Introducing Artificial Intelligence in Interpretation of Foetal Cardiotocography: Medical Dataset Curation and Preliminary Coding—An Interdisciplinary Project
Abstract
1. Introduction
2. Material and Methods
3. Intellispace Perinatal
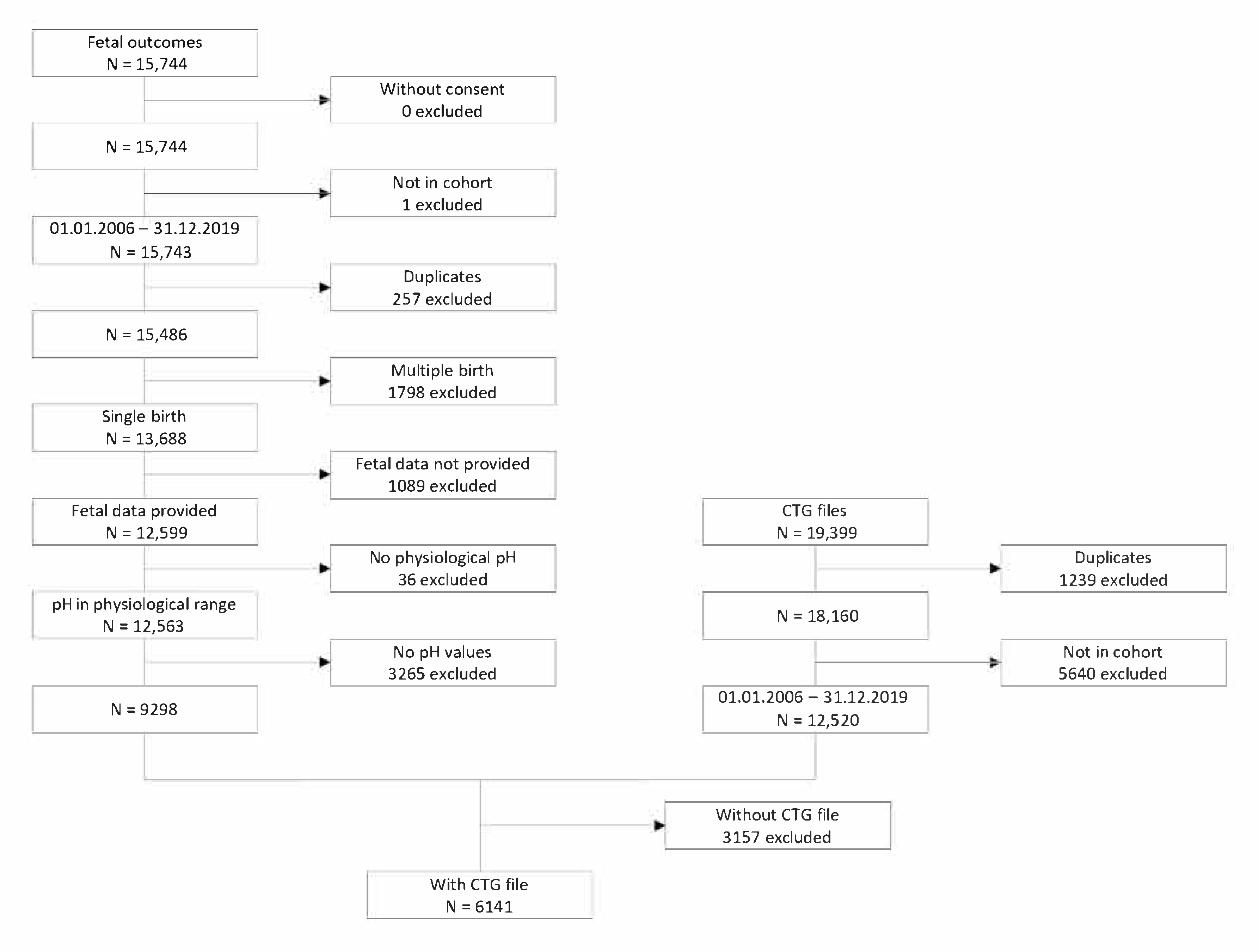
4. Data Collecting
5. Defining the Cohort and Collection of Clinical Data
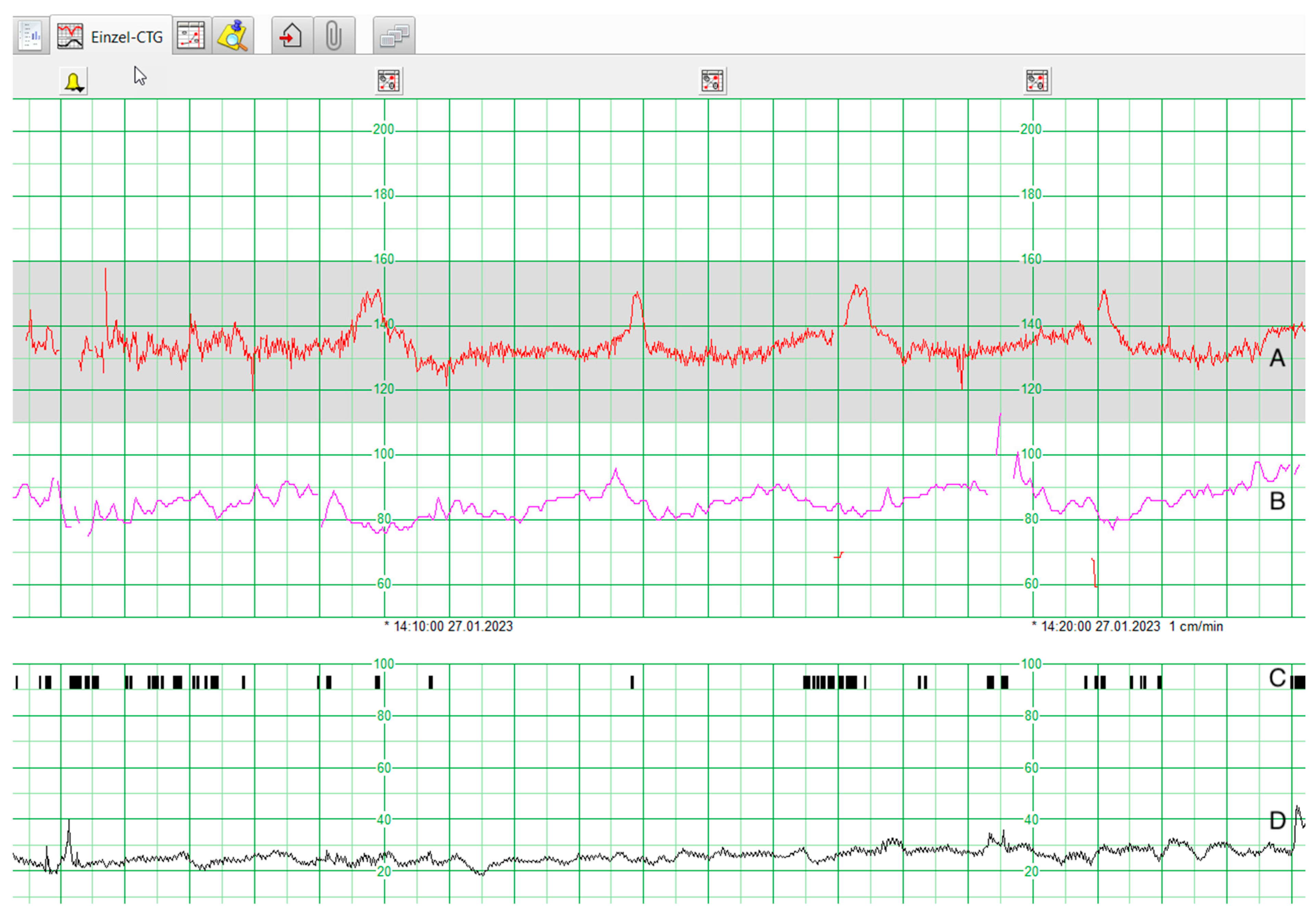
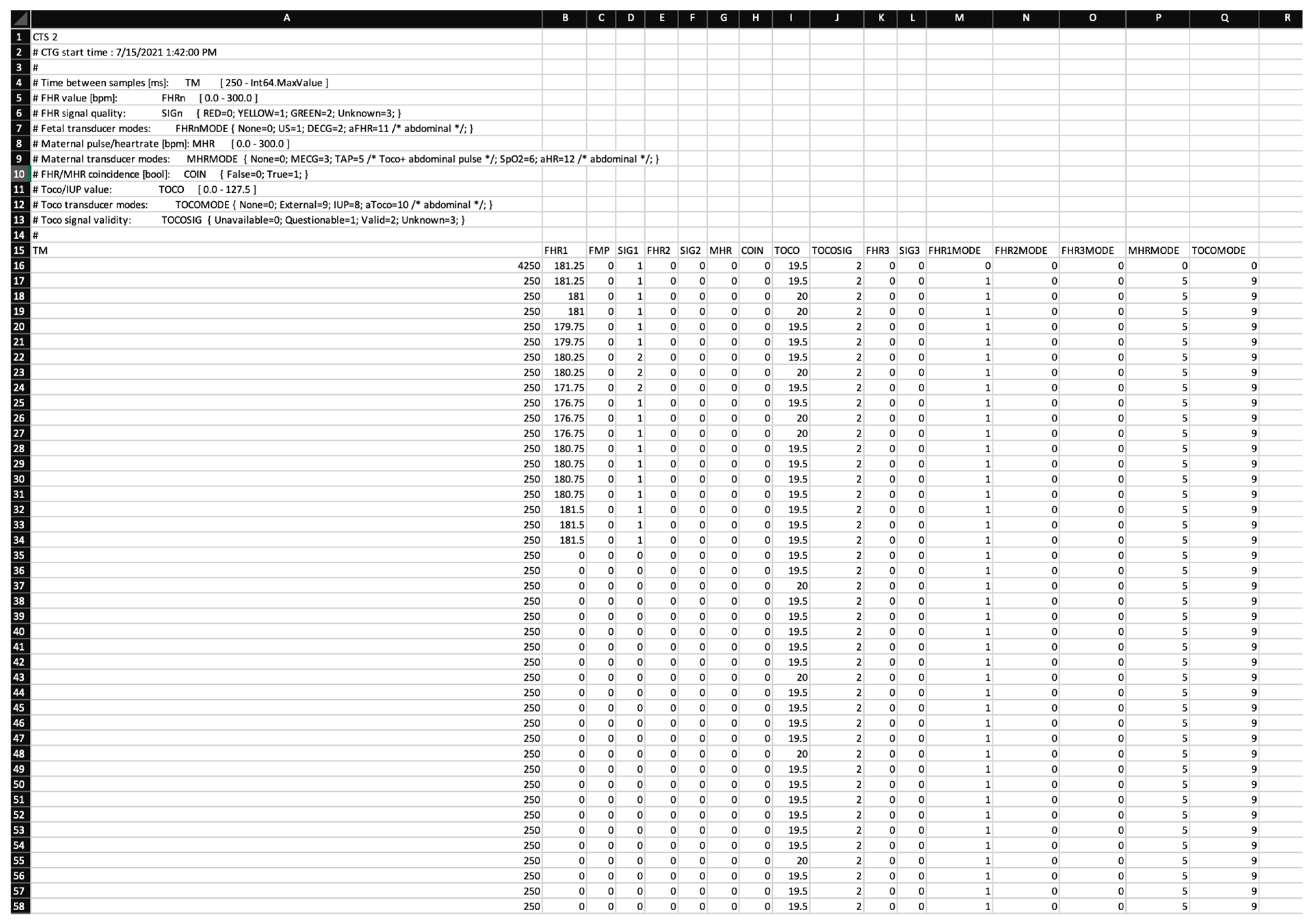
6. CTG Data
7. Eligibility Criteria
8. Results
8.1. CTG Data
8.2. Clinical Data
9. Discussion
10. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| AI | artificial intelligence |
| bpm | beats per minute |
| CNN | convolutional neural network |
| CSEM | Centre Suisse d’Electronique et de Microtechnique |
| CTG | cardiotocography |
| DL | deep learning |
| FHR | foetal heart rate |
| IDC | Insel Data Centre |
| LR | logistic regression |
| LSTM | long short-term memory |
| MHR | maternal heart rate |
| ML | machine learning |
| mmHg | millimetres of mercury |
| NICHD | National Institute of Child Health and Human Development |
| PDA | peridural anaesthesia |
| PID | patient identification number |
| RCT | randomized controlled trial |
| RNN | recurrent neural network |
| SP | signal-processing |
| SVC | support vector classification |
| SVR | support vector regression |
| UC | uterine contractions |
| UCI | University of California Irvine |
| UHB | University Hospital of Bern |
Appendix A
| Item | Label | Exemplification | Datatype & Unit |
|---|---|---|---|
| Name | name | ||
| First name | first_name | ||
| Patient-ID | pid | ||
| Case-ID | fid | ||
| Date of birth mother | dob | ||
| General consent (since 2017) | GC | Binary: 0 = no 1 = yes | |
| General consent vital signs | GCstatus | Binary | |
| Gravidity | gravidity | Birth report, first number from SAFT-Code | Numeric |
| Parity | parity | Birth report, summation of last two numbers from SAFT-Code | Numeric |
| Gestational week | gestation_week | Birth report, number + seventh | |
| Date of birth | delivery_date | Birth report | Date |
| Time of birth | delivery_time | Birth report | Datetime |
| Date cervix at 4 cm | 4 cm_date | Partogram | Date |
| Time cervix at 4 cm | 4 cm_time | Partogram | Datetime |
| Date cervix complete | 10_date | Partogram | Date |
| Time cervix complete | 10 cm_time | Partogram | Datetime |
| PDA | PDA | Partogram | Binary: 0 = no 1 = yes |
| Date PDA insertion | PDA_date | Partogram | Date |
| Time PDA insertion | PDA_time | Partogram | Datetime |
| Date spontaneous or artificial rupture of membranes | ROM_date | Partogram | Date |
| Time spontaneous or artificial rupture of membranes | ROM_time | Partogram | Datetime |
| Delivery mode | delivery_mode | Birth report | Categorical/grouped |
| Foetal sex | sex | Birth report | Categorical/grouped |
| Birth weight | birthweight | Birth report | Numeric (gram) |
| Birth weight percentile | birthweight_p | Birth report | Other |
| Head circumference | head | Birth report | Numeric (centimetre) |
| Head circumference percentile | head_p | Birth report | Other |
| Body length | length | Birth report | Numeric (centimetre) |
| Body length percentile | length_p | Birth report | Other |
| APGAR 1 min | apgar_1 | Birth report | Numeric |
| APGAR 5 min | apgar_5 | Birth report | Numeric |
| APGAR 10 min | apgar_10 | Birth report | Numeric |
| Venous pH | NSV_pH | Birth report | Numeric |
| Arterial pH | NSA_pH | Birth report | Numeric |
| Base excess | BE | Birth report | Numeric |
| Intrauterine growth retardation | IUGR | Diagnoses from birth report: intrauterine Wachstumsrestriktion, intrauterine Wachstumsretardierung, IUWR | Freetext |
| Small for gestational age | SGA | Diagnoses from birth report: small for gestational age, SGA | Freetext |
| Macrosomia | macrosomia | Diagnoses from birth report: Makrosomie | Freetext |
| Amniotic fluid with meconium | meconium | Diagnoses from birth report: mekoniumhaltiges Fruchtwasser, mekoniumhaltiges FW, Mekoniumabgang | Freetext |
| Unsuccessful tocolysis | unsuc_tocolysis | Diagnoses from birth report: Tokolysedurchbruch | Freetext |
| Amnion infection syndrome | AIS | Diagnoses from birth report: Amnioninfektsyndrom, AIS | Freetext |
| Gestational diabetes | GDM | Diagnoses from birth report: Gestationsdiabetes, GDM, iGDM, dGDM | Freetext |
| Preeclampsia/HELLP | PE | Diagnoses from birth report: Präeklampsie, PE, HELLP, HELLP-Syndrom | Freetext |
| Blood loss | bloodloss | Diagnoses from birth report: Blutverlust, BV | Numeric (mL/L) |
References
- Cornet, G. Chapter 4. Robot companions and ethics: A pragmatic approach of ethical design. J. Int. Bioéthique 2013, 24, 49. [Google Scholar] [CrossRef] [PubMed]
- Larson, J.A.; Johnson, M.H.; Bhayani, S.B. Application of Surgical Safety Standards to Robotic Surgery: Five Principles of Ethics for Nonmaleficence. J. Am. Coll. Surg. 2014, 218, 290–293. [Google Scholar] [CrossRef] [PubMed]
- Zhang, P.; Kamel Boulos, M.N. Generative AI in Medicine and Healthcare: Promises, Opportunities and Challenges. Future Internet 2023, 15, 286. [Google Scholar] [CrossRef]
- Beam, A.L.; Kohane, I.S. Big Data and Machine Learning in Health Care. JAMA 2018, 319, 1317. [Google Scholar] [CrossRef] [PubMed]
- Ravi, D.; Wong, C.; Deligianni, F.; Berthelot, M.; Andreu-Perez, J.; Lo, B.; Yang, G.-Z. Deep Learning for Health Informatics. IEEE J. Biomed. Health Inform. 2017, 21, 4–21. [Google Scholar] [CrossRef] [PubMed]
- Hamet, P.; Tremblay, J. Artificial intelligence in medicine. Metabolism 2017, 69, S36–S40. [Google Scholar] [CrossRef] [PubMed]
- Goldberg, D.E.; Holland, J.H. Genetic Algorithms and Machine Learning; Kluwer Academic Publishers: Norwell, MA, USA, 1988. [Google Scholar]
- Ayres-de-Campos, D.; Spong, C.Y.; Chandraharan, E. FIGO Intrapartum Fetal Monitoring Expert Consensus Panel FIGO consensus guidelines on intrapartum fetal monitoring: Cardiotocography. Int. J. Gynecol. Obstet. 2015, 131, 13–24. [Google Scholar] [CrossRef] [PubMed]
- Alfirevic, Z.; Devane, D.; Gyte, G.; Cuthbert, A. Continuous cardiotocography (CTG) as a form of electronic fetal monitoring (EFM) for fetal assessment during labour. Cochrane Database Syst. Rev. 2017, 2017, CD006066. [Google Scholar] [CrossRef] [PubMed]
- Balayla, J.; Shrem, G. Use of artificial intelligence (AI) in the interpretation of intrapartum fetal heart rate (FHR) tracings: A systematic review and meta-analysis. Arch. Gynecol. Obstet. 2019, 300, 7–14. [Google Scholar] [CrossRef] [PubMed]
- Rodriguez-Ruiz, A.; Lång, K.; Gubern-Merida, A.; Broeders, M.; Gennaro, G.; Clauser, P.; Helbich, T.H.; Chevalier, M.; Tan, T.; Mertelmeier, T.; et al. Stand-Alone Artificial Intelligence for Breast Cancer Detection in Mammography: Comparison with 101 Radiologists. J. Natl. Cancer Inst. 2019, 111, 916–922. [Google Scholar] [CrossRef] [PubMed]
- Choy, G.; Khalilzadeh, O.; Michalski, M.; Do, S.; Samir, A.E.; Pianykh, O.S.; Geis, J.R.; Pandharipande, P.V.; Brink, J.A.; Dreyer, K.J. Current Applications and Future Impact of Machine Learning in Radiology. Radiology 2018, 288, 318–328. [Google Scholar] [CrossRef] [PubMed]
- Stirnemann, J.J.; Besson, R.; Spaggiari, E.; Rojo, S.; Loge, F.; Peyro-Saint-Paul, H.; Allassonniere, S.; Le Pennec, E.; Hutchinson, C.; Sebire, N.; et al. Development and clinical validation of real-time artificial intelligence diagnostic companion for fetal ultrasound examination. Ultrasound Obstet. Gynecol. 2023, 62, 353–360. [Google Scholar] [CrossRef] [PubMed]
- Lopes-Pereira, J.; Costa, A.; Ayres-De-Campos, D.; Costa-Santos, C.; Amaral, J.; Bernardes, J. Computerized analysis of cardiotocograms and ST signals is associated with significant reductions in hypoxic-ischemic encephalopathy and cesarean delivery: An observational study in 38,466 deliveries. Am. J. Obstet. Gynecol. 2019, 220, 269.e1–269.e8. [Google Scholar] [CrossRef] [PubMed]
- Koninklijke Philips, N.V. IntelliSpace Perinatal. Available online: https://www.philips.ch/healthcare/product/HCNOCTN177/intellispace-perinatal-kreisaal-datenmanagementsystem (accessed on 2 November 2023).
- Koninklijke Philips, N.V. PHILIPS. Available online: https://www.philips.ch (accessed on 2 November 2023).
- National Institute of Child Health and Human Development NICHD. Available online: https://www.nichd.nih.gov (accessed on 10 October 2023).
- Nunes, I.; Ayres-de-Campos, D. Computer analysis of foetal monitoring signals. Best Pract. Res. Clin. Obstet. Gynaecol. 2016, 30, 68–78. [Google Scholar] [CrossRef] [PubMed]
- Brocklehurst, P.; Field, D.; Greene, K.; Juszczak, E.; Keith, R.; Kenyon, S.; Linsell, L.; Mabey, C.; Newburn, M.; Plachcinski, R.; et al. Computerised interpretation of fetal heart rate during labour (INFANT): A randomised controlled trial. Lancet 2017, 389, 1719–1729. [Google Scholar] [CrossRef] [PubMed]
- Keith, R. The INFANT study—A flawed design foreseen. Lancet 2017, 389, 1697–1698. [Google Scholar] [CrossRef] [PubMed]
- Chudáček, V.; Spilka, J.; Burša, M.; Janků, P.; Hruban, L.; Huptych, M.; Lhotská, L. Open access intrapartum CTG database. BMC Pregnancy Childbirth 2014, 14, 16. [Google Scholar] [CrossRef] [PubMed]
- Dua, D.; Graff, C. UCI Machine Learning Repository. Available online: http://archive.ics.uci.edu/ml (accessed on 2 November 2023).
- Ayres-de-campos, D.; Bernardes, J.; Garrido, A.; Marques-de-sá, J.; Pereira-leite, L. SisPorto 2.0: A Program for Automated Analysis of Cardiotocograms. J. Matern. Fetal Neonatal Med. 2000, 9, 311–318. [Google Scholar] [CrossRef]
- Aeberhard, J.L.; Radan, A.-P.; Delgado-Gonzalo, R.; Strahm, K.M.; Sigurthorsdottir, H.B.; Schneider, S.; Surbek, D. Artificial intelligence and machine learning in cardiotocography: A scoping review. Eur. J. Obstet. Gynecol. Reprod. Biol. 2023, 281, 54–62. [Google Scholar] [CrossRef]
| Year | Number of CTG Episodes |
|---|---|
| 2006 | 0 |
| 2007 | 204 |
| 2008 | 376 |
| 2009 | 526 |
| 2010 | 542 |
| 2011 | 1690 |
| 2012 | 1742 |
| 2013 | 1820 |
| 2014 | 1800 |
| 2015 | 2294 |
| 2016 | 2888 |
| 2017 | 345 |
| 2018 | 271 |
| 2019 | 164 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Aeberhard, J.L.; Radan, A.-P.; Soltani, R.A.; Strahm, K.M.; Schneider, S.; Carrié, A.; Lemay, M.; Krauss, J.; Delgado-Gonzalo, R.; Surbek, D. Introducing Artificial Intelligence in Interpretation of Foetal Cardiotocography: Medical Dataset Curation and Preliminary Coding—An Interdisciplinary Project. Methods Protoc. 2024, 7, 5. https://doi.org/10.3390/mps7010005
Aeberhard JL, Radan A-P, Soltani RA, Strahm KM, Schneider S, Carrié A, Lemay M, Krauss J, Delgado-Gonzalo R, Surbek D. Introducing Artificial Intelligence in Interpretation of Foetal Cardiotocography: Medical Dataset Curation and Preliminary Coding—An Interdisciplinary Project. Methods and Protocols. 2024; 7(1):5. https://doi.org/10.3390/mps7010005
Chicago/Turabian StyleAeberhard, Jasmin Leonie, Anda-Petronela Radan, Ramin Abolfazl Soltani, Karin Maya Strahm, Sophie Schneider, Adriana Carrié, Mathieu Lemay, Jens Krauss, Ricard Delgado-Gonzalo, and Daniel Surbek. 2024. "Introducing Artificial Intelligence in Interpretation of Foetal Cardiotocography: Medical Dataset Curation and Preliminary Coding—An Interdisciplinary Project" Methods and Protocols 7, no. 1: 5. https://doi.org/10.3390/mps7010005
APA StyleAeberhard, J. L., Radan, A.-P., Soltani, R. A., Strahm, K. M., Schneider, S., Carrié, A., Lemay, M., Krauss, J., Delgado-Gonzalo, R., & Surbek, D. (2024). Introducing Artificial Intelligence in Interpretation of Foetal Cardiotocography: Medical Dataset Curation and Preliminary Coding—An Interdisciplinary Project. Methods and Protocols, 7(1), 5. https://doi.org/10.3390/mps7010005






