1. Introduction
The conceptualization of the human body as a complex machine is not new among philosophers and scientists. Descartes, Hobbes, and La Mettrie, for example, are commonly associated with the “mechanical philosophy” of the 17th and 18th centuries [
1,
2,
3]. But it was not until the first half of the 20th century that the mechanical conception of the human body took a different turn: If the human body is a complex machine, maybe the machine could be re-built and improved. This idea emerged in the aftermath of the World War I, not as a matter of philosophical inquiry, but as a matter of necessity and as a problem for policymakers.
As thousands of soldiers returned from the front with missing limbs, engineers and physicians had to work together in order to devise prosthetics limbs that might enable veterans to get back to work. A booklet published by the Red Cross in 1918, for instance, was entitled
Reconstructing the Crippled Soldier [
4]. The booklet contains several pictures of men using prosthetic limbs that functioned as tools such as pliers and hammers. The governments, especially in Germany, were eager to convince the new working force that the “scientific prostheses” [
5]—as they were known—would make the disabled men even better and more productive than before [
6,
7,
8]. Not everyone was convinced, though. In a short essay called “The Prosthetic Economy,” published in 1920, the Austrian artist Raoul Hausmann sarcastically suggested that since the “prosthetic limb never gets tired,” a “25-hour workday” should be adopted. He also went on to say: “The prosthetic man is therefore a better man, raised to a superior class thanks to the world war” [
9]. The philosophical discussion on the ethics of human enhancement, as a topic for academic research, is rather recent, but the ethical concerns over the attempt to re-engineer the human body for the purpose of enhancement is not. It harks back to a broad societal debate that took place during the interwar years [
6,
7,
8,
10].
Now, 100 years later, policymakers, again, have to deal with technologies that have the potential to improve the human body. The conceptualization of the human body as a machine remains in place, but this time the machine is seen in a different light. As the human brain (wrongly or not) is increasingly compared to a supercomputer, and the whole human genome has been sequenced and represented as a six-billion string of letters (A, T, C, and G), the mechanical conception defended in the past has given rise to a new image of the human body, to a vision of human beings not so much as composed of mechanical parts, but as a machine that can be understood at a molecular level, as a source-code that can be read and re-written by means of gene-editing tools. In this article, I intend to focus on the more recent development: I will focus on genetic engineering, rather than on prosthetic limbs, as a means to enhance the human body.
The philosophical debate on the ethics of human enhancement involves a distinction between two basic ideas, which have not always been carefully kept apart. One concerns the means of enhancement, the other concerns the human capacity to be enhanced. These are, for short, the “enhancement by which means question” and the “enhancement of what question.” One can defend a pro-enhancement attitude as regards, for instance, the use of drugs such as modafinil to boost one’s cognitive functions, but argue against the use of drugs for the purpose of enhancing one’s physical capacities in professional sports. One can also have a pro-enhancement stance as regards the use of drugs to improve cognitive and physical capacities, but argue that human moral capacities should not be modulated by means of drugs. Or one can alternatively advance a broader pro-enhancement attitude as regards the use of drugs for improving one’s cognitive, physical, and moral capacities, but still strongly oppose the use of genetic engineering to promote the enhancement of any human capacities. It is possible to think of some other combinations between the means of enhancement, such as prosthetic limbs, deep brain stimulation, or surgeries, and human capacities such as creativity, the propensity to form long-lasting affective bonds with other persons, or aesthetic appearance. Thus, compelling arguments for the use of one specific method of enhancement, targeted at one or more specific human capacities, may not be equally compelling as regards other means of enhancement, targeted at other human capacities. The attempt to advance an all-encompassing approach to the ethics of human enhancement, on the other hand, can obscure important issues or conflate problems that should be dealt with separately. In this article, I intend to examine the use of genetic engineering as a means of pursuing cognitive enhancement. “Genetic engineering” itself is too broad a concept, for it can refer to technologies that, as I intend to show later, give rise to different ethical questions. I will focus here on two such technologies, namely gene editing (henceforth GE) and embryo selection (henceforth ES).
In addition to the “enhancement by which means question” and the “enhancement of what question,” two further questions must be addressed in the human enhancement debate. One is the “enhancement for whom question.” This is especially important in the case of genetic engineering as a means of cognitive enhancement, for the persons who are going to be affected by the enhancement procedure do not yet exist. This means that they cannot give their consent to the use of techniques that will affect not only their personal identities as bearers of certain human capacities, but will ultimately also determine whether or not they are going to come into existence. This, as I intend to show later, raises ethical questions that do not emerge when we deploy other methods of human enhancement such as, for instance, drugs or prosthetic limbs.
Finally, a perennial question in the ethics of human enhancement debate concerns our very understanding of what human enhancement is about:
What is human enhancement and how does it differ from
treatment? In this article, I will not try to draw a clear-cut line between enhancement and treatment, which could be applied to all cases involving the quest for human enhancement. Neither shall I attempt to advance a definition of human enhancement that covers every case that has been described in the philosophical literature as instances of human enhancement [
11].
1 For reasons I am going to spell out later, I suggest we should rather speak of “family resemblance,” as Wittgenstein used the expression in the
Philosophical Investigations, instead of proposing a very comprehensive definition of human enhancement [
12].
I will speak here of “genetic cognitive enhancement” (GCE) to refer to GE and ES as procedures that, in the future, might enable prospective parents to increase the odds of having a child with increased intelligence. To the extent that one has increased intelligence, one can on average perform better in a wide range of cognitive domains such as, for instance, information processing speed, working memory, executive function, episodic memory, sustained attention, selective attention, etc. [
13] (pp. 11–12), [
14] (p. 279), [
15] (pp. 53–54). One can of course have better cognitive performance without having increased intelligence in the sense I will focus on in this article. One can, for example, use some artificial intelligence device in order to improve information processing speed. One can even suggest that the device itself is an extension of the human mind, whether or not it is internal to the human body [
16], (for Reference [
17] see pp. 29–44). The device, however, cannot be genetically inherited. Although different kinds of technologies can be deployed to improve cognitive performance, I will focus here only on GCE.
The question, then, is whether pursuing GCE, whether through GE or ES, is a morally acceptable goal. Different moral theories and philosophical traditions, and even theological ideas, have been deployed in the philosophical debate on the ethics of human enhancement. On the other hand, far less attention has been given to public attitudes related to the development of human enhancement technologies. Some recent surveys on the public perception of human enhancement technologies and human genomics either focus too narrowly on a single country (United States) or on a single region (Europe), or they focus instead too narrowly on public perception of one specific technology (CRISPR, Clustered Regularly Interspaced Short Palindromic Repeats), and not on the public perception of human enhancement technologies at large [
18,
19,
20,
21]. In this article, I intend to focus on two recent surveys conducted by the SIENNA Project, of which the author of this article is a partner.
2 The SIENNA Project has advanced probably the most comprehensive account of public attitudes towards human genomics and human enhancement to date. The SIENNA Project has also delivered a corresponding account of public perception of artificial intelligence and robotics, which I will not take into consideration in this article. The surveys were conducted in 11 countries, spanning across different economic, cultural, and geographical landscapes (France, Germany, Greece, the Netherlands, Poland, Spain, Sweden, Brazil, South Africa, South Korea, and United States). The researchers employed the CATI method (computer assisted telephone interviewing) to interview one thousand respondents in each country [
22,
23].
2. Therapeutic and Non-Therapeutic Uses of Assisted Reproductive Technologies (ART)
Assisted reproductive technologies (henceforth ART) have been developing at a rapid pace over the last decades. These technologies include, for instance:
Contraceptive pills
IVF (in vitro fertilization)
PGD (preimplantation genetic diagnosis)
Egg-freezing (oocyte cryopreservation)
CRISPR (clustered regularly interspaced short palindromic repeats)
In vitro gametogenesis (IVG)
Time-lapse analysis of embryos
Genome-wide association studies (GWAS)
Artificial intelligence (AI)
Some of these technologies (CRISPR, GWAS, and AI) do not qualify as ART in their own right, but in combination with other technologies they constitute some of the most recent developments in the field of ART. Contraceptive pills (or other science-based contraceptive methods) qualify as ART to the extent that they provide a negative control over human reproduction. I call it “negative” because, in this case, one’s primary intention is to avoid a pregnancy. One needs the technology, then, to avert the occurrence of a process that would otherwise naturally occur—or might naturally occur. Some technologies, on the other hand, provide a “positive” control over human reproduction. One needs the technology, then, to stimulate the occurrence of a process that, for some reason or other, would not otherwise naturally occur.
The positive control of human reproduction can be pursued for both therapeutic and non-therapeutic goals. The best known technology for the positive control of human reproduction is IVF. Human embryos generated by means of IVF do not have, though, equal chances of adhering to the uterus, or equal chances of developing into a healthy child. For this reason, the number of embryos generated in each IVF cycle is usually greater than the number of embryos eventually transferred into the uterus. Further development in the field of ART enabled the creation of the PGD, which is usually undertaken by collecting a few cells from a two- to five-day old embryo (blastocyt) for genetic analysis [
24] (p. 34). PGD enable clinicians to screen embryos for health issues such as, for instance, Huntington disease, Marfan Syndrome, cystic fibrosis, and other known genetic disorders, before they can be transferred into the uterus. A new technique called time-lapse analysis has been recently employed to carry out a non-invasive assessment of patterns of development in embryos images in order to help clinicians to decide which embryos have the best prospect for transfer into the uterus [
25,
26,
27]. And more recently still, time-lapse analysis has also been performed in combination with the use of artificial intelligence in order to further improve the assessment of embryo viability [
28]. This procedure enhances the clinicians’ ability to select an embryo that has more chances of adhering to the womb and developing into a healthy child.
Although most ART have been originally developed for medical reasons, it is clear by now that ART can be used for non-therapeutic purposes as well. Yet, in in some cases, a clear-cut distinction between “therapeutic” and “non-therapeutic” uses cannot be easily drawn. This is one of the reasons I will not propose a clear-cut criterion to distinguish enhancement from treatment in this paper. Contraceptive pills, for example, are a pharmaceutical product, but we do not ordinarily think of contraceptive pills as a sort of “medicine,” for they are not usually meant to treat any disease. The IVF and PGD, on the other hand, can indeed be used to address human infertility, which the World Health Organization (WHO) understands to be “a disease of the reproductive system defined by the failure to achieve a clinical pregnancy after 12 months or more of regular unprotected sexual intercourse” [
29] (p. 1522). But when IVF and PGD are deployed to detect genetic disorders in human embryos, it is not entirely clear whether these technologies are being used for therapeutic purpose or as a device for selective contraception. If some genetic disorder is detected, clinicians and prospective parents have to decide whether or not to transfer the embryo into the uterus [
30]. Treating the embryo, though, is not an option, at least not on legally and ethically acceptable grounds, as I am going to explain later. Sometimes, on the other hand, a distinction between “therapeutic” and “non-therapeutic” is more apparent: “Social egg-freezing” and GCE are two such cases. In the remainder of this section, I would like to describe briefly the former, before examining the latter more carefully in the next sections.
A woman may decide to resort to egg-freezing (or oocyte cryopreservation) in order to have some of her egg cells retrieved and frozen during the early years of her adult life. This enables her, for instance, to focus on her professional career while she is young, or while she waits until she has met the partner with whom she wants to have a child. Later, after she has secured the material means to give her child the best start in life, or met a partner she considers suitable to her, one or more egg cells, which had been previously frozen and stored in a fertility clinic, may be fertilized. For this, the woman does not even have to have a male partner. She can simply order a vial of semen from a private sperm bank and have it delivered to a fertility clinic. By the time the egg cells are fertilized, the woman may be less fertile than she was earlier, when the egg cells were retrieved from her ovaries. But this does not mean that she has become ill later, as fertility naturally decreases with age. The procedure, thus, is not used to treat an illness. It could be described, rather, as a technology for the enhancement of human reproduction, even though fertility is not a human capacity usually taken into consideration in the human enhancement debate. In the current literature on oocyte cryopreservation, it has become usual to distinguish “social egg freezing” from “medical egg freezing.” The latter is deployed for medical reasons, when a woman has to undergo, for instance, cancer treatment, which may compromise her fertility after the treatment. Social egg freezing, on the other hand, is undertaken for non-therapeutic reasons [
31,
32,
33,
34,
35].
Now, further development of new technologies such as IVG and CRISPR has the potential to give rise to other non-therapeutic applications of ART, including human enhancement. IVG has never been used for the generation of human gametes intended to start a pregnancy. CRISPR, on the other hand, has been reportedly used for the purpose of human reproduction only once, in 2018 in China, under circumstances that the Chinese government considered illegal, and the international community recognized as ethically unacceptable. The question, then, is whether further development of IVG and CRISPR might give rise to circumstances in which their use as ART, whether for therapeutic or non-therapeutic goals, might be considered ethically acceptable.
3. IVG and CRISPR as a Means of GCE
Embryonic cells, in their early stages of development, can turn into any other types of cells. These cells are also known as stem cells. During the embryonic development, stem cells become specialized cells, such as nerve cells, heart cells, neurons, bone cells, and several other types of cells that make up the human body. Specialized cells are more commonly called somatic cells. Normally, somatic cells can only reproduce and become another somatic cell of the same type, but new technologies now allow researchers to turn somatic cells back into stem cells. Somatic cells that have been induced to behave like stem cells are called “induced pluripotent stem cells” (iPSCs). Because iPSCs can turn into virtually any other type of cell, they can also become reproductive cells or gametes. The generation of reproductive cells from iPSCs is known as in vitro gametogenesis (IVG), or sometimes also as “in vitro generated gametes,” “artificial gametes,” or “synthetic gametes” [
36,
37,
38,
39,
40,
41,
42,
43], (for Reference [
44] see p. 4). IVG was carried out for the first time in Japan, in 2016, on mice. Researchers managed to generate several eggs cells from a small sample of mouse skin cells. The eggs cells were then fertilized in vitro, and the resulting embryos were used to start a pregnancy. The mice produced by the Japanese researchers were healthy and fertile [
45]. Now, further research on IVG might perhaps enable clinicians to use this technology for the generation of human gametes in the future. The range of therapeutic and non-therapeutic applications would be enormous. But then, too, the number and scope of ethical concerns.
A man, for instance, could request the use of IVG to turn a tiny sample of his skin cells into egg cells and, thus, become a mother. In like manner, a woman could use IVG for the generation of sperm cells and, thus, become a father. Two men, or two women, could also have a child genetically related to them. Prospective parents who currently rely on IVF to start a pregnancy would also benefit from IVG. Instead of undergoing a series of hormone injections and a surgical procedure to obtain the eggs cells, the woman would only have to provide a small sample of her skin cells. This would make the entire IVF procedure far less invasive—and possibly less expensive—for the women [
46] (p. 127).
IVG could also disrupt the market for human gametes. Some women who currently undergo the egg retrieval procedure do not intend to become mothers themselves. Their intention is, rather, to donate or sell some of their egg cells, which in turn can be used for research or for human reproduction. This practice, however, has raised some ethical concern relative to informed consent and exploitation of women [
47], (for Reference [
48] see p. 44). Men, too, can donate sperm cells for altruistic or pecuniary reasons [
49,
50,
51,
52], but prohibition of anonymous donation over the last years has led to a shortage of sperm cells in many sperm banks around the world. Recently, some bioethicists have suggested that the institutionalization of presumed consent for post-mortem sperm cells donation might be introduced as a solution for the problem relative to the current shortage of male gametes in sperm banks [
53]. IVG of human cells, on the other hand, could reduce drastically the demand for donation of male and female reproductive cells, making access to human gametes much easier and cheaper for prospective parents and researchers.
Many sperm banks offer some information on the educational background or academic achievements of their individual donors. Prospective parents may use the information, then, as a rough predictor of the cognitive capacities of their offspring. But IVG, in conjunction with IVF and other technologies such as GWAS and AI, could provide prospective parents with a more reliable and effective method for assessing their chances of having a child with increased intelligence. The problem, though, is that the quest for GCE by means of IVG, in conjunction with IVG and GWAS, would require the generation of far more embryos than the amount of embryos that are currently generated in fertility clinics during every IVF cycle. The idea here is that the more embryos from the same parents one has, the more chances one also has of spotting one particular embryo with more odds of developing into a child with increased intelligence [
39,
40,
46,
54] (pp. 191–202), [
55] (pp. 88, 93, 106–110).
Currently, PGD enables clinicians to screen embryos for some genetic diseases before implantation into the womb. Unviable embryos are discarded or, depending on local legislation, used for scientific research. But as genetic sequencing technologies becomes faster and cheaper, clinicians might be able to screen vast amounts of embryos from the same couple for both medical and non-medical issues. And as human intelligence itself is highly inheritable [
56,
57,
58], clinicians could screen the embryos for this specific trait as well. There is not, though, one human gene for intelligence. It is believed that hundreds or thousands of different genes, scattered at different loci of the human genome, are involved in the expression of most psychological, behavioral, and physical human traits. It does not mean, of course, that genes alone determine all features one individual will ultimately have, for the expression of human traits very often depends on diverse degrees of interaction between genome and environment.
GWAS enable researchers to determine which regions of the human genome are associated with the expression of specific psychological, behavioral, or physical traits. This association can be measured and translated in terms of “polygenic scores,” which is a probabilistic—thus not deterministic—estimation of the occurrence of a certain trait as the result of the influence of a large group of genes, [
46] (p. 77). GWAS, thus, could help prospective parents and clinicians to select one embryo, amongst hundreds or thousands of embryos, with increased odds of developing into a child with higher intelligence. On the other hand, as Henry Greely points out, the vast amount of genetic data resulting from the assessment of polygenic scores could also lead prospective parents to make decisions that, in the end, might compromise the well-being of their child. How to decide, for instance, between Embryo
1 and Embryo
2 when Embryo
1 has
x% of having trait
1 (IQ > 100),
y% of developing trait
2 (minor propensity to obesity), and
z% of developing trait
3 (propensity to depression), while Embryo
2 presents different polygenic scores for trait
1, trait
2, and trait
3? The decision becomes increasingly more difficult when the number of embryos to choose from is >100 and the number of eligible traits (both medically and non-medically relevant traits) is also very high [
46] (pp. 193–196). In face of this “information overload”—as Sonia Suter aptly puts the problem—prospective parents would probably have to rely on the expertise of genetic counselors and computer algorithms to assess the polygenic scores their future offspring and adjust their preference for a specific trait like increased intelligence against medically relevant traits [
46] (pp. 193–196), [
54] (pp. 273–276). Once a decision has been made in favor of a specific embryo, the remaining embryos would have to be discarded.
Currently, depending on local legislation, unclaimed viable embryos can be “adopted.” In 2017, for example, a woman in the United States gave birth to a baby that developed from an embryo that had been frozen for 24 years [
59]. The mother was just over one year old when the embryo was generated. In adopting the unclaimed embryo, the woman became the gestational mother, and not the biological mother of the baby. Frozen embryos may remain unclaimed for several reasons: The couple may have divorced after the generation of embryos and, thus, they may have changed their minds as to become father or mother; or the father or mother (or both) may have died after the generation of the embryo; or the couple may have simply decided, after a successful gestation, not to use the remaining embryos for the generation of yet another child. Yet, even if many women would volunteer to adopt surplus embryos, a significant amount of embryos would still have to be discarded.
GCE by means of GE, on the other hand, would not require the generation of vast amounts of human embryos. Instead of selecting one among hundreds of embryos, clinicians could generate only one embryo by means of IVF—with or without the use of IVG. Then, the embryo could be edited with CRISPR to increase the chances that it will later develop into a child with increased intelligence. Thus, no embryo would have to be discarded. It must be emphasized, though, that at its current stage of development CRISPR is not considered a reliable kind of ART—neither for therapeutic nor non-therapeutic goals. Moreover, as mentioned earlier, thousands of genes are involved in the expression of human intelligence. Even if CRISPR becomes safe and reliable for therapeutic goals within the next years, it is still unclear whether it could be deployed for the purpose of GCE without the risk of off-target mutations and mosaicism [
60]. Maybe other forms of human enhancement by means of GE will become acceptable before it can be used for cognitive enhancement, especially in areas where a clear-cut line between enhancement and treatment cannot be drawn.
Consider, for instance, the first actual use of CRISPR as a form of ART, which occurred in November 2018 in China. He Jiankui, a Chinese researcher, edited the genome of two twin girls at an early stage of embryonic development. He Jiankui’s goal was to enable HIV positive men to generate children free of the HIV virus. The usual measure, in these cases, consists in treating the semen with a process known as sperm-wash before proceeding to IVF. This alone should have been enough to enable a man to generate a child uncontaminated by the HIV virus. However, He Jiankui’s goal was not simply to guarantee the birth of an HIV-free child. His intention was to generate a child
immune to the HIV virus. It is clear, then, that the procedure, in this case, was deployed for non-therapeutic reasons. It aimed at the genetic enhancement of two human beings. The whole procedure might be called “immune system genetic enhancement.” Incidentally, some authors have speculated that the procedure might have also indirectly provided the twin girls with some degree of GCE [
61,
62].
The international scientific community vehemently repudiated He Jiankui’s procedure, as it represented a clear violation of the ethical guidelines proposed by many international bodies over the last years [
63,
64,
65]. However, the fact that the procedure was deployed as a means of human enhancement, rather than as a method of treatment for a genetic disorder, was not the main focus of criticism. Many articles, published shortly after He Jiankui announced the birth of the twin girls, did not even take notice of this aspect of his experiment. The main reason for criticism, as I have mentioned above, is the fact that CRISPR cannot yet be considered a safe and reliable kind of ART. It represents a threat not only to the well-being of children whose genome have been edited, but also to the well-being of their descendants.
Now, let us assume, for the sake of argument, that IVG and CRISPR will become available as reliable and safe kinds of ART within the next decades, and that further research on human genomics will provide solid knowledge on the genetics of human intelligence. Would there be good moral reasons, then, to favor one procedure or the other—ES or GE—for the purpose of GCE? Or should we entirely abandon the prospect of pursuing the ES and GE as kinds of ART, whether for therapeutic or non-therapeutic goals? In the next section, I will address these questions by comparing some of the ethical implication each of these technologies entails. Then I will focus on two reports recently published by the SIENNA Project on the public perception of scientific research on human enhancement technologies and human genomics.
4. Public Attitudes towards GCE
How should we assess the ethical implications resulting from the use of technologies such as IVG and CRISPR for the purpose of GCE? Philosophical investigation into the principles of morality often relies on or starts with what we understand by concepts such as duty, virtue, or morality. Consider, for instance, Kant’s statement in the Preface to the Groundwork of the Metaphysic of Morals:
“In this work, I have adopted the method that is, I believe, most fitting if one wants to take one’s route analytically from common cognition [gemeinen Erkenntnisse] to the determination of its supreme principle and in turn synthetically from the examination of this principle and its sources back to common cognition, in which we find it used”
We have to start somewhere, and our common understanding (or “common cognition”) of some elementary moral ideas is a good starting point—indeed, perhaps the only one we have. But Kant also assumes that we all share a “common idea of duty and of moral laws” [
66] (p. 7). The principles of morality, on this analysis, do not have to be agreed upon through a social contract, or through societal debate or similar decisional procedure. The principles have to be, rather, “brought to light” (
aufgeklärt), for they already dwell human beings’ “natural sound understanding” (
natürlichen gesunden Verstande) [
66] (p. 23). More often than not, though,
our common understanding of moral concepts is
common only to the extent that it is shared by some specific group of individuals, living in this or that century, in this or that country, as members of this or that social group. Our current understanding of moral duties and moral laws was certainly not shared, for instance, by Aristotle and other thinkers in the tradition of virtue ethics. And upon examination, we also realize that some ideas at the core of Kant’s moral theory, such as the assumption that “ought implies can,” are not shared by most people [
67,
68]. It might be argued, on the other hand, that Kant was not interested in a psychological account of our common understanding of moral concepts, but in a normative account. If someone, or even a large group of people, happen to lack “natural sound understanding,” or does not recognize what the proper understanding of moral laws amounts to, that person or group of persons should be enlightened. On this account, public attitudes should not be taken into consideration in our analysis of the moral implications resulting from GCE, for moral reasoning itself cannot be enlightened by public opinion.
Philosophical investigation should certainly not pander to public opinion, as public opinion itself can be scrutinized, clarified, and corrected in the light of philosophical analysis, but neither should philosophical investigation underestimate the plurality of points of view at play in the moral debate on issues that matter for the public in general. This is especially important when philosophical expertise is deployed for the elaboration of public policies involving controversial issues, or for the regulation of technologies that may challenge our common understanding about what is ethically acceptable or morally deplorable. As Neil Levy aptly puts the problem:
“Moral expertise is not the exclusive preserve of moral philosophers; instead, it is a domain in which a multitude of thoughtful people outside the academy make important contributions. The arena in which moral debate occurs is, accordingly, not limited to the peer-reviewed journals. Instead moral debate also, and almost certainly more importantly, takes place in newspapers and on television; in novels and films: everywhere moral conflicts are dramatized and explored”
Recall that the first broad societal debate on the ethical implications from the quest for human enhancement by means of new technologies, as I mentioned at the outset of this paper, was mainly driven by stakeholders such as former soldiers, policy-makers, physicians, engineers, and artists rather than by philosophers. The methodological challenge for the assessment of ES and GE as means of GCE consists, then, in our capacity to take into consideration a variety of points of views. From the point of view of a person who was generated by means of new kinds of ART, for instance, it may be important to know which procedure was used before his or her birth: ES or GE? If the embryo has been manipulated with CRISPR, the person has every reason to be grateful or, as the case may be, resentful for any changes that have been made in his or her genome. Suppose that the attempt to promote some cognitive enhancement by means of CRISPR has not been successful and that, for this reason, the person suffers from some minor cognitive disorder such as for instance dyslexia—a condition that makes learning to read more difficult. Let us call that person Person
1. Person
1 could hold her parents or the fertility clinic responsible for her suffering, for the learning disability compromises her chances of success in achieving goals she would like to pursue in life. After all, if the GE procedure had not been used, Person
1 would not have dyslexia. Now, let us suppose there is another person (Person
2) who also suffers from dyslexia and has the same degree of learning difficulty as Person
1. However, Person
2 was not submitted to GE at an embryonic stage of development. Person
2 was generated by means of a combination of IVG and IVF, along with hundreds of sibling embryos, and then selected for implantation into the uterus. All sibling embryos were discarded afterwards. Person
2 may feel as frustrated as Person
1, for the learning disability also compromises her chances of success in achieving goals she would like to pursue. But as some philosophers have noticed, unlike Person
1, Person
2 cannot simply say that some kind of wrongdoing was inflicted on her, or that she has the right to blame her parents or the fertility clinic for being a dyslectic person. For Person
2, the only alternative would be not to have been selected in the first place and, thus, never to have been born. Darek Parfit and Thomas Schwartz are among the first philosophers who drew attention to this kind of paradox [
69,
70,
71]. Schwartz called it the “fallacy of beneficiary-conflation” [
71] (p. 7), but it is better known as the “non-identity-problem,” as Parfit called it early in the 1980s [
70] (p. 359). It is a paradox because a person may apparently present good reasons for wishing that a certain course of actions had never occurred in the past, but without that particular course of actions, which she would prefer had never occurred, she herself would not exist. One might argue, however, that from a legal point of view and as a matter of social justice, we should treat Person
1 and Person
2 the same way: If Person
1 has a right to reparation due to the disorder caused by the GE procedure, then Person
2 should also have the same right, as both cases, at least in the tradition of common law, can be characterized as a tort of wrongful life [
72,
73].
3 A recent survey conducted online among 763 participants also suggests that most people believe that the “non-identity-problem” is not really relevant for an assessment of situations like this, involving the claims of Person
1 and Person
2 [
74].
Alternatively, one might argue that in view of the ethical issues that underlie ES and GE, both procedures should be banned for the purposes of human reproduction. The only non-problematic form of human reproduction—one might suggest—is natural reproduction, that is sexual intercourse of a man and a woman, unaided by ART. However, banning the two procedures does not solve the problem, it just gives rise to a further ethical concern. Consider Person
3, afflicted by dyslexia just as Person
1 and Person
2. Person
3, unlike Person
1 and Person
2, was generated naturally—her conception and birth did not involve the use of ART. Does Person
3 have the right to blame her parents or clinicians for being a person who has dyslexia? The answer to this question will depend on the stage of technological development at the time that Person
3 was conceived, and on the availability of the relevant technologies to her parents. For Person
3, it could be frustrating to learn that her cognitive impairment could have been avoided by means of GE. From the perspective of Person
3, it would have been better if her parents had indeed resorted to GE—even if it had involved some reasonable degree of risk. ES, on the other hand, would not have solved
her problem, as she would not exist if some other embryo, free of dyslexia, had been chosen in her place. The choice for natural reproduction, therefore, is not morally neutral. In circumstances where prospective parents have access to the relevant medical technologies, which allow them to have a child without a condition that, later, may compromise her well-being, the couple is morally accountable both for using the technology and for failing to use it. As Anthony Kenny puts the problem: “As technology increases our knowledge of evils and our power to remove them, it increases our responsibility for not removing them” [
75] (p. 124).
For the time being, though, neither ES (in conjunction with IVG and GWAS) nor GE may be used as a kind of ART—whether for therapeutic or non-therapeutic goals. Leaving aside the question as to which technology will (from a purely technical point of view) mature first, there is some reason to believe that GCE by means of ES might become commercially available prior to GCE by means of GE. This would be a reason, then, to focus the ethical debate on ES rather than on GE.
One reason for the assumption that GCE by means of ES may become commercially available prior to GCE by means of GE is that some fertility clinics, especially in the United States, are now gradually advertising a wider range of medical conditions they can screen for [
76,
77,
78,
79]. Given the thin line between therapeutic and non-therapeutic uses of ART, it may be just a matter of time before intelligence becomes available for screening along with medical conditions such as diabetes, propensity to depression, or intellectual disability, even if the procedure is not overtly advertised as GCE, but rather as therapeutic measure to reduce the odds of having a child with some mental conditions that might compromise his or her well-being later in life. Some people might even argue that ES (whether or not in conjunction with IVG and GWAS) does not qualify as a human enhancement technology at all. Thus, from their perspective, they would be circumventing at least some of the ethical concerns involved in the quest for GCE. Their argument might also be explored by private companies and fertility clinics that cannot advertise their services as genetic enhancement because local legislation explicitly prohibits the pursuit of human enhancement at a genetic level. Whether they argue from genuine philosophical reasoning or from commercial opportunism is not the point. What matters is that they can argue that ES (unlike GE) does not do anything to the embryo. In the normal distribution curve for IQ scores, some people will naturally have IQ > 100 while others will have IQ < 100. GCE by means of ES consists in an attempt to optimize the chances of selecting an embryo that, among other things (e.g., having improved information processing speed, improved working memory, improved executive function, etc.) will develop into a child who will be in the former group. Thus, most definitions of “human enhancement,” discussed in the philosophical literature, will not straightforwardly include ES as a means of human enhancement, for the “enhanced” person, in this case, has not been enhanced at all—i.e., the person herself has not been subjected to an enhancing process. The person, rather, developed from an embryo that had better odds of growing into a person with increased intelligence, quite independently of any modification in the embryo itself. If GCE requires a modification in the embryo, then ES clearly does not qualify as GCE. Yet, there is some reason to believe that GCE does not require embryo modification.
Some questions of social justice will emerge regardless of the procedure one chooses to increase the odds of having a child with increased intelligence. This is the reason why I have stated at the outset of this article that I would eschew the attempt to propose an all-encompassing definition of human enhancement and speak, instead, of “family resemblance.” There are some relevant resemblances between ES and GE that enable us to consider both procedures as human enhancement technologies: (i) Both procedures enable prospective parents to improve the odds of having a child with increased intelligence, even if the child (or the embryo) is not modified when one opts for ES; and (ii) both procedures give rise to the same issues of social justice that require proper regulation from the relevant policy-makers. To see this, let us recall Person1 and Person2.
Person1 was born by means of GE and Person2 was born by means of ES. Let us suppose this time, though, that both procedures (GE and ES) were quite successful, as Person1 and Person2 have, among other things, an IQ of 145. Person1 and Person2 will be able to pursue goals that most people will not be able to pursue, at any rate not as effortlessly as Person1 and Person2. This in turn gives rise to questions of social justice: Is it fair that Person1 and Person2 have these advantages over most people, especially considering that neither Person1 nor Person2 did anything to deserve an IQ of 145? Some influential theories of justice suggest it would not be fair. As John Rawls put the problem in A Theory of Justice:
“The unequal inheritance of wealth is no more inherently unjust than the unequal inheritance of intelligence. It is true that the former is presumably more easily subject to social control; but the essential thing is that as far as possible inequalities founded on either should satisfy the difference principle”
Rawls is not suggesting here, of course, that intelligence itself should be redistributed in line with the difference principle, but that the benefits that people obtain, thanks to their increased intelligence, should also benefit those who have been less fortunate in the natural lottery. After all, they have not deserved their talents—or lack of talents, as the case may be. It is a matter of pure chance that the natural lottery has allotted some people with more intelligence, or that some people were born into wealthier families.
4 But Rawls also suggests, in a another passage, that maybe intelligence, too, could be directly subject to social control: “In the original position, then, the parties want to insure for their descendants the best genetic endowment (assuming their own to be fixed)” [
80] (p. 92). The caveat in parentheses at the end of the sentence may be an indication that Rawls himself did not believe that the genetic endowment of our descendants cannot be changed. Maybe it can (Rawls must have thought) and if it can, it should be subject to social control in line with principles of justice, just in the same way inheritance of wealth is. Rawls, understandably, does not develop this topic further, dismissing it as a “speculative and difficult matter” [
80] (p. 92). But the prospect of using ES and GE as means of bequeathing our descendants a better genetic endowment makes this topic now less speculative, though certainly not less difficult, than it was in 1971, when
A Theory of Justice was published [
81,
82,
83,
84].
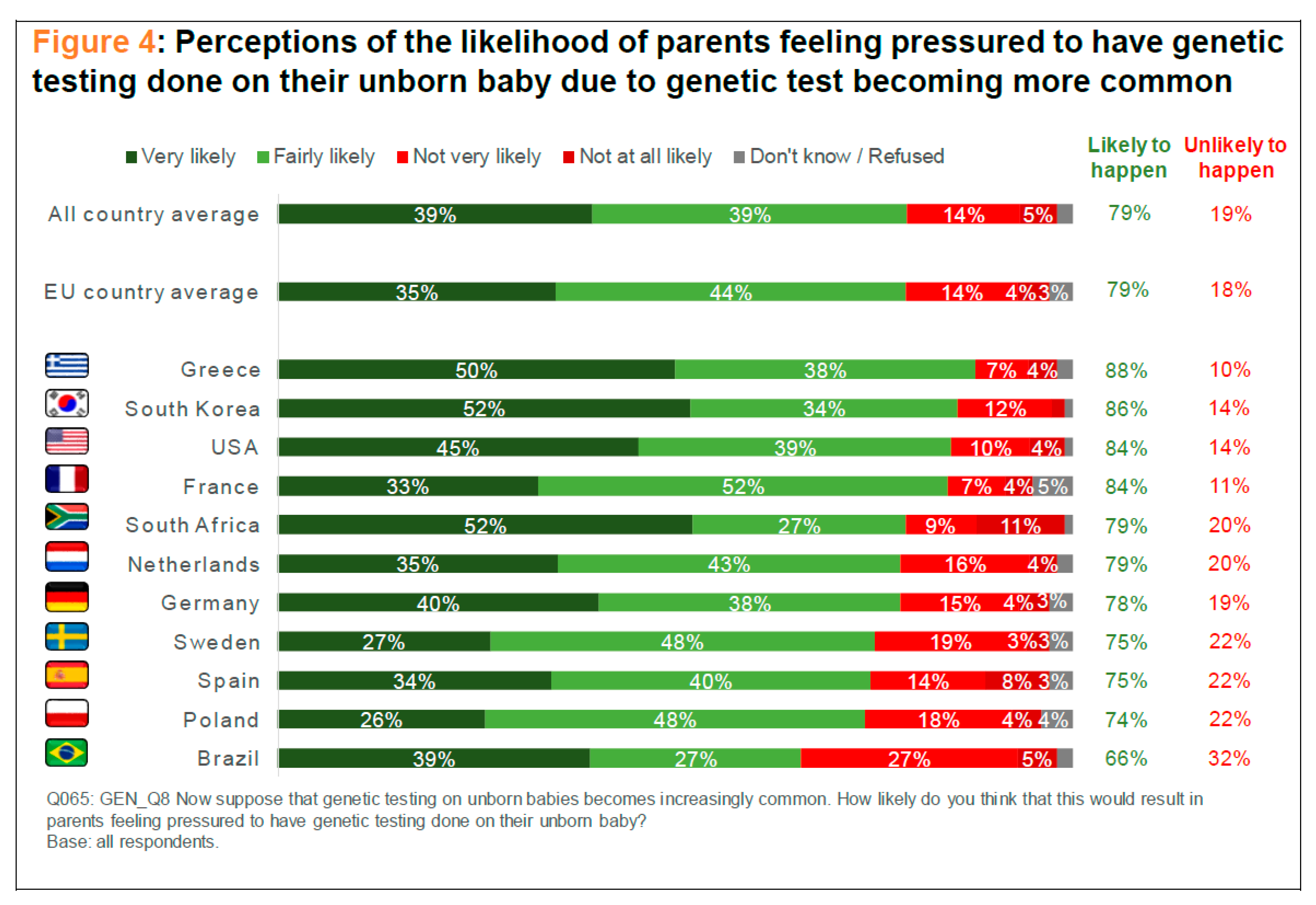
5 If ES and GE become commercially available within the next decades, prospective parents may suffer under the financial and emotional pressure to enhance their children, even if they would otherwise prefer not to enhance them. They may think, for instance, that it would be better to invest their savings in the education rather than in the genetic endowment of their future children. But if, on the other hand, most parents opt for the GCE, they may believe that a non-enhanced child will lag behind in the competition for goods such as university admittance or better positions in the job market. Now, the social pressure here is neither less nor more intense if prospective parents take into consideration that ES (unlike GE) will not modify the genome of their future offspring. Some prospective parents might perhaps favor GE over ES, if they believe, for instance, that abortion is morally wrong, for ES (unlike GE) will require the discarding of human embryos and this, too, may be perceived as equally wrong. But that will be a different kind of pressure: A pressure to favor GE over ES while under the social pressure to enhance their future offspring. From the perspective of policymakers, ES counts as GCE, for ES gives rise to the same questions of social justice that GE does. But policymakers cannot take into consideration here only philosophical arguments. This might lead them to believe, for instance, that the non-identity problem requires that in cases involving wrongful life only the demands of people who were born through GE are valid. This would clearly conflict with the perception that most people, to whom policymakers are accountable, have of the problem. One of the surveys recently conducted by the SIENNA Project revealed, for example, that, on average, 79% of people across 11 countries believe that prospective parents will feel “pressured to have genetic testing done on their unborn baby,” if genetic testing becomes increasingly common (see
Scheme 1). This, too, raises questions of social justice, as many prospective parents may suffer emotionally or financially under the pressure to have their unborn children genetically tested. Some genetic testing may, for instance, reveal an underlying condition that affects not only the unborn baby, but also the living siblings (or the mother, or the father), who might perhaps prefer not to be informed about that particular condition [
85]. It is a matter of societal debate to establish, then, at a national level, how policymakers are expected to address this question. No general philosophical moral principle can be deployed here without failing to do justice to public attitudes towards this problem.
One might suggest now that the quest for GCE has implications for social justice at both national and international levels. At a national level, for reasons I have suggested above, and which are not new in the ethics of human enhancement debate: The rich will bequeath to their children not only their wealth, but also better genetic endowment. Their children, thus, will be better off both financially and cognitively. Lack of appropriate regulation at national level, therefore, might aggravate existing social inequalities. At an international level, countries that are in a position to develop research on human enhancement technologies and human genomics are on average richer than countries that do not have the capability to advance new knowledge and new technologies in these areas. Human enhancement, therefore, might aggravate social inequality at a global level as well.
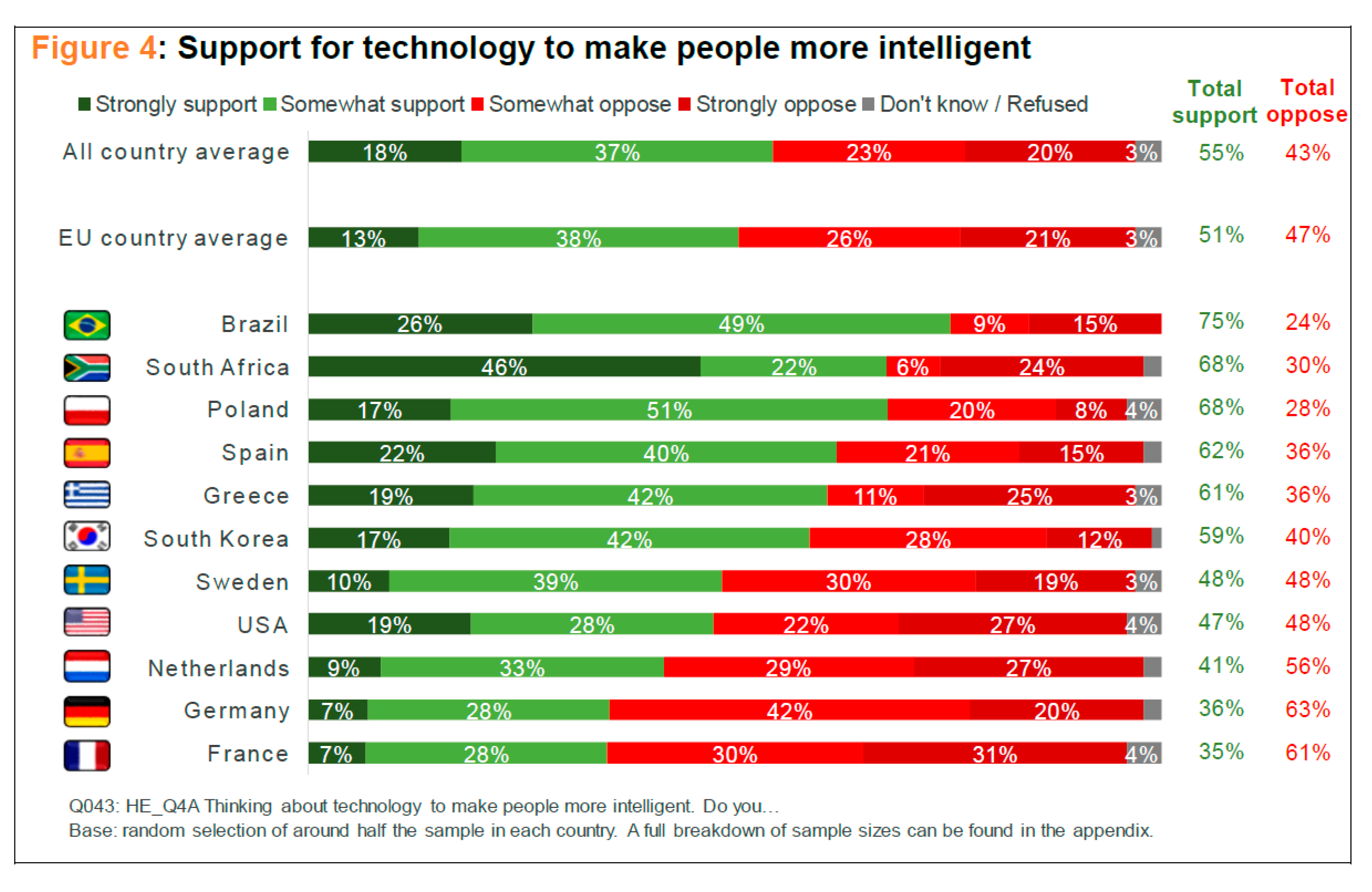
Yet, some of recent findings of the research conducted by the SIENNA Project seem to suggest a different picture. Scientifically and technologically developed countries are not especially supportive of research on human enhancement technologies. Quite the opposite, as one of the reports puts it: “Overall, perceptions of human enhancement technology were most positive in South Africa, Greece and Brazil and least positive in Germany, the USA and France” [
22] (p. 6). As far as support for cognitive enhancement is concerned, a similar pattern emerged: Brazil, South Africa, and Poland were most supportive, while the Netherlands, Germany, and France were least supportive of cognitive enhancement technologies (see
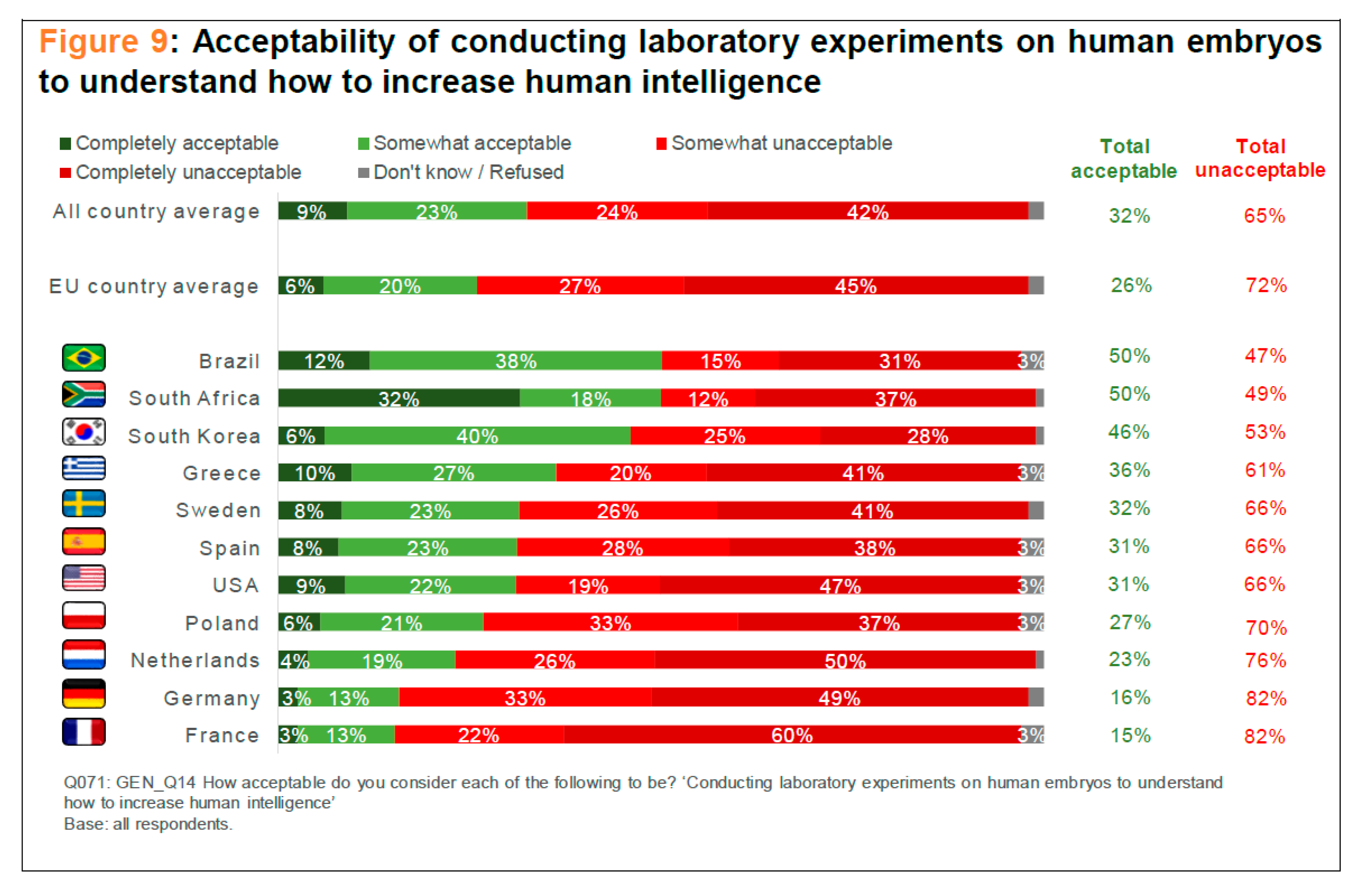
Scheme 2). As for research to better understand the genetics of human intelligence, Brazil, South Africa, and South Korea were most supportive, whereas the Netherlands, Germany, and France, again, were the least supportive countries in this area (see
Scheme 3). As for research on human embryos in order to better understand “how to treat or cure health conditions,” a different pattern emerged: Spain, the Netherlands, and Sweden were most supportive, whereas Greece, the United States, and Germany were least supportive (see
Scheme 4).
One possible implication of these findings is that the unregulated use of GCE technologies might perhaps lead to a greater increase of social inequality at a national level rather than at an international level. Countries such as the United States might perhaps even have more demand for GCE abroad, from countries that are more supportive of human enhancement technologies, than internally. But this tendency will also depend, among other factors, on national laws for the importation of genetic technologies and regulation for the range of procedures that fertility clinics are allowed to perform [
86].
6 This is a topic, though, I cannot delve into in this article, even recognizing that it is worth pursuing for a broader understanding of the ethics of GCE at an international level. My intention here was rather to call attention to the worldwide plurality of public attitudes towards human genomics and human enhancement, as recently surveyed by SIENNA Project, and suggest that these attitudes, too, should be taken into consideration in the debate on the ethics of human enhancement.