Isolation and Cultivation of Adipose-Derived Mesenchymal Stem Cells Originating from the Infrapatellar Fat Pad Differentiated with Blood Products: Method and Protocol
Abstract
1. Introduction
2. Procedure and Experimental Design
2.1. Surgical Cell Harvest
2.2. Ethical Votes
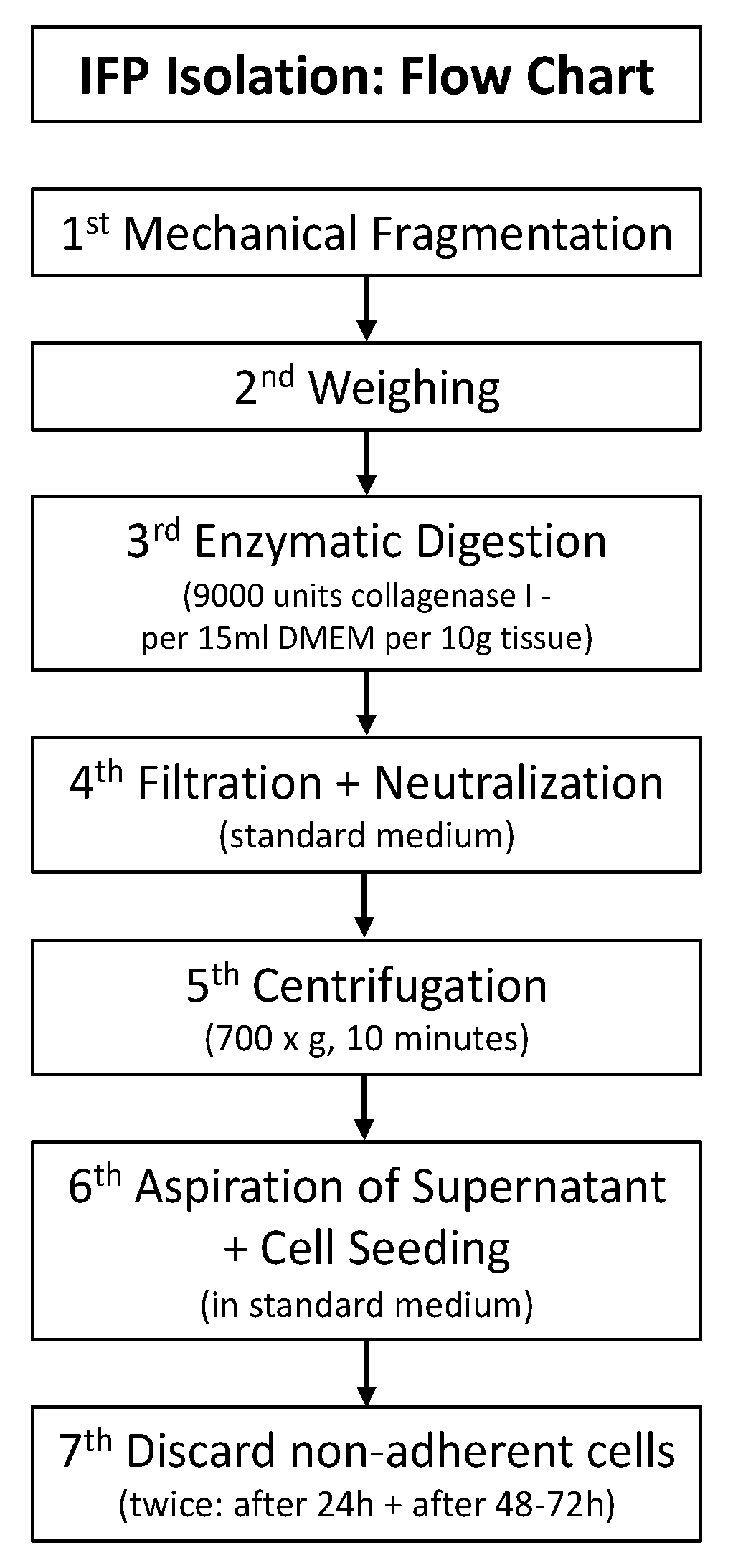
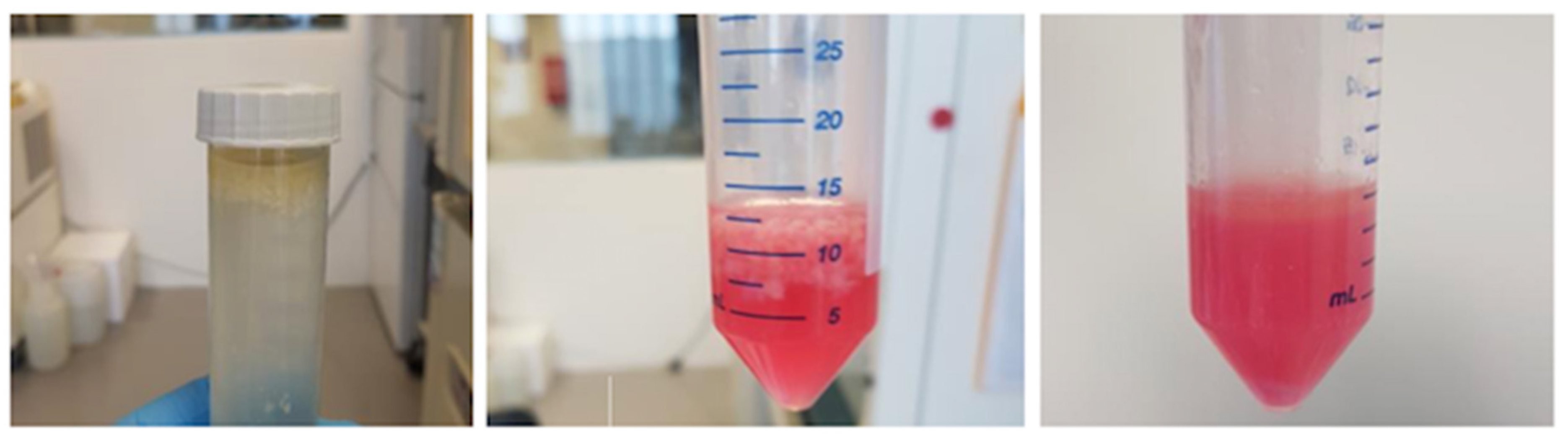
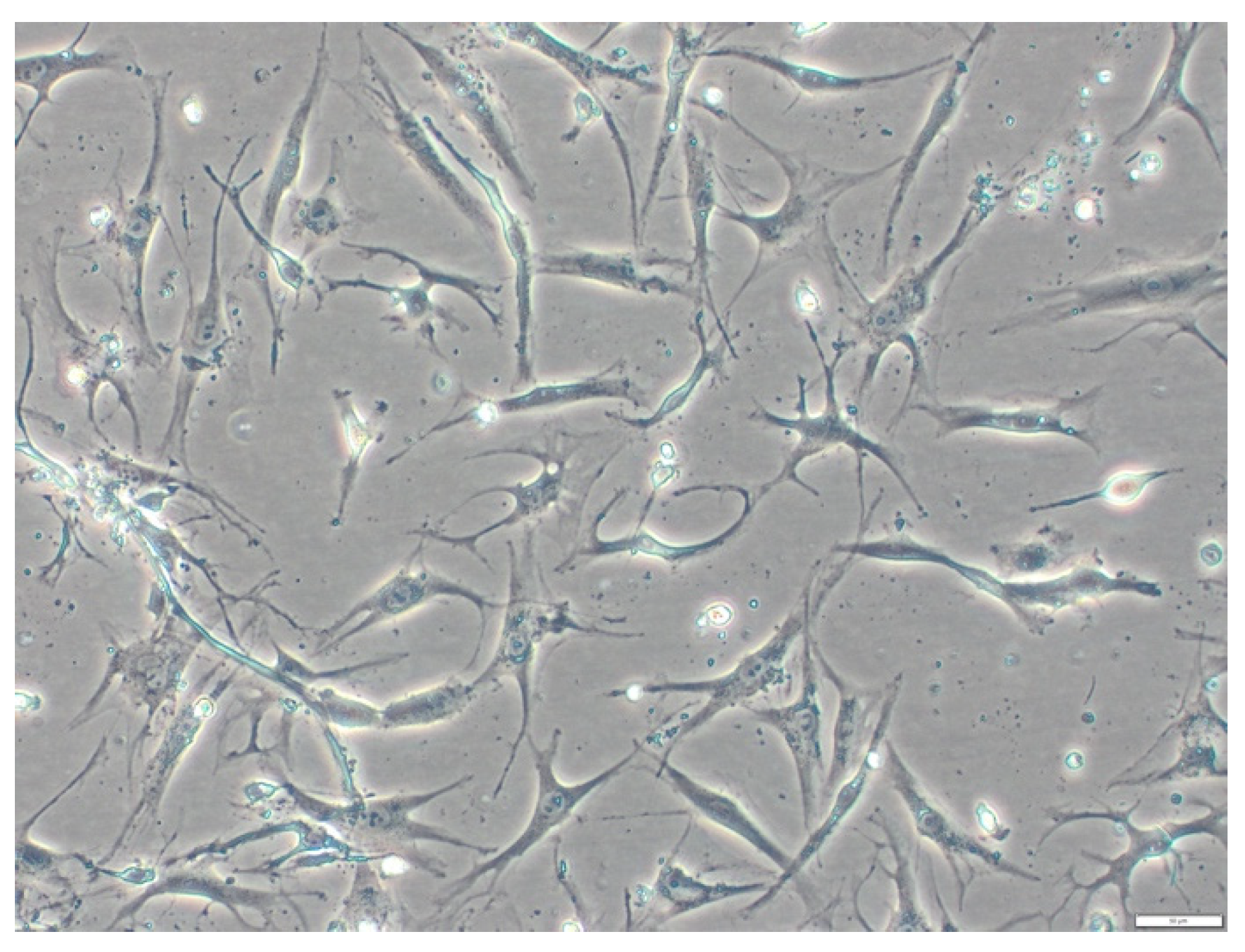
2.3. Cell Isolation and Cell Culture
2.4. Blood Product Preparation Protocol
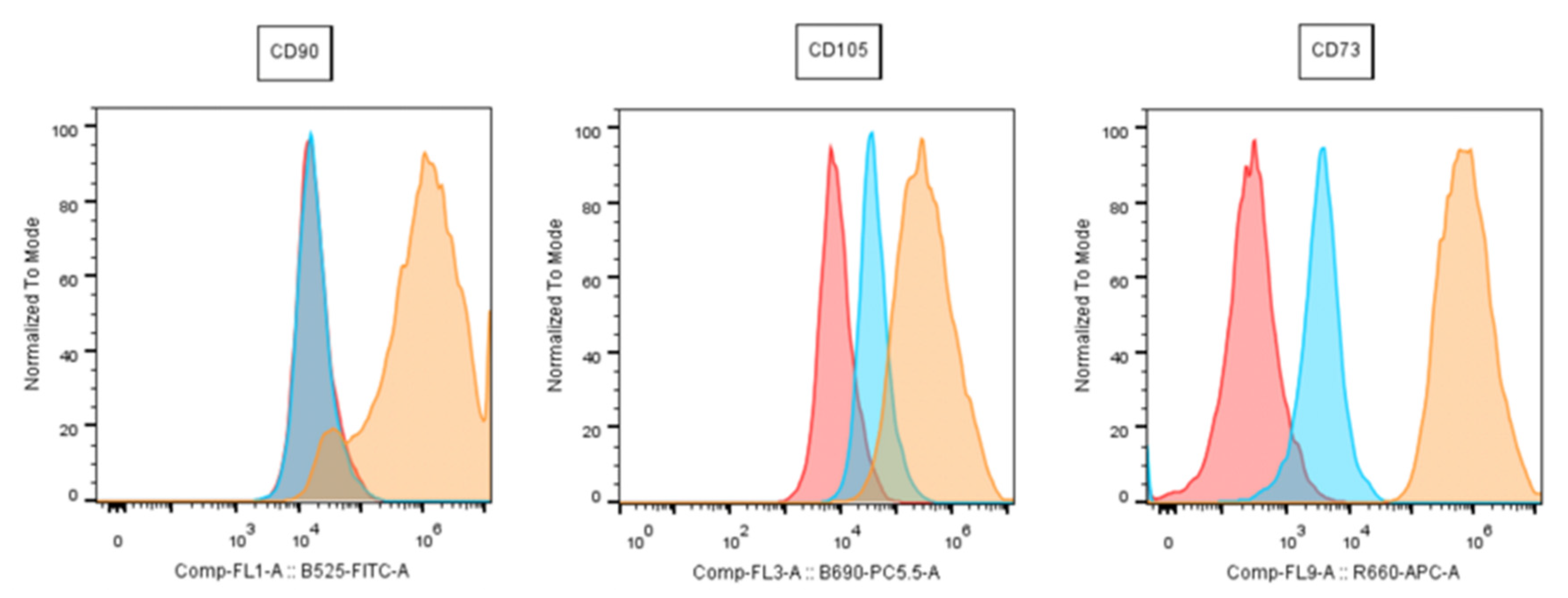
2.5. Flow Cytometry
- Plastic adherent (in standard culture);
- Expression of CD73, CD90 and CD105; Absence of CD34, CD45, CD14, HLA-DR, CD11b and CD19
- Chondrogenic, osteogenic and adipogenic differentiation potential.
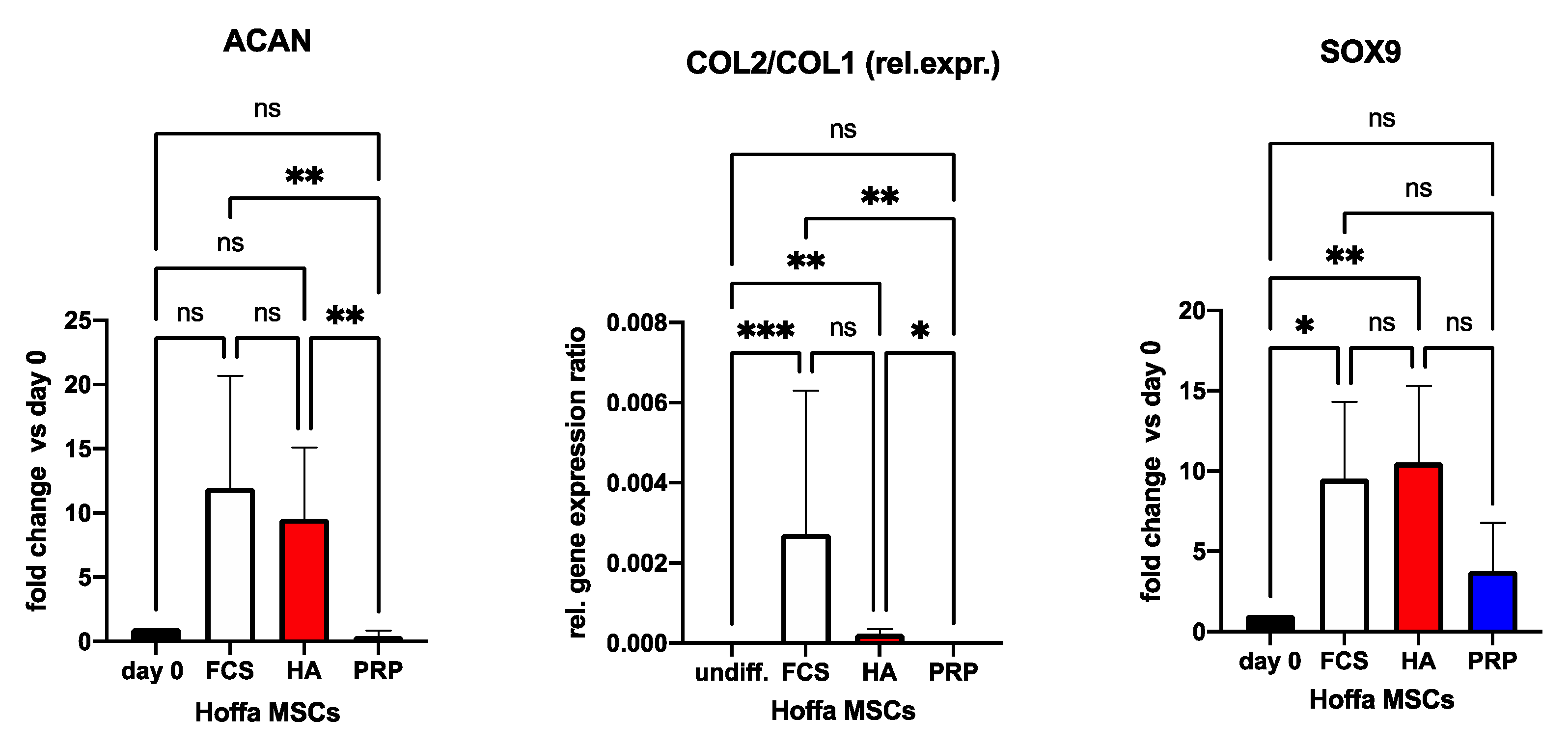
2.6. Blood Product Supplemented Cell Incubation and Chondrogenic Cell Differentiation
3. Discussion
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Barry, F.; Murphy, M. Mesenchymal stem cells in joint disease and repair. Nat. Rev. Rheumatol. 2013, 9, 584–594. [Google Scholar] [CrossRef] [PubMed]
- Neubauer, M.; Jeyakumar, V.; Muellner, T.; Nehrer, S. Bone-marrow-aspirate-concentrate for chondral defects: Surgical techniques, clinical applications and basic science. Ann. Jt. 2018, 3, 107. [Google Scholar] [CrossRef]
- Stoltz, J.-F.; de Isla, N.; Li, Y.P.; Bensoussan, D.; Zhang, L.; Huselstein, C.; Chen, Y.; Decot, V.; Magdalou, J.; Li, N.; et al. Stem Cells and Regenerative Medicine: Myth or Reality of the 21th Century. Stem Cell. Int. 2015, 2015, 734731. [Google Scholar] [CrossRef]
- Pas, H.I.; Winters, M.; Haisma, H.J.; Koenis, M.J.; Tol, J.L.; Moen, M.H. Stem cell injections in knee osteoarthritis: A systematic review of the literature. Br. J. Sport. Med. 2017, 51, 96793. [Google Scholar] [CrossRef] [PubMed]
- Kaoutzanis, C.; Gupta, V.; Winocour, J.; Layliev, J.; Ramirez, R.; Grotting, J.C.; Higdom, K. Cosmetic liposuction: Preoperative risk factors, major complication rates, and safety of combined procedures. Aesthetic Surg. J. 2017, 37, 680–694. [Google Scholar] [CrossRef]
- Park, J.S.; Yang, H.; Woo, D.; Jeon, S.; Park, K.-H. The promotion of chondrogenesis, osteogenesis, and adipogenesis of human mesenchymal stem cells by multiple growth factors incorporated into nanosphere-coated microspheres. Biomaterials 2011, 32, 28–38. [Google Scholar] [CrossRef]
- Mochizuki, T.; Muneta, T.; Sakaguchi, Y.; Nimura, A.; Yokoyama, A.; Koga, H.; Sekiya, I. Higher chondrogenic potential of fibrous synovium- and adipose synovium-derived cells compared with subcutaneous fat-derived cells: Distinguishing properties of mesenchymal stem cells in humans. Arthritis Rheum. 2006, 54, 843–853. [Google Scholar] [CrossRef]
- Wickham, M.Q.; Erickson, G.R.; Gimble, J.M.; Vail, T.P.; Guilak, F. Multipotent Stromal Cells Derived from the Infrapatellar Fat Pad of the Knee. Clin. Orthop. Relat. Res. 2003, 412, 196–212. [Google Scholar] [CrossRef] [PubMed]
- Doner, G.P.; Noyes, F.R. Arthroscopic resection of fat pad lesions and infrapatellar contractures. Arthrosc. Tech. 2014, 3, e413–e416. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Dragoo, J.L.; Johnson, C.; McConnell, J.; Dragoo, J.L. Evaluation and treatment of disorders of the infrapatellar fat pad. Sport. Med. 2012, 42, 51–67. [Google Scholar] [CrossRef] [PubMed]
- Kumar, D.; Alvand, A.; Beacon, J. Impingement of Infrapatellar Fat Pad (Hoffa’s Disease): Results of High-Portal Arthroscopic Resection. Arthrosc. J. Arthrosc. Relat. Surg. 2007, 23, 1180–1186.e1. [Google Scholar] [CrossRef] [PubMed]
- Pak, J. Regeneration of human bones in hip osteonecrosis and human cartilage in knee osteoarthritis with autologous adipose-tissue-derived stem cells: A case series. J. Med. Case Rep. 2011, 5, 296. [Google Scholar] [CrossRef]
- Pak, J.; Lee, J.H.; Pak, N.; Pak, Y.; Park, K.S.; Jeon, J.H.; Jeong, B.C.; Lee, S.H. Cartilage Regeneration in Humans with Adipose Tissue-Derived Stem Cells and Adipose Stromal Vascular Fraction Cells: Updated Status. Int. J. Mol. Sci. 2018, 19, 2146. [Google Scholar] [CrossRef]
- Pak, J.; Lee, J.H.; Kartolo, W.A.; Lee, S.H. Cartilage Regeneration in Human with Adipose Tissue-Derived Stem Cells: Current Status in Clinical Implications. BioMed Res. Int. 2016, 2016, 1–12. [Google Scholar] [CrossRef]
- Hart, P.H.; Ahern, M.; Smith, M.; Finlay-Jones, J.J. Comparison of the suppressive effects of interleukin-10 and interleukin-4 on synovial fluid macrophages and blood monocytes from patients with inflammatory arthritis. Immunology 1995, 84, 536–542. [Google Scholar] [PubMed]
- Neubauer, M.; Kuten, O.; Stotter, C.; Kramer, K.; De Luna, A.; Muellner, T.; Lacza, Z.; Nehrer, S. The Effect of Blood-Derived Products on the Chondrogenic and Osteogenic Differentiation Potential of Adipose-Derived Mesenchymal Stem Cells Originated from Three Different Locations. Stem Cell. Int. 2019, 2019, 1–20. [Google Scholar] [CrossRef] [PubMed]
- Dragoo, J.L.; Chang, W. Arthroscopic Harvest of Adipose-Derived Mesenchymal Stem Cells from the Infrapatellar Fat Pad. Am. J. Sport. Med. 2017, 45, 3119–3127. [Google Scholar] [CrossRef]
- Dominici, M.; Le Blanc, K.; Mueller, I.; Slaper-Cortenbach, I.; Marini, F.C.; Krause, D.S.; Deans, R.J.; Keating, A.; Prockop, D.J.; Horwitz, E.M. Minimal criteria for defining multipotent mesenchymal stromal cells. The International Society for Cellular Therapy position statement. Cytotherapy 2006, 8, 315–317. [Google Scholar] [CrossRef]
- Kohn, D. 17.4—Pathologien Hoffa-Fettkörüer. In Expertise Knie; Thieme, S., Ed.; Thieme: Stuttgart, Germany, 2016. [Google Scholar] [CrossRef]
- Saddik, D.; McNally, E.G.; Richardson, M. MRI of Hoffa’s fat pad. Skelet. Radiol. 2004, 33, 433–444. [Google Scholar] [CrossRef]
- Draghi, F.; Ferrozzi, G.; Urciuoli, L.; Bortolotto, C.; Bianchi, S. Hoffa’s fat pad abnormalities, knee pain and magnetic resonance imaging in daily practice. Insights Imaging 2016, 7, 373–383. [Google Scholar] [CrossRef]
- Jiang, T.; Xu, G.; Wang, Q.; Yang, L.; Zheng, L.; Zhao, J.; Zhang, X. In vitro expansion impaired the stemness of early passage mesenchymal stem cells for treatment of cartilage defects. Cell Death Dis. 2017, 8, e2851. [Google Scholar] [CrossRef] [PubMed]
- FDA. Regulatory Considerations for Human Cells, Tissues, and Cellular and Tissue-Based Products: Minimal Manipulation and Homologous Use. In Guidance for Industry and Drug Administration Staff; FDA: Silver Spring, MD, USA, 2017. [Google Scholar]
- Raposio, E.; Ciliberti, R. Clinical use of adipose-derived stem cells: European legislative issues. Ann. Med. Surg. 2017, 24, 61–64. [Google Scholar] [CrossRef] [PubMed]


| Positive Markers | CD105 PerCP-Cy5.5/CD73 APC/CD90 FITC |
|---|---|
| Additional Positive Drop-In Marker | CD44 PE |
| Negative Cocktail | CD45/CD34/CD11b/CD19/HLA-DR |
| Isotype Control | mlgG1, κ PerCP-Cy5.5/mlgG1, κ APC/mlgG1, κ FITC (for positive markers) mIgG1, κ/mIgG2a, κ PE (for negative cocktail) mIgG2b, κ PE (for CD44 drop in) |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Neubauer, M.; Kramer, K.; Neugebauer, J.; Moser, L.; Moser, A.; Dammerer, D.; Nehrer, S. Isolation and Cultivation of Adipose-Derived Mesenchymal Stem Cells Originating from the Infrapatellar Fat Pad Differentiated with Blood Products: Method and Protocol. Methods Protoc. 2023, 6, 3. https://doi.org/10.3390/mps6010003
Neubauer M, Kramer K, Neugebauer J, Moser L, Moser A, Dammerer D, Nehrer S. Isolation and Cultivation of Adipose-Derived Mesenchymal Stem Cells Originating from the Infrapatellar Fat Pad Differentiated with Blood Products: Method and Protocol. Methods and Protocols. 2023; 6(1):3. https://doi.org/10.3390/mps6010003
Chicago/Turabian StyleNeubauer, Markus, Karina Kramer, Johannes Neugebauer, Lukas Moser, Anna Moser, Dietmar Dammerer, and Stefan Nehrer. 2023. "Isolation and Cultivation of Adipose-Derived Mesenchymal Stem Cells Originating from the Infrapatellar Fat Pad Differentiated with Blood Products: Method and Protocol" Methods and Protocols 6, no. 1: 3. https://doi.org/10.3390/mps6010003
APA StyleNeubauer, M., Kramer, K., Neugebauer, J., Moser, L., Moser, A., Dammerer, D., & Nehrer, S. (2023). Isolation and Cultivation of Adipose-Derived Mesenchymal Stem Cells Originating from the Infrapatellar Fat Pad Differentiated with Blood Products: Method and Protocol. Methods and Protocols, 6(1), 3. https://doi.org/10.3390/mps6010003






