The Frailty Reduction via Implementation of Exercise, Nutrition, and Deprescribing (FRIEND) Trial: Study Protocol and Recruitment Results
Abstract
1. Introduction
1.1. Aim
1.2. Research Objectives
1.2.1. Primary Objectives
1.2.2. Secondary Objective
2. Methods
2.1. Study Design
2.2. Study Population
2.3. Eligibility Criteria
2.3.1. Aged Care Residents
2.3.2. Family Member/Informal Caregivers of Participating Residents
2.3.3. Staff Members
2.4. Recruitment
2.5. Sample Size
2.6. Screening Procedure
2.7. Interventions
2.7.1. Best Practice Frailty Interventions for Residents
Exercise Component of the FRIEND Intervention
Nutritional Component of the FRIEND Intervention
Medication Review Component of the FRIEND Intervention
- Ascertain indications of all drugs the resident was taking;
- Consider the overall risk of drug-induced harm (drug–drug interaction, drug–exercise/nutrition interactions);
- Assess each medication for its eligibility and whether it should be discontinued;
- Prioritise drug discontinuation;
- Monitor drug discontinuation regimens.
2.7.2. Educational Component of the FRIEND Intervention
Residents and Family Members/Informal Caregivers
Participating Staff Members
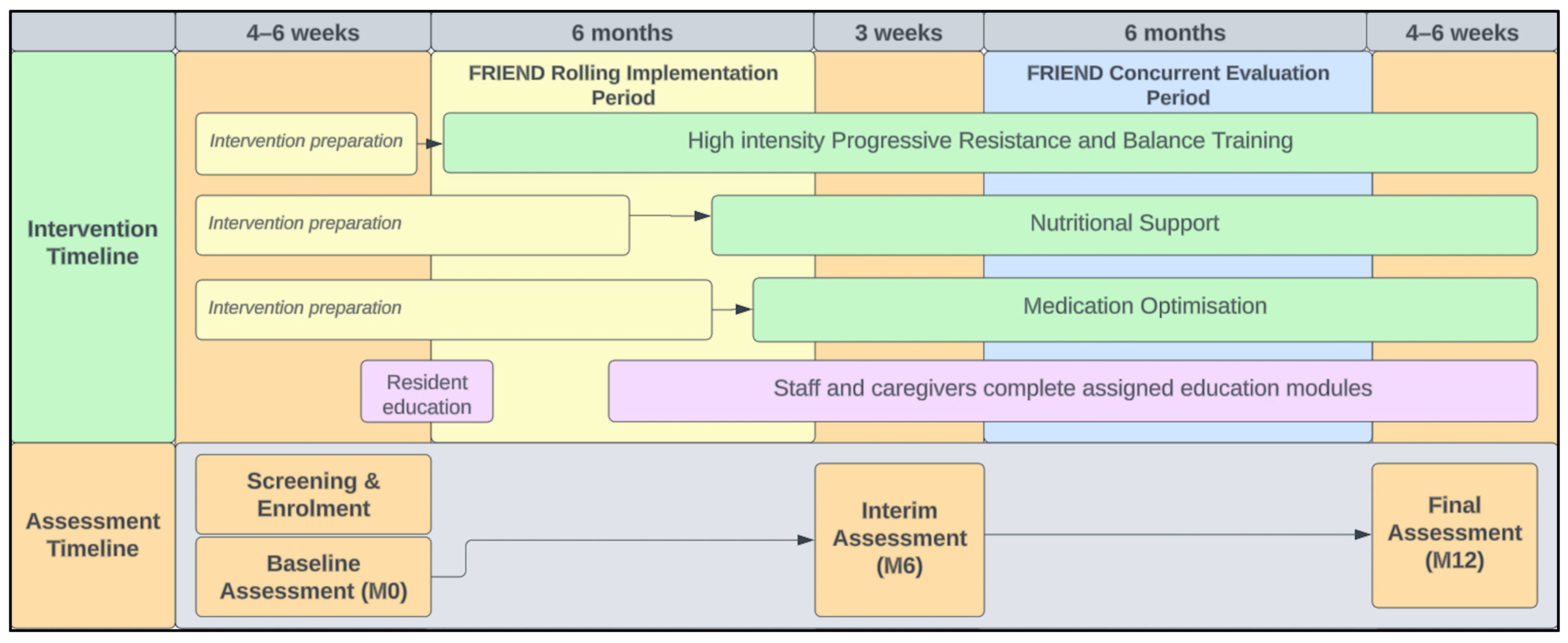
2.7.3. Rolling Implementation of Intervention Components
2.8. Post-Trial Care
2.9. Outcome Measures
2.9.1. Study Participant Data
2.9.2. Facility-Wide Audit Data
2.10. Analysis Plan and Dissemination Plan
3. Baseline Results
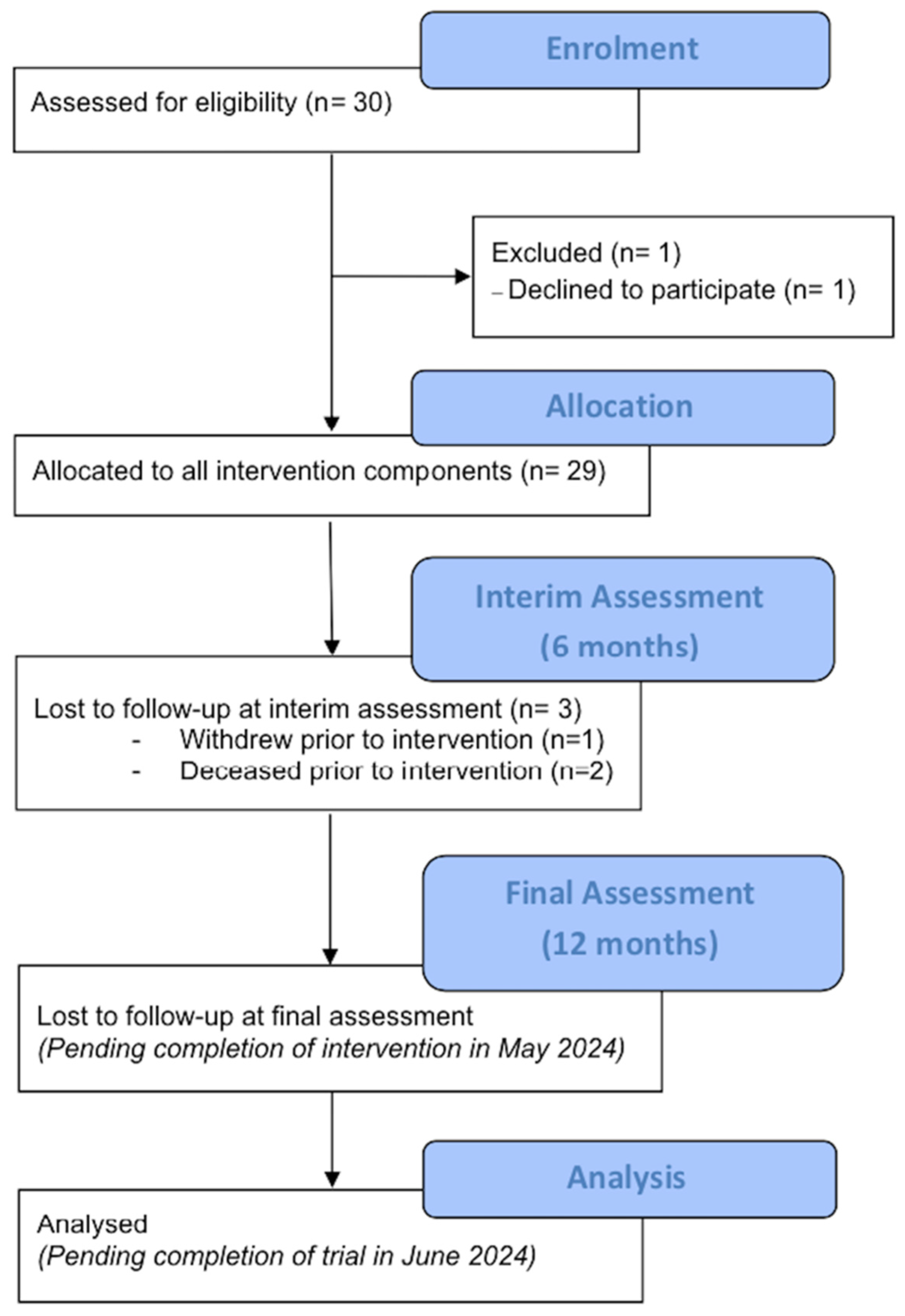
3.1. Recruitment
3.2. Baseline Demographics
| Residents | ||
|---|---|---|
| Sample (n) | Mean (SD) or n | |
| Age (years) | 29 | 88.6 (6.3) |
| Sex (female/male), n | 21/8 | |
| Body Mass Index (kgm−2) | 27 | 26.2 (4.4) |
| Calf circumference (cm) | 23 | 34.1 (3.0) |
| Time since admission to facility (months) | 29 (4–182) * | |
| Prescribed medications and polypharmacy Regular PRN Total prescribed Community definition (≥5), n Aged care definition (≥9), n Hyper-polypharmacy (≥15), n | 29 | 10.7 (4.0) 4.8 (3.5) 15.5 (5.9) 24 21 15 |
| Frailty FRAIL-NH (/14) Fried’s Frailty Phenotype (/5) | 29 23 | 6.3 (2.4) 3.3 (1.1) |
| Cognition MoCA score (/30) Affect GDS-15 (/15) Significant depressive symptoms (>5/15, n) | 29 26 | 13.8 (6.8) 4.3 (3.9) 10 |
| Physical function and capacity SPPB (/12) 6MWT distance (m) | 26 21 | 4.9 (3.1) 222.2 (104.4) |
| Caregivers | ||
| Sample (n) | n | |
| Sex (female/male), n | 30 | 22/8 |
| Relationship to resident Spouse Daughter/daughter-in-law Son/son-in-law Sibling/sibling-in-law Close friend/acquaintance | 0 18 8 2 2 | |
| Staff | ||
| Sample (n) | Mean (SD) or n | |
| Sex (female/male), n | 19 | 17/2 |
| Role Frontline Managerial Senior executive/leadership | 19 | 12 5 2 |
| Time working at facility (months) | 17 | 72 (0–210) * |
| Time working in aged care (months) | 72 (1–408) * | |
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Jadczak, A.D.; on behalf of the FIRST Study Collaborators; Robson, L.; Cooper, T.; Bell, J.S.; Visvanathan, R. The Frailty In Residential Sector over Time (FIRST) study: Methods and baseline cohort description. BMC Geriatr. 2021, 21, 99. [Google Scholar] [CrossRef]
- Dent, E.; Lien, C.; Lim, W.S.; Wong, W.C.; Wong, C.H.; Ng, T.P.; Woo, J.; Dong, B.; de la Vega, S.; Poi, P.J.H.; et al. The Asia-Pacific clinical practice guidelines for the management of frailty. J. Am. Med. Dir. Assoc. 2017, 18, 564–575. [Google Scholar] [CrossRef]
- Liccini, A.P.; Malmstrom, T.K. Frailty and sarcopenia as predictors of adverse health outcomes in persons with diabetes mellitus. J. Am. Med. Dir. Assoc. 2016, 17, 846–851. [Google Scholar] [CrossRef] [PubMed]
- Fried, L.P.; Tangen, C.M.; Walston, J.; Newman, A.B.; Hirsch, C.; Gottdiener, J.; Seeman, T.; Tracy, R.; Kop, W.J.; Burke, G.; et al. Frailty in older adults: Evidence for a phenotype. J. Gerontol. Ser. A Biol. Sci. Med. Sci. 2001, 56, M146–M157. [Google Scholar] [CrossRef] [PubMed]
- Fiatarone, M.A.; Marks, E.C.; Ryan, N.D.; Meredith, C.N.; Lipsitz, L.A.; Evans, W.J. High-intensity strength training in nonagenarians: Effects on skeletal muscle. JAMA 1990, 263, 3029–3034. [Google Scholar] [CrossRef]
- Morris, J.N.; Fiatarone, M.; Kiely, D.K.; Belleville-Taylor, P.; Murphy, K.; Littlehale, S.; Ooi, W.L.; O’Neill, E.; Doyle, N. Nursing rehabilitation and exercise strategies in the nursing home. J. Gerontol. Ser. A Biomed. Sci. Med. Sci. 1999, 54, M494–M500. [Google Scholar] [CrossRef][Green Version]
- Conklin, J.; Farrell, B.; Suleman, S. Implementing deprescribing guidelines into frontline practice: Barriers and facilitators. Res. Soc. Adm. Pharm. 2019, 15, 796–800. [Google Scholar] [CrossRef] [PubMed]
- Kuntz, J.; Kouch, L.; Christian, D.; Peterson, P.L.; Gruss, I. Barriers and Facilitators to the Deprescribing of Nonbenzodiazepine Sedative Medications among Older Adults. Perm. J. 2018, 22, 17–157. [Google Scholar] [CrossRef]
- Djatche, L.; Lee, S.; Singer, D.; Hegarty, S.E.; Lombardi, M.; Maio, V. How confident are physicians in deprescribing for the elderly and what barriers prevent deprescribing? J. Clin. Pharm. Ther. 2018, 43, 550–555. [Google Scholar] [CrossRef]
- Chia, H.S.; Bek, E.S.J. Deprescribing in Primary Care in Singapore: Barriers and Facilitators. Consult. Pharm. 2018, 33, 637–648. [Google Scholar] [CrossRef]
- Luymes, C.H.; van der Kleij, R.M.; Poortvliet, R.K.; de Ruijter, W.; Reis, R.; Numans, M.E. Deprescribing Potentially Inappropriate Preventive Cardiovascular Medication: Barriers and Enablers for Patients and General Practitioners. Ann. Pharmacother. 2016, 50, 446–454. [Google Scholar] [CrossRef]
- Reeve, E.; Bell, J.S.; Hilmer, S.N. Barriers to Optimising Prescribing and Deprescribing in Older Adults with Dementia: A Narrative Review. Curr. Clin. Pharmacol. 2015, 10, 168–177. [Google Scholar] [CrossRef]
- Reeve, E.; To, J.; Hendrix, I.; Shakib, S.; Roberts, M.S.; Wiese, M.D. Patient barriers to and enablers of deprescribing: A systematic review. Drugs Aging 2013, 30, 793–807. [Google Scholar] [CrossRef]
- Paque, K.; Stichele, R.V.; Elseviers, M.; Pardon, K.; Dilles, T.; Deliens, L.; Christiaens, T. Barriers and enablers to deprescribing in people with a life-limiting disease: A systematic review. Palliat. Med. 2019, 33, 37–48. [Google Scholar] [CrossRef]
- Zechmann, S.; Trueb, C.; Valeri, F.; Streit, S.; Senn, O.; Neuner-Jehle, S. Barriers and enablers for deprescribing among older, multimorbid patients with polypharmacy: An explorative study from Switzerland. BMC Fam. Pract. 2019, 20, 64. [Google Scholar] [CrossRef] [PubMed]
- Freiberger, E.; Kemmler, W.; Siegrist, M.; Sieber, C. Frailty and exercise interventions: Evidence and barriers for exercise programs. Z. Gerontol. Geriatr. 2016, 49, 606–611. [Google Scholar] [CrossRef]
- Chodzko-Zajko, W.J.; Proctor, D.N.; Singh, M.A.F.; Minson, C.T.; Nigg, C.R.; Salem, G.J.; Skinner, J.S. Exercise and physical activity for older adults. Med. Sci. Sports Exerc. 2009, 41, 1510–1530. [Google Scholar] [CrossRef]
- Seynnes, O.; Fiatarone Singh, M.A.; Hue, O.; Pras, P.; Legros, P.; Bernard, P.L. Physiological and functional responses to low-moderate versus high-intensity progressive resistance training in frail elders. J. Gerontol. A Biol. Sci. Med. Sci. 2004, 59, 503–509. [Google Scholar] [CrossRef]
- Beck, A.M.; Damkjaer, K.; Tetens, I. Lack of compliance of staff in an intervention study with focus on nutrition, exercise and oral care among old (65+ yrs) Danish nursing home residents. Aging Clin. Exp. Res. 2009, 21, 143–149. [Google Scholar] [CrossRef] [PubMed]
- Krist, L.; Dimeo, F.; Keil, T. Can progressive resistance training twice a week improve mobility, muscle strength, and quality of life in very elderly nursing-home residents with impaired mobility? A pilot study. Clin. Interv. Aging 2013, 8, 443–448. [Google Scholar] [CrossRef] [PubMed]
- Buckinx, F.; Reginster, J.Y.; Petermans, J.; Croisier, J.L.; Beaudart, C.; Brunois, T.; Bruyère, O. Relationship between frailty, physical performance and quality of life among nursing home residents: The SENIOR cohort. Aging Clin. Exp. Res. 2016, 28, 1149–1157. [Google Scholar] [CrossRef]
- Fougère, B.; Kelaiditi, E.; Hoogendijk, E.O.; Demougeot, L.; Duboué, M.; Vellas, B.; Cesari, M. Frailty Index and Quality of Life in Nursing Home Residents: Results From INCUR Study. J. Gerontol. A Biol. Sci. Med. Sci. 2016, 71, 420–424. [Google Scholar] [CrossRef][Green Version]
- Serrano, M.D.; Garrido, M.; Fuentes, R.M.; Simón, M.J.; Díaz, M.J. The impact of biological frailty syndrome on quality of life of nursing home residents. Appl. Nurs. Res. 2017, 35, 112–117. [Google Scholar] [CrossRef] [PubMed]
- Rosenberg, T.; Montgomery, P.; Hay, V.; Lattimer, R. Using frailty and quality of life measures in clinical care of the elderly in Canada to predict death, nursing home transfer and hospitalisation—The frailty and ageing cohort study. BMJ Open 2019, 9, e032712. [Google Scholar] [CrossRef] [PubMed]
- Australian New Zealand Clinical Trials Registry (ANZCTR). Frailty Reduction via Implementation of Exercise, Nutritional Support and Deprescribing Project: The FRIEND Project; NHMRC Clinical Trials Centre, University of Sydney (Australia): Sydney, NSW, Australia, 2022. [Google Scholar]
- Little, M.O.; Sanford, A.M.; Malmstrom, T.K.; Traber, C.; Morley, J.E. Incorporation of Medicare Annual Wellness Visits into the Routine Clinical Care of Nursing Home Residents. J. Am. Geriatr. Soc. 2021, 69, 1100–1102. [Google Scholar] [CrossRef] [PubMed]
- Valenzuela, T. Efficacy of progressive resistance training interventions in older adults in nursing homes: A systematic review. J. Am. Med. Dir. Assoc. 2012, 13, 418–428. [Google Scholar] [CrossRef] [PubMed]
- Mathey, M.-F.A.; Siebelink, E.; de Graaf, C.; Van Staveren, W.A. Flavor enhancement of food improves dietary intake and nutritional status of elderly nursing home residents. J. Gerontol. A Biol. Sci. Med. Sci. 2001, 56, M200–M205. [Google Scholar] [CrossRef] [PubMed]
- Mathey, M.-F.A.; Vanneste, V.G.; de Graaf, C.; de Groot, L.C.; A van Staveren, W. Health effect of improved meal ambiance in a Dutch nursing home: A 1-year intervention study. Prev. Med. 2001, 32, 416–423. [Google Scholar] [CrossRef] [PubMed]
- Borg, G. Borg’s Perceived Exertion and Pain Scales; Human Kinetics; American Psychological Association: Washington, DC, USA, 1998. [Google Scholar]
- Deutz, N.E.; Bauer, J.M.; Barazzoni, R.; Biolo, G.; Boirie, Y.; Bosy-Westphal, A.; Cederholm, T.; Cruz-Jentoft, A.; Krznariç, Z.; Nair, K.S.; et al. Protein intake and exercise for optimal muscle function with aging: Recommendations from the ESPEN Expert Group. Clin. Nutr. 2014, 33, 929–936. [Google Scholar] [CrossRef] [PubMed]
- Pharmaceutical Society of Australia. Guidelines for Quality Use of Medicines (QUM) Services. 2020. Available online: https://www.ppaonline.com.au/wp-content/uploads/2020/04/PSA-Guidelines-for-Quality-Use-of-Medicines-QUM-services.pdf (accessed on 1 February 2024).
- 2019 American Geriatrics Society Beers Criteria® Update Expert Panel. American Geriatrics Society 2019 updated AGS Beers Criteria® for potentially inappropriate medication use in older adults. J. Am. Geriatr. Soc. 2019, 67, 674–694. [Google Scholar] [CrossRef]
- O’Mahony, D.; O’Sullivan, D.; Byrne, S.; O’Connor, M.N.; Ryan, C.; Gallagher, P. STOPP/START criteria for potentially inappropriate prescribing in older people: Version 2. Age Ageing 2014, 44, 213–218. [Google Scholar] [CrossRef]
- Royal Commission into Aged Care Quality and Safety. Final Report: Care, Dignity and Respect. In Summary and Recommendations; Royal Commission into Aged Care Quality and Safety: Adelaide, SA, Australia, 2021. [Google Scholar]
- Kaehr, E.; Visvanathan, R.; Malmstrom, T.K.; Morley, J.E. Frailty in nursing homes: The FRAIL-NH scale. J. Am. Med. Dir. Assoc. 2015, 16, 87–89. [Google Scholar] [CrossRef] [PubMed]
- Vellas, B.; Guigoz, Y.; Garry, P.J.; Nourhashemi, F.; Bennahum, D.; Lauque, S.; Albarede, J.-L. The Mini Nutritional Assessment (MNA) and its use in grading the nutritional state of elderly patients. Nutrition 1999, 15, 116–122. [Google Scholar] [CrossRef]
- Guralnik, J.M.; Simonsick, E.M.; Ferrucci, L.; Glynn, R.J.; Berkman, L.F.; Blazer, D.G.; Scherr, P.A.; Wallace, R.B. A short physical performance battery assessing lower extremity function: Association with self-reported disability and prediction of mortality and nursing home admission. J. Gerontol. 1994, 49, M85–M94. [Google Scholar] [CrossRef] [PubMed]
- Rikli, R.E.; Jones, C.J. The reliability and validity of a 6-minute walk test as a measure of physical endurance in older adults. J. Aging Phys. Act. 1998, 6, 363–375. [Google Scholar] [CrossRef]
- Thorgrimsen, L.; Selwood, A.; Spector, A.; Royan, L.; de Madariaga Lopez, M.; Woods, R.T.; Orrell, M. Whose quality of life is it anyway?: The validity and reliability of the Quality of Life-Alzheimer’s Disease (QoL-AD) scale. Alzheimer Dis. Assoc. Disord. 2003, 17, 201–208. [Google Scholar] [CrossRef]
- Nasreddine, Z.S.; Phillips, N.A.; Bédirian, V.; Charbonneau, S.; Whitehead, V.; Collin, I.; Cummings, J.L.; Chertkow, H. The Montreal Cognitive Assessment, MoCA: A brief screening tool for mild cognitive impairment. J. Am. Geriatr. Soc. 2005, 53, 695–699. [Google Scholar] [CrossRef]
- Lesher, E.L.; Berryhill, J.S. Validation of the geriatric depression scale-short form among inpatients. J. Clin. Psychol. 1994, 50, 256–260. [Google Scholar] [CrossRef]
- NSW Agency for Clinical Innovation (Ed.) Guide to Performing 1 Repetition Maximum Strength Assessment; NSW Government: Sydney, Australia, 2022.
- Schulz, K.F.; Altman, D.G.; Moher, D. CONSORT 2010 statement: Updated guidelines for reporting parallel group randomised trials. J. Pharmacol. Pharmacother. 2010, 1, 100–107. [Google Scholar] [CrossRef]
- Page, A.T.; Falster, M.O.; Litchfield, M.; Pearson, S.-A.; Etherton-Beer, C. Polypharmacy among older Australians, 2006–2017: A population-based study. Med. J. Aust. 2019, 211, 71–75. [Google Scholar] [CrossRef]
- Pavasini, R.; Guralnik, J.; Brown, J.C.; Di Bari, M.; Cesari, M.; Landi, F.; Vaes, B.; Legrand, D.; Verghese, J.; Wang, C.; et al. Short physical performance battery and all-cause mortality: Systematic review and meta-analysis. BMC Med. 2016, 14, 215. [Google Scholar] [CrossRef] [PubMed]
- Australian Government Department of Health. 2020 Aged Care Workforce Census Report; Australian Government: Canberra, Australia, 2020.
- Masnoon, N.; Shakib, S.; Kalisch-Ellett, L.; Caughey, G.E. What is polypharmacy? A systematic review of definitions. BMC Geriatr. 2017, 17, 230. [Google Scholar] [CrossRef] [PubMed]
- Australian Institute of Health and Welfare. Measuring quality in aged care: What is known now and what data are coming. In Australia’s Welfare 2023; Australian Institute of Health and Welfare: Canberra, Australia, 2023. [Google Scholar]
| Outcome | Question/Scope | Measure | Timepoint * |
|---|---|---|---|
| Implementation | |||
| Acceptability | Experience of integrating the intervention | Semi-structured interviews with staff members, residents, and family members/informal caregivers. | M12 |
| Adoption | Satisfaction with amount, content, and format of training provided | Number of staff, residents, and family members/informal caregivers who undertake the educational training modules. | M0–M12 |
| Questionnaire on experience of training and applicability of the skills learned—administered to staff, residents, and family members/informal caregivers. | M12 | ||
| Practice change via the Implementation Behaviour Survey administered to staff members. | M12 | ||
| Audit of intervention components delivered and resident adherence to the intervention components evaluated using self-designed intervention logs. | M0–M12 | ||
| Influences on uptake, integration, and routinisation (roles, relations, context, value systems, barriers/enablers) | Focus groups with managerial and senior representatives of TGSH regarding evaluation of system and process changes to embed the intervention within the organisation’s policies and procedures. | M0, M12 | |
| Monthly implementation team meeting notes. | |||
| Semi-structured interviews with managerial staff and healthcare staff. | M12 | ||
| Knowledge transfer and fidelity | Fidelity to key intervention features; adherence and outcome indictors | Fidelity to key intervention features evaluated via fidelity checks with research and staff members involved in the delivery of the intervention using self-designed fidelity checklists. | M0–M12 |
| Pre- and post-training knowledge acquisition quiz administered to staff, residents, and informal caregivers. | M0, M12 | ||
| Focus group discussions with consumer and consumer representatives, healthcare professionals, and stakeholders to provide feedback on the content of the FRIEND trial website which will be used for ongoing refinement. | M12 | ||
| Cost | Cost of service implementation | Calculation of the cost involved with delivering the FRIEND intervention components (staff time, resources required). | M12 |
| Preliminary effectiveness | |||
| Frailty status | Changes in clinical outcome measures of residents | Frail Nursing Home scale (FRAIL-NH scale). Scored out of 14, with a higher score indicating greater level of frailty [36]. | M0, M6, and M12 |
| Frailty (Fried phenotype) | Derived from existing measures. Weakness (chair stand from SPPB), slowness (gait speed from SPPB), shrinkage (weight change from institutional data), fatigue (Q13 GDS-15), sedentariness (not meeting weekly exercise guidelines of 150/mins of at least moderate physical activity each week). Scored out of 5, with a score of 1–2 indicating pre-frailty and 3–5 indicating frailty [4]. | ||
| Nutritional status | Mini-nutritional Assessment (MNA)—including weight, height, mid-arm, and mid-calf circumferences. A score of 24–30/30 indicates normal nutrition, 17 to 23.5 indicates at risk of malnutrition, and <17 points indicates malnourished state [37]. | M0, M6, and M12 | |
| Medication count and use of potentially inappropriate medications (PIMs) | Medication audit. | ||
| Functional mobility and capacity | Short Physical Performance Battery (SPPB). Scored out of 12, with scores < 10 indicating greater risk of disability and adverse events [38]. Six-minute walk test (6MWT). Higher distance covered in a six-minute period indicates better function [39]. | M0, M6, and M12 | |
| Maximal dynamic muscle strength | One-repetition maximum (1RM) strength on leg press and knee extension machines ** | M0, M6, and M12 | |
| Quality of life | Quality of Life–Alzheimer’s Disease scale (QoL-AD scale). Scored out of 52, with a higher score indicating better quality of life [40]. | M0, M6, and M12 | |
| Cognitive function | Montreal Cognitive Assessment (MoCA). Scored out of 30, with a higher score indicating better cognitive function and scores ≤ 26/30 indicating cognitive impairment [41]. | M0, M6, and M12 | |
| Depressive symptoms | Geriatric Depression Scale—Short Form 15 Item (GDS-15). A higher score out of 15 indicates greater depressive symptoms, with a score > 5/15 indicating significant depressive symptoms [42]. | M0, M6, and M12 | |
| Health status | Past medical history | Past medical diagnosis and symptoms (physical, mental, or emotional), past treatments, medications, past falls. | M0 |
| Ongoing health status | New medical diagnosis, new (or change) in symptoms, new (or change) in medications, falls, visits to healthcare practitioners. Gathered using a weekly health check questionnaire | Weekly | |
| Clinical trial requirements | |||
| Adverse events | All adverse events | All adverse events related and not related to the intervention. Gathered using weekly health status check and reporting of events throughout the study period. | M0–M12 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Inskip, M.; Almendrales Rangel, C.; Njoku, C.M.; Barnett, F.; Shih, I.; O’Neill, L.; Fiatarone Singh, M.A.; Valenzuela, T. The Frailty Reduction via Implementation of Exercise, Nutrition, and Deprescribing (FRIEND) Trial: Study Protocol and Recruitment Results. Methods Protoc. 2024, 7, 26. https://doi.org/10.3390/mps7020026
Inskip M, Almendrales Rangel C, Njoku CM, Barnett F, Shih I, O’Neill L, Fiatarone Singh MA, Valenzuela T. The Frailty Reduction via Implementation of Exercise, Nutrition, and Deprescribing (FRIEND) Trial: Study Protocol and Recruitment Results. Methods and Protocols. 2024; 7(2):26. https://doi.org/10.3390/mps7020026
Chicago/Turabian StyleInskip, Michael, Carolina Almendrales Rangel, Chidiamara Maria Njoku, Fiona Barnett, Isabel Shih, Leonie O’Neill, Maria A. Fiatarone Singh, and Trinidad Valenzuela. 2024. "The Frailty Reduction via Implementation of Exercise, Nutrition, and Deprescribing (FRIEND) Trial: Study Protocol and Recruitment Results" Methods and Protocols 7, no. 2: 26. https://doi.org/10.3390/mps7020026
APA StyleInskip, M., Almendrales Rangel, C., Njoku, C. M., Barnett, F., Shih, I., O’Neill, L., Fiatarone Singh, M. A., & Valenzuela, T. (2024). The Frailty Reduction via Implementation of Exercise, Nutrition, and Deprescribing (FRIEND) Trial: Study Protocol and Recruitment Results. Methods and Protocols, 7(2), 26. https://doi.org/10.3390/mps7020026









