J.; Jaroszewska, M.; Dabrowski, K.; Czesny, S.; Rinchard, J. Effects of dietary vitamin B-1 (thiamine) and magnesium on the survival, growth and histological indicators in lake trout (Salvelinus namaycush) juveniles. Comp. Biochem. Phys. A 2012, 162, 219–226. [Google Scholar] [CrossRef]
- Depeint, F.; Bruce, W.R.; Shangari, N.; Mehta, R.; O’Brien, P.J. Mitochondrial function and toxicity: Role of the B vitamin family on mitochondrial energy metabolism. Chem. Biol. Interact. 2006, 163, 94–112. [Google Scholar] [CrossRef] [PubMed]
- Gibson, G.E.; Zhang, H. Interactions of oxidative stress with thiamine homeostasis promote neurodegeneration. Neurochem. Int. 2002, 40, 493–504. [Google Scholar] [CrossRef]
- Vollset, K.W.; Urdal, K.; Utne, K.; Thorstad, E.B.; Sægrov, H.; Raunsgard, A.; Skagseth, Ø.; Lennox, R.J.; Østborg, G.M.; Ugedal, O.; et al. Ecological regime shift in the Northeast Atlantic Ocean revealed from the unprecedented reduction in marine growth of Atlantic salmon. Sci. Adv. 2022, 8, abk2542. [Google Scholar] [CrossRef] [PubMed]
- Vuorinen, P.J.; Rokka, M.; Nikonen, S.; Juntunen, E.-P.; Ritvanen, T.; Heinimaa, P.; Keinänen, M. Model for estimating thiamine deficiency-related mortality of Atlantic salmon (Salmo salar) offspring and variation in the Baltic salmon M74 syndrome. Mar. Freshw. Behav. Physiol. 2021, 54, 97–131. [Google Scholar] [CrossRef]
- Lonsdale, D. A review of the biochemistry, metabolism and clinical benefits of thiamin(e) and its derivatives. Evid.-Based Complement. Altern. Med. 2006, 3, 49–59. [Google Scholar] [CrossRef]
- Vuorinen, P.J.; Rokka, M.; Ritvanen, T.; Käkelä, R.; Nikonen, S.; Pakarinen, T.; Keinänen, M. Changes in thiamine concentrations, fatty acid composition, and some other lipid-related biochemical indices in Baltic Sea Atlantic salmon (Salmo salar) during the spawning run and pre-spawning fasting. Helgol. Mar. Res. 2020, 74, 10. [Google Scholar] [CrossRef]
- Keinänen, M.; Käkelä, R.; Ritvanen, T.; Pönni, J.; Harjunpää, H.; Myllylä, T.; Vuorinen, P.J. Fatty acid signatures connect thiamine deficiency with the diet of the Atlantic salmon (Salmo salar) feeding in the Baltic Sea. Mar. Biol. 2018, 165, 161. [Google Scholar] [CrossRef]
- Long, J.; Liu, C.; Sun, L.; Gao, H.; Liu, J. Neuronal mitochondrial toxicity of malondialdehyde: Inhibitory effects on respiratory function and enzyme activities in rat brain mitochondriae. Neurochem. Res. 2009, 34, 786–794. [Google Scholar] [CrossRef]
- Del Rio, D.; Stewart, A.J.; Pellegrini, N. A review of recent studies on malondialdehyde as toxic molecule and biological marker of oxidative stress. Nutr. Metab. Cardiovasc. Dis. 2005, 15, 316–328. [Google Scholar] [CrossRef]
- Cheng, J.; Wang, F.; Yu, D.-F.; Wu, P.-F.; Chen, J.-G. The cytotoxic mechanism of malondialdehyde and protective effect of carnosine via protein cross-linking/mitochondrial dysfunction/reactive oxygen species/MAPK pathway in neurons. Eur. J. Pharmacol. 2011, 650, 184–194. [Google Scholar] [CrossRef]
- Balon, E.K. Terminology of intervals in fish development. J. Fish. Res. Board Can. 1975, 32, 1663–1670. [Google Scholar] [CrossRef]
- Amcoff, P.; Börjeson, H.; Landergren, P.; Vallin, L.; Norrgren, L. Thiamine (vitamin B1) concentrations in salmon (Salmo salar), brown trout (Salmo trutta) and cod (Gadus morhua) from the Baltic sea. Ambio 1999, 28, 48–54. [Google Scholar]
- Kumar, E.; Koponen, J.; Rantakokko, P.; Airaksinen, R.; Ruokojärvi, P.; Kiviranta, H.; Vuorinen, P.J.; Myllylä, T.; Keinänen, M.; Raitaniemi, J.; et al. Distribution of perfluoroalkyl acids in fish species from the Baltic Sea and freshwaters in Finland. Chemosphere 2022, 291, 132688. [Google Scholar] [CrossRef] [PubMed]
- Haugland, M.; Holst, J.C.; Holm, M.; Hansen, L.P. Feeding of Atlantic salmon (Salmo salar L.) post-smolts in the Northeast Atlantic. ICES J. Mar. Sci. 2006, 63, 1488–1500. [Google Scholar] [CrossRef]
- Shimidzu, N.; Goto, M.; Miki, W. Carotenoids as singlet oxygen quenchers in marine organisms. Fisheries Sci. 1996, 62, 134–137. [Google Scholar] [CrossRef]
- Faulkner, C.W.; Ittinuar, S.J.; Tartak, C.U.; L’Herault, V.; Harris, L.N.; Davoren, G.K.; Yurkowski, D.J. You are the colour of what you eat: Higher invertebrate consumption correlates with redder muscle pigmentation in anadromous Arctic char (Salvelinus alpinus) along western Hudson Bay, Nunavut, Canada. Mar. Biol. 2025, 172, 179. [Google Scholar] [CrossRef]
- Tou, J.C.; Jaczynski, J.; Chen, Y.-C. Krill for Human Consumption: Nutritional Value and Potential Health Benefits. Nutr. Rev. 2007, 65, 63–77. [Google Scholar] [CrossRef]
- Pasternack, M.; Salminen, M.; Heinimaa, P. Physiological Condition and Migratory Readiness of Hatchery-Reared Neva Stock Salmon Smolts in 2007–2009. In Riista-ja Kalatalous Selvityksiä 16; Finnish Game and Fisheries Research Institute: Helsinki, Finland, 2010; 33p, Available online: http://urn.fi/URN:ISBN:978-951-776-788-0 (accessed on 16 September 2024). (In Finnish)
- Salminen, T. Kilohaili- ja Silakkaperaisen Ravinnon Vaikutus Itameren Lohen (Salmo salar) Kasvuun ja Rasvahappokoostumukseen Sekä Yhteydet M74-Oireyhtymaan. Master’s Thesis, Helsingin Yliospisto, Biotieteellinen Tiedekunta, Bio-ja Ymparistotieteiden Laitos, Helsinki, Finland, 2018. (In Finnish) [Google Scholar]
- Rand, P.S.; Ruggerone, G.T. Biennial patterns in Alaskan sockeye salmon ocean growth are associated with pink salmon abundance in the Gulf of Alaska and the Bering Sea. ICES J. Mar. Sci. 2024, 81, 701–709. [Google Scholar] [CrossRef]
- Kendall, N.W.; Nelson, B.W.; Losee, J.P. Density-dependent marine survival of hatchery-origin Chinook salmon may be associated with pink salmon. Ecosphere 2020, 11, e03061. [Google Scholar] [CrossRef]
- Dunlop, K.; Eloranta, A.P.; Schoen, E.; Wipfli, M.; Jensen, J.L.A.; Muladal, R.; Christensen, G.N. Evidence of energy and nutrient transfer from invasive pink salmon (Oncorhynchus gorbuscha) spawners to juvenile Atlantic salmon (Salmo salar) and brown trout (Salmo trutta) in northern Norway. Ecol. Freshw. Fish 2021, 30, 270–283. [Google Scholar] [CrossRef]
- Luke. Salmon Run Monitoring, River Teno, Accumulated Numbers of Fish. Available online: https://luonnonvaratieto.luke.fi/numerotieto/raportit?panel=nousulohiseuranta&inits=REGION_ID%3D3&lang=en (accessed on 9 September 2025).
- Karlsson, L.; Karlström, Ö. The Baltic salmon (Salmo salar L.): Its history, present situation and future. Dana 1994, 10, 61–85. [Google Scholar]
- Orell, P.; Erkinaro, J.; Svenning, M.A.; Davidsen, J.G.; Niemelä, E. Synchrony in the downstream migration of smolts and upstream migration of adult Atlantic salmon in the subarctic River Utsjoki. J. Fish Biol. 2007, 71, 1735–1750. [Google Scholar] [CrossRef]
- Morera, F.J.; Castro-Guarda, M.; Nualart, D.; Espinosa, G.; Muñoz, J.L.; Vargas-Chacoff, L. The biological basis of smoltification in Atlantic salmon. Austral J. Vet. Sci. 2021, 53, 73–82. [Google Scholar] [CrossRef]
- Garnås, E.; Hvidsten, N.A. The food of Atlantic salmon Salmo salar L. and brown trout Salmo trutta L. smolts during migration in the Orkla river, Norway. Fauna Nor. 1985, 6, 24–28. [Google Scholar] [CrossRef]
- Erkinaro, J.; Orell, P.; Pohjola, J.P.; Kytökorpi, M.; Pulkkinen, H.; Kuusela, J. Development of invasive pink salmon (Oncorhynchus gorbuscha Walbaum) eggs in a large Barents Sea river. J. Fish Biol. 2022, 101, 1063–1066. [Google Scholar] [CrossRef]
- Marchenko, S.L. Pink salmon Oncorhynchus gorbuscha (Salmoniformes, Salmonidae) from the continental coast of the Sea of Okhotsk. Communication 2: Juveniles. Russ. J. Mar. Biol. 2023, 49, 569–578. [Google Scholar] [CrossRef]
- Bengtsson, O.; Lydersen, C.; Christensen, G.; Węsławski, J.M.; Kovacs, K.M. Marine diets of anadromous Arctic char (Salvelinus alpinus) and pink salmon (Oncorhynchus gorbuscha) in Svalbard, Norway. Polar Biol. 2023, 46, 1219–1234. [Google Scholar] [CrossRef]
- Andreassen, P.M.R.; Martinussen, M.B.; Hvidsten, N.A.; Stefansson, S.O. Feeding and prey-selection of wild Atlantic salmon post-smolts. J. Fish Biol. 2001, 58, 1667–1679. [Google Scholar] [CrossRef]
- Stefansson, S.O.; Björnsson, B.T.; Sundell, K.; Nyhammer, G.; McCormick, S.D. Physiological characteristics of wild Atlantic salmon post-smolts during estuarine and coastal migration. J. Fish Biol. 2003, 63, 942–955. [Google Scholar] [CrossRef]
- Iverson, S.J.; Frost, K.J.; Lang, S.L.C. Fat content and fatty acid composition of forage fish and invertebrates in Prince William Sound, Alaska, factors contributing to among and within species variability. Mar. Ecol. Prog. Ser. 2002, 241, 161–181. [Google Scholar] [CrossRef]
- Utne, K.R.; Meier, S.; Rasinger, J.D.; Nikolioudakis, N.; Stenevik, E.K.; Slotte, A.; Engberg, K.; Wennevik, V. Fatty acids and lipid levels in Atlantic salmon (Salmo salar) change with sea-age and differ from other pelagic fish in the Norwegian Sea. Res. Square 2025. [Google Scholar] [CrossRef]
- Erkinaro, J.; Orell, P.; Falkegård, M.; Kylmäaho, M.; Domaas, S.; Pohjola, J.-P.; Johansen, N.; Haantie, J.; Foldvik, A.; Kuusela, J. Status of Atlantic Salmon Stocks in the Rivers Teno/Tana and Näätämöjoki/Neidenelva, Finland/Norway; International Council for the Exploration of the Sea/North Atlantic Salmon Working Group: Copenhagen, Denmark, 2021; 17p. [Google Scholar]
- Jensen, I.J.; Mæhre, H.K.; Tømmerås, S.; Eilertsen, K.E.; Olsen, R.L.; Elvevoll, E.O. Farmed Atlantic salmon (Salmo salar L.) is a good source of long chain omega-3 fatty acids. Nutr. Bull. 2012, 37, 25–29. [Google Scholar] [CrossRef]
- Nomura, T.; Myers, K.W.; Kondzela, C.M.; Murphy, J.M.; Honma, H.; Carlson, H.R. Variation in lipid content in the muscle of chum and pink salmon in the Gulf of Alaska in May 1999. Bull. Natl. Salmon Resour. Cent. 2001, 4, 13–18. Available online: https://salmon.fra.affrc.go.jp/kankobutu/bulletin/BNSRC04-3s.pdf (accessed on 8 August 2025).
- Kaga, T.; Sato, S.; Morita, K.; Fukuwaka, M.-A.; Chiba, T.; Takasaki, D. Seasonal Changes in the Total Lipid Contents of Chum and Pink Salmon in the North Pacific Ocean and Bering Sea During the Spring and Summer of 2005–2006; Doc. 1040; North Pacific Anadromous Fish Commission: Vancouver, BC, Canada, 2007; 20p, Available online: http://www.npafc.org (accessed on 18 June 2025).
- Tømmerås, S. Fat, Fatty Acids and Fat Soluble Nutrients in Fillet of Farmed and Wild Atlantic Salmon (Salmo salar L.). Master’s Thesis, University of Tromsø, Tromsø, Norway, 2011. [Google Scholar]
- Utne, K.R.; Pauli, B.D.; Haugland, M.; Jacobsen, J.A.; Maoileidigh, N.; Melle, W.; Broms, C.T.; Nøttestad, L.; Holm, M.; Thomas, K.; et al. Poor feeding opportunities and reduced condition factor for salmon post-smolts in the Northeast Atlantic Ocean. ICES J. Mar. Sci. 2021, 78, 2844–2857. [Google Scholar] [CrossRef]
- Utne, K.R.; Skagseth, Ø.; Wennevik, V.; Broms, C.T.; Melle, W.; Thorstad, E.B. Impacts of a changing ecosystem on the feeding and feeding conditions for Atlantic salmon during the first months at sea. Front. Mar. Sci. 2022, 9, 824614. [Google Scholar] [CrossRef]
- Millane, M.; Kelly, S.; Gallagher, C. Introduction to Pink Salmon in the North Atlantic and Arctic (Why Are They Here and Where?); North Atlantic Salmon Conservation Organization (NASCO): Edinburgh, UK, 2024; 12p. [Google Scholar]
- Fokina, N.; Ruokolainen, T.; Efremov, D.; Murzina, S.A. Changes in the composition of lipids and their fatty acids in pink salmon, Oncorhynchus gorbuscha, during the spawning migration. Environ. Biol. Fishes 2025, 108, 109–130. [Google Scholar] [CrossRef]
- Gallagher, E.J.; Harter, T.S.; Brauner, C.J. The ontogeny of Na+ balance during rapid smoltification in pink salmon (Oncorhynchus gorbuscha). J. Comp. Physiol. B 2021, 191, 17–28. [Google Scholar] [CrossRef] [PubMed]
- Gallagher, Z.S.; Bystriansky, J.S.; Farrell, A.P.; Brauner, C.J. A novel pattern of smoltification in the most anadromous salmonid: Pink salmon (Oncorhynchus gorbuscha). Can. J. Fish. Aquat. Sci. 2013, 70, 349–357. [Google Scholar] [CrossRef]
- Fitzgerald, K.A.; Bellmore, J.R.; Fellman, J.B.; Cheng, M.L.H.; Boyles-Muehleck, N.; Delbecq, C.E.; Falke, J.A. Juvenile coho salmon growth differences track biennial pink salmon spawning patterns. Freshw. Biol. 2024, 69, 1583–1595. [Google Scholar] [CrossRef]
- Heinimaa, S.; Heinimaa, P. Effect of the female size on egg quality and fecundity of the wild Atlantic salmon in the sub-arctic River Teno. Boreal Environ. Res. 2004, 9, 55–62. [Google Scholar]
- Armstrong, J.B. Comment on “Egg consumption in mature Pacific salmon (Oncorhynchus spp.)” Appears in Can. J. Fish. Aquat. Sci. 66(9): 1546–1553. Can. J. Fish. Aquat. Sci. 2010, 67, 2052–2054. [Google Scholar] [CrossRef]
- Meier, G.M.; Meyer, E.I.; Meyns, S. Lipid content of stream macroinvertebrates. Arch. Hydrobiol. 2000, 147, 447–463. [Google Scholar] [CrossRef]
- Dvorak, M.; Dittmann, I.L.; Pedrini-Martha, V.; Hamerlik, L.; Bitusik, P.; Stuchlik, E.; Vondrák, D.; Füreder, L.; Lackner, R. Energy status of chironomid larvae (Diptera: Chironomidae) from high alpine rivers (Tyrol, Austria). Comp. Biochem. Phys. A 2023, 284, 111477. [Google Scholar] [CrossRef] [PubMed]
- Murzina, S.A.; Nefedova, Z.A.; Pekkoeva, S.N.; Veselov, A.E.; Efremov, D.A.; Ruokolainen, T.R.; Nemova, N.N. Dynamics of lipid and fatty acid content at early ontogenesis stages in pink salmon Oncorhynchus gorbuscha (Walbaum, 1792) in a natural environment (Indera River, Kola Peninsula). Russ. J. Dev. Biol. 2019, 50, 180–188. [Google Scholar] [CrossRef]
- Bledsoe, G.E.; Bledsoe, C.D.; Rasco, B. Caviars and fish roe products. Crit. Rev. Food Sci. Nutr. 2003, 43, 317–356. [Google Scholar] [CrossRef]
- Nomura, T.; Urawa, S.; Ueno, Y. Variations in muscle lipid content of high-seas chum and pink salmon in winter. N. Pac. Anadromous Fish Comm. Bull. 2000, 2, 347–352. Available online: https://cir.nii.ac.jp/crid/1570009750225900288 (accessed on 12 June 2025).
- Vuorinen, P.J.; Paasivirta, J.; Koistinen, J.; Rantio, T. Organochlorines in salmon (Salmo salar L.) from the Teno River. In Proceedings of the Symposium on the State of the Environment and Environmental Monitoring in Northern Fennoscandia and the Kola Peninsula, Rovaniemi, Finland, 6–8 October 1992; Tikkanen, E., Varmola, M., Katermaa, T., Eds.; Arktisen Keskuksen Julkaisuja 4; Arktinen Keskus: Rovaniemi, Finland, 1992; pp. 186–188. [Google Scholar]
- Kaivarainen, E.I.; Rendakov, N.L.; Khurtina, S.N.; Manoylova, D.I.; Efremov, D.A.; Murzina, S.A. Changes in Na+/K+-ATPase activity and biomembrane lipid composition in smolts of pink salmon Oncorhynchus gorbuscha (Salmonidae) during their downstream migration in the Indera River (White Sea Basin). J. Ichthyol. 2025, 65, 724–736. [Google Scholar] [CrossRef]
- Jobling, M.; Larsen, A.V.; Andreassen, B.; Sigholt, T.; Olsen, R.L. Influence of a dietary shift on temporal changes in fat deposition and fatty acid composition of Atlantic salmon post-smolt during the early phase of seawater rearing. Aquac. Res. 2002, 33, 875–889. [Google Scholar] [CrossRef]
- Cornet, V.; Wanderscheid, A.; Carpentier, C.; Erraud, A.; Larondelle, Y.; Lefrançois, C.; Kestemont, P. Natural lipid profile mimicking diets to enhance survival and fitness of restocked salmon parr in river ecosystems? Aquaculture 2026, 612, 743259. [Google Scholar] [CrossRef]
- Bell, J.G.; Ghioni, C.; Sargent, J.R. Fatty acid compositions of 10 freshwater invertebrates which are natural food organisms of Atlantic salmon parr (Salmo salar): A comparison with commercial diets. Aquaculture 1994, 128, 301–313. [Google Scholar] [CrossRef]
- Chen, J.; Liu, H. Nutritional indices for assessing fatty acids: A mini-review. Int. J. Mol. Sci. 2020, 21, 5695. [Google Scholar] [CrossRef]
- Honeyfield, D.C.; Peters, A.K.; Jones, M.L. Thiamine and lipid utilization in fasting Chinook salmon. In North Pacific Anadromous Fish Commission Bulletin 6; North Pacific Anadromous Fish Commission: Vancouver, BC, Canada, 2016; pp. 13–19. [Google Scholar] [CrossRef]
- Todisco, V.; Fridolfsson, E.; Axén, C.; Dahlgren, E.; Ejsmond, M.J.; Hauber, M.M.; Hindar, K.; Tibblin, P.; Zöttl, M.; Söderberg, L.; et al. Thiamin dynamics during the adult life cycle of Atlantic salmon (Salmo salar). J. Fish Biol. 2024, 104, 807–824. [Google Scholar] [CrossRef]
- Murzina, S.A.; Nefedova, Z.A.; Pekkoeva, S.N.; Veselov, A.E.; Efremov, D.A.; Nemova, N.N. Age-specific lipid and fatty acid profiles of Atlantic salmon juveniles in the Varzuga River. Int. J. Mol. Sci. 2016, 17, 1050. [Google Scholar] [CrossRef]
- Jobling, M.; Andreassen, B.; Larsen, A.V.; Olsen, R.L. Fat dynamics of Atlantic salmon Salmo salar L. smolt during early seawater growth. Aquac. Res. 2002, 33, 739–745. [Google Scholar] [CrossRef]
- Bell, J.G.; Tocher, D.R.; Farndale, B.M.; Cox, D.I.; McKinney, R.W.; Sargent, J.R. The effect of dietary lipid on polyunsaturated fatty acid metabolism in Atlantic salmon (Salmo salar) undergoing parr-smolt transformation. Lipids 1997, 32, 515–525. [Google Scholar] [CrossRef] [PubMed]
- Honda, K.; Hasegawa, K.; Ban, M.; Yano, Y.; Ogura, Y. Massive stocking of chum salmon (Oncorhynchus keta) fry fattens non-native brown trout (Salmo trutta) in Hokkaido, Japan. PLoS ONE 2024, 19, e0307552. [Google Scholar] [CrossRef] [PubMed]
- Nomura, T.; Carlson, H.R.; Urawa, S.; Mayama, H.; Fukuwaka, M.; Ueno, Y.; Ishida, Y. Variations in Lipid Content of High-Seas Chum and Pink Salmon; National Salmon Resources Center, Fisheries Agency of Japan: Sapporo, Japan, 1999; 9p. [Google Scholar]
- Ota, T.; Yamada, M. Lipids of masu salmon—II. Seasonal variations in the lipids of masu salmon parr during the life in fresh-water. Nippon Suisan Gakkaishi 1974, 40, 699–706. [Google Scholar] [CrossRef]
- Vuorinen, P.J.; Paasivirta, J.; Piilola, T.; Surma-Aho, K.; Tarhanen, J. Organochlorine compounds in Baltic salmon and trout. I. Chlorinated hydrocarbons and chlorophenols 1982. Chemosphere 1985, 14, 1729–1740. [Google Scholar] [CrossRef]
- Jobling, M.; Larsen, A.V.; Andreassen, B.; Olsen, R.L. Adiposity and growth of post-smolt Atlantic salmon Salmo salar L. Aquac. Res. 2002, 33, 533–541. [Google Scholar] [CrossRef]
- Kattner, G.; Hagen, W. Lipids in marine copepods: Latitudinal characteristics and perspective to global warming. In Lipids in Aquatic Ecosystems; Arts, M.T., Brett, M.T., Kainz, M.J., Eds.; Springer: New York, NY, USA, 2009; pp. 257–280. [Google Scholar]
- Thorne, R.E.; Thomas, G.L. Acoustic monitoring of the juvenile pink salmon food supply and predators in Prince William Sound, Alaska. In Proceedings of the OCEANS 2007, Aberdeen, UK, 29 September–4 October 2007; pp. 1–7. [Google Scholar]
- Auel, H.; Harjes, M.; da Rocha, R.; Stübing, D.; Hagen, W. Lipid biomarkers indicate different ecological niches and trophic relationships of the Arctic hyperiid amphipods Themisto abyssorum and T. libellula. Polar Biol. 2002, 25, 374–383. [Google Scholar] [CrossRef]
- Lee, R.F.; Hagen, W.; Kattner, G. Lipid storage in marine zooplankton. Mar. Ecol. Prog. Ser. 2006, 307, 273–306. [Google Scholar] [CrossRef]
- Hyvönen, L.; Koivistoinen, P. Fatty acid analysis, TAG equivalents as net fat value, and nutritional attributes of fish and fish products. J. Food Compos. Anal. 1994, 7, 44–58. [Google Scholar] [CrossRef]
- Khurtina, S.N.; Voronin, V.P.; Manoilova, D.I.; Efremov, D.A.; Nemova, N.N.; Murzina, S.A. Fatty Acid Composition of Juvenile Pink Salmon Oncorhynchus gorbuscha during Downstream Migration from the River Indera to the White Sea. Dokl. Biol. Sci. 2025. [Google Scholar] [CrossRef] [PubMed]
- Jacobson, P. Size-Dependent Predator-Prey Interactions, Distribution and Mortality in Salmon: Effects on Individuals and Populations. Ph.D. Thesis, Swedish University of Agricultural Sciences, SLU, Öregrund, Sweden, 2020. [Google Scholar]
- Jacobson, P.; Gårdmark, A.; Östergren, J.; Casini, M.; Huss, M. Size-dependent prey availability affects diet and performance of predatory fish at sea: A case study of Atlantic salmon. Ecosphere 2018, 9, e02081. [Google Scholar] [CrossRef]
- Hellenbrecht, L.M.; Utne, K.R.; Karlsen, Ø.; Glover, K.A.; Wennevik, V. Diet analysis of Atlantic salmon (Salmo salar) post-smolts after the ecological regime shift in the Northeast Atlantic. Fish. Res. 2023, 262, 106672. [Google Scholar] [CrossRef]
- Budge, S.M.; Penney, S.N.; Lall, S.P. Response of tissue lipids to diet variation in Atlantic salmon (Salmo salar): Implications for estimating diets with fatty acid analysis. J. Exp. Mar. Biol. Ecol. 2011, 409, 267–274. [Google Scholar] [CrossRef]
- Thurow, F. On Food, Behaviour and Population Mechanism of Baltic Salmon; Report 4; Swedish Salmon Research Institute: Älvkarleby, Sweden, 1968; pp. 1–16. [Google Scholar]
- Dessen, J.E.; Weihe, R.N.; Hatlen, B.; Thomassen, M.S.; Rorvik, K.A. Different growth performance, lipid deposition, and nutrient utilization in in-season (S1) Atlantic salmon post-smolt fed isoenergetic diets differing in protein-to-lipid ratio. Aquaculture 2017, 473, 345–354. [Google Scholar] [CrossRef]
- Gélineau, A.; Corraze, G.; Boujard, T.; Larroquet, L.; Kaushik, S. Relation between dietary lipid level and voluntary feed intake, growth, nutrient gain, lipid deposition and hepatic lipogenesis in rainbow trout. Reprod. Nutr. Dev. 2001, 41, 487–503. [Google Scholar] [CrossRef]
- Power, M.; Thorstad, E.B.; Forseth, T.; Fiske, P. Temporal shifts in the marine feeding of individual Atlantic salmon inferred from scale isotope ratios. Ecol. Evol. 2023, 13, e10656. [Google Scholar] [CrossRef]
- Crozier, L.G.; Siegel, J.E. A comprehensive review of the impacts of climate change on salmon: Strengths and weaknesses of the Literature by life stage. Fishes 2023, 8, 319. [Google Scholar] [CrossRef]
- Salminen, M.; Erkamo, E.; Salmi, J. Diet of post-smolt and one-sea-winter Atlantic salmon in the Bothnian Sea, northern Baltic. J. Fish Biol. 2001, 58, 16–35. [Google Scholar] [CrossRef]
- Paspatis, M.; Boujard, T. A comparative study of automatic feeding and self-feeding in juvenile Atlantic salmon (Salmo salar) fed diets of different energy levels. Aquaculture 1996, 145, 245–257. [Google Scholar] [CrossRef]
- Anonymous. Status of the Tana/Teno River Salmon Populations in 2019; Report from the Tana Monitoring and Research Group nr 1/2019; Tana Monitoring and Research Group: Tromsö, Norway; Trondheim, Norway; Oulu, Finland, 2019; 98p, Available online: https://jukuri.luke.fi/handle/11111/12017 (accessed on 11 August 2025).
- Johnson, A.F.; Gorelli, G.; Jenkins, S.R.; Hiddink, J.G.; Hinz, H. Effects of bottom trawling on fish foraging and feeding. Proc. R. Soc. B 2015, 282, 20142336. [Google Scholar] [CrossRef] [PubMed]

 ; yolk-sac fry
; yolk-sac fry  ; alevin
; alevin  ; parr
; parr  ; smolt
; smolt  ; post-smolt
; post-smolt  . The parr phase of Atlantic salmon, which lasts for several years in the river, is absent in pink salmon. The feeding migration of Atlantic salmon from post-smolt to adult in the sea can take several years, but the feeding migration of pink salmon from post-smolt to adult takes a little over a year. The total life cycle of a salmon is 4–10 years and that of pink salmon is 2 years. (Photos: Mikko Kytökorpi, Ari Savikko, Ville Vähä, and Pekka J. Vuorinen).
. The parr phase of Atlantic salmon, which lasts for several years in the river, is absent in pink salmon. The feeding migration of Atlantic salmon from post-smolt to adult in the sea can take several years, but the feeding migration of pink salmon from post-smolt to adult takes a little over a year. The total life cycle of a salmon is 4–10 years and that of pink salmon is 2 years. (Photos: Mikko Kytökorpi, Ari Savikko, Ville Vähä, and Pekka J. Vuorinen).
 ; yolk-sac fry
; yolk-sac fry  ; alevin
; alevin  ; parr
; parr  ; smolt
; smolt  ; post-smolt
; post-smolt  . The parr phase of Atlantic salmon, which lasts for several years in the river, is absent in pink salmon. The feeding migration of Atlantic salmon from post-smolt to adult in the sea can take several years, but the feeding migration of pink salmon from post-smolt to adult takes a little over a year. The total life cycle of a salmon is 4–10 years and that of pink salmon is 2 years. (Photos: Mikko Kytökorpi, Ari Savikko, Ville Vähä, and Pekka J. Vuorinen).
. The parr phase of Atlantic salmon, which lasts for several years in the river, is absent in pink salmon. The feeding migration of Atlantic salmon from post-smolt to adult in the sea can take several years, but the feeding migration of pink salmon from post-smolt to adult takes a little over a year. The total life cycle of a salmon is 4–10 years and that of pink salmon is 2 years. (Photos: Mikko Kytökorpi, Ari Savikko, Ville Vähä, and Pekka J. Vuorinen).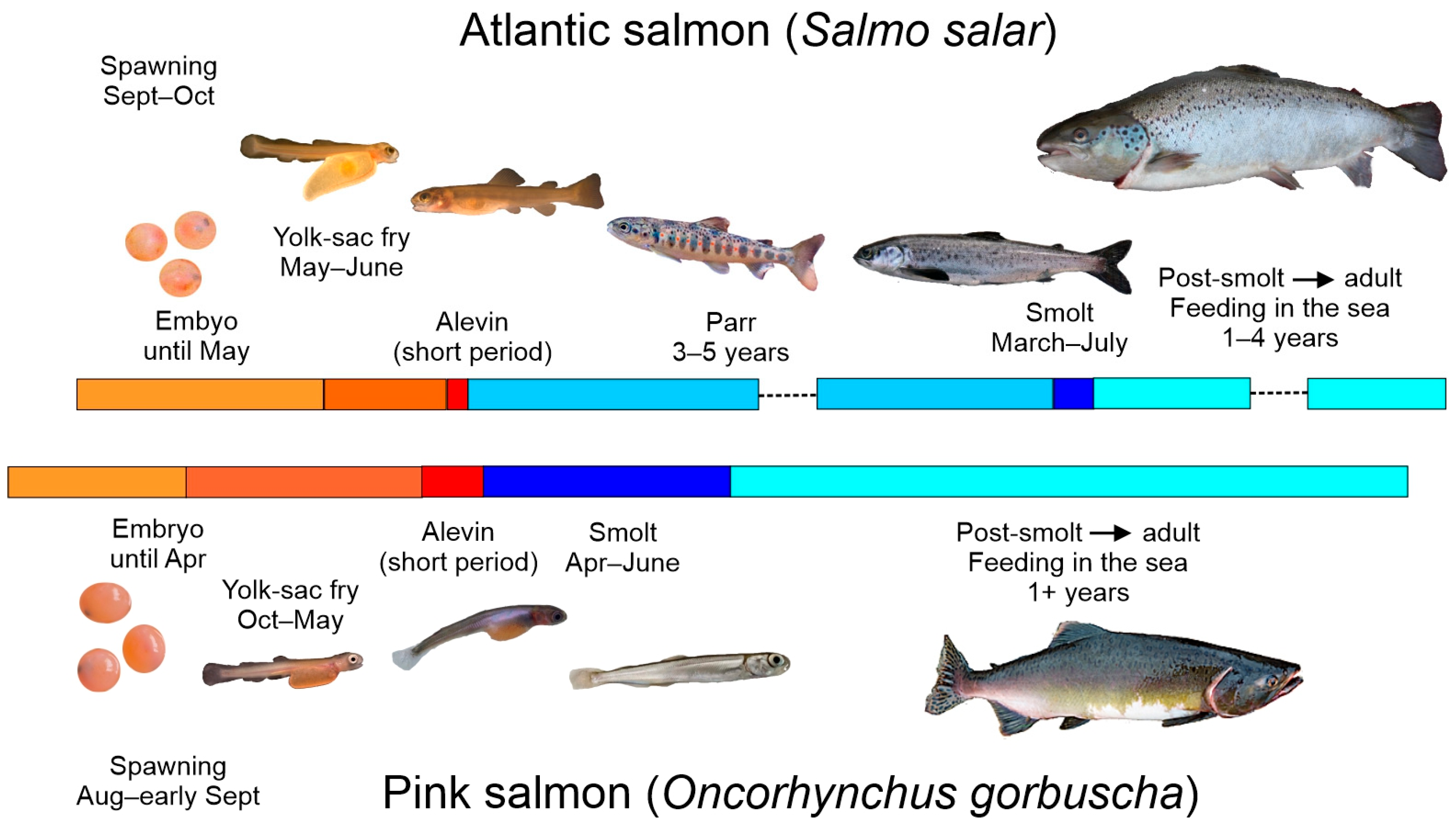












Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Keinänen, M.; Vuorinen, P.J. How May the Increase in Pink Salmon (Oncorhynchus gorbucsha) Cause the Populations of the Atlantic Salmon (Salmo salar) to Decline? Fishes 2026, 11, 17. https://doi.org/10.3390/fishes11010017
Keinänen M, Vuorinen PJ. How May the Increase in Pink Salmon (Oncorhynchus gorbucsha) Cause the Populations of the Atlantic Salmon (Salmo salar) to Decline? Fishes. 2026; 11(1):17. https://doi.org/10.3390/fishes11010017
Chicago/Turabian StyleKeinänen, Marja, and Pekka J. Vuorinen. 2026. "How May the Increase in Pink Salmon (Oncorhynchus gorbucsha) Cause the Populations of the Atlantic Salmon (Salmo salar) to Decline?" Fishes 11, no. 1: 17. https://doi.org/10.3390/fishes11010017
APA StyleKeinänen, M., & Vuorinen, P. J. (2026). How May the Increase in Pink Salmon (Oncorhynchus gorbucsha) Cause the Populations of the Atlantic Salmon (Salmo salar) to Decline? Fishes, 11(1), 17. https://doi.org/10.3390/fishes11010017







